Abstract
Lipid-rich carcinoma is a rare histotype of canine mammary tumors with cytoplasmic vacuolation. In humans, glycogen-rich carcinoma, secretory carcinoma, and myoepithelial neoplasms are included in the differential diagnosis for lipid-rich carcinoma. The aim of the study was to investigate the existence of histotypes other than lipid-rich in canine mammary carcinomas with vacuolated cytoplasm using a diagnostic algorithm based on histopathology, histochemistry, immunohistochemistry, and ultrastructure and to evaluate the molecular phenotype of these neoplasms. Ten mammary carcinomas were collected, histologically reviewed, and subjected to histochemistry (PAS, PAS with diastase, Alcian blue, Sudan III [1 case], and Congo red [1 case]); immunohistochemistry for CK19, CK5/6, CK14, p63, calponin, vimentin, ER, PR, and HER2; and transmission electron microscopy (TEM). Cytokeratin immunolabeling demonstrated the epithelial origin of all tumors. Sudan III and TEM confirmed the diagnosis of lipid-rich carcinoma in 8 tumors (one amyloid-producing). One tumor was reclassified as a glycogen-rich carcinoma based on PAS reactivity that was diastase-labile, and a second tumor was reclassified as a carcinoma-and-malignant myoepithelioma based on the differentiation markers. Lipid-rich carcinomas were basal-like (5/8), null-type (2/8), and luminal A phenotype (1/8). The glycogen-rich carcinoma was basal-like, while the carcinoma-and-malignant myoepithelioma was luminal A. Vacuolated morphology of neoplastic cells in canine mammary carcinoma can indicate either a neoplasm of luminal epithelial origin with cytoplasmic lipid or glycogen, or vacuolated neoplastic suprabasal myoepithelial cells. Glycogen-rich carcinoma is a novel histological type that should be considered in the differential diagnosis for canine mammary carcinomas with vacuolated cytoplasm.
Keywords
Clear cell carcinomas of the breast were originally described as tumors with vacuolated, optically empty, clear cell cytoplasm. 17 Exceptionally rare types and variants of breast carcinoma with vacuolated cytoplasm are classified as secretory, sebaceous, lipid-rich, glycogen-rich, and acinic cell carcinomas and myoepithelial neplasms. 12 In the classification of canine mammary carcinomas, lipid-rich carcinoma is the only histological type with morphological features of large cytoplasmic vacuoles. 13,33 Lipid-rich carcinoma is extremely rare, usually found in young intact bitches, and has an unfavorable biological behavior with lymphatic invasion and nodal and distant metastasis. 10,13,33 Similarly, human lipid-rich carcinoma is frequently classified as histological grade III with triple negative phenotype, nodal metastasis at presentation, and a high first-year mortality rate. 12 Lipid-rich carcinoma of the human breast is diagnosed when no fewer than 90% of the cells contain abundant, cytoplasmic neutral lipid, either on histochemical or ultrastructural evaluation. 12 By contrast, in dogs, a diagnosis of lipid-rich carcinoma should only be made when more than 50% of the neoplastic cells have vacuolated cytoplasm. 33
The differential diagnosis for human lipid-rich carcinoma includes breast glycogen-rich, histiocytoid, secretory, signet-ring, myoepithelial and metastatic renal carcinomas, and carcinomas modified by hormonal therapy and chemotherapy. 12 Glycogen-rich carcinoma is a rare subtype of human invasive mammary gland carcinoma, in which at least 90% of the neoplastic cells have abundant, clear cytoplasm containing glycogen. 12 Several reports suggest that glycogen-rich carcinoma in women is an aggressive type, with a high incidence of axillary lymph node metastasis, high histological grade, and short disease-free survival and overall survival. 34
In humans, secretory carcinoma is a rare, translocation-associated (ETV6-NTRK3 gene), invasive carcinoma with a solid, microcystic and tubular architecture composed of cells that specifically produce intracellular and extracellular secretory material. 2,12 Secretory carcinoma has a low-grade clinical course with extremely rare distant metastasis and a favorable prognosis in younger human patients. 12 Secretory carcinoma is a rarely described canine mammary carcinoma composed of lobules and tubules of neoplastic cells with clear cytoplasm and prominent vacuoles, together with luminal spaces filled with eosinophilic secretion. 8
The aim of the study was to investigate the existence of histological subtypes other than lipid-rich carcinoma in canine mammary carcinomas with vacuolated cytoplasm, using a diagnostic algorithm based on histopathology, histochemical staining, immunohistochemical markers of differentiation, and ultrastructural characterization. The second aim was to evaluate the molecular phenotype of these neoplasms. 4 –6,24,33
Materials and Methods
Case Collection and Clinical Data
Ten formalin-fixed paraffin-embedded (FFPE) canine mammary carcinomas were retrospectively collected (2007–2012) from the database of the Pathology Service of the Department of Veterinary Medical Science of Bologna University, from the Department of Veterinary Sciences of Pisa University, and from the Department of Veterinary Medicine, University of Milan. The selection criterion was a diagnosis of lipid-rich carcinoma.
Tumor size data were collected during the trimming of the samples. Lymph node metastases were histologically investigated. Systemic metastases were histologically or cytologically and radiographically documented. Tumor-specific survival was clinically confirmed, and a follow-up period of 2 years was recorded.
Histology and Histochemistry
Sections 4-µm thick were cut from FFPE tissue and routinely stained with hematoxylin and eosin for the histological review of tumors. The slides were reviewed by 2 board-certified pathologists (LVM and BB). The tumors were graded according to the canine-adapted Nottingham system, based on the histological features of tubule formation, nuclear pleomorphism, and mitotic count. 25
Histochemical stains including periodic acid–Schiff (PAS), PAS with diastase, and Alcian blue were performed on all the collected neoplasms. Wet tissue was available in 1 case, and a portion of the neoplasm was snap-frozen in isopentane and cooled at −196 °C, cryosectioned at 5 μm, and stained with Sudan III. 3 In one case, where there was a morphological suspicion of amyloid, 8-μm-thick sections were stained with Congo red.
Immunohistochemistry
Seven consecutive sections from each block underwent immunohistochemistry (IHC) using antibodies to estrogen receptor (ER), progesterone receptor (PR), human epidermal growth factor receptor 2 (HER2), cytokeratin 5/6 (CK5/6), cytokeratin 14 (CK14), cytokeratin 19 (CK19), p63, calponin, and vimentin (Supplemental Table S1).
Sections were dewaxed and rehydrated. Endogenous peroxidase was blocked by immersion in 0.3% H2O2 in methanol for 30 minutes. Sections were then rinsed in Tris buffer and antigen retrieval was performed by incubation in citrate buffer (pH 6.0 except for sections labelled for CK5/6, which were incubated with EDTA, pH 8.0) and heated for two 5-minute periods in a microwave oven at 750 W, followed by cooling at room temperature for 20 minutes.
All antibodies were incubated with the tissue sections overnight at 4 °C. Binding sites were revealed by secondary biotinylated antibodies (dilution 1:200) and amplified using a commercial avidin-biotin peroxidase kit (VECTASTAIN ABC Kits). The chromogen DAB (3,3′-diaminobenzidine; 0.05% for 3 minutes at room temperature) was used (ACH500-IFU, ScyTek Laboratories). Slides were counterstained with Harris’ hematoxylin. The primary antibody was omitted in the negative control. As positive controls to assess the cross reactivity with canine tissues and the specificity of the immunohistochemical procedure, sections of normal canine mammary gland (for anti-ER and -PR, -CK5/6, -CK14, -CK19, -p63, -calponin, and -vimentin), normal canine uterus (for anti-ER and -PR antibodies), and canine skin (for anti-CK5/6, -CK14, -CK19, -p63 antibodies) were used following the same protocols. A human mammary ductal carcinoma (kindly provided by P. Viacava, Department of Oncology, University of Pisa, Italy) known to react with HER2 antibody and a scored 3+ 32 canine mammary carcinoma were used as positive controls for HER2. HER2 was evaluated according to the current ASCO/CAP (American Society of Clinical Oncology/College of American Pathologists) guidelines (score from 0 to 3+ where only 3+ are considered positive). 32 The immunohistochemical panel was evaluated following the recommended guidelines for canine mammary tumors. 26 The tumors were classified into 5 molecular subtypes according to the previous literature. 4 –6,24,33 Immunohistochemical expression of luminal and basal cytokeratins in the different subtypes was interpreted based on the criteria for human breast tumor investigation. 1
Transmission Electron Microscopy (TEM)
All the tumors were processed for TEM. 14 FFPE tissues were available for review. From all FFPEs, 5 mm 3 areas of interest were selected by comparison with related hematoxylin and eosin–stained sections and dewaxed in xylene, washed in a graded series of ethanol (100%, 95%, 70%), rehydrated rapidly in distilled water, and then rinsed in 0.15 M cacodylate buffer overnight. Rehydrated tissue samples were postfixed in 1% OsO4 in cacodylate buffer, dehydrated in graded ethanol, and embedded in Araldite. Ultrathin sections, stained with uranyl acetate and lead citrate, were examined with a Philips TEM CM100 Transmission Electron Microscope.
Histochemical, Immunohistochemical, and Ultrastructural Algorithm
Based on the human breast cancer literature on the differential diagnosis of carcinoma with clear vacuoles, 12 we created a diagnostic algorithm to investigate the chemical components and differentiation lineage of canine carcinoma with clear vacuoles (Table 1 and Supplemental Figure S1). The histological entities that we investigated were lipid-rich carcinoma, glycogen-rich carcinoma, secretory carcinoma, and carcinoma-and-malignant myoepithelioma.
Diagnostic Algorithm for Canine Mammary Carcinoma With Vacuolated Cytoplasm.
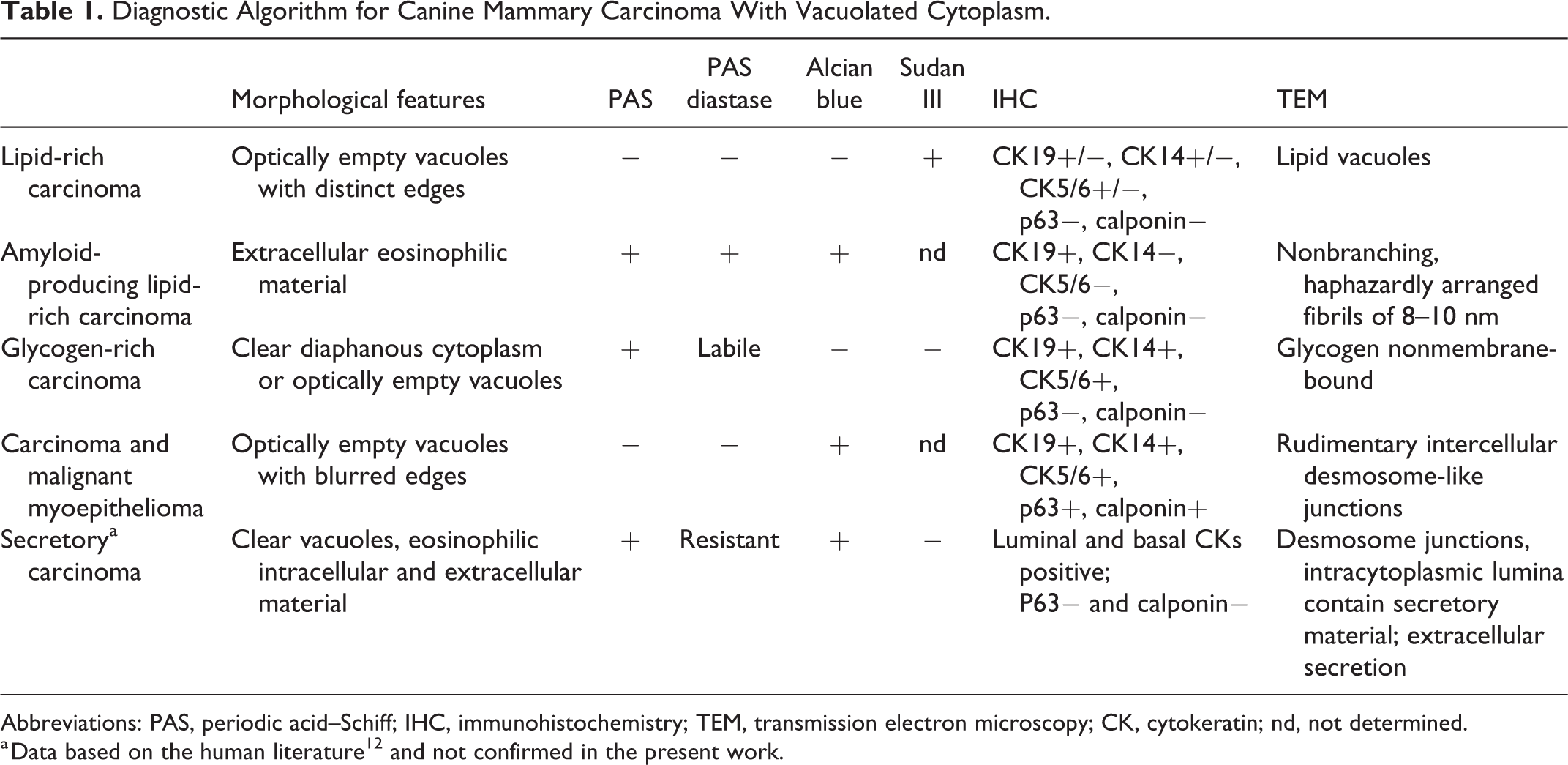
Abbreviations: PAS, periodic acid–Schiff; IHC, immunohistochemistry; TEM, transmission electron microscopy; CK, cytokeratin; nd, not determined.
a Data based on the human literature 12 and not confirmed in the present work.
The diagnostic scheme was based on the following:
Histological features: presence of optically empty vacuoles with distinct or indistinct edges and/or diffuse diaphanous (translucent) cytoplasm. Vacuoles were morphologically characterized and semiquantitatively scored as the percentage of neoplastic cells affected (from 50% to 100%).
Histochemical stains: PAS, PAS with diastase, Alcian blue, and Sudan III (the latter only on frozen samples).
Immunohistochemistry: p63, calponin, cytokeratin 14, cytokeratin 5/6, and cytokeratin 19.
Ultrastructural features: presence of intercellular junctions, lipid vacuoles, and glycogen.
Results
Revised Diagnosis Based on the Proposed Algorithm
Based on the proposed algorithm, by integration of morphological, histochemical, immunohistochemical, and ultrastructural data, the 10 lipid-rich carcinomas were reclassified as 8 lipid-rich carcinomas (one of which was amyloid-producing), 1 glycogen-rich carcinoma, and 1 carcinoma-and-malignant myoepithelioma.
Lipid-Rich Carcinoma
The 8 cases of lipid-rich carcinoma included 7 females (3 intact and 4 spayed) and 1 male with a concurrent testicular interstitial cell tumor. The mean age at the time of diagnosis was 7 years. The breeds represented were mixed breed (2), German Shepherd, Rottweiler, English Setter, Maltese, Bolognese, and Bulldog (1 for each breed). The mean tumor size was 3.8 cm, and the affected mammary gland were the IV and V. A clinical 2-year-follow-up was available in 4 cases, with tumor-specific survival times of 3, 17, 18, and 24 months. Systemic metastases were histologically confirmed in one case (case 6, Supplemental Table S2) and radiographically and cytologically confirmed in one other case (case 7, Supplemental Table S2).
Lipid-rich carcinomas were composed of polygonal cells with a moderate amount of cytoplasm containing single or multiple, optically empty vacuoles with distinct edges (Fig. 1). The percentage of neoplastic cells with vacuoles in their cytoplasm ranged from 60% to 100%. Mitotic count ranged from 3 to 24 in 10 high-power fields (2.37 mm 2 ).
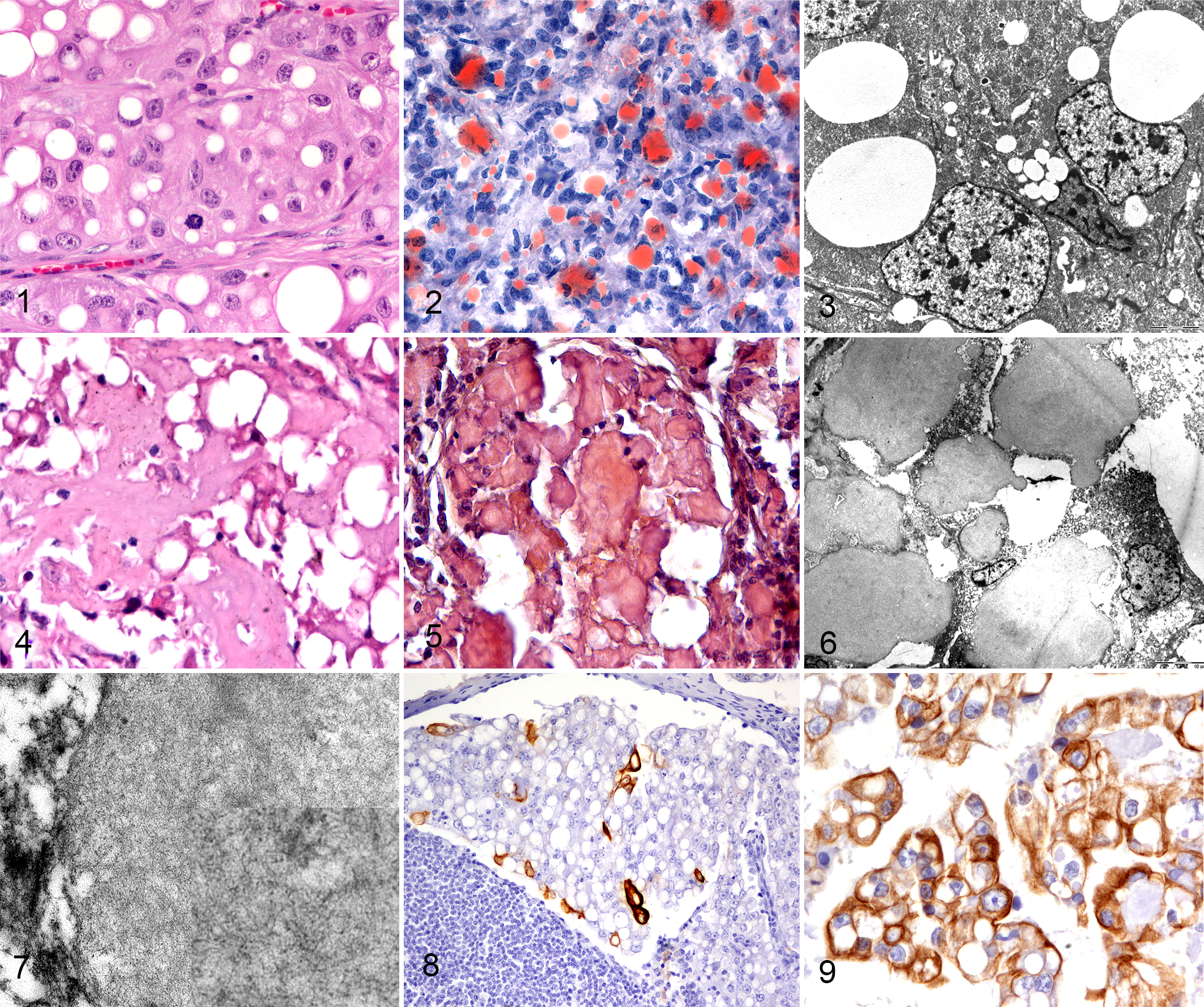
Lipid-rich carcinoma, mammary gland, dog.
On the one formalin-fixed frozen sample available, neoplastic cells stained positively with Sudan III (Fig. 2). Clear vacuoles were negative by PAS and Alcian blue stains in all cases. The electron-lucent vacuoles were ultrastructurally identified as lipid vacuoles, confirming the diagnosis of lipid-rich carcinoma (Fig. 3).
In one carcinoma, in addition to the lipid content, abundant extracellular eosinophilic material was detectable (Fig. 4). The eosinophilic material was PAS-positive and diastase-resistant, and Alcian blue and Congo red positive (Fig. 5), and therefore was interpreted as amyloid. The ultrastructural features of both the intracytoplasmic and extracellular material (Fig. 6) were of haphazardly arranged, 8 to 10 nm diameter, nonbranching fibrils, consistent with amyloid (Fig. 7). The neoplasm was classified as amyloid-producing lipid-rich carcinoma.
Three lipid-rich carcinomas were grade III, and 5 were grade II. Lymphovascular invasion was observed in 2 cases. One case (case 6, Supplemental Table S2) had lymph node metastases and systemic metastasis to the liver, lung, and spleen. Another case (case 7, Supplemental Table S2) had metastases to liver and lung.
In all the examined cases, the epithelial origin of the neoplasms was confirmed by the presence of intercellular junctions by TEM and immunohistochemical expression of cytokeratins. Vimentin was negative in all but one case that was also characterized by concurrent immunolabeling for CK14.
The immunophenotype of lipid-rich carcinoma was triple negative basal-like in 5 of 8 primary mammary tumors, based on positive labeling of basal cytokeratins (CK14, CK5/6; Fig. 8). All the neoplasms were negative for ER, but one lipid-rich carcinoma showed expression of PR and was therefore classified as luminal A. None of the examined neoplasms showed overexpression of HER2. Two neoplasms were classified as null type, displaying negativity to all antibodies in the panel. CK19 was expressed in the cytoplasm of neoplastic cells in 6 of 8 lipid-rich carcinomas (Fig. 9). Co-expression of luminal and basal cytokeratin was present in 3 of 5 basal-like carcinomas, defining a mixed subtype. There was no immunolabeling of neoplastic epithelial cells for myoepithelial markers (p63, calponin). p63 and calponin labeling were diffusely lost in all 8 carcinomas, confirming stromal invasion. The immunophenotype in one lymph node metastasis was concordant with the primary site (basal-like), while the other lymph node metastasis was discordant with the primary tumor (null-type and basal-like, respectively). Overall results are summarized in Supplemental Table S2.
Glycogen-Rich Carcinoma
The single case of glycogen-rich carcinoma was an 11-year-old, intact female Boxer dog with a 1.5 cm mass in the fifth mammary gland. No follow-up information was available. The glycogen-rich carcinoma was composed of lobules, tubules, and cords of polygonal cells with sharply distinct borders, moderate to abundant amounts of clear, diaphanous cytoplasm (Fig. 10) or with optically empty vacuoles and an intermediate N/C ratio. There were 25 mitoses in 10 high-power fields (2.37 mm 2 ). The percentage of neoplastic cells with diffusely vacuolated, diaphanous cytoplasm was 60%. The neoplasm was grade III.
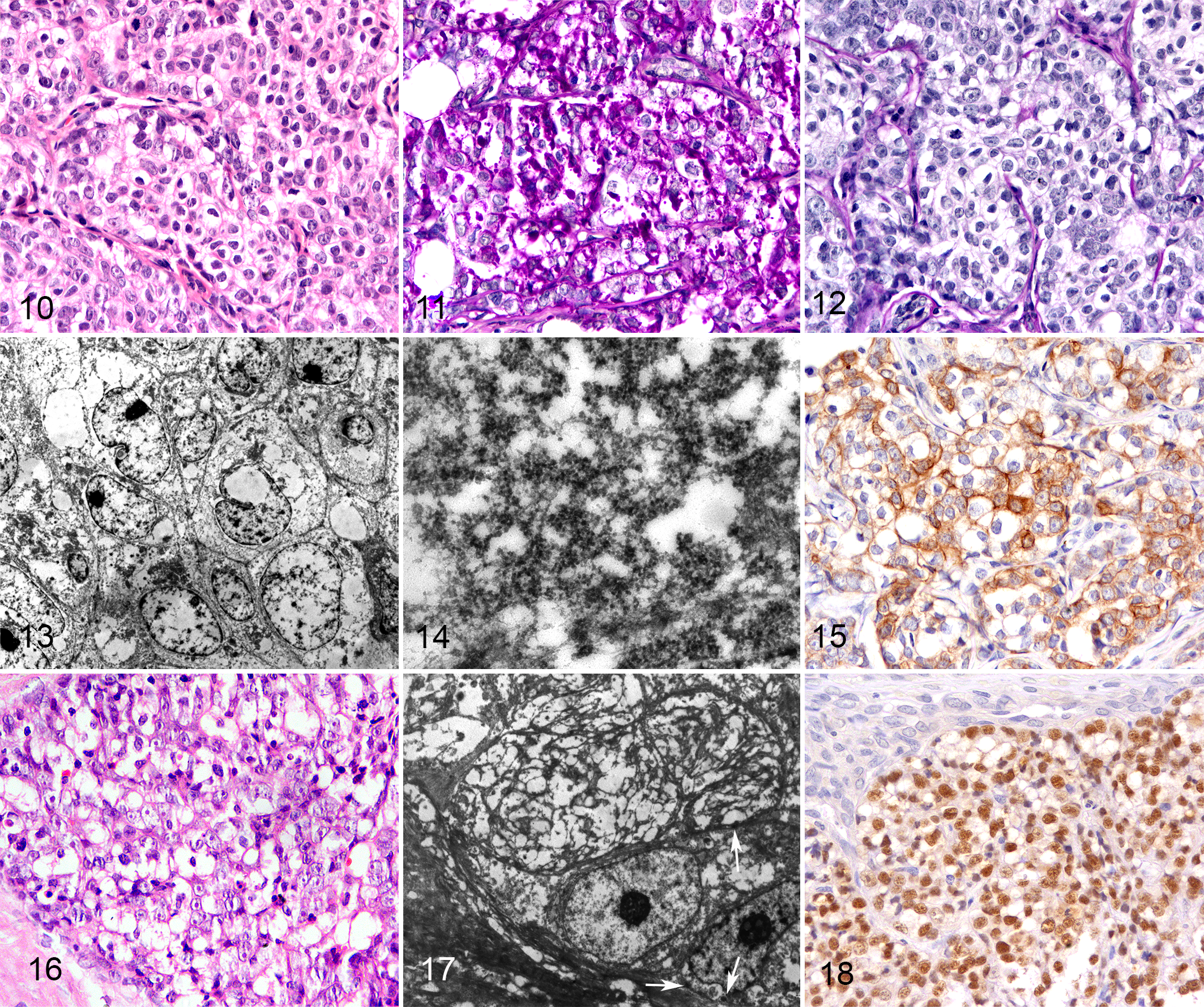
Glycogen-rich carcinoma, mammary gland, dog.
The cytoplasm of neoplastic cells was diffusely and strongly positive for PAS (Fig. 11) and diastase-labile (consistent with glycogen; Fig. 12). Ultrastructural analysis confirmed the presence of abundant, granular, electron-dense cytoplasmic material (glycogen) in the neoplastic cells, supporting the diagnosis of glycogen-rich carcinoma. However, glycogen was only visible in a few areas (Figs. 13, 14) due to partial extraction during routine processing of FFPE specimens for TEM study.
A draining lymph node was massively invaded by the glycogen-rich carcinoma with the same morphological and histochemical features seen in the primary tumor. The glycogen-rich carcinoma was immunolabeled for CK19 and CK5/6 (Fig. 15), scattered expression of CK14, and no labeling for ER, PR, or HER2. It had a basal-like mixed phenotype in the primary tumor and lymph node metastasis. No immunoexpression of p63 or calponin was present.
Carcinoma-and-Malignant Myoepithelioma
The single case of carcinoma-and-malignant myoepithelioma was a 10-year-old, intact female, mixed breed. Carcinoma-and-malignant myoepithelioma was composed of tubules, papillae, and solid sheets of polygonal cells. The biphasic nature of the tumor was subsequently defined by immunohistochemistry. The solid component was characterized by optically empty cytoplasmic vacuoles with blurred edges (Fig. 16) and an intermediate nucleus to cytoplasm ratio. There were 17 mitoses in 10 high-power fields (2.37 mm 2 ). The neoplasm was grade III. Transmission electron microscopy demonstrated rudimentary intercellular desmosome-like junctions between cells featuring an elongated nucleus (Fig. 17). Basal lamina and remnant cytoplasmic myofibrils were not clearly recognizable due to retrieval from FFPE specimens.
The immunophenotype of the carcinoma-and-malignant myoepithelioma revealed the biphasic nature of the neoplasm. The tubular and papillary components were CK19-positive. The solid sheets of vacuolated cells expressed CK14, CK5/6, p63 (Fig. 18), and calponin. There was expression of PR and no staining for ER and HER2, compatible with a luminal A immunophenotype.
Discussion
According to the current classification system for dogs, lipid-rich carcinoma is the only mammary carcinoma histotype that can be diagnosed in the presence of cytoplasmic vacuolation of neoplastic cells. 33 By contrast, in humans, breast cancers with clear cells include several tumor types: lipid-rich carcinoma, glycogen-rich carcinoma, apocrine carcinoma, secretory carcinoma, and myoepithelial neoplasms. 12 A diagnostic algorithm was applied to investigate other histotypes of mammary carcinoma with vacuolated cytoplasm in dogs. In the present study, 8 lipid-rich carcinomas (one of which was amyloid-producing), 1 glycogen-rich carcinoma, and 1 carcinoma-and-malignant myoepithelioma were identified, confirming that canine mammary carcinomas with vacuolated cytoplasm can include different tumor entities other than lipid-rich carcinoma, as in the human counterpart.
The first tumor parameter to be evaluated in the diagnostic algorithm is morphological. In fact, based on the data obtained and on the reviewed classification, the morphological characteristics of the clear cytoplasm can be useful for guiding the diagnostic procedure. Lipid-rich tumors always have cytoplasm with optically empty vacuoles that have distinct margins. On the contrary, the solid myoepithelial component of carcinoma-and-malignant myoepithelioma has optically empty cytoplasmic vacuoles with blurred, poorly defined margins. Finally, glycogen-rich carcinomas have diffusely clear cytoplasm with a central nucleus and prominent cell margins (colloquially referred to as “fried eggs appearance”). 11,16,17,19,23,28 Histochemistry and immunohistochemistry are supportive in characterizing the different histological subtypes, and ultrastructural analysis can confirm the diagnosis.
Demonstration of the lipid content can be achieved with Sudan III stain on frozen sections. In the absence of frozen samples, ultrastructural analysis can be performed on FFPE material. 9 Based on our results, the diagnosis of lipid-rich carcinoma should require the presence of vacuoles in at least 50% of neoplastic cells as well as the demonstration of lipid content within the vacuoles, as defined by the current classification. 33
Localized amyloid deposition has been reported in canine mammary carcinoma 29 and amyloid-producing lipid-rich carcinoma is a very rarely described variant of lipid-rich carcinoma in which the amyloid and the neoplastic cells are β-casein positive. 30 Amyloid is ultrastructurally defined by the presence of aggregates of nonbranching fibrils with a diameter of 8 to 10 nm. 21 The present case of amyloid-producing lipid-rich carcinoma was Congo red–positive and had the canonical ultrastructural features reported in the literature. 21
According to the literature, the epithelial cells of lipid-rich carcinoma can express both luminal and basal cytokeratins 27 and usually do not express estrogen and progesterone receptors. 10
Lipid-rich carcinomas in our study expressed luminal cytokeratin (CK19), basal cytokeratins (CK5/6 and CK14), no hormone receptors (ER, PR; with one exception for PR), and no HER2, in line with the canine literature. 10,27 In humans 12,20 and dogs, 10 lipid-rich carcinoma has a triple negative immunophenotype with loss of hormone receptors, and its behavior is typically aggressive with nodal or distant metastasis. In the present study, the lipid-rich neoplasms were triple negative in 7 of 8 cases (5 basal-like and 2 null-type). Three of 5 basal-like carcinomas co-expressed the luminal CK19, defining a mixed subtype. In human breast cancer, expression of CK19 is suggestive of maintained glandular differentiation; 1 it is almost always expressed by carcinomas regardless of the phenotype 15,31 and is a useful marker in the detection of micrometastasis in lymph node core biopsies. 31 A mixed luminal/basal subtype characterized by co-expression of luminal and basal cytokeratins has been described in human breast cancer with a biological behavior more similar to the basal-like phenotype. 1 In only one case was there expression of PR, and the neoplasm classified as luminal A. Both concordant and discordant immunophenotypes were observed in the metastatic sites compared to the primary tumors, as previously described in the dog. 4,5
Secretory carcinoma is an additional histological type that has been reported in dogs in one previous publication. 8 In the current canine classification system, the term secretory carcinoma is used as a synonym of lipid-rich carcinoma. 33 By contrast, in humans, secretory carcinoma is considered a separate entity characterized by secretory material within neoplastic tubules and translocation of ETV6-NTRK3 gene. As our study confirms the presence of several analogies between canine and human carcinomas, we believe that the use of the term secretory as a synonym for lipid-rich carcinoma should be discouraged, and that it should be restricted to those neoplasms showing both clear vacuoles, eosinophilic intracellular and extracellular material that is PAS positive, PAS diastase resistant, and Alcian blue positive. 12
Glycogen-rich clear cell carcinoma is a rare breast carcinoma accounting for 0.9% to 3% of human breast neoplasms. 11,23 The typical cellular feature of human glycogen-rich clear cell carcinoma of the breast is the “fried eggs appearance” with clear cytoplasm and small, dark, punctate central nuclei. 11,16,17,19,23,28 The diagnosis in humans is confirmed by PAS-positive, diastase-labile, intracytoplasmic glycogen granules in more than 90% of neoplastic cells 11,16,17,23,28 Ultrastructural analysis confirms the presence of tight junctions and abundant nonmembrane bound glycogen in the neoplastic cells. 17 In the present canine case, histological features were similar to the human cases, but glycogen was present in only 60% of neoplastic cells. A canine adapted cutoff value should be considered, similar to lipid-rich carcinoma in dog. 33 Moreover, the glycogen component was maintained in the lymph node metastasis. In this case, a basal-like phenotype was confirmed, suggesting a poor prognosis. 18 Prognosis of glycogen-rich carcinoma of the human breast is also generally poor and is typically associated with a high-grade, advanced-stage, triple-negative hormone receptor status, lymph node metastasis, and high mortality rate. 17,22,34 Glycogen-rich mammary carcinoma has also been reported in cats. 7
Canine carcinoma-and-malignant myoepithelioma is a biphasic neoplasm characterized by the presence of malignant luminal epithelial and malignant myoepithelial cells. 33 The myoepithelial cells may have different morphology 5,33 ranging from spindle to stellate and polygonal. The presence of cytoplasmic vacuoles can occur in myoepithelial neoplasms; therefore, further immunohistochemical characterization in mammary carcinoma with clear vacuoles should be considered to investigate a myoepithelial origin of the neoplasm. 33
In conclusion, the proposed algorithm can be useful in distinguishing different histological entities among mammary carcinomas with vacuolated cell morphology, which may be associated with different biologic behavior. According to the results of this study, vacuolated morphology in canine mammary carcinoma can indicate either an epithelial phenotype with lipid or glycogen content or a myoepithelial phenotype. As expected, the lipid-rich carcinoma was the most prevalent. Glycogen-rich carcinoma is a newly described, rare histological subtype of canine mammary carcinoma, characterized by clear cytoplasm, diastase-sensitive PAS positivity, and a triple negative phenotype. Lipid-rich carcinoma and glycogen-rich carcinoma should be considered as a potential differential diagnosis for mammary carcinoma with vacuolated cytoplasm in dogs.
Supplemental Material
Combined_supplemental_materials-Muscatello_et_al_v2 - Canine Mammary Carcinoma With Vacuolated Cytoplasm: Glycogen-Rich Carcinoma, a Histological Type Distinct From Lipid-Rich Carcinoma
Combined_supplemental_materials-Muscatello_et_al_v2 for Canine Mammary Carcinoma With Vacuolated Cytoplasm: Glycogen-Rich Carcinoma, a Histological Type Distinct From Lipid-Rich Carcinoma by Luisa Vera Muscatello, Valentina Papa, Francesca Millanta, Giuseppe Sarli, Barbara Bacci, Giovanna Cenacchi, Alessandro Poli, Chiara Giudice and Barbara Brunetti in Veterinary Pathology
Footnotes
Acknowledgement
We thank Professor Maria Pia Foschini for the precious discussion about molecular classification of human breast cancer and Dr Arianna Pasquini for the excellent technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
