Abstract
A 21-year-old thoroughbred mare had a 35 X 14 X 10 cm mass involving the mammary gland. Metastases were found in the kidneys, lungs, skeletal muscles, and regional lymph nodes. Histopathologic examination of the tumor revealed a ductal solid carcinoma with extensive intraductal and intralobular involvement and focal infiltration of the adjacent stroma. The intralobular neoplasms were divided into irregularly shaped islands and sheets of polygonal and spindle-shaped epithelial cells by thick or thin fibrous connective tissue bundles. The neoplastic cells had a small or moderate amount of cytoplasm that stained faintly with eosin and round or oval hyperchromatic nuclei. Immunohistochemically, the neoplastic cells were strongly positive for Lu-5, weakly positive for AE1/AE3, vimentin, and glial fibrillary acidic protein, and negative for cytokeratin 8, cytokeratin 14, a-smooth muscle actin, calponin, and S100. The neoplasm was diagnosed as an invasive ductal carcinoma of the mammary gland with multiple metastases.
Neoplasms derived from the mammary gland are common in carnivores but are very uncommon in herbivores. 8,14 In horses, there are only a few reported cases of mammary neoplasms in the last 30 years, and the histopathologic and immunohistochemical characteristics of the neoplasms have been described in only a few cases. 6 , 9–11 In this report, a malignant mammary neoplasm in a mare was examined by histopathologic and immunohistochemical techniques.
A 21-year-old nonlactating nonpregnant thoroughbred mare had a large, painful, ulcerated mass involving both mammary glands and was suspected of having chronic suppurative mastitis. The mare was euthanatized because of unsuccessful treatment for the mastitis.
This firm neoplastic mass measured 35 × 14 × 10 cm and involved both mammary glands with an ulcerated surface discharging purulent material. The cut surface of the mass was yellow-white with variably sized lobules. Metastases were found in kidneys, lungs, skeletal muscles, and lumbar lymph nodes.
Samples for histologic and immunohistochemical investigations were collected from the mammary glands, including the neoplasm, and from liver, spleen, kidneys, heart, lungs, skeletal muscles, and lymph nodes. The tissues were fixed in 10% neutral buffered formalin and progressively dehydrated in a series of alcohols. Sections for histopathologic examination were cut 4 μm thick from paraffin embedded blocks and stained with hematoxylin and eosin.
Histopathologic examination of the mammary tumor revealed a ductal solid carcinoma with extensive intraductal (Fig. 1) and intralobular involvement and focal infiltration of the adjacent stroma. The neoplasm was divided into irregularly shaped islands and sheets of epithelial cells by thick and thin branches of fibrous connective tissue, and mainly arranged as lobular and solid nests (Fig. 2). Sheets of neoplastic cells invaded the surrounding stromal tissues (Fig. 2). The neoplastic cells frequently invaded the surrounding residual gland and ductal tissues and occasionally formed tubules with intraluminal neoplastic epithelial cells (Fig. 3). The neoplastic cells were epithelial mostly polygonal and fusiform in some areas. They had a small or moderate amount of cytoplasm, stained faintly with eosin, and had round or oval hyperchromatic nuclei (Fig. 4). Three to five mitotic figures were seen per high power field. Many blood and lymphatic vessels contained intraluminal clusters of neoplastic cells. In the metastatic lesions of the kidneys, lungs, skeletal muscles, and lymph nodes, the neoplastic cells had the same appearance of the primary mammary neoplasm, but did not form the lobular or solid nests.
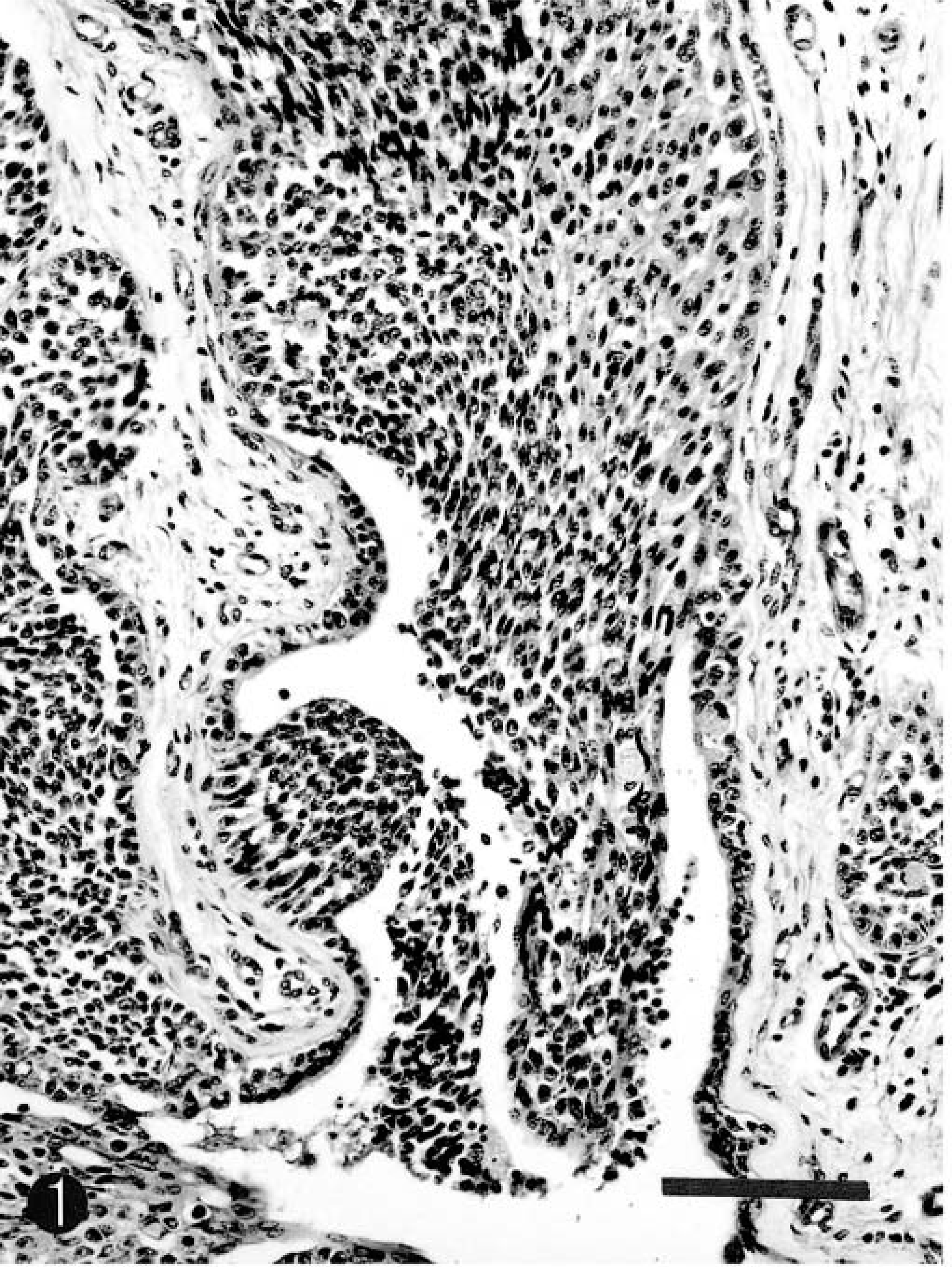
Mammary carcinoma; horse. Ductal solid carcinoma with extensive intraductal involvement. HE. Bar = 130 μm.

Mammary carcinoma; horse. Neoplastic cells mainly forming solid nests in some areas (above), and frequently invading the surrounding stromal tissues, are arranged as sheets elsewhere (below). HE. Bar = 130 μm.

Mammary carcinoma; horse. Neoplastic cells occasionally forming tubules with intraluminal epithelial cells. HE. Bar = 20 μm.

Mammary carcinoma; horse. Neoplastic epithelial cells are mostly polygonal with small to moderate amounts of homogeneous cytoplasm and round to oval hyperechromatic nuclei. HE. Bar = 50 μm.
Serial sections of the neoplasm were immunohistochemically examined by the avidin-biotin–peroxidase complex (ABC) procedure (Vectastain Elite ABC Kit; Vector Laboratories, Burlingame, CA, USA). Details of the primary antibodies used in this study are summarized in Table 1. The deparaffinized sections were treated with 3% H2O2 for 10 minutes to neutralize endogenous peroxidases. All sections were incubated with the primary antibody at 4 C for 16 hours, with the biotinylated secondary antibody for 30 minutes at room temperature and with avidin–peroxidase conjugate for 30 minutes. Finally, 0.05% 3,3′-diaminobenzidine solution was added to the sections and then counterstained with Mayer's hematoxylin. The expression and distribution of CK14 and S-100 was detected simultaneously in the same section using a double immunolabeling technique. The sections immunostained with CK14 antibody and DAB were treated with 0.005% biotin in phosphate buffer saline to mask avidin for 30 minutes at room temperature, and then incubated with S100 antibody as described above. The section was covered with biotinylated goat anti-rabbit IgG, and then incubated with alkaline phosphatase conjugated streptoavidin and alkaline phosphatase substrate Fast blue (SAB-AP Kit; Nichirei Co., Tokyo, Japan), which produces a blue precipitate. Control procedure included the use of non-immune serum instead of the primary antibody; omission of the primary antibody resulted in the absence of specific labeling. Tissues from nonlactating normal mammary gland of two horses were used as positive controls.
Antibodies used for immunohistochemical examination of equine mammary carcinoma.
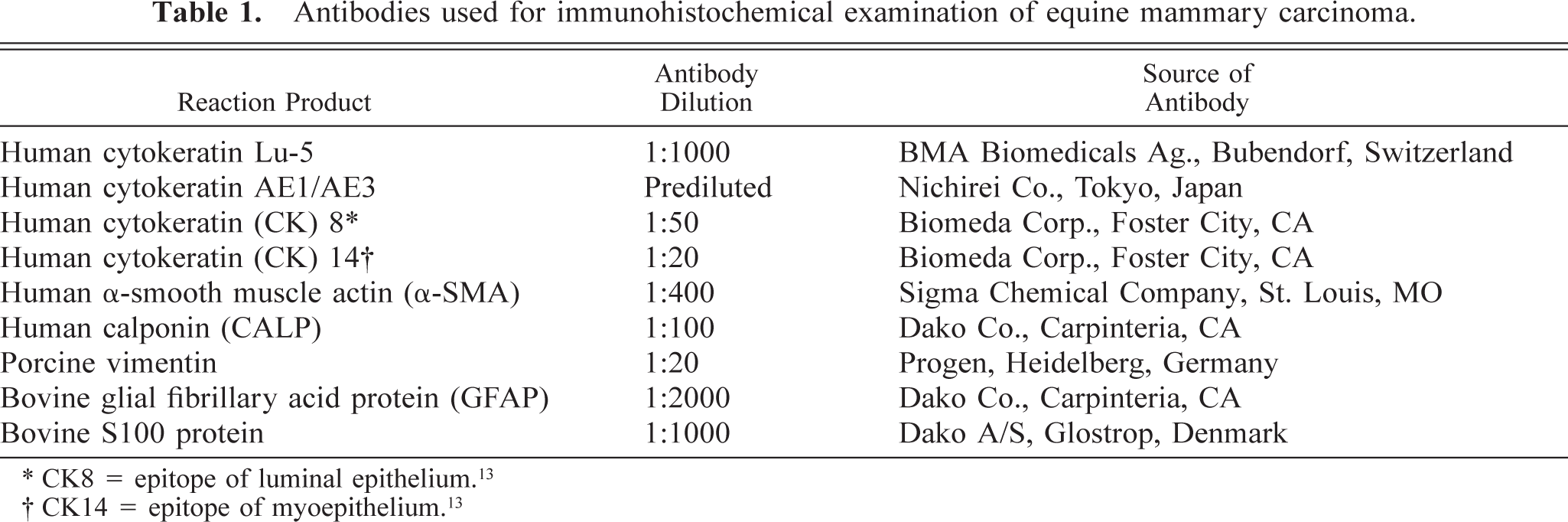
∗ CK8 = epitope of luminal epithelium.13
† CK14 = epitope of myoepithelium.13
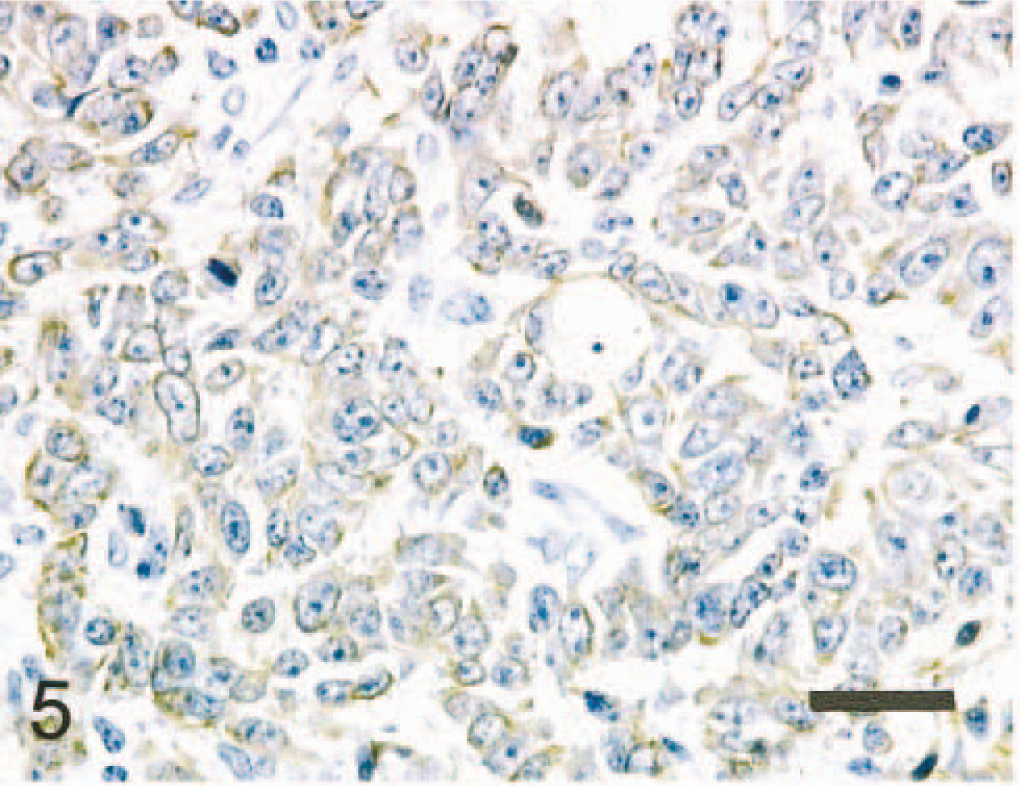
The immunohistochemical results of normal and neoplastic mammary gland are summarized in Tables 2 and 3. In the normal mammary gland, the alveolar and ductal luminal cells were strongly positive for Lu-5, AE1/AE3, cytokeratin 8 (CK8), and occasionally for S100. The alveolar and ductal myoepithelial cells were strongly positive for Lu-5, AE1/AE3, CK14, α-smooth muscle actin (α-SMA), calponin (CALP), and vimentin. In the neoplasm, neoplastic cells were strongly positive for Lu-5 (Fig. 5), weakly positive for AE1/AE3 and vimentin, and negative for CK8, CK14, α-SMA, and S100. Lobular nests were surrounded by a single peripheral layer of CALP positive cells (Fig. 6). Major solid nests had a single peripheral layer staining for CALP and CK14 (Fig. 7) and α-SMA indicating residual myoepithelium of the gland tissue. CALP positive cells of the peripheral layer disappeared frequently in solid nests (Fig. 8). The neoplastic cells invaded the surrounding stromal tissues and were arranged as sheets (Fig. 8). In the double labeling sections, luminal cells in the tubular nests were strongly positive for S-100 (Fig. 9). A single peripheral layer strongly reacted to CALP and CK14 (Fig. 9), and this was interpreted as residual glandular myoepithelium of the gland tissue. Neoplastic cells that weakly expressed glial fibrillary acidic protein (GFAP) were occasionally found in solid nests (Fig. 10). Although the metastatic neoplastic cells had similar immunohistochemical properties of those of the mammary neoplasm, a single peripheral cell layer that expressed CALP, CK14, and α-SMA, luminal cells that expressed CK8, and S100 were not observed.
Immunohistochemical properties of horse, dog, cat, and human normal mammary cells.∗
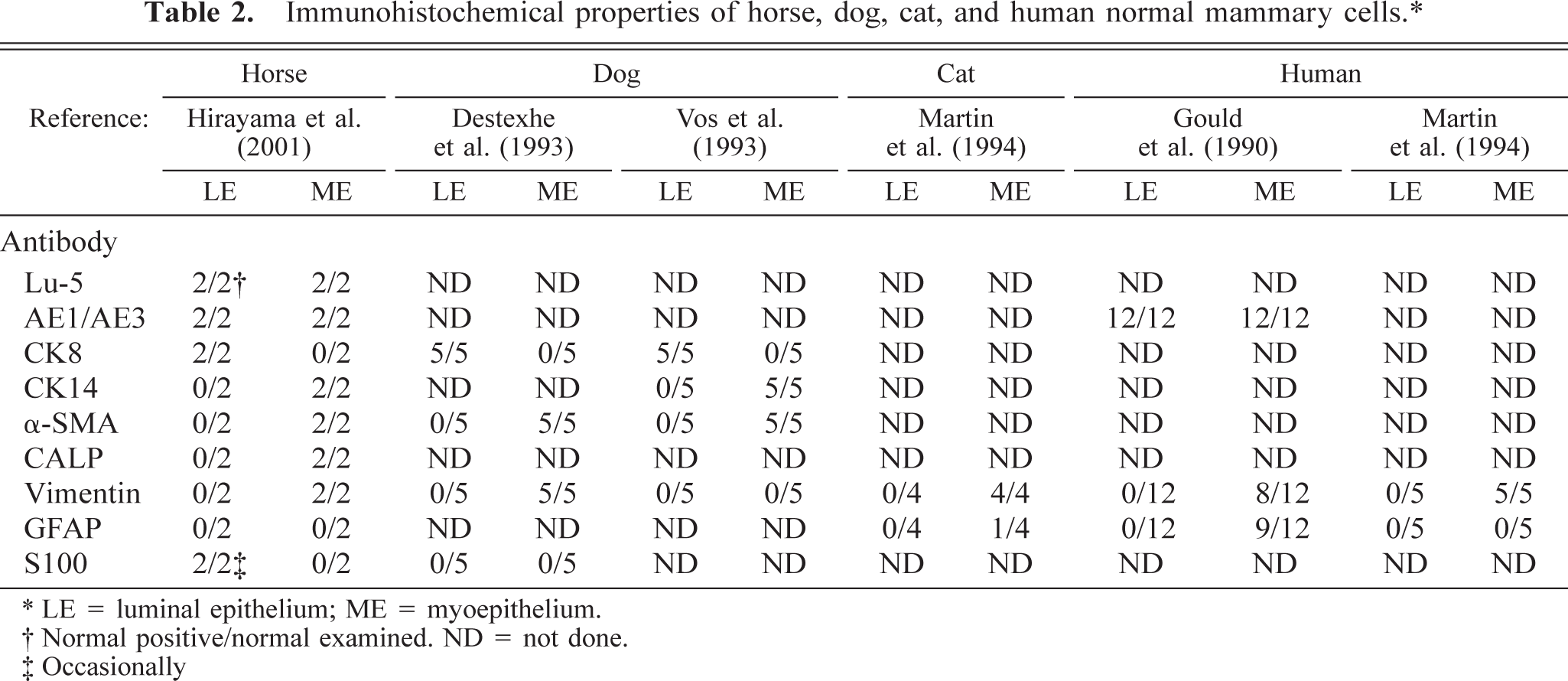
∗ LE = luminal epithelium; ME = myoepithelium.
† Normal positive/normal examined. ND = not done.
‡ Occasionally
Immunohistochemical properties of horse, dog, cat, and human mammary neoplastic cells.
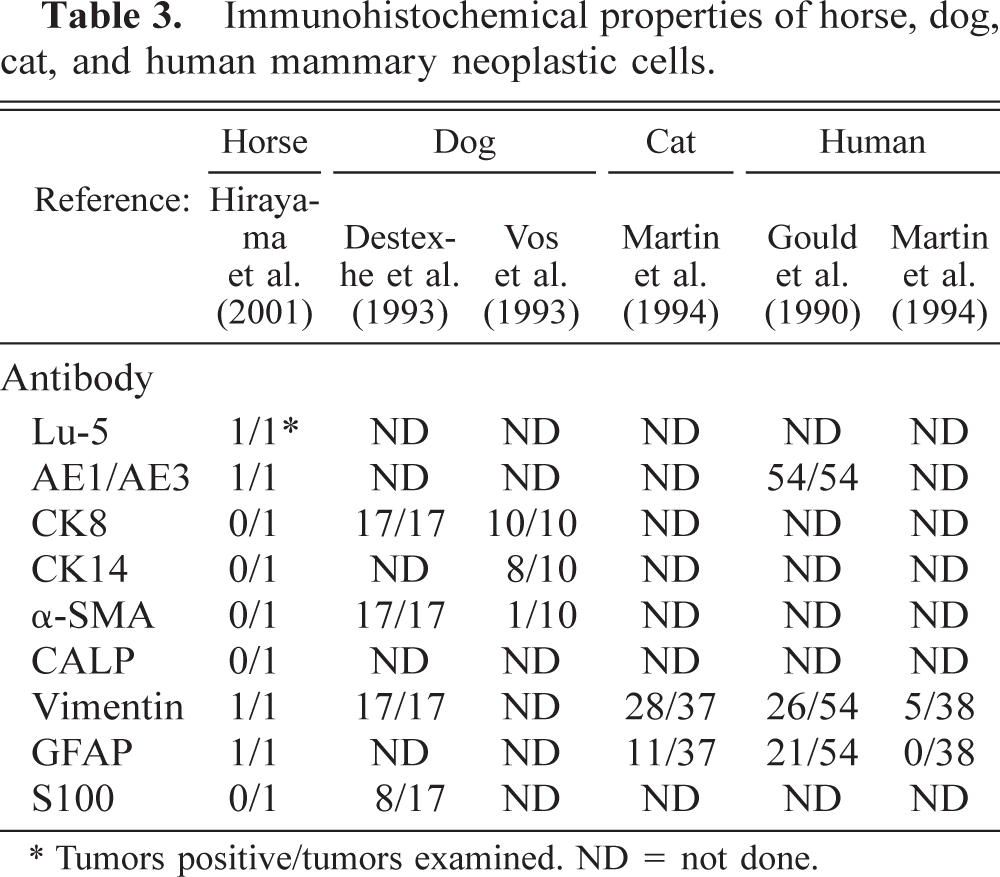
∗ Tumors positive/tumors examined. ND = not done.

Mammary carcinoma; horse. Neoplastic epithelial cells are strongly positive for human cytokeratin Lu-5. Bar = 40 μm.

Mammary carcinoma; horse. Lobular neoplasm (∗) is surrounded by a single peripheral layer of human calponin positive cells. ABC complex method, Mayer's hematoxylin counterstain. Bar = 100 μm.

Mammary carcinoma; horse. Neoplastic nests are clearly surrounded by a single peripheral layer of human CK14 positive cells, suggesting myoepithelial cells. ABC complex method, Mayer's hematoxylin counterstain. Bar = 70 μm.

Mammary carcinoma; horse. Calponin positive cells of the peripheral layer in some neoplastic solid nests disappear (arrows). Neoplastic cells invade the surrounding stromal tissues and are arranged as sheets (right). ABC complex method, Mayer's hematoxylin counterstain. Bar = 70 μm.

Mammary carcinoma; horse. The central luminal cells in the tubular nests are strongly positive for S100 (blue; arrow). A single peripheral layer of cytokeratin 14 positive cells (brown; arrowhead) is present. ABC complex method with double labeling, without counterstain. Bar = 40 μm.
Mammary carcinoma; horse. The neoplastic cells are frequently positive for GFAP (arrows). ABC complex method, Mayer's hematoxylin counterstain. Bar = 20 μm.
Equine mammary carcinomas have been occasionally reported. 8 The horses were between 12 and 25 years of age, 6 , 9–11 and usually had widespread metastases in many organs. 6 , 9–11 , 14 These neoplasms were solid carcinomas, tubular adenocarcinomas, or papillary ductal adenocarcinomas. 6 , 9–11 In the present study, the neoplasm is an invasive ductal carcinoma of the mammary gland with multiple metastases.
Immunohistochemical examinations of the intermediate filament proteins of normal and neoplastic mammary epithelial cells have been performed in cats, 7 dogs, 2,12,13 and humans, 3,7 but not horses. Normal feline and canine mammary glands express CK8 and CK14, in luminal cells and myoepithelial cells, respectively. 7,13
In feline neoplasms, immunoreactivity toward CK8 occurs in 63–89% of the tubular, papillary, mucinous, and combined carcinomas. Vimentin and GFAP immunoreactivity occurs in 76% and 30% of these carcinomas, respectively. 7 GFAP immunoreactivity is seen in luminal epithelial cells of all types of carcinomas and in a few cells at the peripheral layer of some neoplastic nests. 7 A single, continuous layer of cells at the epithelial stromal junction of some neoplastic nests is α and γ muscle actin–positive (57%) (Table 3). 7 Most epithelial cells of the canine tubular adenocarcinoma express CK8 and CK14 and are negative for α-SMA. 2,13 Some epithelial cells are positive for vimentin. 2 Most epithelial cells in solid carcinomas are positive for CK8 but negative for CK14 and α-SMA. A few cells are positive for vimentin (Table 3). 2,13 In humans, immunoreactivity toward CK8 occurs in 95–100% of infiltrative ductal and lobular carcinomas. Vimentin immunoreactivity is seen in the luminal epithelial cells of 13% of infiltrative ductal carcinomas. GFAP immunopositive cells are not seen in any cases of infiltrative ductal and lobular carcinomas (Table 3). 3, 7
The coexpression patterns of vimentin and GFAP with cytokeratin in human mammary neoplasm has been described. 3 A small number of ductal (n = 1/20), papillary (n = 1/1), and lobular carcinomas (n = 1/7) had scattered cells with coexpression of cytokeratin, vimentin, and GFAP. Vimentin expression is possibly a sign of decreased differentiation and may be a potential predictor of aggressive behavior in a mammary neoplasm. 3 GFAP is described in the myoepithelial cells of the parotid gland and in pleomorphic adenomas. 1 The coexistence of cytokeratin-vimentin-GFAP in pleomorphic adenomas and in a small group of carcinomas of the salivary gland has also been described in humans. 4 In dogs, GFAP is expressed in the myoepithelial cells of pleomorphic adenomas of the lacrimal gland but not in myoepithelial cells of the normal gland. 5
In the present study, normal alveolar and ductal epithelial cells of the equine mammary gland were strongly positive for AE1/AE3, CK8, and S100 but negative for CK14, vimentin, α-SMA, and GFAP. Myoepithelial cells were positive for AE1/AE3, CK14, vimentin, α-SMA, and occasionally for GFAP. The neoplastic cells were weakly positive for AE1/AE3, vimentin, and GFAP but negative for CK8, CK14, α-SMA, and S100. The peripheral cells in the solid nests expressing CK14 and α-SMA, and the central luminal cells in the tubular nests expressing CK8 and S100, may represent non-neoplastic residual alveolar and ductal luminal epithelial and myoepithelial cells (of the acinar and ductal mammary tissues) because these cells were not seen in metastatic lesions. These immunohistochemical findings may suggest that neoplastic cells developed from the ductal epithelial cells, 3 invaded acini and ducts, infiltrated the interstitium, and metastasized.
In summary, the present case is diagnosed as having an invasive ductal carcinoma of the mammary gland with multiple metastases.
