Abstract
Hepatobiliary neuroendocrine carcinoma was diagnosed in 17 cats in a period of 10 years. Seven tumors were of intrahepatic origin, one of which was a composite containing components of epithelial and neuroendocrine carcinoma. Nine tumors were of extrahepatic origin, and one tumor was located in the gallbladder The cats were adult and geriatric, and the male: female ratio varied according to tumor group. Hepatomegaly, anorexia, weight loss, and vomiting were the most common clinical signs observed in the cats with hepatic neuroendocrine carcinoma. The cats with extrahepatic neuroendocrine carcinoma showed these signs plus icterus (5/9) and high concentrations of hepatic enzymes. Histologically, the hepatic neuroendocrine carcinomas had two patterns, one with acinar structures separated by vascular stroma lined by cuboidal or columnar cells and the other solid with groups of anaplastic cells separated by vascular stroma. The composite tumor consisted of both bile duct carcinoma and neuroendocrine carcinoma. The extrahepatic neuroendocrine carcinomas and the gallbladder neuroendocrine carcinoma were characterized by solid sheets or groups of round to oval cells with vascular or fibrovascular stroma. Immunohistochemical examination of 10 of the neuroendocrine carcinomas revealed that all 10 stained with neuron-specific enolase; one bile duct carcinoma and the gallbladder carcinoma stained with chromogranin; four of five bile duct carcinomas and the gall bladder carcinoma stained with synaptophysin; and one bile duct carcinoma stained with gastrin. one cat with hepatic carcinoma had duodenal ulcer; in this cat, ultrastructural studies showed neurosecretory granules leading to the diagnosis of Zollinger-Ellison syndrome. In four cats in which necropsy was permitted, carcinomatosis (4/4), lymph nodes (4/4), lungs (2/4), and intestines (1/4) were the metastatic sites. Fourteen of the 17 cats were euthanatized during or immediately after surgery.
Hepatic neoplasms in cats, other than primary and secondary lymphoma, are uncommon. The prevalence varies from 1.5 to 2.3% of feline neoplasms. 22, 34, 41, 43 In a 10-year study of hepatic neoplasms in cats, epithelial neoplasms and neoplasms arising from the biliary system were more common than nonepithelial and heptocellular carcinomas, respectively; single cases of hepatic and extrahepatic bile duct neuroendocrine carcinoma were reported. 34 Neuroendocrine carcinoma of the hepatobiliary system has not been otherwise described. Hepatic neuroendocrine carcinoma in dogs was described in 1981. 39 Subsequently, a few reports, mostly single cases, of hepatic and gallbladder neuroendocrine carcinoma in dogs have been published. 10, 27, 28, 34
Neuroendocrine carcinoma arises from the dispersed neuroendocrine system. 4, 9, 21 Although uncommon, neuroendocrine carcinoma has been described in a wide range of organs in humans. The gastrointestinal and bronchiopulmonary systems are the most common sites. 1, 2, 4, 6, 11, 17, 21, 24, 25, 42, 53
In dogs and cats, neuroendocrine carcinoma has been reported in the intestines, 37 liver, 38 bile duct, 34 lungs, 16 gallbladder, 27, 28 stomach, 47 esophagus, 36 nasal cavity, 33 nasopharynx, 40 and skin. 35 Immunocytochemical and electron microscopic examinations have rarely been performed. 34–36, 40
In this study, we describe the clinicopathologic, gross, histopathologic, immunohistochemical, and electron microscopic findings of 17 cases of hepatobiliary neuroendocrine carcinoma in cats.
Materials and Methods
Hepatobiliary neuroendocrine carcinoma was diagnosed in 17 cats during the period 1993–2002. The sites included intrahepatic (seven cats), extrahepatic bile duct (nine cats), and gallbladder (one cat). Tissues were collected during surgery except in one cat in which tissues were collected during necropsy. The tissues were fixed in 10% buffered formalin, processed routinely, and stained with hematoxylin and eosin.
Clinical, gross pathologic, and follow-up information was compiled from the medical and pathology department records. Selected slides from 10 cases (4 intrahepatic, 5 extra-hepatic, and 1 gallbladder) were studied immunohistochemically using a supersensitive immunodetection system (Biogenics Corp., Sam Ramon, CA), an improved biotin-streptavidin amplification system, according to the instructions provided by the manufacturer. The slides were stained for synatophysin, chromogranin, somatostatin, neuron-specific enolase (NSE), serotonin, gastrin, glucagon, insulin, and cytokeratin (AE1/AE3), with appropriate positive and negative controls. The staining was graded as 3+ when more than 50% of cells stained, 2+ when 20–50% of the cells stained, and 1+ when focal cell groups or scattered cells stained.
For electron microscopic examination, tissues were fixed in a mixture of buffered formalin and glutaraldehyde, post-fixed in osmium tetroxide, dehydrated in graded alcohol, and embedded in an epoxy resin. Specimens for transmission electron microscopy were selected from thick epoxy sections, stained with toluidine blue, and sequentially stained with uranyl acetate and lead citrate.
Results
Seven of the 17 hepatobiliary neuroendocrine carcinomas were intrahepatic, 6 of which were hepatic in origin. Of these six cats, five were Domestic Shorthair and one was Siamese. Five were castrated males, and one was a spayed female (male : female ratio, 5 : 1). The age range was 7–10 years (mean, 9.1 years), with most cats close to 10 years of age. The seventh tumor was a composite neoplasm with components of bile duct adenoma, adenocarcinoma, and neuroendocrine carcinoma with distinct transition from one to the other. This was found in a 19-year-old, spayed, female Domestic Shorthair cat.
Nine of the 17 hepatobiliary neuroendocrine carcinomas were extrahepatic, arising from the bile ducts. The breed distribution of these cats was seven Domestic Shorthair, one Domestic Longhair, and one Siamese. There were six castrated males and three spayed females (male : female ratio, 2 : 1). The age range was 3–17 years (mean, 9 years). The cat with gall bladder neuroendocrine carcinoma was an 11-year-old, castrated, male Domestic Shorthair cat.
Clinical signs
The most common clinical signs observed in the six cats with hepatic neuroendocrine carcinoma were hepatomegaly or hepatic mass on physical or ultrasound examination (5/6), anorexia and weight loss (2/6), vomiting (1/6), and dementia (1/6). The cat with mixed neuroendocrine carcinoma had a history of vomiting after eating. The most common signs observed in the cats with bile duct neuroendocrine carcinoma were different degrees of icterus (5/9), vomiting (3/9), anorexia (3/9), and high activity of liver enzymes (1/9). Ultrasound examination showed dilated bile ducts or dilated gallbladder, or both, in four of nine cats and a mass lesion in the bile duct in two. The cat with the gall-bladder carcinoma had a history of vomiting for 2 weeks, diarrhea, and icterus.
Liver function was more remarkable in the cats with the bile duct and gallbladder carcinoma than in the cats with hepatic neoplasms, in which the values were normal or close to normal. In the former group, the total bilirubin (range, 2.0–10.6 mg/dl; normal range, 0.02–0.04 mg/dl), alkaline phosphatase (range, 31–97 IU-L; normal range, 0–62 IU-L), aspartate transaminase (range, 38–2,789 IU-L; normal range, 28–76 IU-L), and alanine transaminase (range, 21–1,431 IU-L; normal range, 5–55 IU-L) were all high. All cats were jaundiced.
Gross findings
In the cats with hepatic neuroendocrine carcinoma, multiple lobes were involved in five cats and a single lobe was involved in one cat. In the cat with the mixed tumor, the medial lobe was involved. This neoplasm had solid and cystic areas containing thick mucoid material. In the cats with extrahepatic neuroendocrine carcinoma, different segments of bile duct and gallbladder were dilated depending on the location of strictures (7/9) or the presence of a mass lesion (2/9). The masses were 1 cm or less in size. In two cats, the neck of the gallbladder and the cystic ducts were involved. In the cat with the gallbladder neoplasm, a 3-cm intraluminal mass was seen infiltrating the wall and adjoining the liver.
Histology
Intrahepatic neuroendocrine carcinoma
Two different histologic patterns were seen in the hepatic carcinomas. Five neoplasms were characterized by small acinar structures lined by cuboidal or columnar cells, some having eosinophilic secretion in the lumen (Fig. 1). Transition to solid areas with a typical neuroendocrine pattern was seen. The cells had small hyperchromatic nuclei with indistinct nucleoli, minimal eosinophilic cytoplasm, and rare mitotic figures.
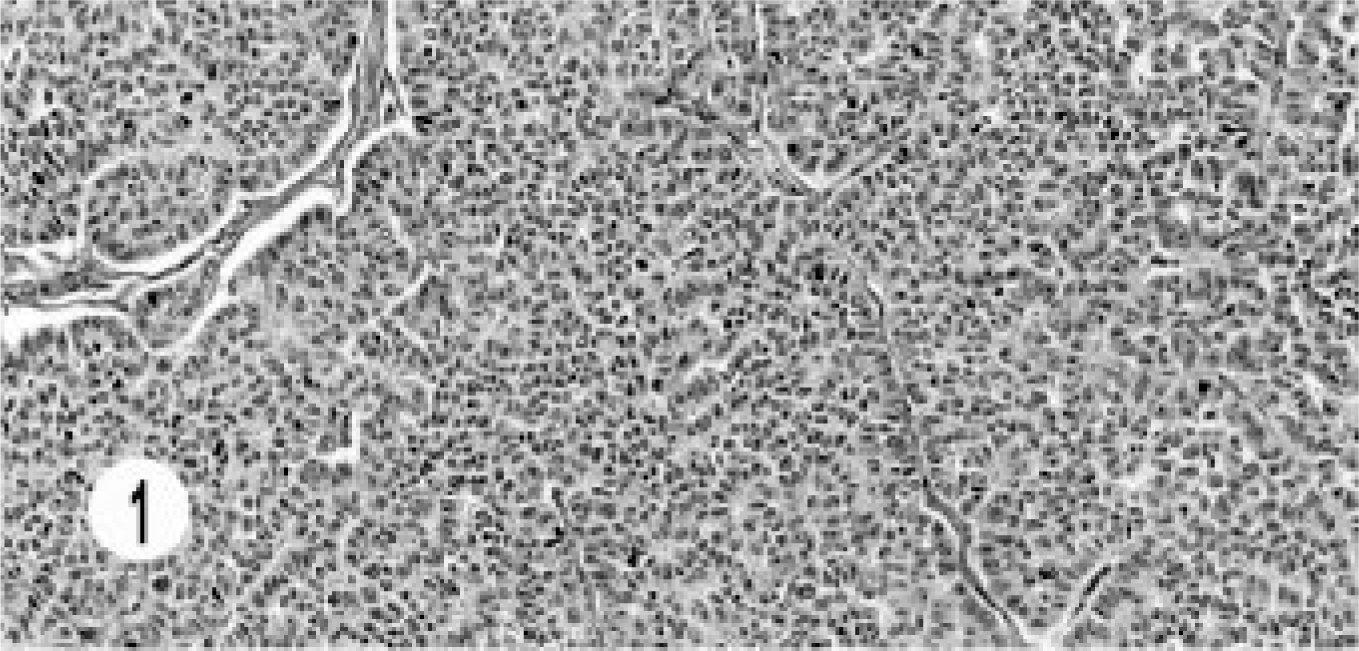
Hepatic neuroendocrine carcinoma; cat. Small acinar structures are lined by cuboidal and columnar cells with thin vascular stroma.
The other hepatic neuroendocrine carcinoma had characteristic solid groups or sheets of oval to round cells with vascular stroma. The cells had vesiculated nuclei and prominent nucleoli (Fig. 2). The mitotic figures were 3–5 per high-power field. The cytoplasm was pale, eosinophilic, granular, and scanty. Areas of sclerosis and intravascular invasion were seen.
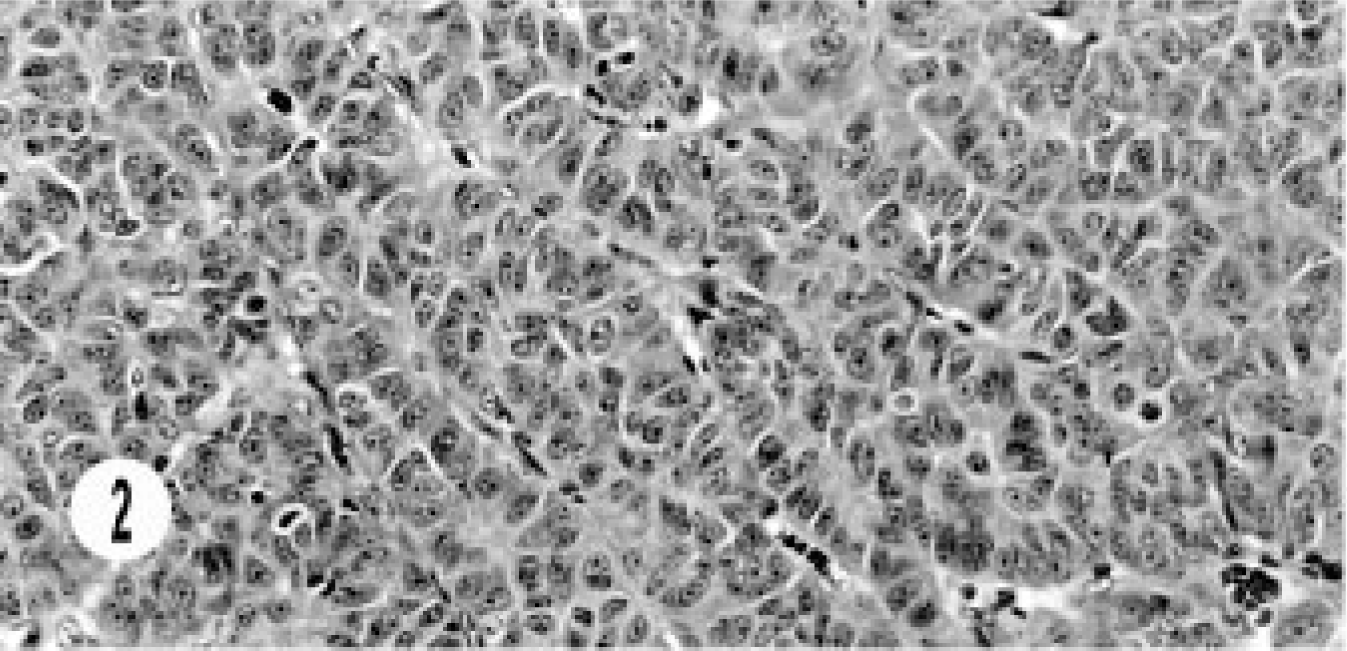
Hepatic neuroendocrine carcinoma; cat. Solid groups of anaplastic cells with prominent nucleoli and scattered mitotic figures are separated by vascular stroma.
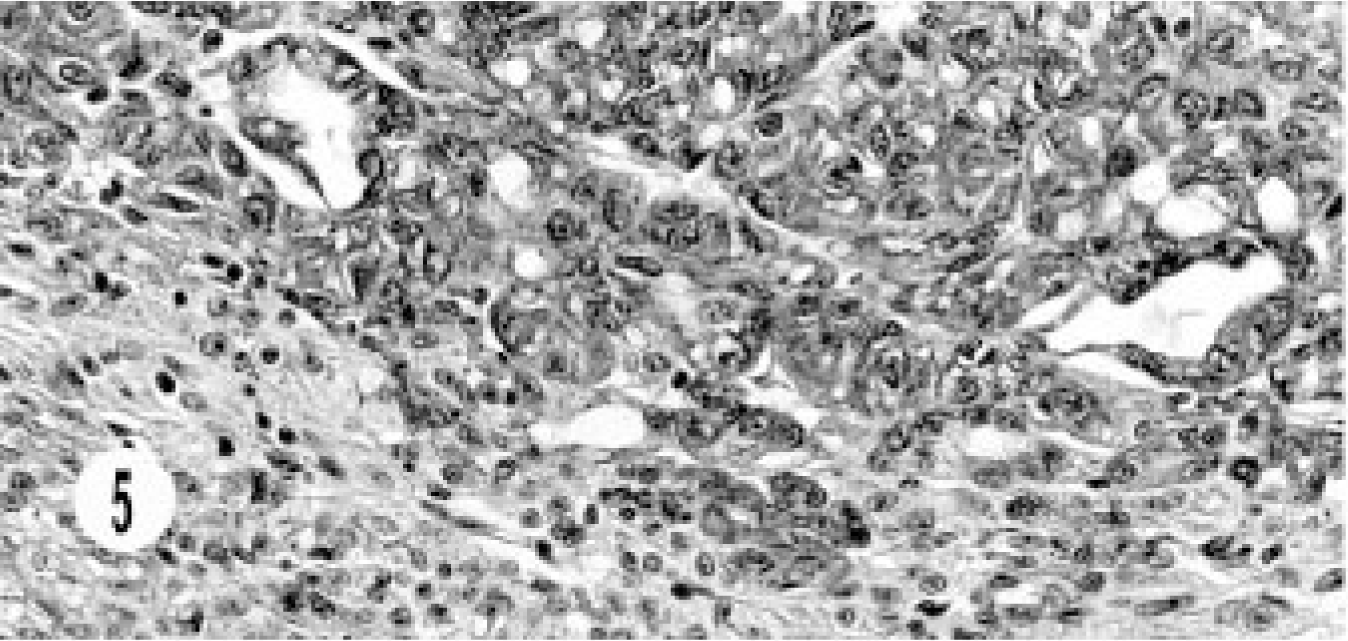
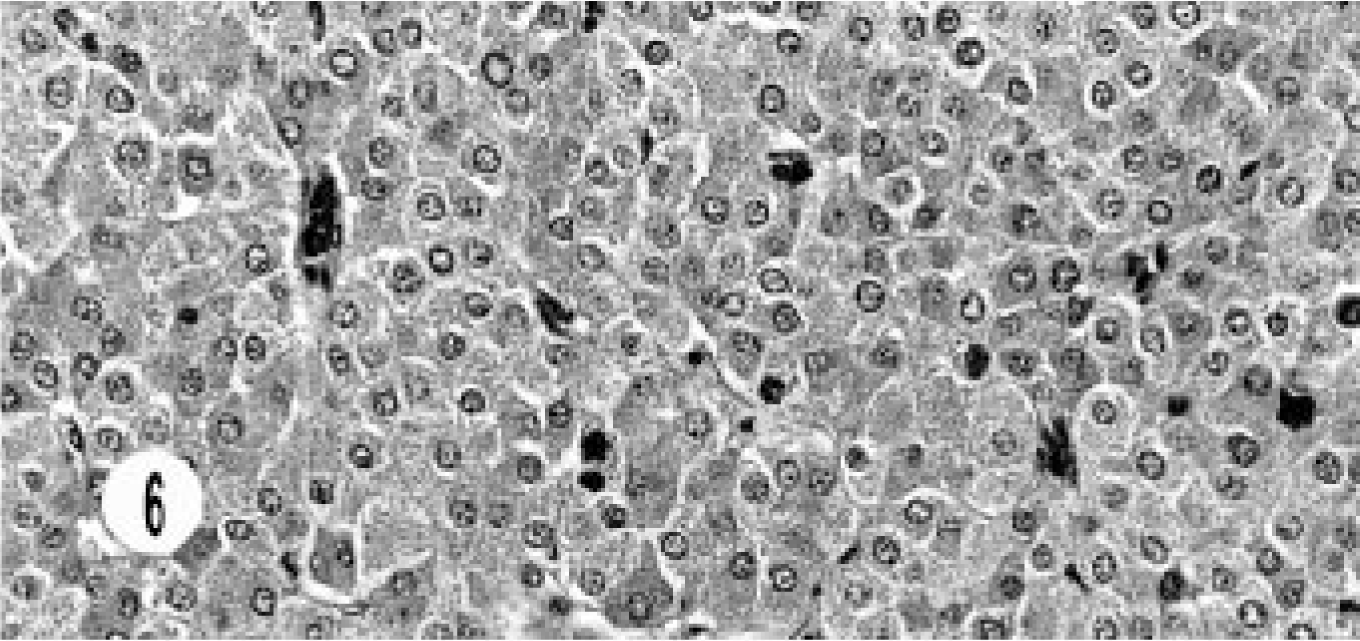
The histology of the composite carcinoma was unusual and different from that of the other neoplasms. This tumor was characterized by continuation from benign cyst adenoma to adenocarcinoma and neuroendocrine carcinoma and showed a morphologic transition from the benign lesion to neuroendocrine carcinoma. The benign lesion ranged from cystic structures lined by cuboidal to flattened epithelial cells, to a mixture of columnar and cuboidal epithelial cells, to oval to round cells replacing the cystic structures, and then to areas of typical, solid, endocrine pattern characterized by groups of small, oval to round cells in a peri-vascular pattern (Figs. 3, 4). The cells had hyperchromatic or vesiculated nuclei and small nucleoli. Mitotic figures were 0–1 per high-power field. The neoplasm extended to the adjoining sinuses. This tumor was unique in its immunohistology in that besides the neuroendocrine markers it stained for AE1/AE3, especially in cystic areas (Fig. 5). The neuroendocrine components stained with NSE (Fig. 6).
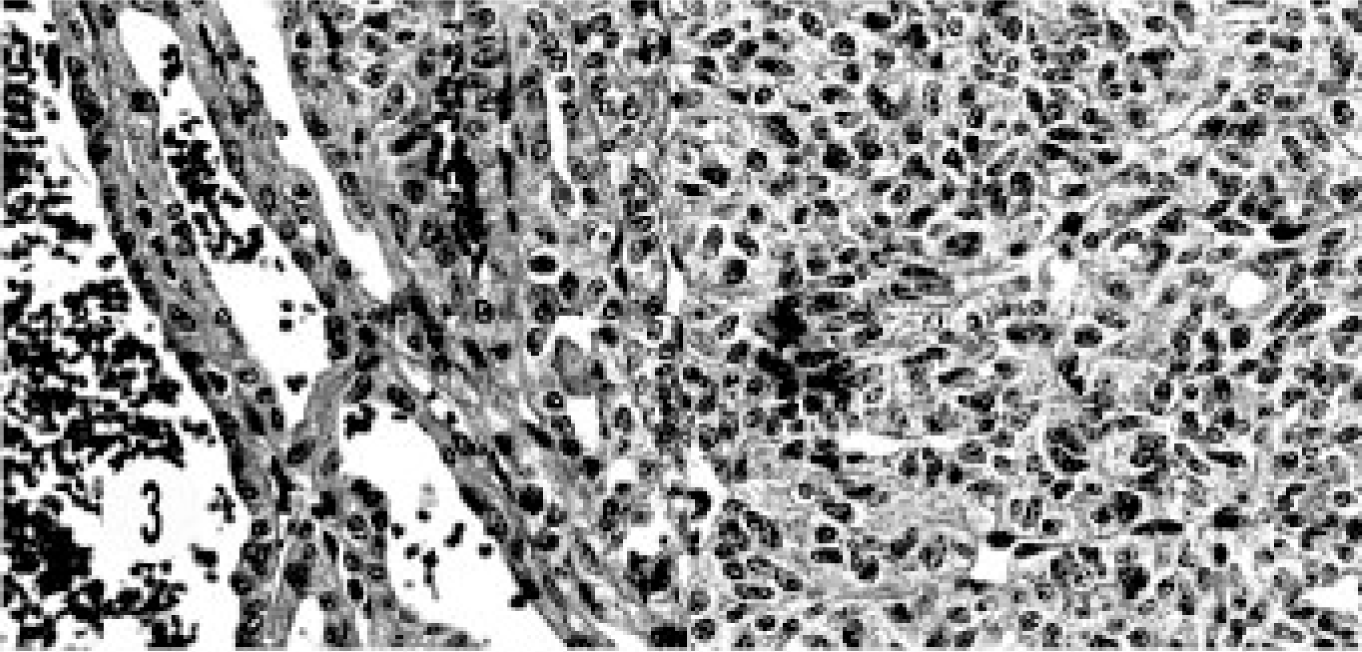
Composite carcinoma; cat. Cystic bile duct carcinoma adjoins neuroendocrine carcinoma consisting of solid groups of cells with vascular stroma.
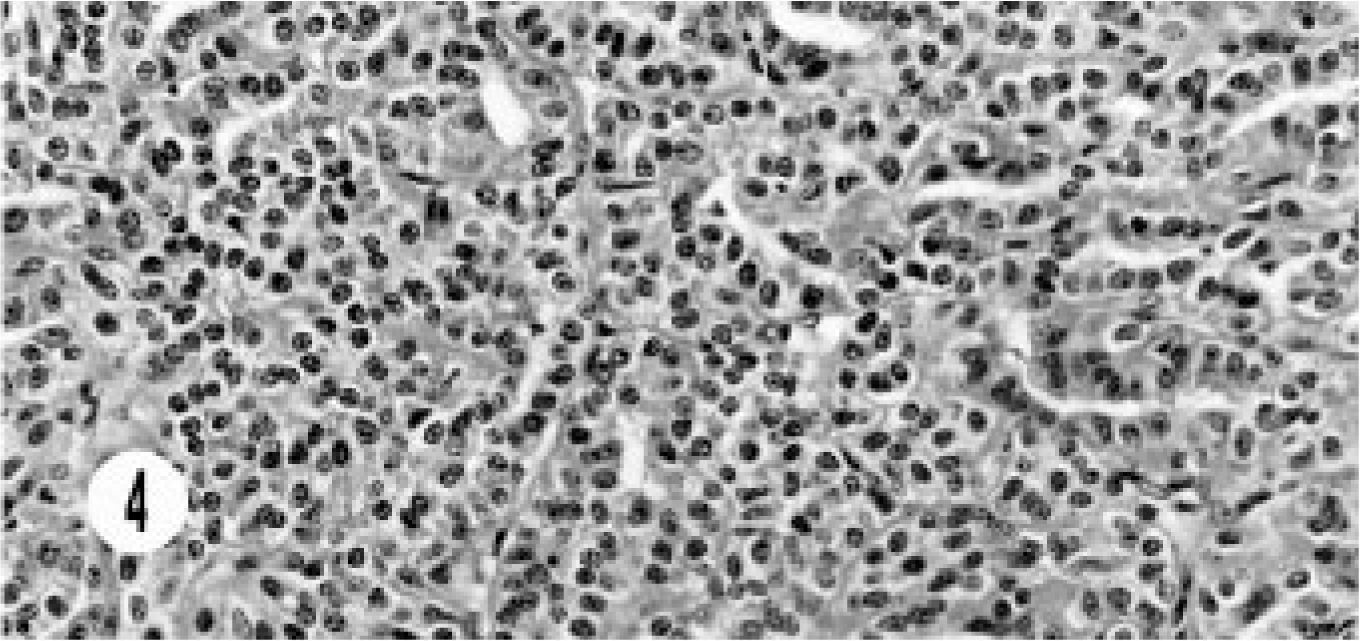
Composite carcinoma; cat. Neuroendocrine carcinoma component shows solid groups of round to oval cells with vascular stroma.
Composite carcinoma; cat. Immunohistochemical labeling for AE1/AE3 demonstrates diffuse intracytoplasmic staining of the epithelial component and nonstaining neuroendocrine cells. Supersensitive multilink immunodetection system; diaminobenzadine chromogen Gill III counter stain.
Composite carcinoma; cat. Immunohistochemical labeling for NSE demonstrates neuroendocrine cells with intra-cytoplasmic staining of granules. Supersensitive multilink immunodetection system; diaminobenzadine chromogen Gill III counter stain.
Extrahepatic bile duct neuroendocrine carcinoma
All these neoplasms were characterized by solid groups or nests of cells with fibrovascular stroma, vesiculated nuclei, and single to multiple (3–4) nucleoli (Fig. 7). Mitotic figures varied from 0 to 1 per high-power field. Transition from the adjoining epithelium was seen in most cases. Local infiltration and sclerosis were common. Areas with acinar structures were seen in three neoplasms.
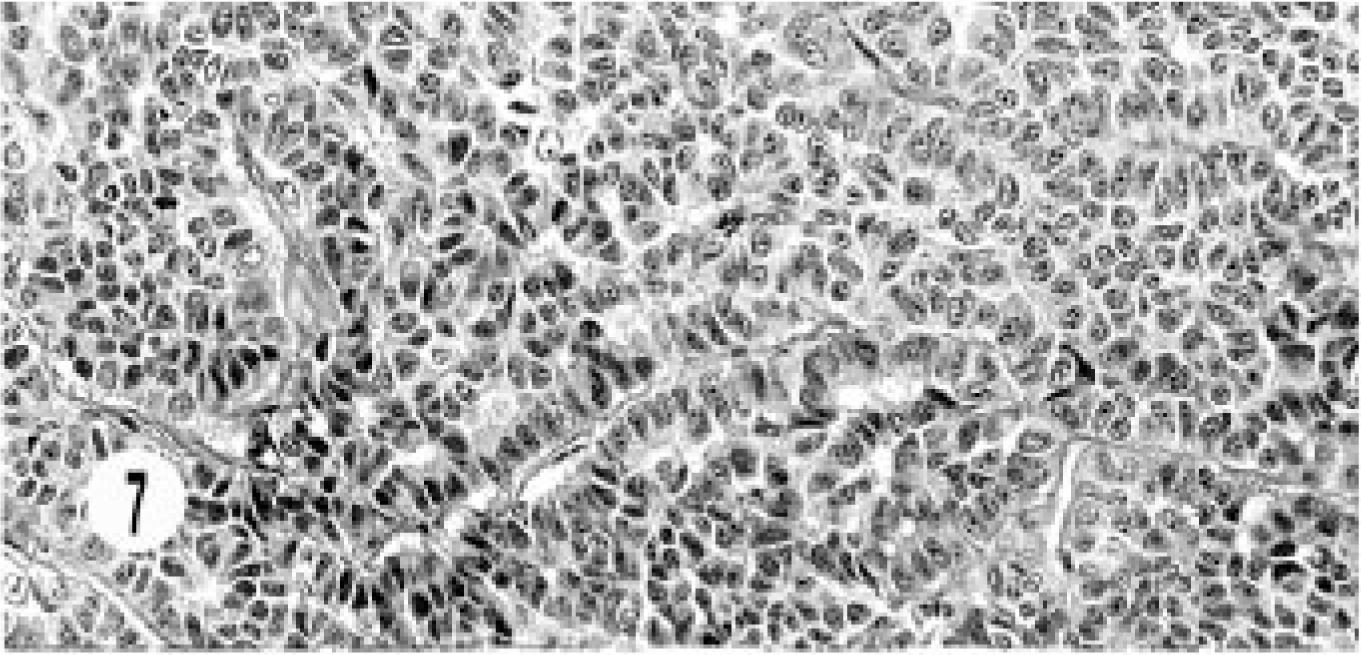
Hepatic bile duct neuroendocrine carcinoma; cat. Solid groups of round to oval cells are separated by fibrovascular stroma.
Gallbladder neuroendocrine carcinoma
The morphologic pattern of this neoplasm was similar to that of the extrahepatic bile duct carcinomas (Fig. 8).
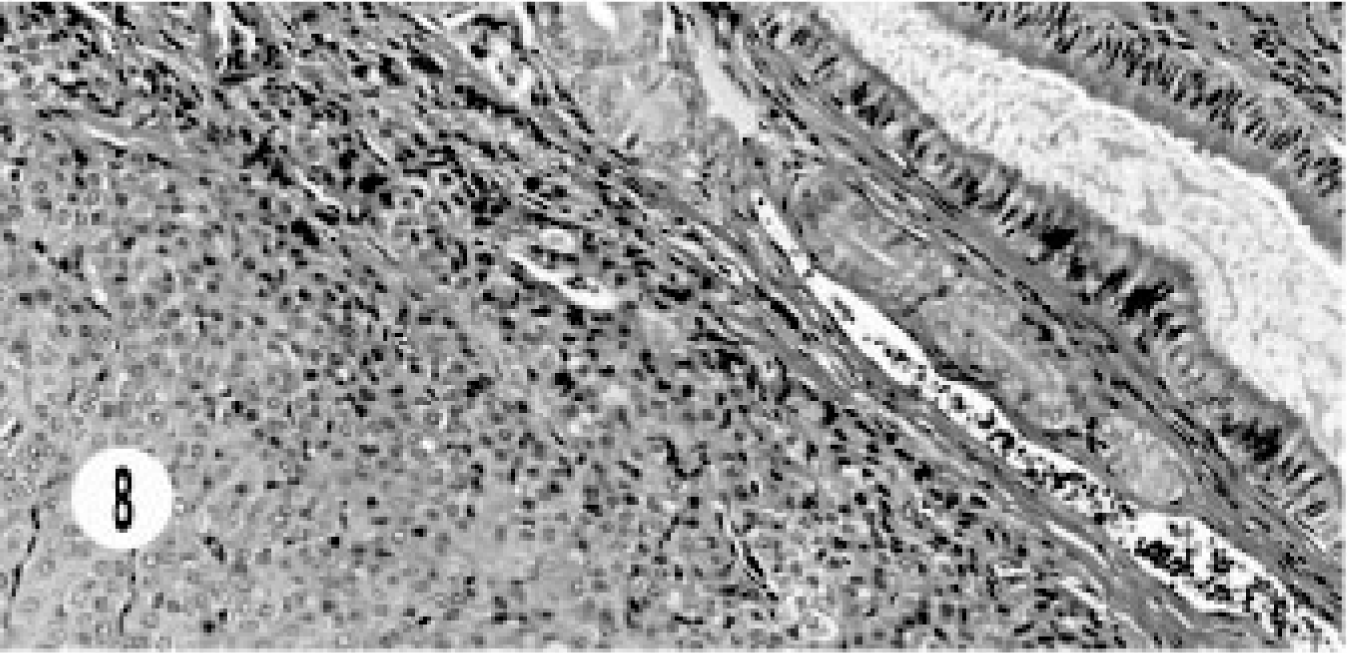
Extrahepatic gallbladder neuroendocrine carcinoma; cat. Solid groups of cells with vascular stroma are close to the gallbladder epithelium.
Immunohistochemistry
Immunohistochemical studies were done in 10 of 17 cases—one mixed, three hepatic, five extrahepatic bile duct, and one gallbladder neuroendocrine carcinoma. None of the tumors stained for somatostatin, serotonin, glucagon, or insulin. All tumors stained for NSE (Fig. 9). All hepatic tumors were negative for chromogranin A and synaptophysin. One extrahepatic bile duct carcinoma (3+) and the gallbladder carcinoma (2+) stained for chromogranin (Fig. 10), and one bile duct carcinoma (2+) stained for gastrin (Fig. 11). Only the epithelial component of the mixed tumor stained for AE1/AE3. Most of the extrahepatic bile duct (4/5) and the gallbladder carcinoma stained for synaptophysin (Fig. 12).
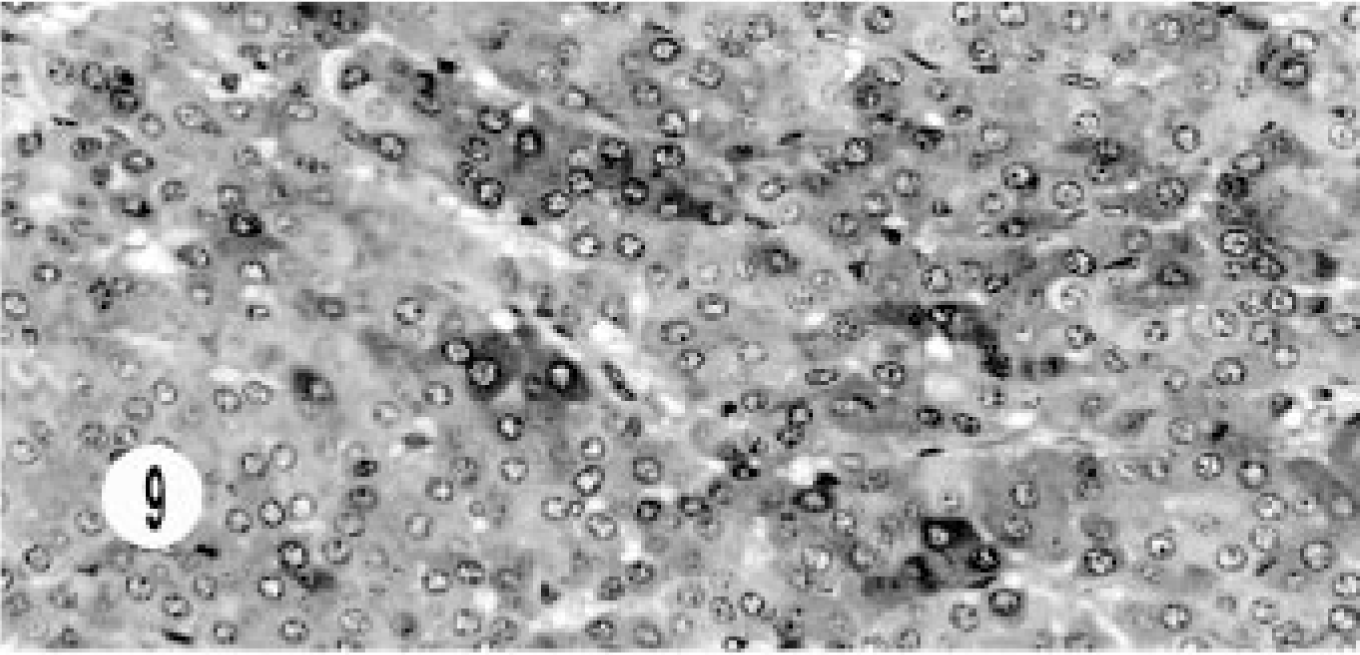
Hepatic neuroendocrine carcinoma; cat. Immunohistochemical labeling for NSE demonstrates groups of cells with deep intracytoplasmic staining. Supersensitive multilink immunodetection system; diaminobenzadine chromogen Gill III counter stain.

Extrahepatic bile duct neuroendocrine carcinoma; cat. Immunohistochemical labeling for chromogranin A shows intracytoplasmic staining. Supersensitive multilink immunodetection system; diaminobenzadine chromogen Gill III counter stain.
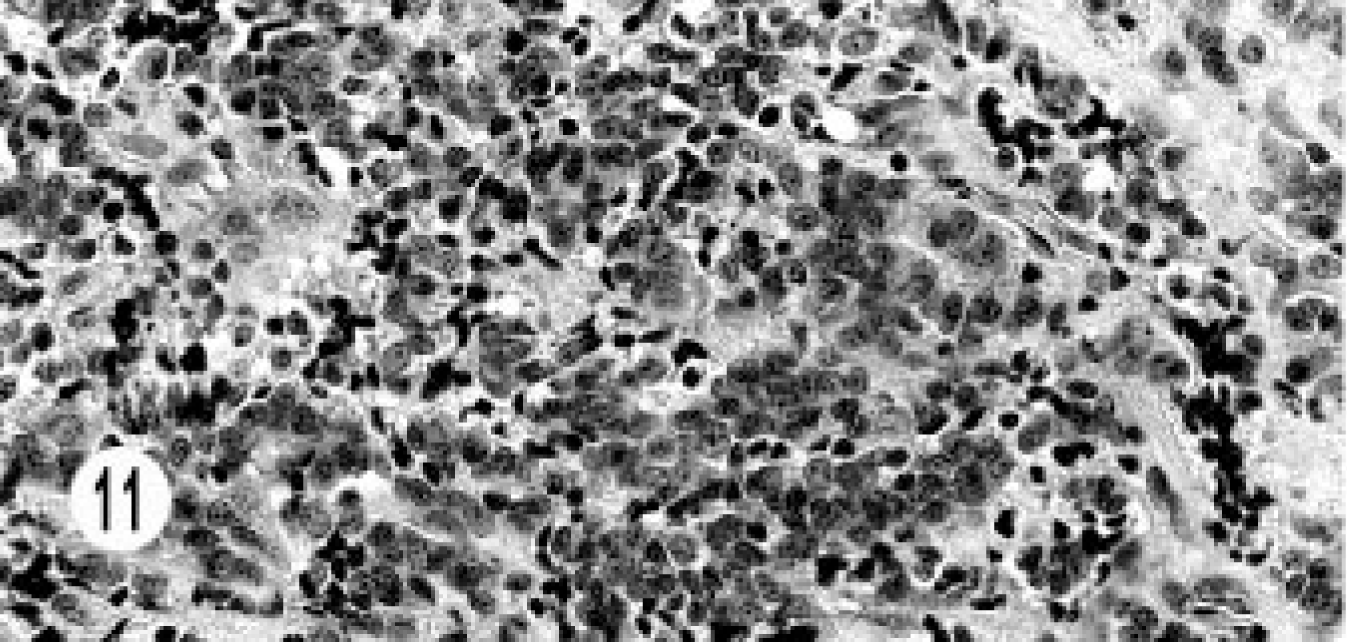
Extrahepatic bile duct neuroendocrine carcinoma; cat. Immunohistochemical labeling for gastrin shows pale, intracytoplasmic staining. Supersensitive multilink immunodetection system; diaminobenzadine chromogen Gill III counter stain.
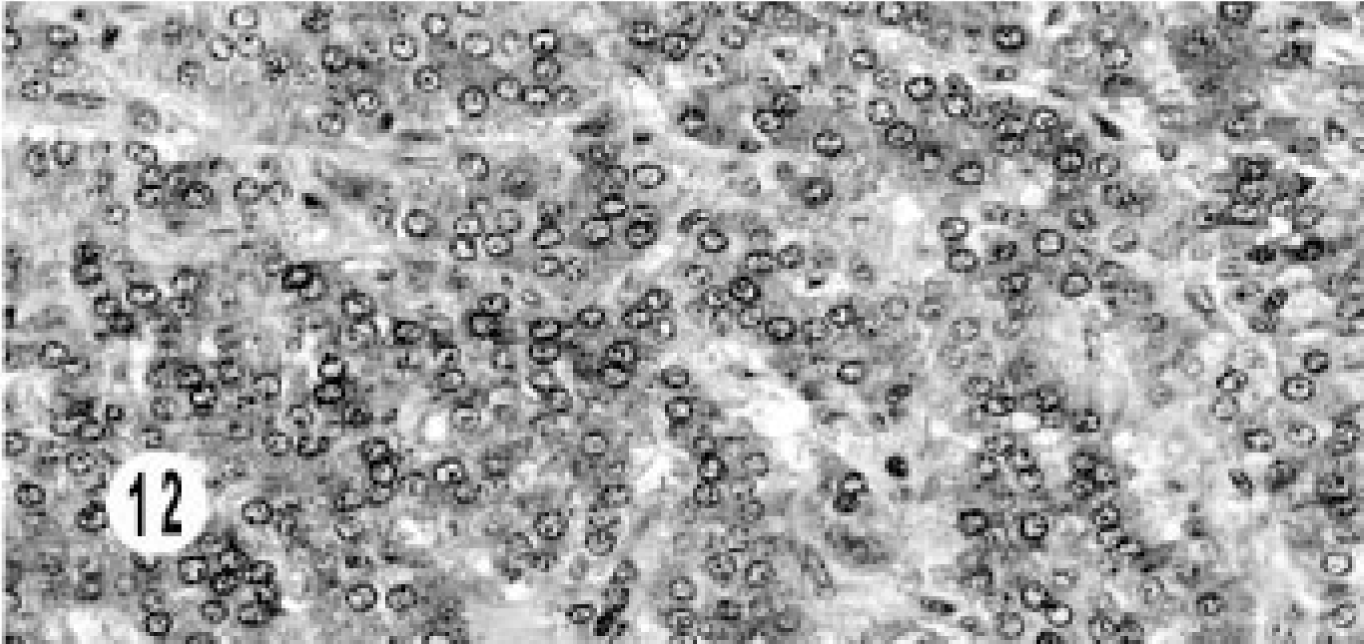
Extrahepatic bile duct neuroendocrine carcinoma; cat. Immunohistochemical labeling for synaptophysin shows intracytoplasmic staining. Supersensitive multilink immunodetection system; diaminobenzadine chromogen Gill III counter stain.
Electron microscopy
Ultrastructural studies were done in two cases, one hepatic and one extrahepatic neuroendocrine carcinoma. The hepatic carcinoma was characterized by cords of epithelial cells surrounded by continuous basement membrane separated from the fibrovascular stroma. Numerous, round, dense-core granules filled the cytoplasm and desmosomes. The features were similar to those of gastrinoma (Figs. 13, 14).
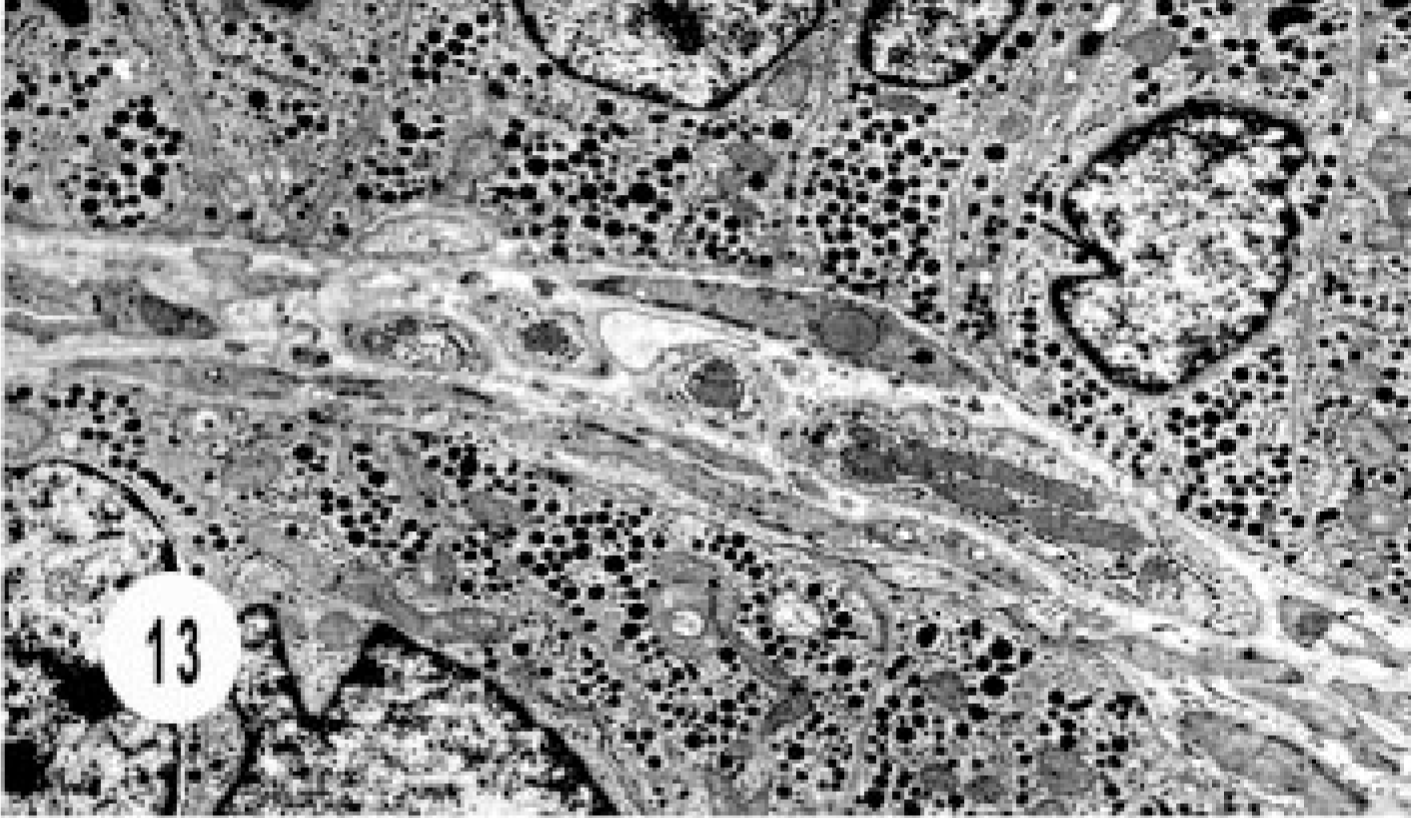
Electron micrograph, hepatic neuroendocrine carcinoma; cat. Cords of epithelial cells are separated by fibrovascular stroma and intracytoplasmic granules.
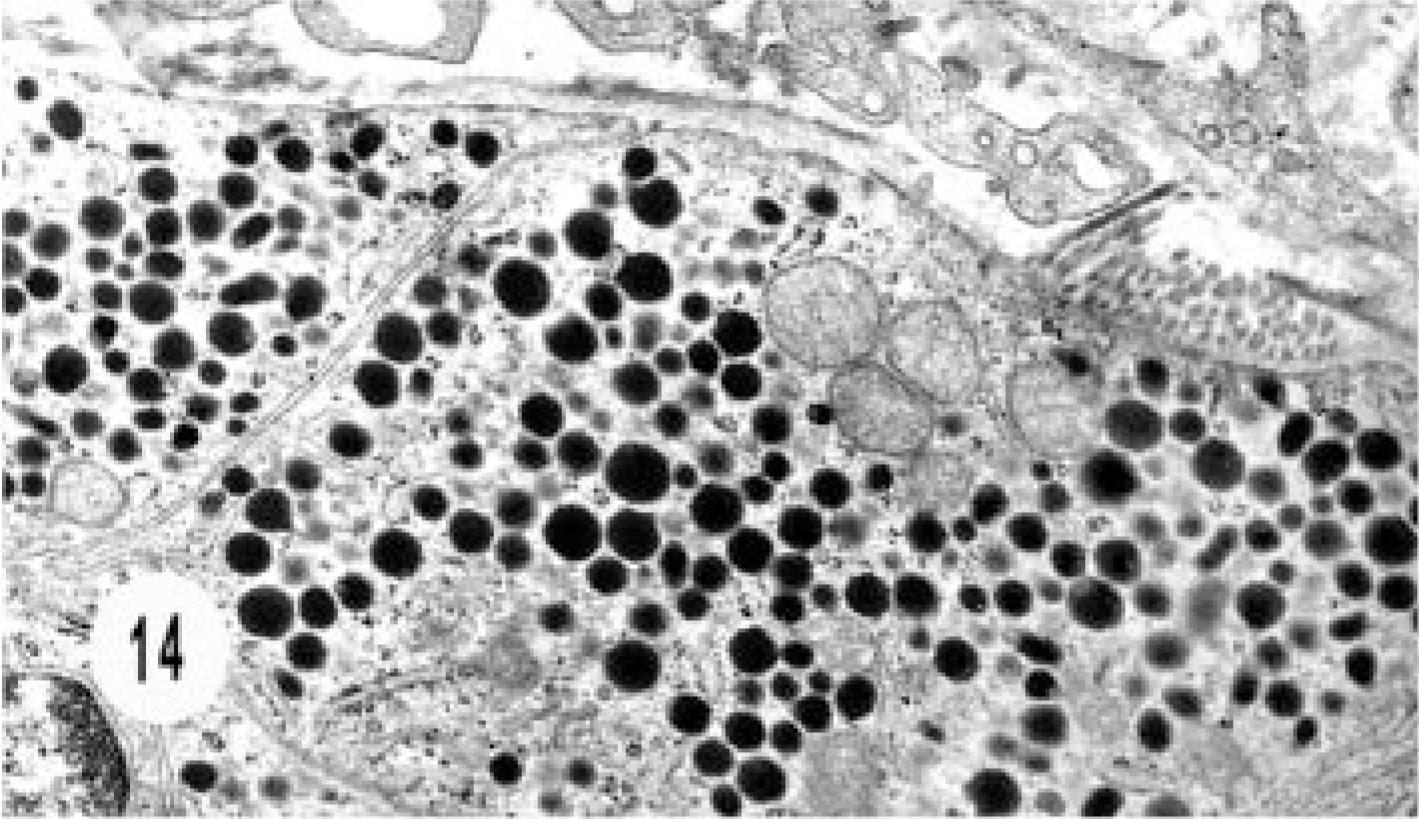
Electron micrograph, hepatic neuroendocrine carcinoma; cat. Higher magnification of Fig. 13 shows numerous intracytoplasmic dense-core granules similar to gastrinoma.
The bile duct carcinoma had solid nests of tightly juxtaposed epithelial cells with variable numbers of intracytoplasmic, dense-core neurosecretory granules (Figs. 15, 16).
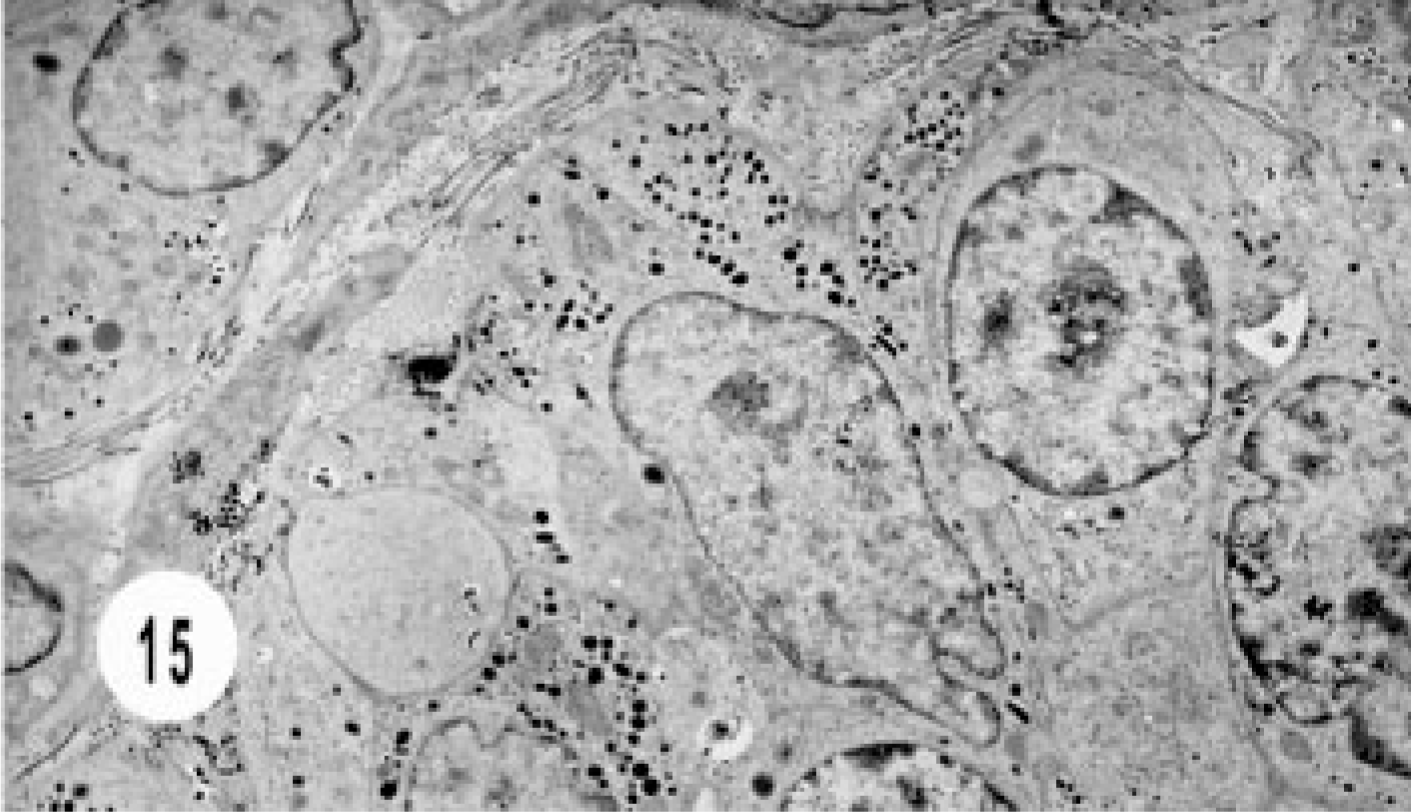
Electron micrograph, extrahepatic bile duct neuroendocrine carcinoma; cat. Note solid nest of cells with variable number of intracytoplasmic granules.
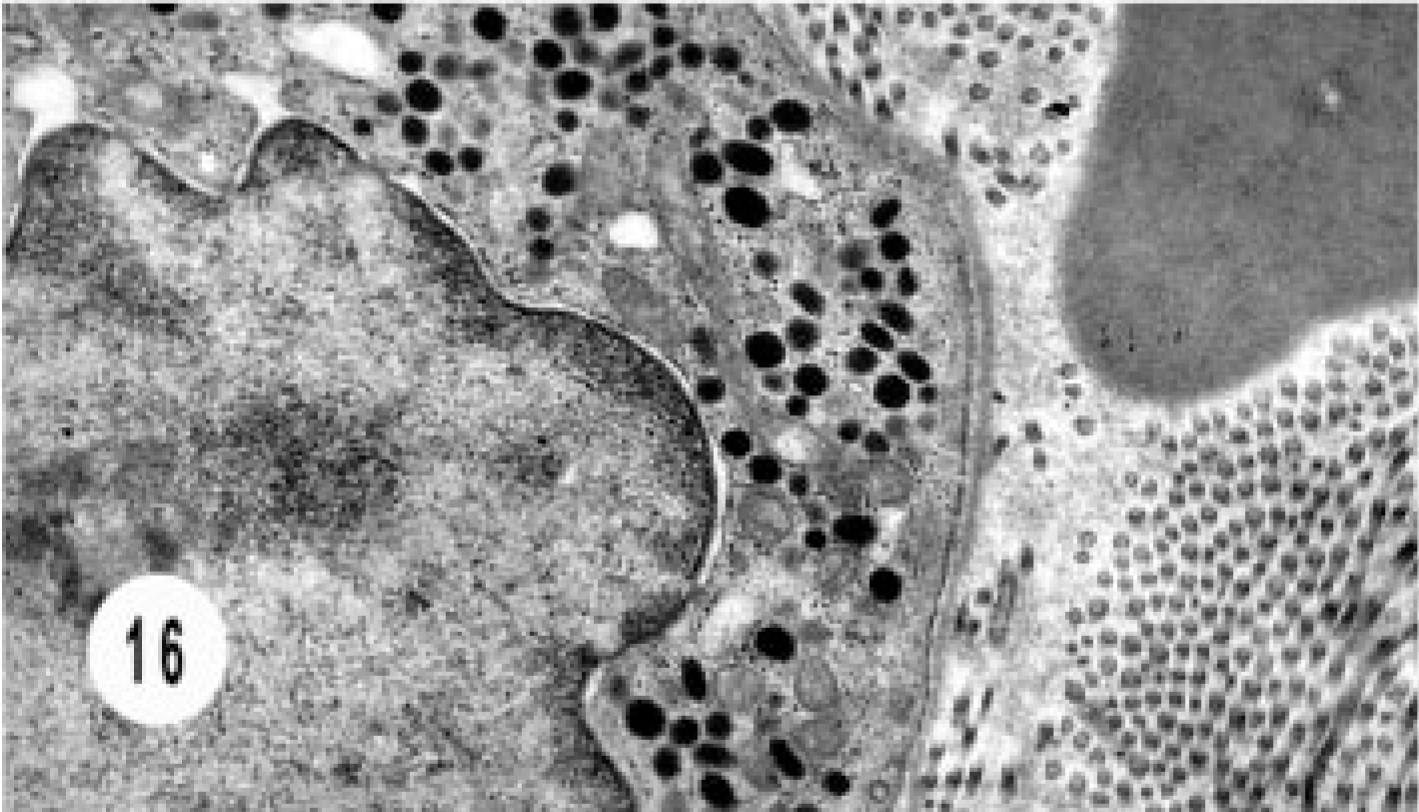
Electron micrograph, extrahepatic bile duct neuroendocrine carcinoma; cat. Higher magnification of Fig. 15 shows the dense core granules.
Zollinger-Ellison syndrome
In a 7-year-old, spayed, female Domestic Shorthair cat of this series, a 2-cm, nonperforating duodenal ulcer was seen during surgery for hepatic tumor. The hepatic neoplasm was diagnosed as neuroendocrine carcinoma (Fig. 2) and was stained for NSE (Fig. 9). Electron microscopy demonstrated typical dense-core granules similar to those seen with gastrinoma (Figs. 13, 14), leading to a diagnosis of Zollinger-Ellison syndrome.
Follow-up
The cat with composite tumor had lived for 370 days with this tumor when it developed a malignant nerve sheath tumor in the lower spinal cord extending to the peripheral nerves and was euthanatized. Two of the cats with bile duct carcinoma lived for 1 year and were then lost to follow-up. The other cats were euthanatized during or soon after surgery. Necropsy was permitted in four cats, including one with hepatic carcinoma. The sites of metastasis were lymph nodes (4), lungs (2), and intestines (1); all four had carcinomatosis.
Discussion
Hepatic neuroendocrine carcinoma is extremely rare in humans and animals. In humans, hepatic neuroendocrine carcinoma was first described in 1958. 11 Only 53 cases have been described in the English literature as of 2001. 17 These tumors were found mostly in middle-aged patients and more frequently in females. 6, 17, 42 The first reports of neuroendocrine carcinoma in the liver of dogs and cats were published in 1981 39 and 1992, 34 respectively. No other reports have been published in these animals except for one case report in a dog. 10 In the 1981 study, neuroendocrine carcinomas constituted 10% of hepatic neoplasms; the mean age of the dogs with hepatic neuroendocrine carcinoma was lower than that of the dogs with other hepatic neoplasms, and there was no sex predilection. 38 In the six cats with hepatic neuroendocrine carcinoma in this study, the male : female ratio was 5 : 1, which differs from that in humans and dogs. 11, 17, 38, 39 The mean age of the cats was 9 years, which might be considered middle age, similar to the age of humans and dogs with this neoplasm 38, 39, 42 The gross and histomorphologic patterns were similar to those reported for human and canine hepatic neuroendocrine carcinoma. The trabecular pattern was common. 17, 34, 39
Nine of the 17 cats of this study had extrahepatic neuroendocrine carcinoma affecting different segments of the bile duct. To the best of our knowledge, there are no reports of extrahepatic neuroendocrine carcinoma in dogs and only a single case report in a cat. 34 In humans, extrahepatic neuroendocrine carcinoma is extremely rare and accounts for 0.1–0.21% of all carcinoids of the gastrointestinal tract. 5, 21, 24 They are more common in females, and the most common site is the bile duct. Jaundice is the most common presenting complaint. 8, 24 In this study, the nine cats with extra-hepatic neuroendocrine carcinoma had a male predominance (2 : 1), and the mean age was 9 years. The most common presenting sign, as in humans, was jaundice. Clinically significant chronic inflammation or sclerosis was not observed in any of the nine cats. In a recent study of a cat with biliary obstruction, the pathogenesis was complex inflammatory disease, including pancreatitis, cholangiohepatitis, cholelithiasis, and cholecysitis, and biliary and pancreatic adenocarcinoma. 26
Neuroendocrine carcinoma of the gallbladder is uncommon in humans. Only limited numbers of individual cases and short series have been reported since its original description in 1981. 2, 7, 13, 25, 27, 30, 31, 52 Most epithelial tumors of the gallbladder are adenocarcinoma, accounting for 60–80% of malignant tumors. 2, 13, 25 Only 4% of epithelial tumors of the gallbladder are neuroendocrine carcinoma, which is reported to have a poor prognosis. 2, 25
Adenocarcinoma of the gallbladder in dogs is extremely rare. 38 Single case reports of neuroendocrine carcinoma of the gallbladder have recently been published. 27, 28 To the best of our knowledge, this report is the first description of neuroendocrine carcinoma of the gallbladder in a cat.
Neoplasms of dual differentiation to adenocarcinoma and neuroendocrine carcinoma are divided into three different histologic categories: composite or mixed, collision, and amphicrine. 12, 14, 19, 23, 32 The composite or mixed tumors arise from a common precursor cell and are characterized by an intricate mixture of both elements. In collision tumors, the adenocarcinoma and neuroendocrine carcinoma arise from two different precursor cells and differentiate simultaneously without mixture of cell components. In the amphicrine tumors, the dual differentiation occurs in the same stem cell, and the neoplasms have morphologic features of both.
In this study, we report a composite tumor in the liver of a cat with transition from bile duct adenocarcinoma with a benign component to neuroendocrine carcinoma with areas of intimate mixture of both components. Morphologically, the components were distinct. Immunohistochemically, this is the only tumor in the series that stained with AE1/AE3 in the transitional areas. In humans, this type of dual differentiation is rare and has been reported in the colon, 29 gall-bladder, 12 and stomach, 53 in addition to other sites. It is extremely rare in the biliary system. 14, 19, 32, 51 We did not find any reports of this tumor in the liver in humans. This type of mixed tumor, i.e., adenocarcinoma and neuroendocrine carcinoma, has not been reported in dogs and cats, although mixed tumors of hepato-cellular and bile duct carcinoma have been reported in dogs. 38 Genetic studies of gastric composite adenocarcinoma and neuroendocrine carcinoma suggest that these tumors sequentially evolve from glandular precursor cells to a genetically heterogeneous adenocarcinoma and then to a neuroendocrine differentiation. 20 Morphologically, the transition was obvious in this case.
Zollinger-Ellison syndrome has become a well-recognized syndrome in humans since its description in 1955. 54 There are a few reports in dogs. 15, 18, 46, 48 The syndrome is characterized in humans by recurrent peptic ulcer, hypersecretion of gastric acid, and a non–B-cell tumor. The syndrome has not been described as such in cats, although ulcer associated with gastric tumor and gastrinoma has been reported. 47 This series included one cat with neuroendocrine carcinoma of the liver and duodenal ulcer. Electron microscopic study showed typical dense-core granules of gastrinoma. Gastric acid evaluation was not done in this cat, and thus we could not confirm the diagnosis. One of the bile duct carcinomas in this series was gastrin positive, but the cat did not have a history of gastroduodenal ulcer.
Regarding the results of immunohistochemical studies, it is observed that chromogranin, which is the most commonly used neuroendocrine marker in human medicine, was not diagnostically useful in the cats of this study. Only one of the bile duct carcinomas plus the gallbladder tumor was positive for chromogranin (2/17). All the neoplasms of this study were positive for NSE, and most of the bile duct (4/5) and the gall bladder neoplasm were positive for synaptophysin.
Fourteen of the 17 cats of this study were euthanatized during surgery or immediately after surgery. The prognosis in cats with these tumors is not favorable.
The origin of hepatic neuroendocrine carcinoma is the early hepatic stem cells, which give rise to the bile ductules and heptocytes. 3, 49 The neuroendocrine cells originate in the bile ductules. 44, 45, 50 The bile duct and gallbladder neuroendocrine carcinomas arise from pre-existing neuroendocrine cells in the epithelium. 19
