Abstract
A 35-mo-old spayed female mixed-breed cat with continuous vomiting, emaciation, and abdominal distention for 2 wk was presented to a private veterinary clinic for evaluation. At 71 d after the initial visit, the cat died with anemia, jaundice, and hypoalbuminemia, and was subjected to autopsy. Grossly, numerous firm masses, 0.5–2.5 cm diameter, were randomly located in the left lobe of the pancreas. Histologic examination revealed that the pancreatic mass consisted of 2 tumor cell types: mostly small round cells with a minority of epithelial cells. The small cells were arranged in nests of various sizes, which were separated by thin fibrous stroma, and had small, round, hyperchromatic nuclei, scant cytoplasm containing argyrophilic granules, and often formed rosettes. The epithelial cells formed luminal structures. Metastases were observed in the liver, greater omentum, and pancreatic, gastric, pulmonary, and mediastinal lymph nodes. Immunohistochemical examination revealed that the small cells were positive for vimentin, neuron-specific enolase, chromogranin A, cytokeratin (CK) AE1/AE3, and trypsin, whereas the epithelial cells were positive for AE1/AE3, trypsin, CK19, and nestin. Ultrastructurally, the small cells contained abundant electron-dense granules, ~200 nm diameter, whereas the epithelial cells had apical microvilli and numerous zymogen granules, ~300 nm diameter. These findings indicated that the tumor was a pancreatic neuroendocrine carcinoma with exocrine differentiation and systemic metastases.
Primary neoplasia of the pancreas is rare and is associated with a poor prognosis in cats.14,30 Malignant exocrine tumors include ductal adenocarcinoma, acinar cell carcinoma, and undifferentiated carcinoma, whereas malignant endocrine tumors are often islet cell carcinoma, according to the World Health Organization (WHO) classification of tumors of domestic animals. 14 Other types, including carcinosarcoma and squamous cell carcinoma, have also been described in cats.11,32
Neuroendocrine carcinomas are malignant tumors that originate from neuroendocrine cells and are known to produce hormones and peptides. 6 In humans, neuroendocrine tumors developing in the pancreas have a predominant neuroendocrine differentiation and can be classified as various types, including well-differentiated neuroendocrine tumors, poorly differentiated neuroendocrine carcinomas, and mixed adenoneuroendocrine carcinoma (MANEC), which include mixed acinar–neuroendocrine carcinoma, mixed ductal–neuroendocrine carcinoma, and mixed acinar–neuroendocrine–ductal carcinoma. 6 On immunohistochemical examination, pancreatic neuroendocrine tumors can be identified using chromogranin A (CgA), neuron-specific enolase (NSE), and synaptophysin, along with morphologic features. In cats, neuroendocrine carcinomas have been reported in several organs, including the bile duct, gallbladder, skin, colon, trachea, esophagus, and liver.1,2,10,17,26–31 However, pancreatic neuroendocrine carcinoma has not been described in the pancreas of cats to our knowledge.
A 35-mo-old spayed female mixed-breed cat suffering from continuous vomiting, emaciation, and abdominal distention for 2 wk was presented to a private veterinary clinic for evaluation. A detailed ultrasonographic examination revealed mild ascites and multiple hepatic nodules. Serologic analyses for antibodies against feline immunodeficiency virus and coronavirus, and feline leukemia virus antigens were negative, and against feline panleukopenia virus antigens were positive. Additional radiographic examination and aggressive therapy were not performed by the owner’s request. At 71 d after the initial visit, the cat died with multiple hepatic masses, with anemia, jaundice, and hypoalbuminemia, and was subjected to autopsy on the same day.
Numerous firm masses, 0.5–2.5 cm diameter, were located randomly in the left lobe of the pancreas (Fig. 1A). Both the external and cut surfaces of the masses were homogeneously gray-white. There were multifocal gray- or yellow-white masses, 0.5–2.0 cm diameter, in the liver (Fig. 1B). Some masses had a centrally depressed surface or central cysts containing yellow-brown cloudy fluid. The serosa of the stomach and greater omentum exhibited similar masses. The gastric, pulmonary, and mediastinal lymph nodes were enlarged. Jaundice and pleural and peritoneal effusion were also observed.
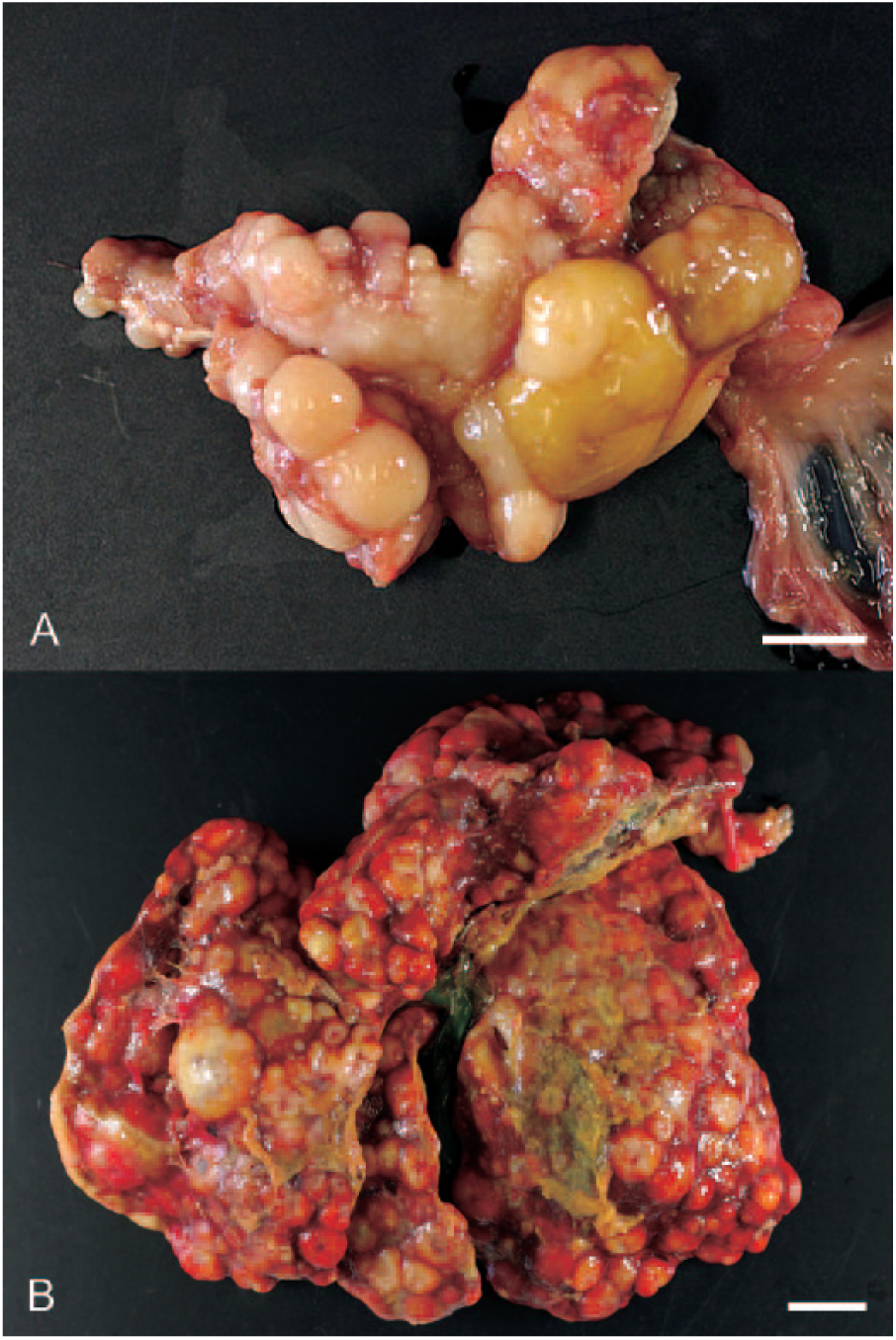
Gross appearance of pancreatic neuroendocrine carcinoma in the pancreas and liver of a 35-mo-old female mixed-breed cat.
Tissues were fixed in 10% neutral-buffered formalin, embedded in paraffin wax, sectioned at 4 μm, and stained with hematoxylin and eosin, periodic acid–Schiff (PAS), alcian blue (AB) at pH 2.5, and Grimelius stains. Serial sections were immunostained using a labeled streptavidin–biotin method. The primary antibodies used in immunohistochemical staining were as follows: cytokeratin (CK) AE1/AE3, CK7, vimentin, NSE, CgA, synaptophysin, gastrin, carcinoembryonic antigen (CEA), S100 protein, cluster of differentiation (CD)117, CD3, CD79α, CD10, α-fetoprotein (AFP), hepatocyte, and Ki-67 (Dako Denmark, Glostrup, Denmark); insulin, glucagon, and somatostatin (Biomeda, Foster City, CA); trypsin (Millipore, Temecula, CA); CK19 (Boehringer Mannheim, Mannheim, Germany); CK14 (BioGenex, San Ramon, CA); CK5 (NeoMarkers, Fremont, CA); and nestin (Immuno-Biological Laboratories, Gunma, Japan) (Table 1). Sections were treated in 0.03% H2O2 in 33% methanol at room temperature for 30 min to block endogenous peroxidase and subsequently underwent antigen retrieval treatment. The reaction to each antigen was visualized by adding 3,3′-diaminobenzidine tetrahydrochloride chromogen and counterstaining with hematoxylin. The validation of these antibodies was confirmed by a positive reaction with normal tissues or by a negative reaction on replacement of the antibodies with normal immunoglobulins of mouse, rabbit, and guinea pig. The Ki-67 index was assessed by counting both Ki-67–positive and Ki-67–negative neoplastic cells in randomly selected fields at 400× magnification and by expressing the count as a percentage (Ki-67–positive cells/total number of cells counted). For electron microscopic examination, small pieces of the formalin-fixed neoplastic tissues were re-fixed in 1% osmium tetroxide, followed by 0.2 M phosphate buffer, and then embedded in epoxy resin. Ultrathin sections were examined using an electron microscope after staining with uranyl acetate and lead citrate.
Primary antibodies used in the current study.*
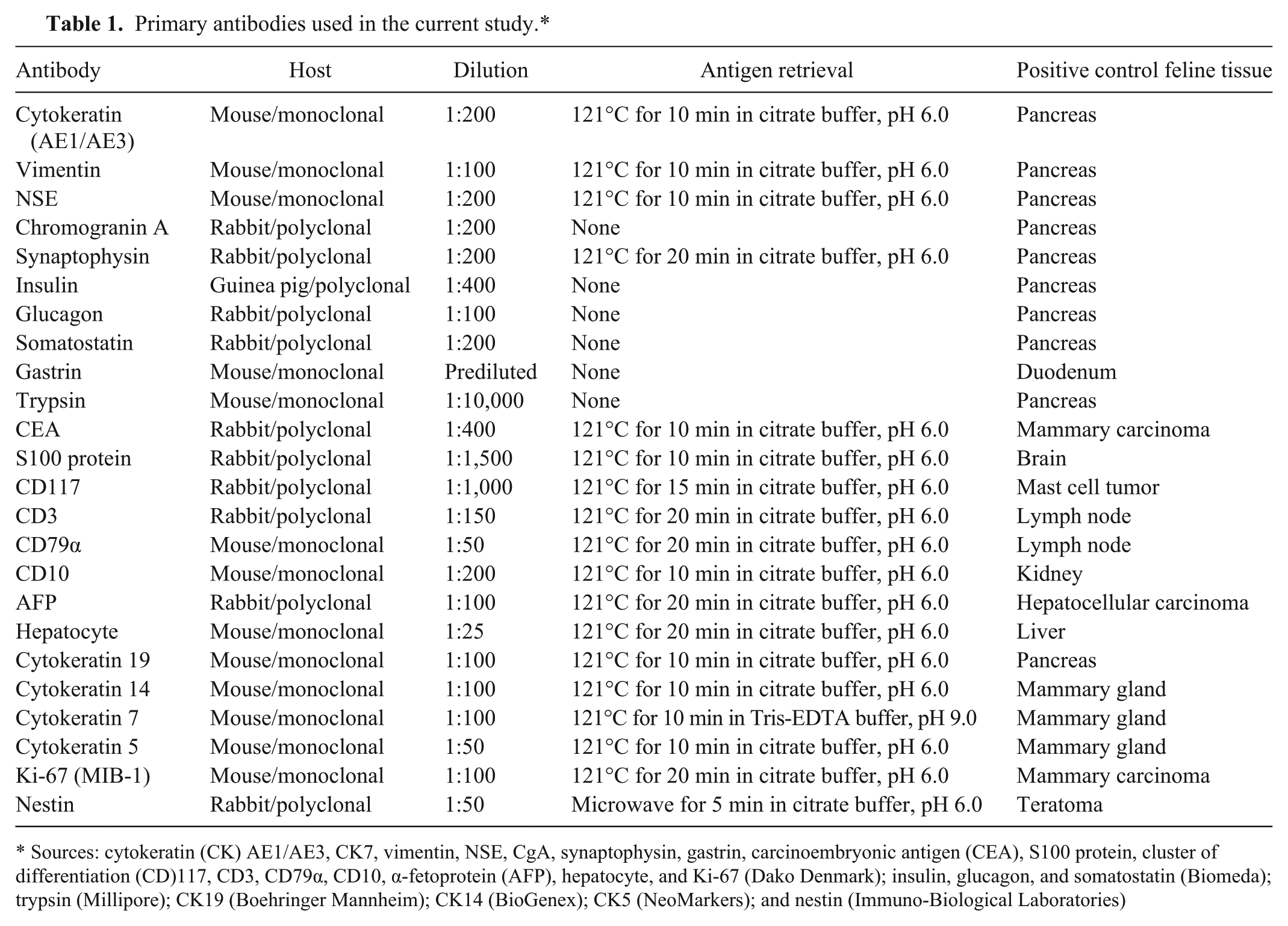
Sources: cytokeratin (CK) AE1/AE3, CK7, vimentin, NSE, CgA, synaptophysin, gastrin, carcinoembryonic antigen (CEA), S100 protein, cluster of differentiation (CD)117, CD3, CD79α, CD10, α-fetoprotein (AFP), hepatocyte, and Ki-67 (Dako Denmark); insulin, glucagon, and somatostatin (Biomeda); trypsin (Millipore); CK19 (Boehringer Mannheim); CK14 (BioGenex); CK5 (NeoMarkers); and nestin (Immuno-Biological Laboratories)
Histologic examination revealed that the pancreatic mass was comprised of 2 tumor cells: mostly small round cells and a minority of epithelial cells. The small cells were arranged in nests of various sizes, separated by thin fibrous stroma, and had small, round hyperchromatic nuclei, scant cytoplasm containing argyrophilic granules positive by Grimelius stain, and often formed rosettes (Fig. 2A). In contrast, epithelial cells formed luminal structures (Fig. 2A) and were negative for AB and PAS stains. The frequency of mitotic figures was 0–1 per high-power field. The neoplastic cells invaded the pancreatic lymph node.
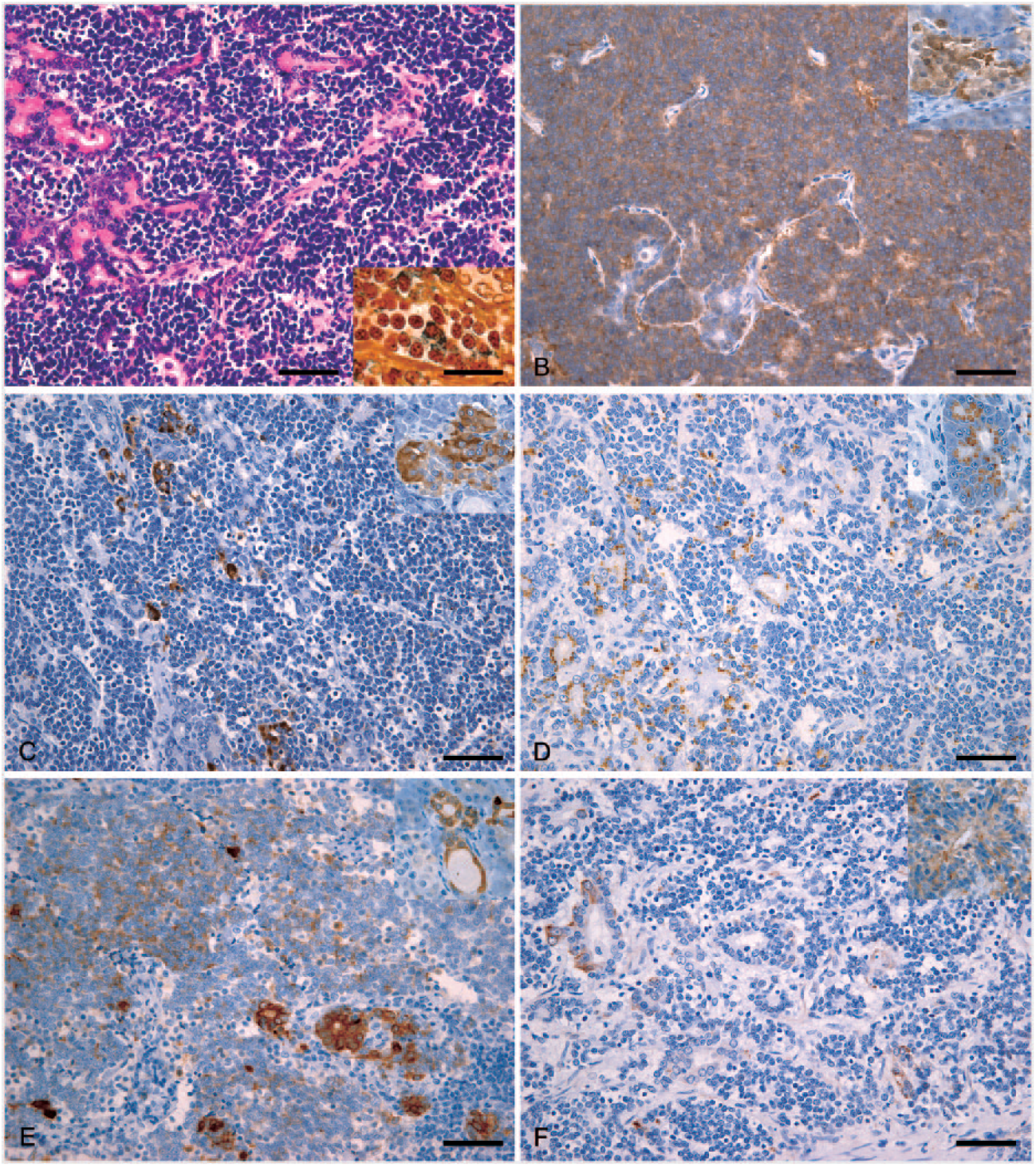
Pancreatic neuroendocrine carcinoma in a young mixed-breed cat.
Immunohistochemical examination revealed that the small cells were mostly positive for vimentin and NSE (Fig. 2B) and partially positive for CgA (Fig. 2C), cytokeratin AE1/AE3, and trypsin (Fig. 2D). In contrast, the epithelial cells were positive for AE1/AE3, trypsin (Fig. 2D), CK19 (Fig. 2E), CEA, CK7, and nestin (Fig. 2F). Both tumor cell types were negative for synaptophysin, insulin, glucagon, somatostatin, CD10, AFP, S100 protein, CK5, CK14, CD117, CD3, CD79α, and hepatocyte. The Ki-67 index of the small and epithelial cells was 16.1% and 3.2%, respectively.
Ultrastructurally, the cytoplasm of the small cells contained a few electron-dense granules of ~200 nm diameter (Fig. 3A), whereas the epithelial cells had apical microvilli and numerous zymogen granules of ~300 nm diameter, in the cytoplasm (Fig. 3B).
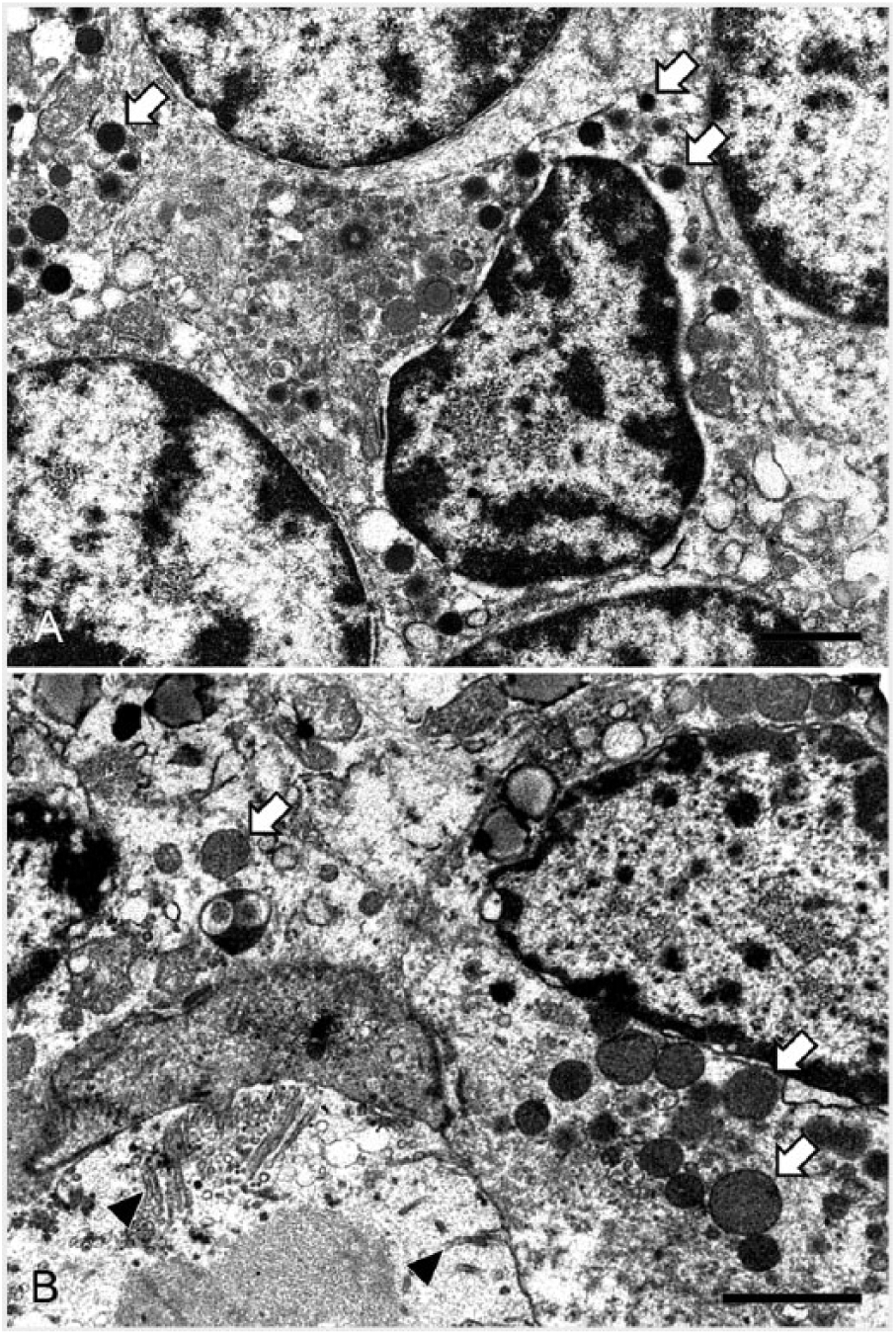
Electron micrographs of small cells and epithelial cells.
Metastatic lesions containing both neoplastic cell types were observed in the liver, greater omentum, and pancreatic, gastric, pulmonary, and mediastinal lymph nodes. Immunohistochemical features of the metastatic tumor cells were similar to those of the primary pancreatic tumor cells. Exocrine hyperplasia was also observed in the pancreas.
On the basis of the morphologic, immunohistochemical, and ultrastructural findings, the tumor was diagnosed as pancreatic neuroendocrine carcinoma with exocrine differentiation. This case should be distinguished from pancreatoblastoma and MANEC, which are characterized by neoplastic cells with acinar, endocrine, and ductal differentiation. The characteristic feature of pancreatoblastoma is the formation of squamoid nests, which are circumscribed islands of larger, spindle-shaped cells that may show keratinization. 6 However, no squamoid nests were observed in this case. In contrast, MANECs have both exocrine and endocrine components, with each component exceeding 30%. 6 The mass in our case was composed of a majority of tumor cells with neuroendocrine differentiation and a minority with exocrine differentiation. Therefore, both tumors were excluded.
Neuroendocrine markers, such as CgA, synaptophysin, and NSE, are useful for the diagnosis of neuroendocrine carcinoma, which is also characterized by morphologic features, Grimelius staining, and ultrastructural features. Regarding hepatic neuroendocrine tumors, synaptophysin and CgA were not expressed in 17 cases of neuroendocrine carcinomas that were positive for NSE. 28 In contrast, a case of neuroendocrine carcinoma expressing CgA, but not confirmed for the expression of NSE and synaptophysin, has been reported. 17 However, the expression of CgA in pancreatic neuroendocrine carcinoma remains elusive. In our case, the tumor was positive for neuroendocrine markers, such as CgA and NSE, although the expression of synaptophysin was not confirmed. Moreover, the use of CK19, CK7, and CEA for ductal cells and trypsin for acinar cells is crucial to characterize a pancreatic tumor with neuroendocrine and exocrine tumor cell proliferation, as in our case.
Pancreatic neuroendocrine tumors in humans are classified as either a functional type, such as insulinoma, gastrinoma, glucagonoma, and VIPoma, or nonfunctional types, depending on their ability to secrete biologically active hormones and polypeptides and elicit characteristic symptomatology. 8 Nonfunctional tumors frequently occur in hepatic metastases and form multifocal lesions. 33 In cats, paraneoplastic signs have been reported in cases of hypoglycemia with seizures by insulinoma, duodenal ulceration by gastrin-secreting neoplasia, and necrolytic migratory erythema associated with glucagon-producing hepatic neuroendocrine carcinoma.1,9,12,13,23 In addition, hyperinsulinemic and hypoglycemic episodes of general shaking, also caused by mixed acinar–endocrine carcinoma, were reported in a dog. 4 In contrast, the most common clinical presentations of exocrine pancreatic carcinoma in cats were weight loss, decreased appetite, vomiting, palpable abdominal mass, and diarrhea. 20 In our case, clinical signs, such as jaundice and hypoalbuminemia, were caused by the multiple hepatic metastases of the pancreatic tumor, but paraneoplastic signs were absent. Unfortunately, in this case, although we confirmed the existence of neuroendocrine and zymogen granules, we could not identify a specific substance, suggesting that this case may have been a nonfunctional neuroendocrine carcinoma.
In humans, pancreatic neuroendocrine carcinoma is a high-grade tumor, defined by the presence of >20 mitoses per high-power field and >20% of Ki-67 indices. 6 However, the grading system regarding mixed tumors with both endocrine and exocrine differentiation remains unclear. This tumor was a high-grade type, exhibiting aggressive behavior, such as metastases to the lymph nodes and liver leading to hepatic failure, although both the majority of tumor cells with neuroendocrine differentiation and the minority of cells with exocrine differentiation had a Ki-67 index of <20%.
Pancreatic tumors composed of neuroendocrine and exocrine tumor cells have been reported as neuroendocrine carcinoma with exocrine differentiation, acinar cell carcinoma with neuroendocrine differentiation, or MANEC.15,18,24,25 Evidence from a 2012 study suggests that pancreatic tumors in humans develop in tumor cells with stemness features, also termed cancer stem cells, which are capable of differentiating into exocrine and endocrine cells. 3 Cancer stem cells are involved in tumorigenesis, tumor progression, and metastasis, and may be dependent on the phenotype for epithelial–mesenchymal transition, acquiring an increased migratory capacity and invasiveness. 5 Although pancreatic cancer stem cells have been identified as CD44+ CD24+ epithelial-specific antigen (ESA)+19 as well as ALDHhigh CD133+ cells, 16 their differentiation capacity toward acinic, ductal, and endocrine cells remains elusive. In humans, nestin, a marker of stem/progenitor cells, is expressed in 30% of ductal adenocarcinomas and is a key regulator of not only cell migration, invasion, and metastasis in pancreatic tumors, but also self-renewal in pancreatic cancer stem cells.21,22 In the adult human pancreas, nestin-positive cells can be differentiated into endocrine and exocrine cell phenotypes in vitro, whereas most exocrine cells are derived from nestin-expressing progenitors during embryonic pancreas development.7,34 Therefore, nestin is a useful marker for cancer stem cells and pancreatic adenocarcinoma progression. In our case, nestin may have played a crucial role in feline pancreatic tumor formation, in particular exocrine carcinoma, but the tumorigenesis underlying this case remains unknown.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by Grants-in-Aid for Scientific Research (26292161 to M. Michishita) from the Ministry of Education, Culture, Sports, Sciences and Technology, Japan (MEXT).
