Abstract
Human epidermal growth factor receptor 2 (HER2) is a tyrosine kinase receptor overexpressed in a subset of breast cancer due to HER2 gene amplification. HER2 protein is expressed in feline mammary carcinomas, but little is known about its cytogenetic alterations. The aim of this study was to evaluate HER2 gene amplification status and its correlation with HER2 protein expression in feline mammary carcinomas. Feline mammary carcinomas were retrospectively selected and immunohistochemically (IHC) evaluated for HER2 protein expression. All the HER2 IHC-positive (3+) and equivocal (2+) cases and a subset of negative cases (0/1+) were selected for fluorescence in situ hybridization (FISH). Dual-core tissue microarrays were prepared for FISH. IHC and FISH were evaluated according to the 2013 American Society of Clinical Oncology/College of American Pathologists guidelines. The study included 107 feline mammary carcinomas from 88 queens. HER2 protein expression was positive (3+) in 7 cases (6.5%), equivocal (2+) in 48 cases (45%), and negative (0/1+) in 52 cases (48.5%). HER2 status was indeterminate in 8 feline mammary carcinomas (12%), amplified in 3 (4%), equivocal in 4 (6%), and nonamplified in 53 (78%). HER2 gene amplification and protein expression were significantly positively correlated (R = 0.283; P < .0001). HER2 gene is amplified in a subset of feline mammary carcinomas despite the HER2 positive or equivocal protein expression, but it remains to be determined if the HER2 amplification is a gene alteration that drives mammary tumor carcinogenesis or only a bystander passenger mutation.
Keywords
Tyrosine kinase receptor mutations or structural alterations are responsible for neoplastic transformation 2 since they become oncoproteins and acquire intrinsic tyrosine kinase activities. 13 Oncoproteins are activated in tumors through numerous chromosomal changes like point mutations, gene rearrangements, and gene amplification. 13 Human epidermal growth factor receptor 2 (HER2) gene amplification is widely described in humans. The increased copy numbers of HER2 gene trigger overexpression of its receptor tyrosine kinase product, which is implicated in the poor prognosis of breast cancer and ovarian carcinomas. 13 Epidermal growth factor receptors (EGFRs) are a class of mutated and overexpressed receptors in several human tumors. 14 HER2 is a transmembrane receptor with kinase activity and is considered an orphan receptor that fails to form the tetrahedral structure normally present in the extracellular region of the other EGFR receptors. 4 HER2-positive tumors represent 20% of breast cancers in women, second in incidence to tumors expressing hormone receptors. 15 The introduction of trastuzumab (Herceptin), a humanized monoclonal antibody that targets, binds, and inhibits HER2, ensures ample therapeutic successes in around a third of patients with HER2-positive tumors. 15 Before the advent of this drug, HER2-positive tumors were considered to have a highly unfavorable clinical course and poor survival rate of <10 years. 15 The exceptional therapeutic efficacy of trastuzumab demonstrates the importance of HER2 as an oncogenetic tumor conductor. 15 HER2 gene was first sequenced in cats in 2005. 7 Since then, many studies have investigated HER2 protein expression in feline mammary tumors, reporting variable results. 3,5,7,17,25,28 The standard guidelines for HER2 evaluation in canine mammary tumor propose considering positive cases as only those with a 3+ score by immunohistochemistry (IHC). 24 In humans, based on the American Society of Clinical Oncology/College of American Pathologists (ASCO/CAP) guidelines, equivocal IHC cases (2+) are subjected to fluorescence in situ hybridization (FISH) analysis. 33 Only 1 study detected HER2 gene amplification by chromogenic in situ hybridization in feline mammary tumors, 23 whereas no study in veterinary medicine has detected HER2 amplification using the FISH technique.
The aim of this study was to evaluate HER2 gene amplification status in feline mammary carcinoma using FISH and correlate it with the protein expression of its tyrosine kinase receptor.
Materials and Methods
Samples, History, and Histological Diagnosis
This retrospective study included 107 feline mammary carcinomas from 88 animals. The cases were consecutively selected from the archives of the Anatomic Pathology Service of the Department of Veterinary Medical Sciences, University of Bologna on the basis of a diagnosis of carcinoma with stromal invasion and excluding carcinoma in situ. The samples were available as formalin-fixed, paraffin-embedded material, and histological sections were routinely stained with hematoxylin and eosin (HE).
All methods were performed in accordance with relevant guidelines and regulations. The experiments were in compliance with the current national legal treatment of animal tissue samples and subsequent to the clinical informed consensus from the animal’s owners.
The anamnestic and clinical data, such as breed, age, sex, and anatomical location and size of the tumor, were collected. Histological diagnosis was based on the World Health Organization (WHO) classification of feline mammary tumors 19 and the current literature. 30 The histological grade was based on lymphatic invasion, nuclear form, and mitotic count. 18
Immunohistochemistry
Histologic sections (3-micron-thick) were dewaxed and rehydrated. Endogenous peroxidase was blocked by immersion in 3% H2O2 in methanol for 30 minutes at room temperature. Antigen retrieval was performed by incubation in citrate buffer, pH 6.0, for 10 minutes in a microwave oven at 750 W, followed by cooling at room temperature for 20 minutes. An antibody to HER2 (polyclonal, A0485, dilution 1/200; Dako, Glostrup, Denmark) known to be cross-reactive with the feline antigen 7 was incubated with the tissue sections overnight at 4°C. Binding sites were revealed by secondary biotinylated antibody (dilution 1:200) and amplified using a commercial avidin-biotin-peroxidase kit (VECTASTAIN; ABC Kits, Peterborough, UK). The chromogen 3,3′-diaminobenzidine (0.05% for 3 minutes at room temperature) was used. Slides were counterstained with Papanicolaou’s hematoxylin. Primary antibody was replaced with an irrelevant, isotype-matched antibody as a negative control. Sections of normal feline mammary gland and feline mammary carcinoma score 3+ 24,33 were used as controls to assess the specificity of the IHC procedure.
Evaluation of HER2 protein IHC expression was recorded by 2 methods:
The 2013 ASCO/CAP recommendations for IHC divide immunoreactivity to HER2 into the following 4 classes: 0 and 1+ (negative), 2+ (equivocal), and 3+ (positive). The ASCO/CAP algorithm is based on the circumferential membrane staining (complete, incomplete), membranous intensity of staining (strong, moderate, weak), and the percentage of positive tumor cells (cutoff value of 10%). The immunoreaction should be estimated using a low-power objective (10×) and observed within a homogeneous and contiguous invasive cell population. 33
Like the current recommendations, 33 the Consensus of Standard Guidelines on HER2 evaluation in canine mammary tumors, based on the 2007 ASCO/CAP recommendations, 34 group immunoexpression in the same categories (0, 1+, 2+, 3+) but recommend considering positive only cases with score 3+ characterized by a strong, complete, homogeneous membrane labeling (chicken-wire pattern) in >30% of cells. Cases reaching this pattern in ≤30% of cells or with a weak or moderate heterogeneous complete membrane labeling in at least 10% of cells are considered with the score 2+. Cases with no labeling (0) or weak, incomplete membrane labeling (1+) in any percentage of cells are considered negative. 24
Analysis of Gene Sequences
To assess the identity of the nucleotide sequence of the HER2 gene between feline and human species, the sequence alignment was verified using the BLAST (Basic Local Alignment Search Tool—NCBI) database 35 (http://blast.ncbi.nlm.nih.gov/Blast.cgi). The alignment showed a 92% identity to the transcripts (messenger RNAs [mRNAs]) and an 82% identity to the genomic sequence between feline and human species.
Tissue Microarray
Based on the IHC results, 68 tumors were selected for FISH assay. All 3+ and 2+ scored samples and a subset of randomly selected 1+ and 0 scored samples underwent FISH. Two of the 68 tumors were examined as whole sections, and the remaining 66 cases were included in 11 tissue microarrays (TMAs). Each TMA was made up of 12 cores, with 2 cores per case (TMA dual core), and a control tissue.
First, a sector map was designed depicting the layout, which specified the location within the tissue array for each core sample, and this was used to guide both assembly and subsequent scoring. 21,32 The sector map consisted of an Excel (Microsoft, Redmond, WA) sheet containing the exact position of each case on the slide. The layout was asymmetrically designed, and a different tissue (lung) was included on each TMA as a landmark (orientation core) and negative control. Tissue microarray is based on the method of Kononen 12 of extracting a cylindrical core of paraffin wax–embedded “donor” block and inserting it into a “recipient” wax block. 10 The 13 cores for each TMA were extracted manually with a skin biopsy punch of 3 mm in diameter. Before extraction, the area of interest was selected on the IHC slide by light microscopy examination of the most representative area. We included all the positive cases (3+), all equivocal (2+) cases, and 13 negative (1+ and 0) cases. The selection criteria were the presence of neoplastic tissue with HER2 expression, evaluated according to the score, the most representative of the same section, absence of necrosis, inflammation, and desmoplasia. After extraction from donor blocks, the tissue cores were reintegrated in the previously created empty cylinders in recipient blocks and subjected to reinclusion. Two serial sections were cut from each TMA: the first stained with HE as quality control to review the array and assess its quality, 21,26 confirming the presence of neoplastic tissue; the second used to perform FISH.
FISH
FISH was standardized and carried out at the Cytogenetics Laboratory of Bellaria Hospital (Bologna, Italy). Eleven TMA sections constituted by a total of 66 cases dual core and 2 whole sections were subjected to FISH analysis. The sections were incubated at 60°C for 10 minutes in an oven and in a thermal plate for 10 minutes at 75°C. The sections were dewaxed in xylene with three 20-minute steps and rehydrated in absolute ethanol with 2-minute steps and air-dried at room temperature (RT). 9 Incubation was performed in Tris EDTA (ethylenediaminetetraacetic acid) pH 9.00 in a heating bath at 98°C for 1 hour, followed by cooling at RT for 20 minutes. The sections were then washed in 2× SSC (pH 7.00 buffer sodium citrate) for 2 minutes.
The dehydration was performed with 2-minute steps in 70%, 85%, and 100% ethanol. The sections were air-dried at RT; the buffer solution (Smart-ISH solve; OaCP IE LTD, Cork, Ireland) and the probe for HER2 were applied based on the manufacturer’s instructions (Her2/Neu (17q12)/SE 17, KreatechTM FISH probes). The HER2 probe is a dual-color probe designed for the human species to identify the HER2 gene (red fluorescence) and the α-satellite sequences of the centromeric region of the chromosome 17q12 (green fluorescence). The already tested BAC probe 662A1H2 that hybridized the p1.12–p1.11 regions of the chromosome E1, including the gene NOR, 6 was used in HER2 equivocal and amplified cases to verify the presence/absence of polysomy of feline chromosome E1 in which feline HER2 is located. The hybridization area was covered with a coverslip and sealed with rubber cement. The slides were incubated at 75°C for 10 minutes for DNA denaturation and at 37°C overnight for hybridization. The slides were washed in NP40 0.5%/2× SSC (pH 7.0–7.5) at 75°C for 2 minutes and in 2× SSC for 2 minutes at RT. The samples were then dehydrated in ethanol. Chromatin counterstaining was obtained using 4′,6′-diamidino-2-phenylindole suspended in Antifade diluents. Human breast cancer with amplified HER2 was used as a positive control. The specificity of the in situ hybridization was further evaluated considering the euploidy of the fibroblasts and lymphocytes adjacent to the neoplastic cells and of the pulmonary tissue core used as a TMA landmark. Tissue slides were evaluated using an epifluorescence microscope. Cytogenetic-specific image analysis software (CytoVision, Leica Biosystems, Nussloch, Germany.) was used to count the number of gene copies per nucleus in at least 60 tumor nuclei for each case.
The evaluation system used to interpret FISH sections is based on the 2013 ASCO/CAP guidelines.
33
Because of the differences in the α-satellite sequences of the centromeric region between the human and feline species, the green signal was considered not specific. Therefore, the HER2 test was evaluated as an ISH single-probe array. The 2013 ASCO/CAP guidelines categorize HER2 testing in situ hybridization (ISH) in 3 classes, examining the average gene copy number:
33
ISH positive: average HER2 copy number ≥6.0 signals/cell ISH equivocal: average HER2 copy number of ≥4 and <6 signals/cell ISH negative: average HER2 copy number of <4 signals/cell Samples were considered indeterminate for HER2 if technical issues prevented 1 or both tests (IHC or ISH) from being classified as positive, negative, or equivocal. The conditions of technical errors include artifacts and preanalytic conditions.
33
Statistical Analysis
Statistical analysis was performed with Statistica software (Statsoft, Inc., Tulsa, Oklahoma, United States.). The normality distribution of the data was assessed with Shapiro and Wilk’s W test. The mean and standard deviation were specified for normally distributed data; for data not normally distributed, the median has been indicated. Continuous not normally distributed data were analyzed with the Spearman test. Categorical data were examined with the Pearson χ2 test. P value ≤.05 was considered significant.
Results
Animals, Histological Diagnosis, and Grading
The study included 107 mammary carcinomas from 88 cats. The mean age at diagnosis was 11.8 years ± 2.8 (mean and standard deviation). The main breed represented was the Domestic Short Hair (74/88), followed by Persian (6/88), Siamese (4/88), Burmese (1/88), Tabby (1/88), Chartreux (1/88), and Norwegian (1/88) breeds. The animals examined were entire (49/88) and spayed (39/88) females. The thoracic mammary glands were the most common tumor sites (62/107). Median tumor size was 22.7 mm (range, 2–120 mm). The most frequent diagnosis was simple tubulopapillary carcinoma (51/107), followed by simple solid carcinoma (29/107), mucinous carcinoma (8/107), micropapillary infiltrating carcinoma (5/107), ductal carcinoma (5/107), cribriform carcinoma (5/107), anaplastic carcinoma (3/107), and adenosquamous carcinoma (1/107). Tumors were grade 1 in 21 cases (20%), grade 2 in 37 (35%), and grade 3 in 49 cases (46%).
HER2 Protein Expression
The 2 evaluation systems used 24,33 for the immunoreactivity score of HER2 showed overlapping results in all mammary tumors examined (R = 1; P < .0000001). Among the 107 cases, HER2 expression was positive (3+) (Fig. 1) in 7 (6.5%), with the typical chicken-wire membranous pattern visible at low magnification. HER2 immunoreactivity was equivocal (2+) (Fig. 2) in 48 cases (45%) and negative (0/1+) in 52 (48.5%), of which 36 (33.6%) had a 1+ score (Fig. 3) and 16 (14.9%) had a 0 score (Fig. 4). Cytoplasmic staining was present but was considered nonspecific. The equivocal group (2+) was the most represented, followed by the negative (0/1+) and positive (3+) groups. All tumors with HER2 IHC 3+ were grade 3, but the association between tumor grade and the IHC expression of HER2 was not statistically significant (Pearson χ2 P = .17).
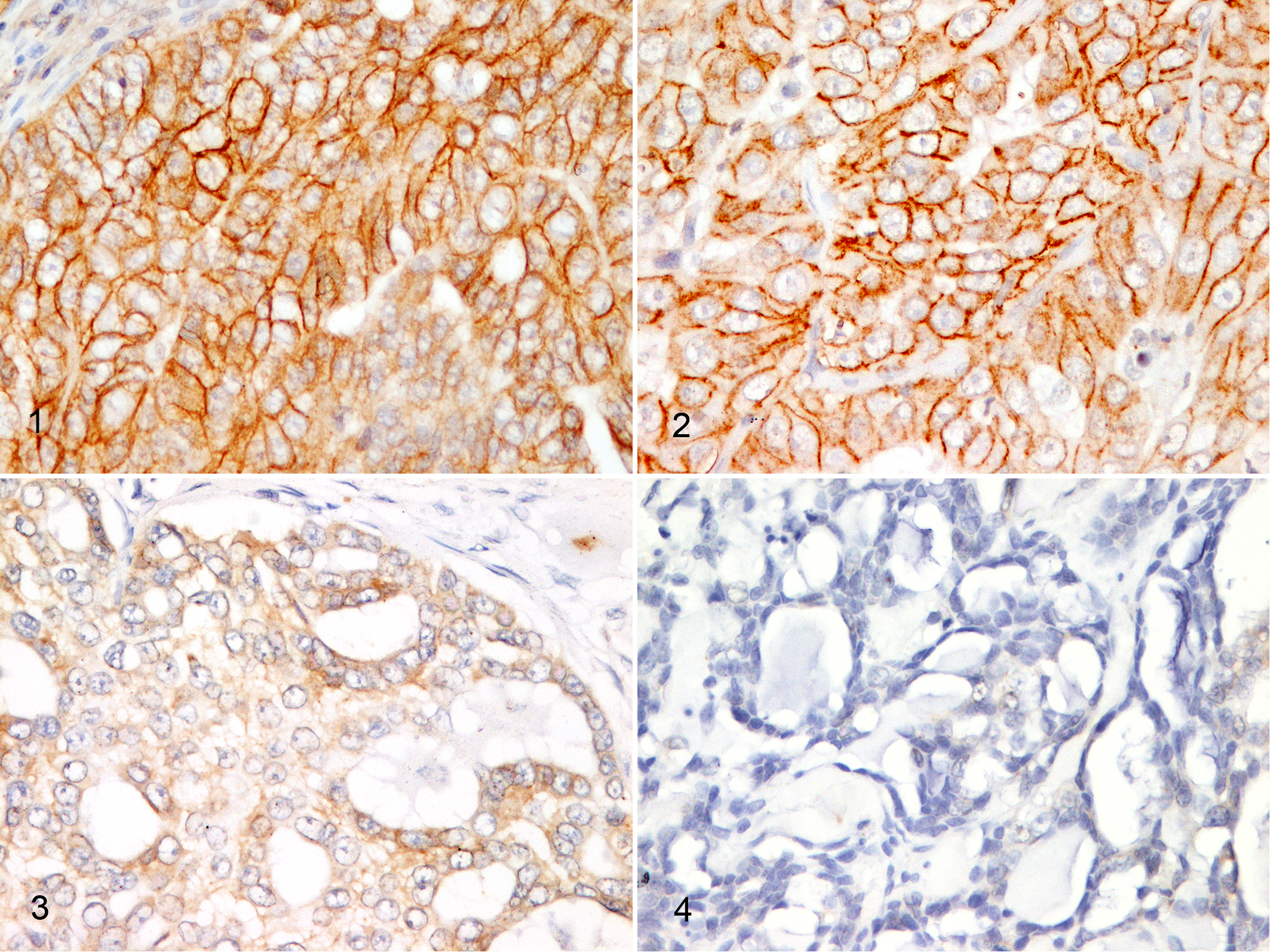
Human epidermal growth factor receptor 2 (HER2) protein expression in invasive carcinoma, mammary gland, cat, immunohistochemistry (IHC) for HER2.
HER2 Gene Amplification Status
A total of 68 tumors were subjected to FISH analysis, including 66 cases with TMA assay and 2 whole sections. Of 68 tumors examined, 8 (12%) were considered HER2 indeterminate because they were technically inadequate and therefore excluded.
HER2 status was amplified (Fig. 5) in 3 tumors (4%), corresponding to 2 HER2 positive (3+) and 1 equivocal (2+) on IHC. The amplification pattern was cluster type in 2 (Fig. 6) of the 3 amplified cases. HER2 status was equivocal (Fig. 7) in 4 tumors (6%), corresponding to 2 HER2 positive (3+) and 2 equivocal (2+) on IHC. HER2 status was nonamplified (Fig. 8) in 53 tumors (78%), corresponding to 3 HER2 positive (3+), 40 equivocal (2+), and 10 negative (1+) on IHC.
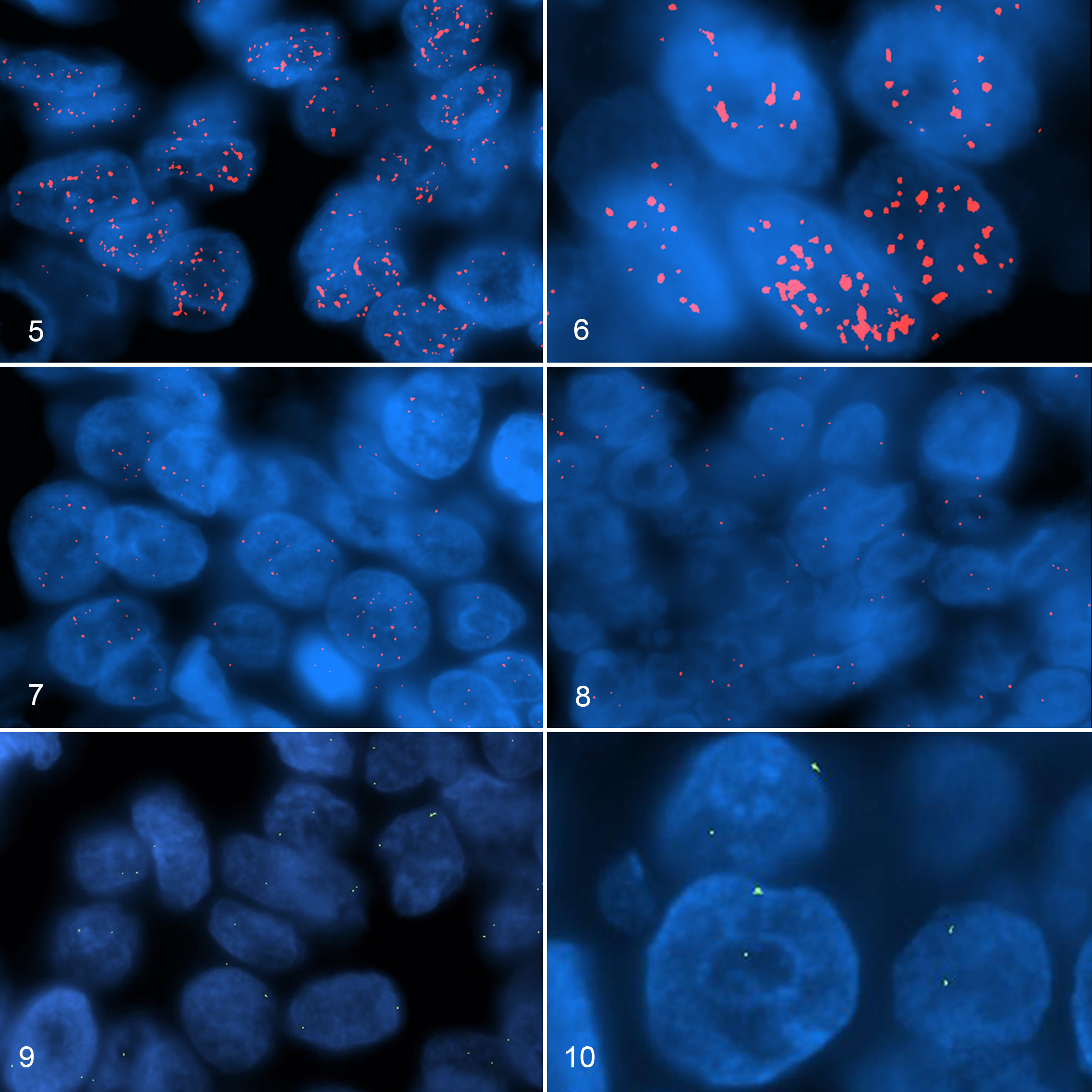
Human epidermal growth factor receptor 2 (HER2) gene amplification status in invasive carcinoma, mammary gland, cat. Red: hybridization of HER2 gene. Green: hybridization signals of chromosome E1 p1.12–p1.11 regions.
The BAC probe 662A1H2 that hybridized the p1.12–p1.11 regions of chromosome E1 produced a diploid signal in each case (Figs. 9, 10). Seven IHC-positive (3+) carcinomas had HER2 gene amplification in 2 (29%), equivocal gene expression in 2 (29%), and nonamplification in 3 cases (42%) (Fig. 11).
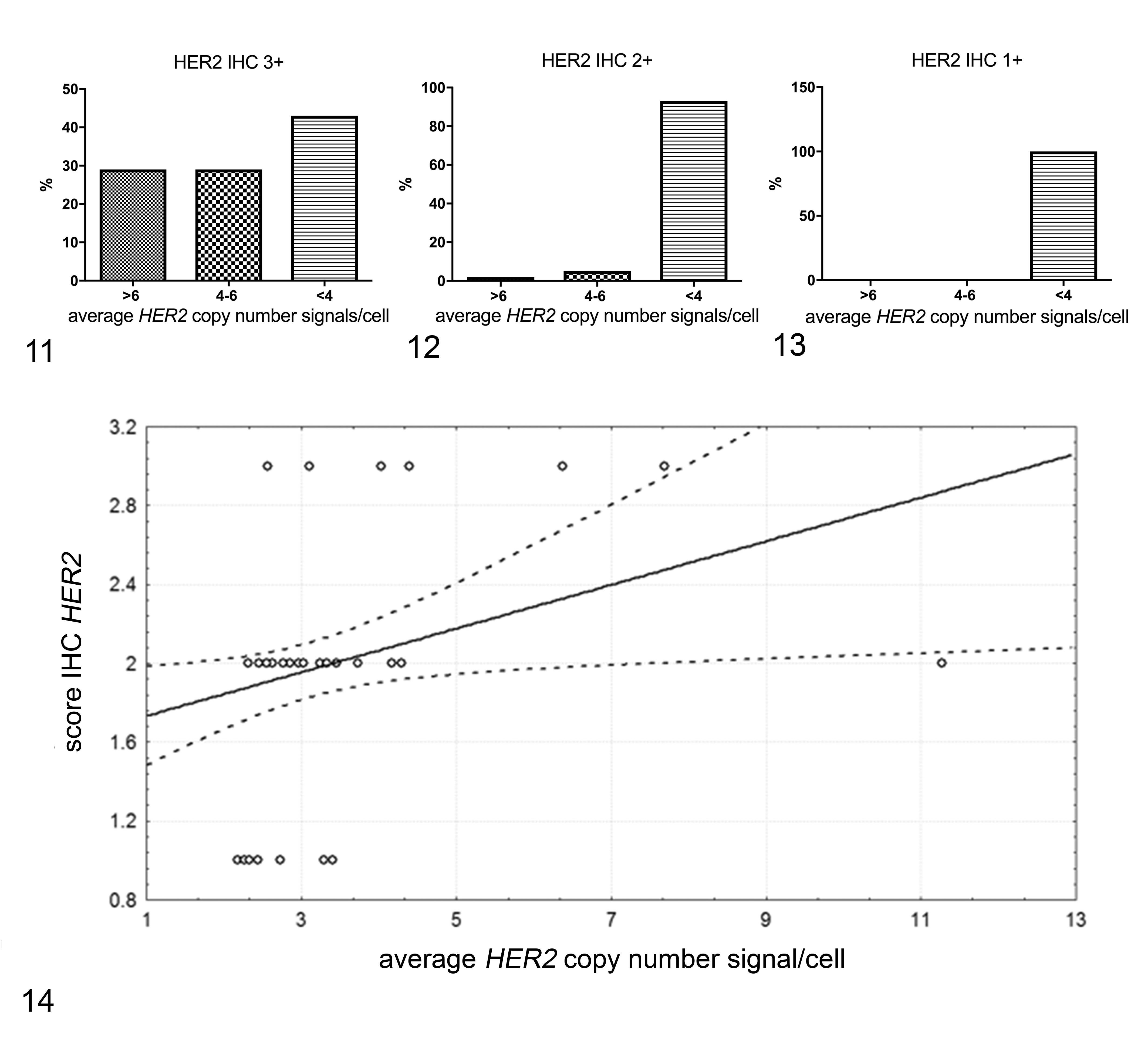
Comparison between human epidermal growth factor receptor 2 (HER2) protein expression and gene amplification in feline mammary carcinomas, measured by average HER2 copy number signals/cell (>6, 4–6, <4).
Thirty-nine of 42 (93%) equivocal cases for IHC expression were not amplified. Two of 42 cases (5%) with equivocal protein expression (2+) had HER2 FISH equivocal and 1 (2%) had HER2 cluster-type amplification (Fig. 12). All IHC-negative cases (1+) were nonamplified on FISH analysis (Fig. 13).
No statistical association was found between tumor grade and HER2 amplification status (Pearson χ2 P = .37). HER2 gene amplification and HER2 protein expression were significantly positively correlated (R = 0.283; P < .0001) (Fig. 14).
Discussion
The feline ortholog of the human HER2 gene was sequenced in 2005 and discovered to have a kinase domain homology of 92%. 7 The same study found an increase in HER2 mRNA in both cell cultures and feline mammary carcinoma, but no gene amplification was detected. 7 Since then, HER2 protein expression levels have been widely studied in feline mammary carcinoma, reporting variable results. 3,5,7,17,25,28 Recently, standard guidelines have been proposed for the immunohistochemical evaluation of epithelial and myoepithelial markers of HER2 and hormone receptor status of canine mammary carcinoma. 24 Based on the ASCO/CAP recommendation of 2007 34 for the assessment of HER2 in canine mammary tumor, this consensus 24 suggests considering positive tumors that have a complete and intense membrane with a chicken-wire pattern in >30% of neoplastic cells. 24 In 2013, ASCO/CAP guidelines for human breast cancer provided an evaluation algorithm categorizing the IHC expression of HER2 as negative (0/1+), equivocal (2+), and positive (3+) in ≥10% of the invasive neoplastic cells. 33 According to these guidelines, IHC equivocal tumors (2+) are routinely subjected to FISH to assess HER2 gene amplification status. 33 Therefore, FISH is considered the gold standard method to assess HER2 amplification status. 33
The amplification status of HER2 in feline mammary tumors is controversial, both in terms of biological significance and technical detection. The literature is divided, with some reports supporting the hypothesis that HER2 is amplified 23 and others finding no amplification. 7,31 This could be caused by technical variables, mainly due to the preanalytical unknowns and the lack of commercially available specific probes for the feline gene sequence.
Soares et al 31 investigated HER2 status using FISH, chromogenic in situ hybridization (CISH), and silver in situ hybridization (SISH) of feline mammary tumors with HER2 protein overexpression but found no amplification. Another study used CISH to evaluate HER2 amplification status in 30 feline mammary tumors, identifying overexpression in 16% of cases. 23
The present experimental study performed HER2 IHC on 107 feline mammary carcinomas, evaluating the tumors according to 2013 ASCO/CAP guidelines 33 and the system suggested for canine mammary tumors. 24 The 2 evaluation systems yielded overlapping results. All the immunohistochemical positive and equivocal cases and some negative cases underwent FISH examination to assess HER2 amplification status. HER2 amplification was found in 29% of IHC HER2-positive cases (3+), while another 29% of IHC HER2-positive cases were HER2 ISH equivocal (with average copy numbers, ≥4 and <6, higher than the normal diploid). If an equivocal ISH amplification is obtained during the human diagnostic procedure, the sampling is repeated in view of HER2 heterogeneity 33 and patients are still deemed eligible for target therapy.
Furthermore, a subset of tumors (42%) with IHC 3+ protein expression showed no gene amplification of HER2. In humans, the concordance rate between gene amplification by FISH analysis and protein overexpression by IHC is 80%, 27 although different studies report a higher correlation in 0 and 1+ (negative) tumors, compared to an increased discordance between FISH and IHC in 2+ (equivocal) and 3+ (positive) tumors. 8 The discordance between IHC and FISH in the subset of IHC 3+ tumors and no gene amplification is attributable to a possible excess of HER2 protein in the absence of gene amplification due to a posttranslational modification of receptor nondegradation. Preanalytical technical problems such as the time of sample fixation may play a role as well. According to the ASCO/CAP guidelines, sample fixation should not exceed 72 hours, 33 a condition not always known in our caseload.
FISH detection of HER2 status in tumors with IHC score 3+ decreases the percentage of false positives in human breast tumors. The same technique increased the accuracy HER2 status in 2+ equivocal cases, also reducing the number of false negatives 8 as in our series where 2% of IHC 2+ equivocal cases were amplified by FISH and 5% showed equivocal amplification.
The identification of HER2 amplification in mammary carcinoma of cat indicates that it is a possible cytogenetic alteration of a subgroup of feline mammary tumors, but whether this is a driver mutation or a simple bystander passenger mutation without phenotypic consequence remains to be elucidated with prospective prognostic studies and further mechanistic investigations on the molecular pathway of HER2.
In human beings, HER2, when overexpressed and amplified, drives the carcinogenesis, and therefore a target therapeutic approach for HER2-positive breast cancer has led to the formulation of a wide range of therapies directed against the p185 HER2 oncoprotein. 22 Herceptin, the first humanized monoclonal antibody directed against HER2, exerts its antitumor effect by blocking HER2 cleavage, stimulating a cell-mediated antibody-dependent immunotoxicity and inhibiting mitogenic activity. 11,20,29 Trastuzumab in combination with pertuzumab, a monoclonal antibody that binds to an epitope different from that of trastuzumab, increases the overall survival of patients with HER2-positive metastatic breast cancer. 1 Oncolytic virotherapy is an emerging new treatment modality that uses viral vectors capable of conveying a tumor vaccine directly to target molecules. It is administered prior to cancer development for the purposes of primary prevention. 16,22
Confirmatory studies with further molecular biology techniques are needed to establish a potential role of HER2 in the carcinogenesis of feline mammary tumors. Furthermore, in vitro models could properly establish a possible therapeutic predictivity in the feline species.
FISH is a specific method to identify the HER2 amplification in positive and equivocal feline mammary carcinomas assessed by IHC. The future use of cat-specific probes will enhance the specificity of cytogenetic alterations in veterinary oncology.
HER2 is amplified in a subset of feline mammary carcinomas despite the HER2-positive or equivocal expression, but whether the HER2 amplification is a gene alteration that drives the mammary tumor carcinogenesis or is only a bystander passenger mutation remains to be determined.
Footnotes
Acknowledgements
We are grateful to Professor Terje Raudsepp, who gave us expert cytogenetics consults and kindly provided the BAC probe (662A1H2). We also thank Professor Leslie A. Lyons for her constructive genetic advice and for addressing us to Professor Rausdepp. Anne Collins edited the English text.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
