Abstract
Twenty-one orally inoculated and seven naturally infected sheep with scrapie were examined for PrPSc in peripheral tissues and in the central nervous system (CNS), using immunohistochemistry. In the inoculated group, VRQ (valine at codon 136, arginine at codon 154 and glutamine at codon 171)/VRQ sheep generally had a greater accumulation of the pathologic form of prion protein (PrPSc) in peripheral tissues, as compared with VRQ/ARQ (alanine at codon 136, arginine at codon 154, and glutamine at codon 171) animals at corresponding time points after inoculation. PrPSc was not detected in the ileal Peyer's patch, the spleen, the superficial cervical lymph node, and peripheral nervous tissues of several inoculated VRQ/ARQ animals. All inoculated VRQ/VRQ sheep, but only one of eight inoculated VRQ/ARQ animals, were PrPSc-positive in the CNS. Thus, the propagation of PrPSc seemed slower and more limited in VRQ/ARQ animals. Tissue and cellular localization of PrPSc suggested that PrPSc was disseminated through three different routes. PrPSc-positive cells in lymph node sinuses and in lymphatics indicated spreading by lymph. The sequential appearance of PrPSc in the peripheral nervous system and the CNS, with satellite cells as early targets, suggested the periaxonal transportation of PrPSc through supportive cells. Focal areas of vascular amyloid-like PrPSc in the brain of five sheep, suggested the hematogenous dissemination of PrPSc. There was a poor correlation between the amount of PrPSc in the CNS and clinical signs. One subclinically affected sheep showed widespread PrPSc accumulation in the CNS, whereas three sheep had early clinical signs without detectable PrPSc in the CNS. A VV136 (homozygous for valine at codon 136) sheep inoculated with ARQ/ARR (alanine at codon 136, arginine at codon 154, and arginine at codon 171) tissue succumbed to disease, demonstrating successful heterologous transmission. Less susceptible sheep receiving VRQ/VRQ or ARQ/ARR material were PrPSc-negative by immunohistochemistry, enzyme-linked immunosorbent assay, and western blot.
Keywords
Transmissible spongiform encephalopathies, or prion diseases, are progressive neurodegenerative disorders, of which scrapie is the prototype. These diseases are characterized by the accumulation of a pathologic form of a host-encoded prion protein (PrP) termed PrPSc, which, according to the prion hypothesis, is the infectious agent. 41 Generation of PrPSc is dependent on the presence of the normal form of PrP (PrPC), 12 which is a glycosylphosphatidylinositol-anchored cell surface protein. 44 The conversion of PrPC to PrPSc most likely occurs at the plasma membrane or along the endocytic pathway of the cell. 7, 13, 14
In sheep, susceptibility to this disease is highly dependent on polymorphism in the PrP gene (PRNP). Valine (V) at codon 136 and glutamine (Q) at codon 171 are scrapie promoting, whereas arginine (R) at position 171 is associated with resistance to the disease. Codon 154 is also a polymorphic site in the sheep PRNP but less related to disease susceptibility. 19 Homozygous V136R154Q171 sheep (VRQ) are regarded as the most susceptible to the disease. The R171 allele is influential, and VRQ/ARR sheep are less susceptible to scrapie (A=alanine). ARR/ARR sheep are the most resistant to the disease.
Oral infection is the most plausible means of natural transmission of scrapie in sheep. The early presence of PrPSc has been found in the gut-associated lymphoid tissue (GALT), the mesenteric lymph nodes, the medial retropharyngeal lymph node (RPLN), and the tonsil in sheep that were infected naturally as well as experimentally. 1, 25, 31, 49 The lymphoreticular phase is prominent in sheep with susceptible PrP genotypes. In contrast, lymphoid tissues appear to be minimally affected in sheep with the ARR haplotype, 1, 43, 48 although we recently reported PrPSc in follicles of the ileal Peyer's patch (IPP) of a naturally infected lamb with the PrP genotype VRQ/ARR. 16
Accumulation of PrPSc in the enteric nervous system (ENS) occurs later than in the GALT. 1 The infectious agent seems to be transported to the central nervous system (CNS) along two nervous pathways. One route is through the splanchnic nerves, connected to the intermediolateral column (IMLC) of the distal half of the thoracic and the rostral part of the lumbar spinal cord. The other route is through the vagus nerve, connected to the dorsal motor nucleus of the vagus nerve (DMNV) in the medulla oblongata. 4, 33, 38, 49 PrPSc can also be hematogenously transported, and blood from sheep infected with either bovine spongiform encephalopathy (BSE), or scrapie, has been used to experimentally transmit the disease. 28
In the CNS, characteristic pathologic changes in prion diseases are vacuolation of neuropil and neuronal perikarya, accompanied by degenerative features such as chromatolysis, cell shrinkage, neuronal necrosis, and gliosis. 18 The distribution of histopathologic lesions 36, 50 and the relative magnitude of different types of PrPSc accumulation 20 in the brains of clinically affected animals can vary among sheep from different sources. The distribution and the magnitude of these lesions and PrPSc accumulation are considered to be related to the infecting scrapie strain as well as to the PrP genotype of the sheep. The transmission of natural scrapie from different sheep sources to inbred mice is associated with reproducible biological characteristics, incubation periods, and neuropathology. These features are consistent with the isolation of specific strains of scrapie. 10, 11
There are few sequential studies of the dissemination of PrPSc in ovine disease, but they include sheep that are naturally infected with scrapie 31, 47, 49 and sheep infected orally with BSE. 32 On the other hand, mapping of PrPSc propagation throughout peripheral tissues and CNS has not been done previously in experimental ovine scrapie. We have developed an experimental model of scrapie in sheep and recently described the early uptake and dissemination of PrPSc with emphasis on study of the lymphoid tissues. 25 In this study, we use the same model to study further the dissemination of PrPSc as well as the progression of the disease in peripheral tissue and CNS in sheep of different PrP genotypes. To evaluate whether the experimental model mimics natural scrapie, we compare PrPSc distribution in naturally infected sheep in preclinical and clinical phases with the distribution in experimentally inoculated animals.
Materials and Methods
PrP-genotyping
PrP-genotyping was performed by direct sequencing of a polymerase chain reaction–generated product covering codons 93–216 of the PrP open reading frame, as described by Tranulis et al. (1999). 45 The polymorphisms at codons 136, 154, and 171 in the ovine PrP gene are indicated with the appropriate amino acid single-letter code.
Animals
Twenty-one lambs between the ages of 46 and 61 days (with a mean age of 55.6 days, SD 4.7) were inoculated with brain material from clinical cases of scrapie, using a stomach tube passed into the rumen or abomasum (Table 1). The lambs were in an early phase of rumination, and regurgitation to the mouth was expected in all. Each lamb was given a single dose of 15 ml of a 30% (wt/vol) brain tissue homogenate (5 g tissue). The inoculum for each PrP genotype contained tissues from cerebellum, thalamus, and the hippocampus region from several scrapie-affected sheep. Frozen, infected tissues were pooled and mixed with physiologic saline in a stomacher. Scrapie was confirmed in the donor sheep by use of immunohistochemistry (VRQ/VRQ and VRQ/ARQ sheep) or western blot (ARQ/ARR sheep). The amount of infectivity in each inoculum was not known. Inocula from VRQ/VRQ, VRQ/ARQ, and ARQ/ARR donor sheep were given to recipient lambs with the same PrP genotypes. These inoculations were defined as homologous. In addition, inocula from VRQ/VRQ donor sheep were given to VRQ/ARR, AHQ/AHQ, and ARQ/ARR recipient lambs, and an inoculum from ARQ/ARR donor sheep was given to a VRQ/VRQ recipient lamb. These inoculations were defined as heterologous. Fifteen control lambs, matched for age and PrP genotype, received an oral dose of physiologic saline with a stomach tube on the same day as the inoculated lambs. The inoculated and control animals were kept in separate isolation facilities. Infected sheep were continuously monitored by video cameras. All animal experiments adhered to legal and ethical national requirements and codes of conduct.
Details of experimentally infected sheep included in the study.∗
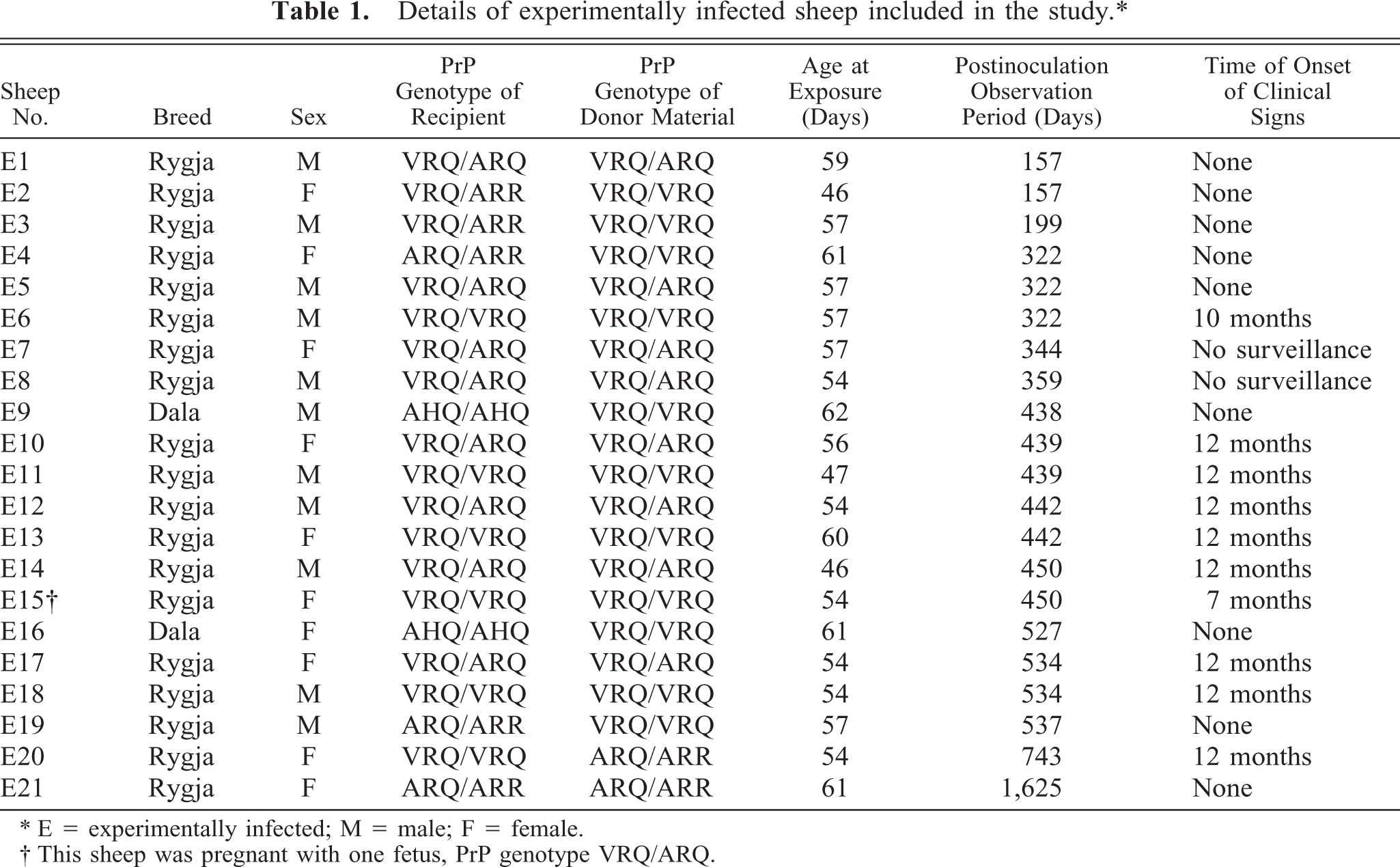
∗ E = experimentally infected; M = male; F = female.
† This sheep was pregnant with one fetus, PrP genotype VRQ/ARQ.
Seven naturally infected sheep in preclinical and clinical phases of the disease were also included in this study (Table 2). The sheep originated from four different farms and varied in age from 10 months to 7 years. Sheep N25, N26, and N28 were pregnant. One fetus from sheep N26 and one fetus from sheep N28 were PrP genotyped, and they were found to be of ARQ/ARR and VRQ/ARR genotypes, respectively.
Details of naturally infected ewes with scrapie included in the study.∗

∗ N = naturally infected; NE = not examined.
† Offspring of sheep N28.
Sampling of material
The inoculated animals and the corresponding controls were examined sequentially postmortem, between 157 and 1,625 days postinoculation (dpi) (Table 1). The animals were put in deep pentobarbital anesthesia, euthanatized by exsanguination, and then examined. Sheep E3 was found dead and examined approximately 5 hours later. This animal was subsequently diagnosed as having focal symmetrical encephalomalacia. Postmortem examination and tissue sampling were performed in the same manner for inoculated, naturally infected, and control animals. Table 3 presents an overview of all tissues collected. Tissue samples prepared for histology and immunohistochemistry were fixed by immersion in 4% formaldehyde immediately after death. Samples were subsequently dehydrated in graded ethanol solutions and embedded in paraffin. Tissue samples for enzyme-linked immunosorbent assay (ELISA) and western blot were frozen at −20 C.
Collected tissues and examined areas.
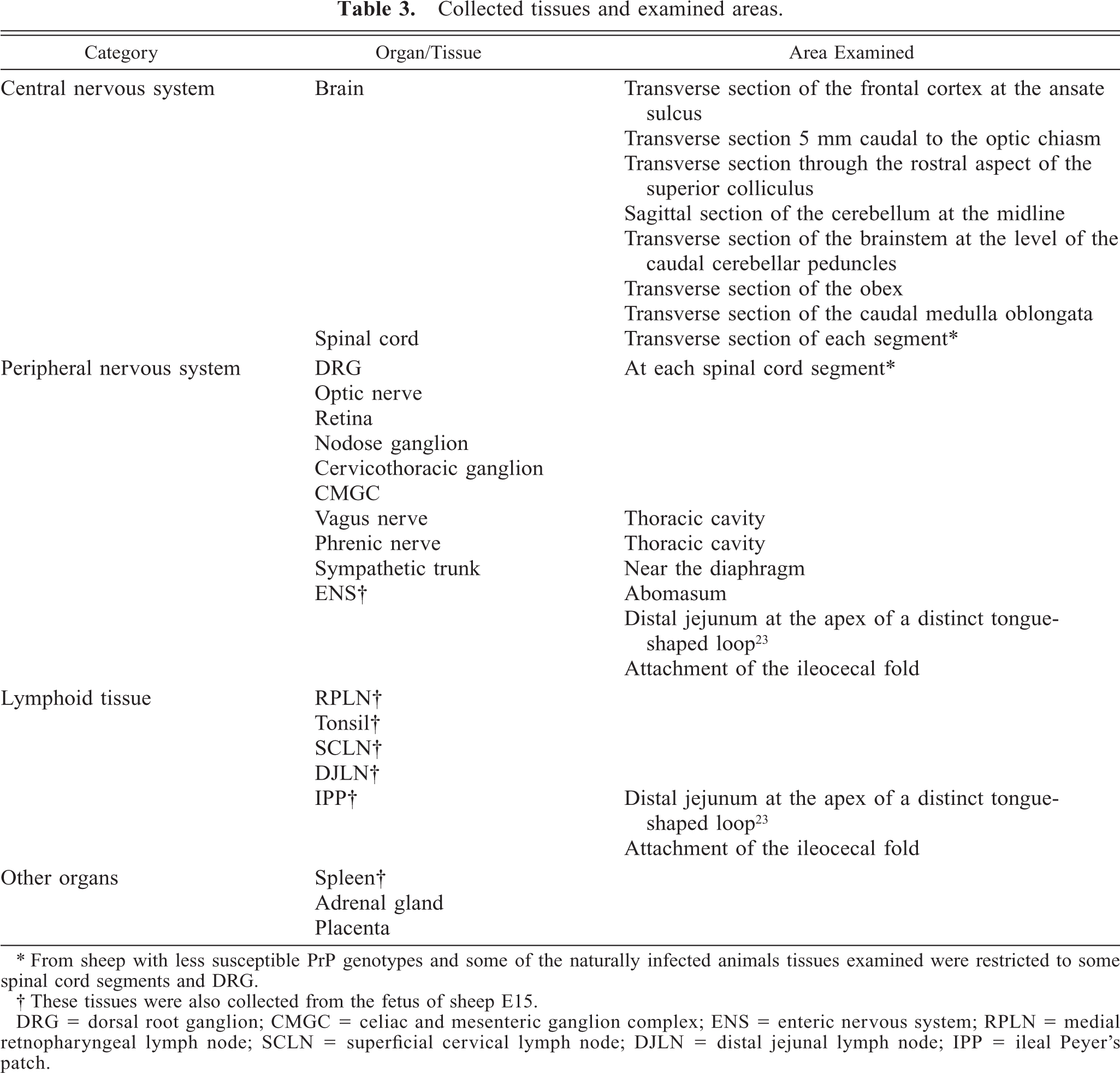
∗ From sheep with less susceptible PrP genotypes and some of the naturally infected animals tissues examined were restricted to some spinal cord segments and DRG.
† These tissues were also collected from the fetus of sheep E15.
DRG = dorsal root ganglion; CMGC = celiac and mesenteric ganglion complex; ENS = enteric nervous system; RPLN = medial retnopharyngeal lymph node; SCLN = superficial cervical lymph node; DJLN = distal jejunal lymph node; IPP = ileal Peyer's patch.
Immunohistochemistry
Sections were mounted on Superfrost® Plus slides (Menzel-Gläser, Braunsdawag, Germany) and dried for 48 hours at 37 C. Immunohistochemistry was performed mainly using the monoclonal anti-PrP antibody F89/160.1.5 (VMRD Inc., Pullman, Washington, DC), covering the peptide sequence 142–145, and diluted 1 : 2,000. Sections from peripheral tissues of infected, susceptible sheep that were initially negative were reexamined with at least one of the following PrP antibodies: p4 (peptide sequence 89–104, monoclonal, kindly provided by M. Groschup), L42 (peptide sequence 145–163, monoclonal, kindly provided by M. Groschup), and R486 (peptide sequence 217–231, polyclonal, kindly provided by R. Jackman). After rehydration, tissue sections were immersed in formic acid and autoclaved at 121 C in 0.01 M citric acid (pH 6.1). Endogenous peroxidase was blocked by the use of 0.3% H2O2 in methanol for 30 minutes at room temperature. Tissue sections were then incubated with 20% normal goat serum in phosphate-buffered saline, pH 7.2, for 20 minutes, to block nonspecific binding sites. Sections were subsequently incubated with primary antibodies at room temperature for 1 hour. Further steps were performed with Envision™+ kit (DakoCytomation, K4005, DAKO Corporation, Carpinteria, CA). All runs included at least one positive brain section and negative control tissue sections from the same organs as were included from the infected sheep. The positive controls were confirmed clinical scrapie cases. The intensity of the PrPSc labeling on the positive brain section was evaluated in each run to compare immunolabeling between runs. Negative tissue samples came from the controls matched for age and PrP genotype. To characterize non-specific immunolabeling, mouse immunoglobulin G1–negative control serum (DakoCytomation, X 0931, DAKO Corporation, Glostnip, Denmark) was used to replace the primary antibody on one section in each run. Digital images of sections were captured using a digital camera (Spot RT230-2 Slider, Diagnostic Instruments, Sterling Heights, MI) mounted on a microscope (Leica DM RA/RXA, Leica Microsystems Wetzlar GmbH, Wetzlar, Germany).
Enzyme-linked immunosorbent assay
Tissue samples included in the ELISA investigation were the distal jejunal lymph node (DJLN), the RPLN, the spleen, the eighth thoracic segment of the spinal cord, the medulla oblongata at the obex, the cerebellum, and the midbrain. These tissue samples were derived from the heterologous inoculations (except sheep E9), the homologue ARQ/ARR inoculation, and the negative control animals. PrPSc was detected using an enzyme immunometric assay (Platelia test; Bio-Rad, Sundbyberg, Sweden). This test is used for the diagnosis of BSE in bovine tissue. However, it is also suitable for the diagnosis of scrapie because both the monoclonal antibodies used recognize ovine PrP. The method has been described previously and is summarized briefly in this study. 6 Samples from the CNS and lymphoid tissues were homogenized with a ribolyser (Savant Instruments, Holbrook, NY) at about 20% (wt/vol) in a 5% glucose solution. The homogenates were treated with proteinase K for 10 minutes at 37 C and then centrifuged. The pellets were resuspended in a denaturing solution, heated to 100 C, and then diluted threefold in Tris buffer, which contained 3% bovine serum albumin. Thereafter, 100 μl from each sample was applied to the antibody-coated wells of the Bio-Rad ELISA test. The microplates were washed three times, incubated with the 100 μl enzyme-conjugated antibody for 1 hour at 37 C, and then washed again. Finally, the 100 μl substrate was added and allowed to react in darkness for 30 minutes before the addition of 1 N of sulfuric acid. Immediately thereafter, levels of absorbance were measured at 450/620 nm with a microplate reader (PR 2100; Bio-Rad, Marres-la-Coquette, France). A cutoff value was calculated. That value was the mean of the measurements of the four negative controls included in the kit, plus 0.090 absorbance units. 21
Western blot
Tissue samples included in the western blot investigation were the DJLN, the RPLN, the spleen, the eighth thoracic segment of the spinal cord, the medulla oblongata at the obex, the cerebellum, and the midbrain. These tissues were derived from the heterologous inoculations (except sheep E3 and E9), the homologue ARQ/ARR inoculation, and the negative control animals. The method has been described previously and is summarized briefly in this study. 6 Samples were treated with proteinase K and centrifuged. Pellets were resuspended in Laemmli buffer, heated, and centrifuged, before being analyzed with sodium dodecylsulphate–polyacrylamide gel electrophoresis, using a 12% gel. After transfer to the nitrocellulose membrane, the monoclonal PrP antibody p4 was applied. PrPSc was revealed by using a chemiluminescent substrate (SuperSignal West Dura, Pierce, Rockford, IL), and the signal was captured with an imager (Fluorchem 8000, Alpha Innotech, San Leandro, CA).
Evaluation of histopathology and immunohistochemistry
One section from each peripheral tissue, from each of the seven areas in the brain and from the spinal cord segments (Table 3), was stained with hematoxylin and eosin. A parallel section was immunolabeled for PrP. Defined neuroanatomic sites in each CNS section were separately scored for histopathologic changes and for the amount and type of PrPSc deposits present. The intensity and amount of PrPSc immunolabeling in both peripheral tissues and CNS were evaluated subjectively and classified using the following scale: 0, negative; ±, sparse; +, mild; ++, moderate; +++, marked.
Results
Clinical signs
Experimentally infected sheep
With the exception of two sheep (Table 1), all animals were continuously monitored using video cameras after experimental infection. The first clinical sign to be registered was pruritus and was often characterized by scratching of the head or the lumbar area of the back. This progressed to include scratching of the flanks and was further manifested by biting at the legs. The earliest signs were detected 7 months after infection, in a VRQ/VRQ sheep (E15). The other susceptible sheep showed clinical signs between 10 and 12 months after inoculation (Table 1). In sheep E20, the pruritus did not progress significantly during the period between 12 and 23 months after infection. However, just after 23 months, sheep E20 suddenly developed severe neurologic signs including involuntary movements, disorientation, nervousness, and ataxia. Twenty-four months after inoculation, the ewe became paretic and was euthanatized.
Naturally infected sheep
Three of the sheep showed no clinical signs of scrapie (Table 2). The other four sheep were all in poor bodily condition and exhibited smacking of the lips. In three of the sheep, the main sign was pruritus with bilateral wool loss. Sheep N25 had neurologic signs including severe tremor and ataxia.
Immunohistochemical detection of PrPSc in peripheral tissues
A summary of the results is shown in Table 4.
PrPSc in peripheral tissues in experimentally and naturally infected sheep.∗
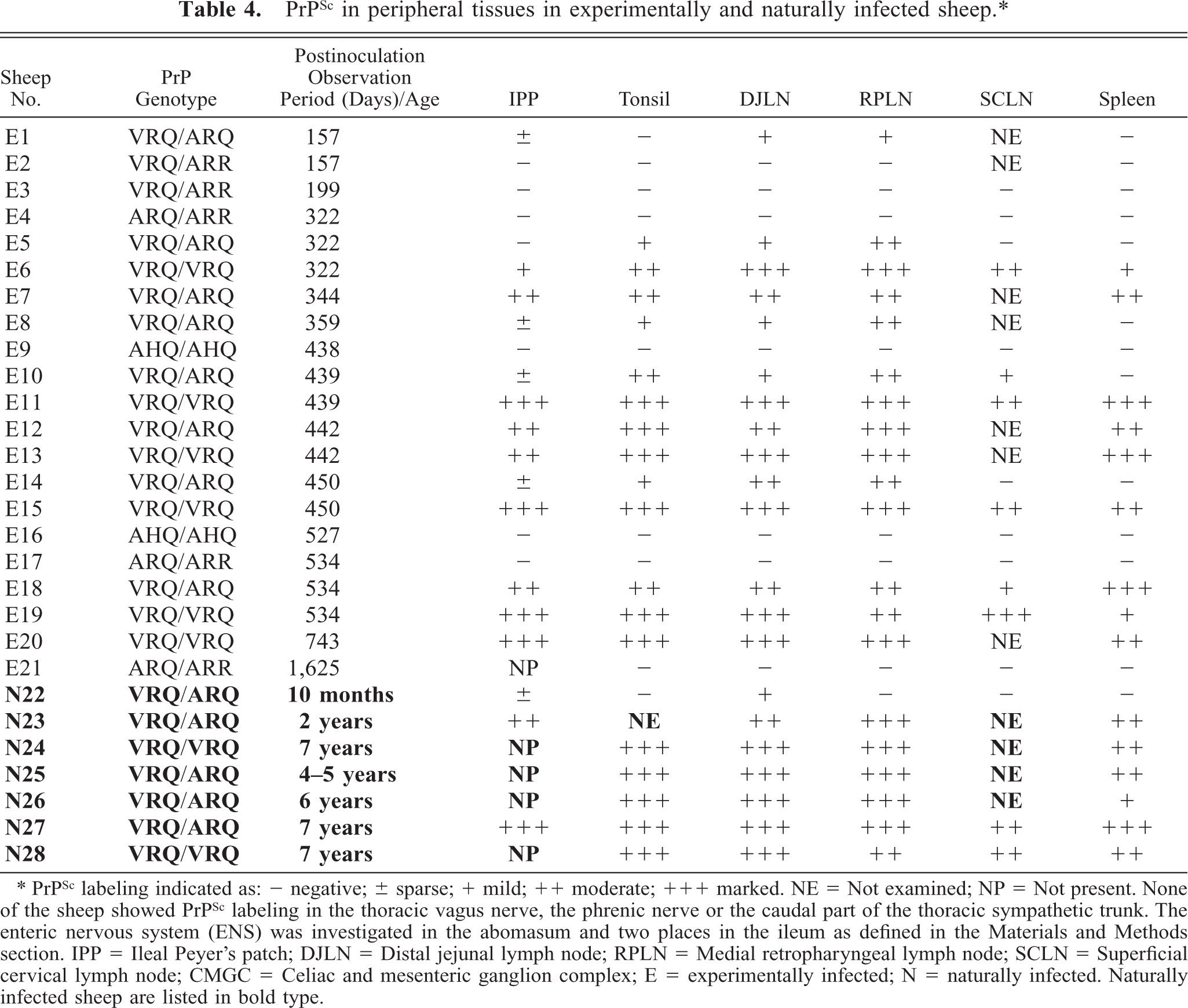
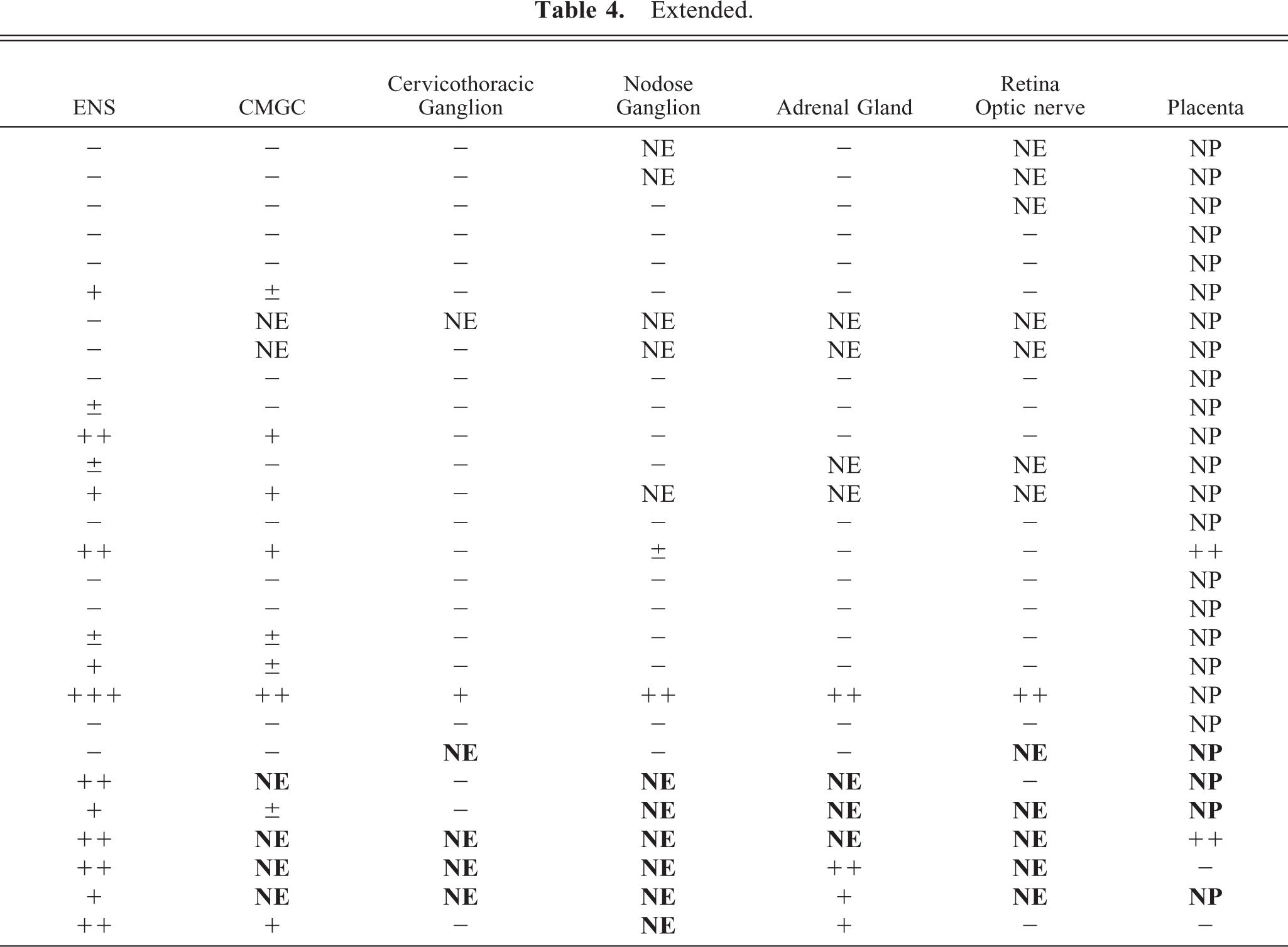
∗ PrPSc labeling indicated as: − negative; ± sparse; + mild; ++ moderate; +++ marked. NE = Not examined; NP = Not present. None of the sheep showed PrPSc labeling in the thoracic vagus nerve, the phrenic nerve or the caudal part of the thoracic sympathetic trunk. The enteric nervous system (ENS) was investigated in the abomasum and two places in the ileum as defined in the Materials and Methods section. IPP = Ileal Peyer's patch; DJLN = Distal jejunal lymph node; RPLN = Medial retropharyngeal lymph node; SCLN = Superficial cervical lymph node; CMGC = Celiac and mesenteric ganglion complex; E = experimentally infected; N = naturally infected. Naturally infected sheep are listed in bold type.
Experimentally infected sheep
All sheep with PrP genotypes, associated with reduced susceptibility for scrapie (VRQ/ARR, AHQ/AHQ, ARQ/ARR), proved negative in the peripheral tissues tested. In susceptible VRQ/VRQ and VRQ/ARQ sheep, the RPLN and the DJLN were the only organs where PrPSc was found at all points in time. Immunohistochemistry also revealed that there was a gradual increase in the number of positive lymphoid follicles in the RPLN and the DJLN, as the disease progressed. In the least affected lymph nodes, the positive follicles appeared to occur in clusters. In general, VV136 animals had more PrPSc deposits than VA136 sheep at the same point in time after inoculation. There was primarily labeling of tingible body macrophages (TBMs) in less affected lymphoid follicles with small amounts of PrPSc, which were often seen in the early stages of infection. A reticular pattern of labeling, indicative of extracellular PrPSc associated with follicular dendritic cells (FDCs), was found particularly in follicles with large amounts of PrPSc in the sheep that were in the later stages of infection. In the RPLN and the DJLN of all susceptible sheep, there was labeling of some cells in the inter-follicular area, as well as in the peritrabecular and medullary sinuses, near positive follicles (Fig. 1). With the exception of one sheep (E1), all animals had PrPSc-labeled cells in the subcapsular sinus of the RPLN. A total of 9 out of 14 sheep had PrPSc-labeled cells in the subcapsular sinus of the DJLN (Fig. 2). The morphology of many of these cells was consistent with that of macrophages. There were PrPSc-positive mono-nuclear cells in several lymphatic vessels in the RPLN of the VA136 (E14) sheep 450 days dpi. The superficial cervical lymph node (SCLN) was less affected than the other two lymph nodes examined and was negative in two of the VA136 sheep. All sheep that were positive in the SCLN had PrPSc-labeled cells in the subcapsular sinus and other sinuses near positive lymphoid follicles.
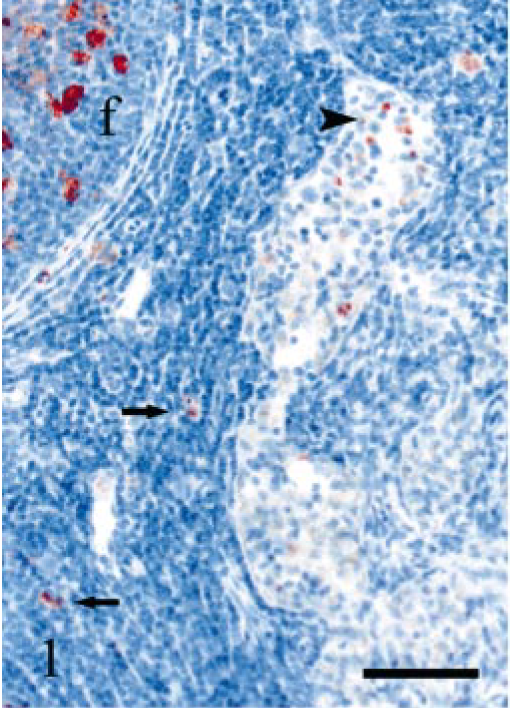
DJLN; sheep E6, 322 dpi, PrP genotype VRQ/VRQ. Several PrPSc-positive macrophages (arrowhead) in a peritrabecular sinus and a lymphoid follicle (f) with immunolabeled TBMs. A few PrPSc-positive cells (arrows) are located in the interfollicular area. PrP antibody F89/160.1.5, Envision™ + kit, Mayer's hematoxylin counterstain. Bar = 60 μm.
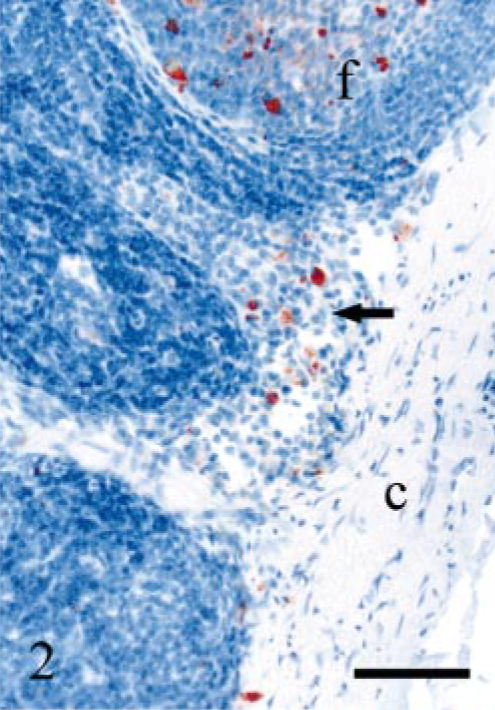
DJLN; sheep E6, 322 dpi, PrP genotype VRQ/VRQ. A group of PrPSc labeled macrophages (arrow) are seen in the subcapsular sinus. Note a PrPSc-positive lymphoid follicle (f) nearby. The capsule of the lymph node is indicated (c). PrP antibody F89/160.1.5, Envision™ + kit, Mayer's hematoxylin counterstain. Bar = 60 μm.
Immunolabeling in the tonsil correlated well with the RPLN, except in the VRQ/ARQ sheep (E1) 157 dpi. Sheep E1 proved negative in the tonsil and positive in the RPLN. The labeling pattern in the tonsillar follicles resembled that of the lymph nodes, and the number of PrPSc-positive cells in the interfollicular area varied. In several of the VRQ/VRQ sheep, but not in the VRQ/ARQ heterozygotes, PrPSc-labeled cells were found beneath the epithelium, as well as between epithelial cells. These PrPSc-positive cells were most often found in association with subepithelial and intraepithelial accumulations of leucocytes (Fig. 3).
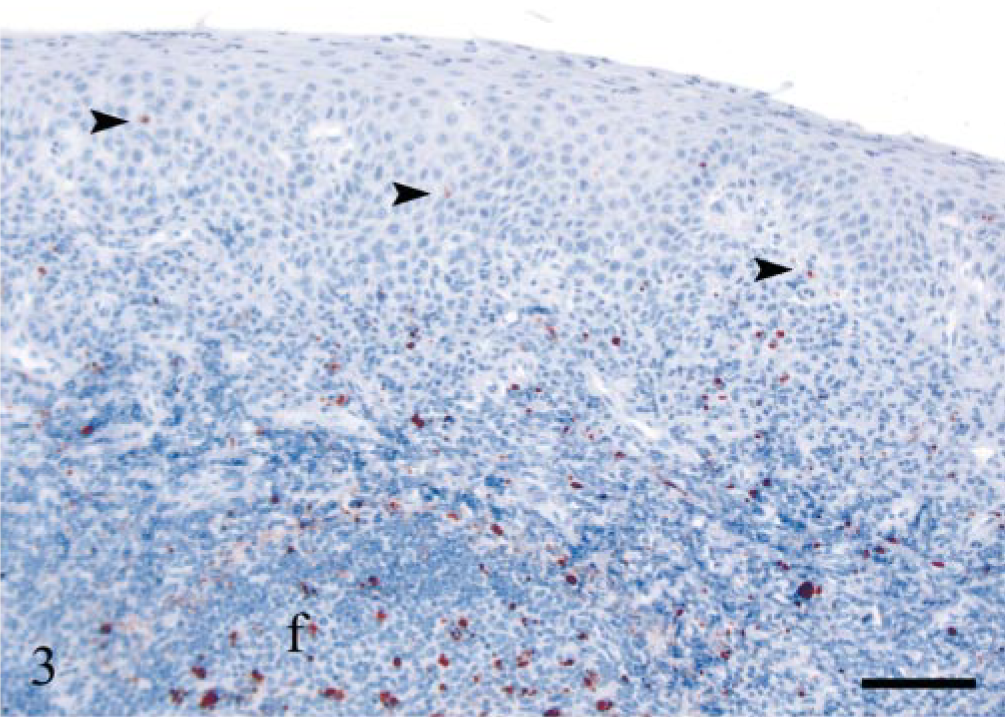
Tonsil; sheep E13, 442 dpi, PrP genotype VRQ/VRQ. Numerous PrPSc-positive leucocytes are seen beneath the epithelium and a few immunolabeled cells in the epithelium (arrowheads). There is mainly granular PrPSc labeling of macrophages in the lymphoid follicle (f). PrP antibody F89/160.1.5, Envision™ + kit, Mayer's hematoxylin counterstain. Bar = 80 μm.
The sheep included in the study had variable numbers of lymphoid follicles in the IPP, which reflected individual variation in the degree of lymphoid involution. Many of the VRQ/ARQ sheep had sparse or no PrPSc labeling in the IPP. Generally, the V136 homozygotes had more positive follicles with a higher number of PrPSc-labeled cells within each follicle than was the case with the VA136 sheep. The follicles of the IPP mainly showed TBM labeling (Fig. 4). There was also an extensive reticular PrPSc pattern in the most affected and less involuted follicles, especially of VRQ/VRQ sheep. There were few positive cells in the dome and neck areas of the follicles.
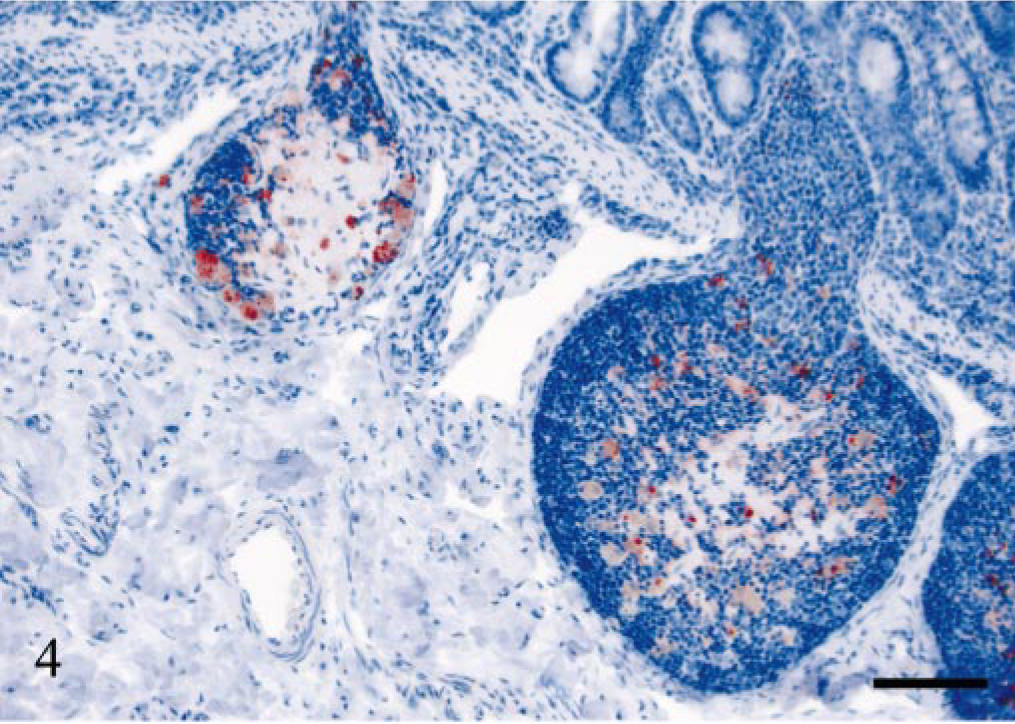
Ileal Peyer's patch; sheep E17, 534 dpi, PrP genotype VRQ/ARQ. Granular intracellular PrPSc deposits in TBMs of two lymphoid follicles undergoing involution. PrP antibody F89/160.1.5, Envision™ + kit, Mayer's hematoxylin counterstain. Bar = 80 μm.
The spleen was PrPSc-negative in five of eight VRQ/ARQ sheep and generally showed lower accumulation of PrPSc compared with the lymph nodes examined. There was mainly labeling of macrophages, but there was also a reticular pattern of labeling in the most affected germinal centers. Macrophages were also PrPSc labeled in the periarterial lymphatic sheaths. Small granular PrPSc labeling was found in the marginal zone of all PrPSc-positive animals, and in some sheep, this labeling was rather extensive (Fig. 5).
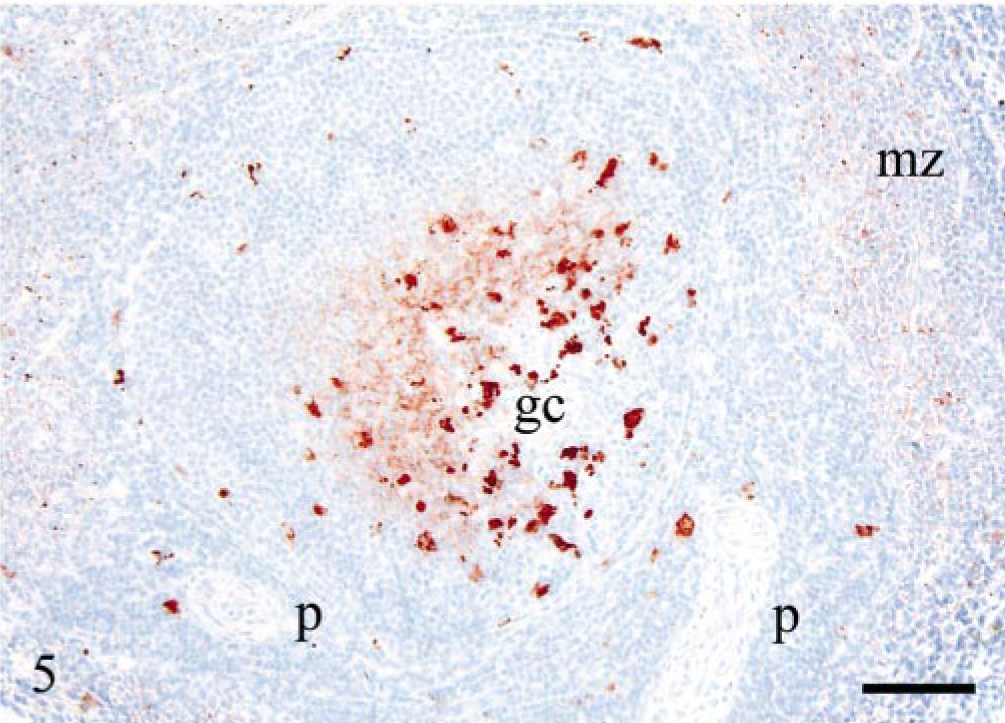
Spleen; sheep E17, 534 dpi, PrP genotype VRQ/ARQ. Reticular PrPSc pattern indicative of labeling of FDCs and granular PrPSc deposits of macrophages in the germinal center (gc). Single macrophages are labeled in the periarterial lymphatic sheaths (p). There are numerous small immunolabeled granules in the marginal zone (mz). PrP antibody F89/160.1.5, Envision™ + kit, Mayer's hematoxylin counterstain. Bar = 80 μm.
Varying levels of PrPSc labeling were observed in the peripheral nervous system. All VRQ/VRQ sheep and three VRQ/ARQ sheep were PrPSc-positive in the ENS, which was present in the form of granular PrPSc labeling in neuronal perikarya and of satellite cells. In some of the V136 homozygotes, the labeling of the ENS was mild, with few or no intraneuronal deposits and with more prominent satellite cell labeling (Fig. 6). There was sparse PrPSc labeling in the ENS of VA136 heterozygotes, and this was primarily associated with satellite cells and restricted to the distal jejunum. The celiac and mesenteric ganglion complex (CMGC) was PrPSc-positive in all VRQ/VRQ animals and in only one VRQ/ARQ sheep (E18), 534 dpi. In the VRQ/ARQ animal, only granular PrPSc deposits associated with satellite cells were detected, and the neurons were not labeled. In the VRQ/VRQ sheep, with the exception of sheep E20 (which was terminally ill), few CMGC neurons were labeled. However, sheep E20 showed numerous PrPSc-positive neuronal perikarya and additional sparse labeling in the periphery of nerve fibers. There were scattered PrPSc-labeled satellite cells in all these animals, and in many areas, this labeling was present together with PrPSc-negative neurons. Only sheep E20 showed accumulation of PrPSc in the cervicothoracic ganglion. Two V136 homozygotes (E20 and E15) were PrPSc-positive in the nodose ganglion. The labeling pattern in these two ganglia was the same as in the CMGC. Only the sheep that was terminally ill proved positive in the adrenal medulla, where many groups of chromaffin cells revealed granular and short linear PrPSc labeling (Fig. 7).
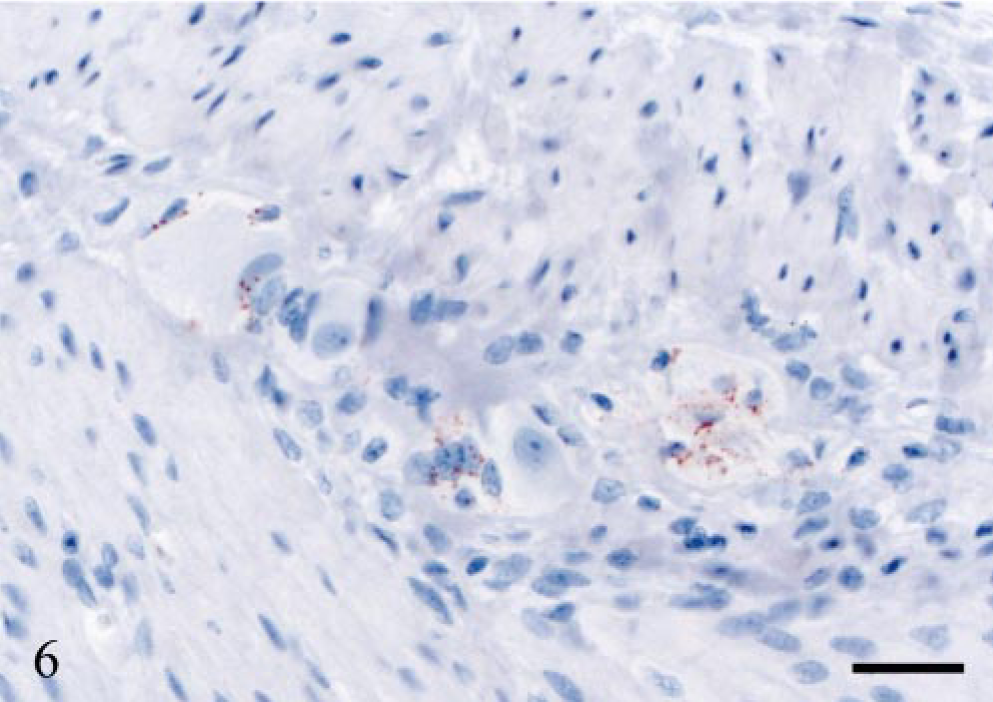
ENS; sheep E13, 442 dpi, PrP genotype VRQ/VRQ. Granular PrPSc labeling of satellite cells. The neuronal perikarya are PrPSc-negative. PrP antibody F89/160.1.5, Envision™ + kit, Mayer's hematoxylin counterstain. Bar = 30 μm.
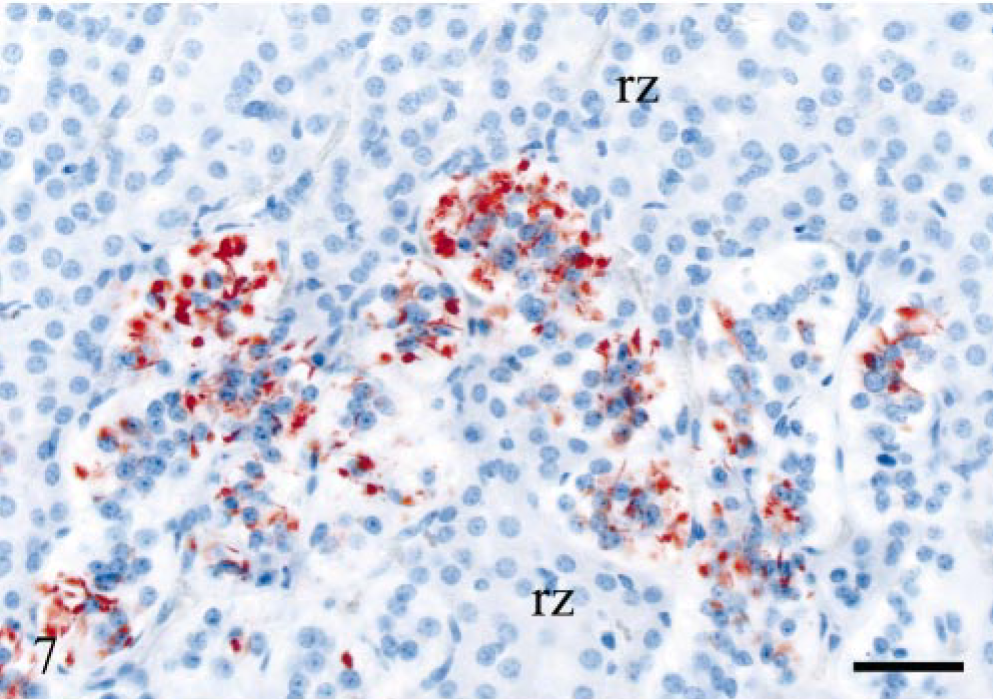
Adrenal gland; sheep E20, 743 dpi, PrP genotype VRQ/VRQ. Granular and short linear PrPSc labeling of chromaffin cells in the adrenal medulla. Cells of the reticular zone (rz) are PrPSc-negative. PrP antibody F89/160.1.5, Envision™ + kit, Mayer's hematoxylin counterstain. Bar = 40 μm.
None of the sheep showed PrPSc labeling in the thoracic vagus nerve, the phrenic nerve, or the caudal part of the thoracic sympathetic trunk. PrPSc labeling in all layers of the retina was identified in the terminally ill sheep. In the same sheep, granular PrPSc deposits were found in glial cells of the optic nerve.
Sheep E15 was pregnant with a VRQ/ARQ fetus at the time of autopsy. There were multiple PrPSc-labeled areas in the placentome. Most of the positive cells were judged to be maternal epithelium, but fetal trophoblasts were also immunolabeled. The tonsil, the RPLN, the DJLN, the SCLN, ileum, and the spleen from the fetus were examined, and all tissues were PrPSc-negative.
Naturally infected sheep
The same labeling patterns as described for the experimentally infected sheep were also present in the naturally infected sheep. Sheep N22 was weakly PrPSc-positive only in the DJLN and the IPP. In the follicles of lymph nodes, the PrPSc labeling was mainly of TBMs with a weaker FDC pattern. All animals, except sheep N22, had PrPSc-positive cells in lymph node sinuses. Sheep N23 had PrPSc-positive leucocytes in medullary lymphatic vessels in the RPLN. In general, the tonsil showed greater accumulation of PrPSc than the lymph nodes, and there was often a prominent FDC-labeling pattern in the follicles. All PrPSc-positive sheep had some PrPSc-labeled cells beneath the tonsillar epithelium as described in the experimentally infected sheep. However, this was most evident in the VRQ/VRQ animals. Less PrPSc labeling was observed in the spleen as compared with the lymph nodes and the tonsil. Lymphoid tissue was not identified in the ileum in most of the animals. There was mild to moderate immunolabeling, mainly of satellite cells, and fewer neurons in the ENS. In the CMGC, a small number of neurons and satellite cells had granular PrPSc deposits, whereas the cervicothoracic ganglion was negative in all sheep investigated. PrPSc labeling of the adrenal medulla was mild or moderate. Multifocal PrPSc-positive areas were found in a placentome of sheep N25. The accumulation of PrPSc was evident in two morphologically different cell populations, interpreted to be fetal trophoblasts and maternal epithelial cells. A few large intra-cellular PrPSc granules were found in the fetal trophoblasts, whereas there were multiple, smaller granules and more diffuse labeling in the maternal epithelium (Fig. 8).
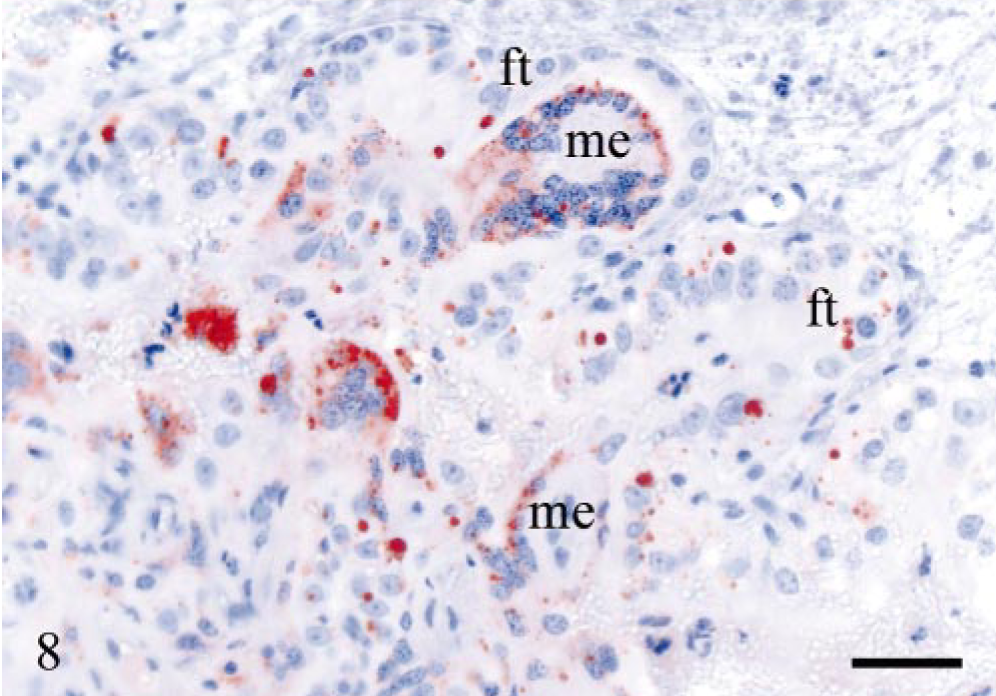
Placenta; sheep N25, 4–5 years of age, PrP genotype VRQ/ARQ. Numerous PrPSc granules in maternal epithelial cells (me). Fetal trophoblasts (ft) are less involved, but several have large intracellular, often single, PrPSc granules. PrP antibody F89/160.1.5, Envision™ + kit, Mayer's hematoxylin counterstain. Bar = 40 μm.
Histopathology of brain and spinal cord
Experimentally infected sheep
Only the terminally ill sheep (E20) showed extensive degenerative changes, including marked neuropil vacuolation throughout the brain. Additional moderate intraneuronal vacuolation was observed in the obex. In the spinal cord, there was moderate, and in some segments marked, neuropil and intraneuronal vacuolation in the IMLC and ventral horn, as well as mild neuropil vacuolation in the dorsal horn. These spongiform changes were often accompanied by gliosis. Other changes in this animal included sporadic central chromatolysis, eosinophilic, necrotizing neurons, especially evident in cerebellar Purkinje cells (Fig. 9) and some spheroids. Some vacuolation was also found in the lateral and ventral funiculi of the spinal cord. These vacuoles contained lipid-laden macrophages, degenerative axons, or were empty. Sheep E13 had a few neuropil vacuoles in the DMNV, whereas the other PrPSc-positive sheep showed no changes at this neuroanatomic site.
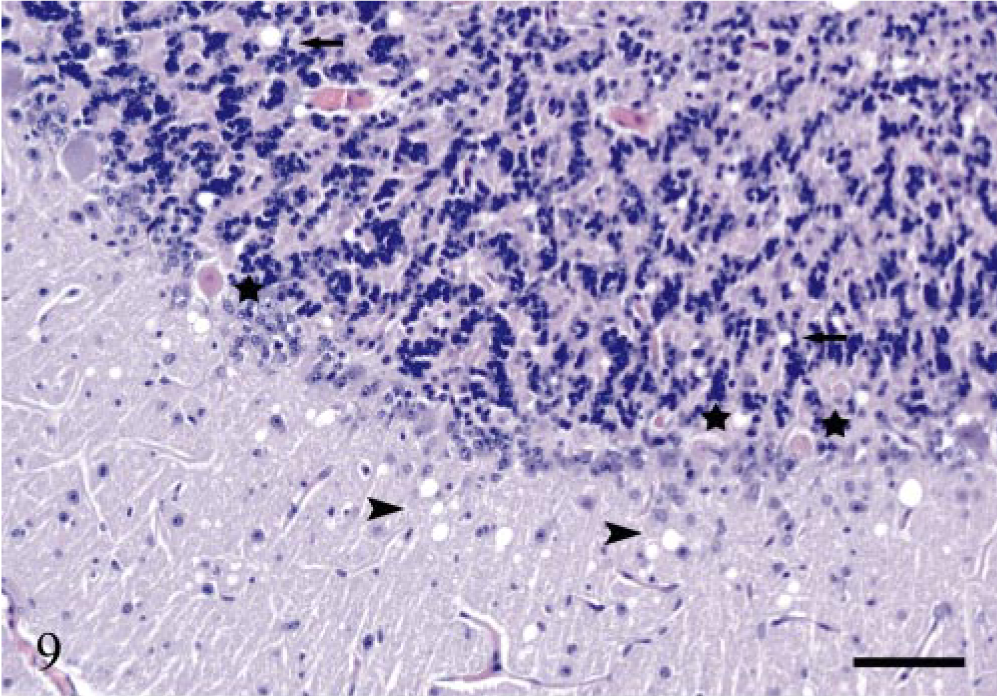
Cerebellum; sheep E20, 743 dpi, PrP genotype VRQ/VRQ. Several Purkinje cells (stars) are necrotic, showing pale, eosinophilic cytoplasm and shrunken nuclei. There are numerous vacuoles in the molecular layer (arrowheads) and fewer vacuoles in the granular layer (arrows). Hematoxylin and eosin. Bar = 80 μm.
Naturally infected sheep
Subclinically affected sheep N23 had multiple areas with moderate or marked neuropil vacuolation and occasional intraneuronal vacuoles in the brainstem and in the thalamus. There were some vacuoles in the spinal cord in this sheep. Sheep N24 had sparse vacuolation in the brainstem. All clinically affected sheep (Table 2) had marked neuropil vacuolation and occasional intraneuronal vacuoles in the brainstem. There were focal areas of neuropil vacuolation in the cerebellar and cerebral cortices. Gliosis was evident in the most affected areas. Sheep N28 had several focal areas with perivascular plaques and vacuoles in the surrounding neuropil in the thalamus (Fig. 10). In sheep N25 and N28, perivascular plaques were evident in the cerebral cortex. Sheep N26 had abundant neuropil vacuolation in the dorsal horn throughout the spinal cord, moderate vacuolation in the IMLC, and sparse vacuolation in the ventral horn. Sheep N25 showed sparse vacuolation in the lateral funiculus. The other clinically affected sheep had few vacuoles in the spinal cord.
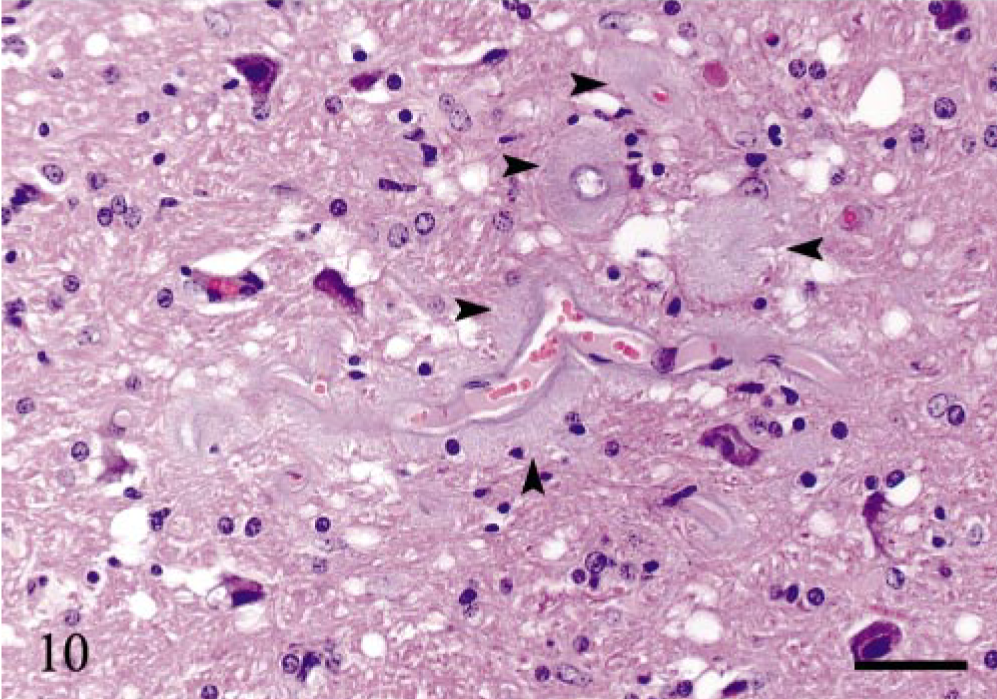
Thalamus; sheep N28, 7 years of age, PrP genotype VRQ/VRQ. Several perivascular plaques of small blood vessels (arrowheads). There are numerous vacuoles and gliosis in the surrounding neuropil. Hematoxylin and eosin. Bar = 40 μm.
Immunohistochemical detection of PrPSc in brain, spinal cord, and dorsal root ganglia
Experimentally infected sheep
The distribution of PrPSc accumulation in the brain, spinal cord, and dorsal root ganglia (DRG) is illustrated in Fig. 11. All six V136 homozygotes and only one of eight VRQ/ARQ sheep were PrPSc-positive in the brain. Of these, the terminally ill sheep (E20) had immunolabeling throughout the brain, which was dominated by intra-neuronal and intraglial granular PrPSc. Heavy punctate and coalescing neuropil labeling was observed in several areas, especially in the brainstem and in the thalamus. There were granular PrPSc deposits at the apical side of the ependyme, as well as subependymal labeling distributed throughout the ventricular system. The other six sheep were PrPSc-positive in the ventral part of the DMNV. In three of the animals, this was the only site of immunolabeling. In these early CNS stages, PrPSc was found associated with both neurons and glial cells (Fig. 12). Sheep E13, 442 dpi, showed PrPSc deposits in several nuclei in the obex and in the vestibular complex. Another VV136 sheep (E15), 450 dpi, had perivascular and mural amyloid-like deposits of PrPSc in one area in a sagittal section of the cerebellum (Fig. 13). A VRQ/ARQ sheep (E18), 534 dpi, had PrPSc deposits in the lateral periphery of nuclues ambiguus in addition to the DMNV in the obex.
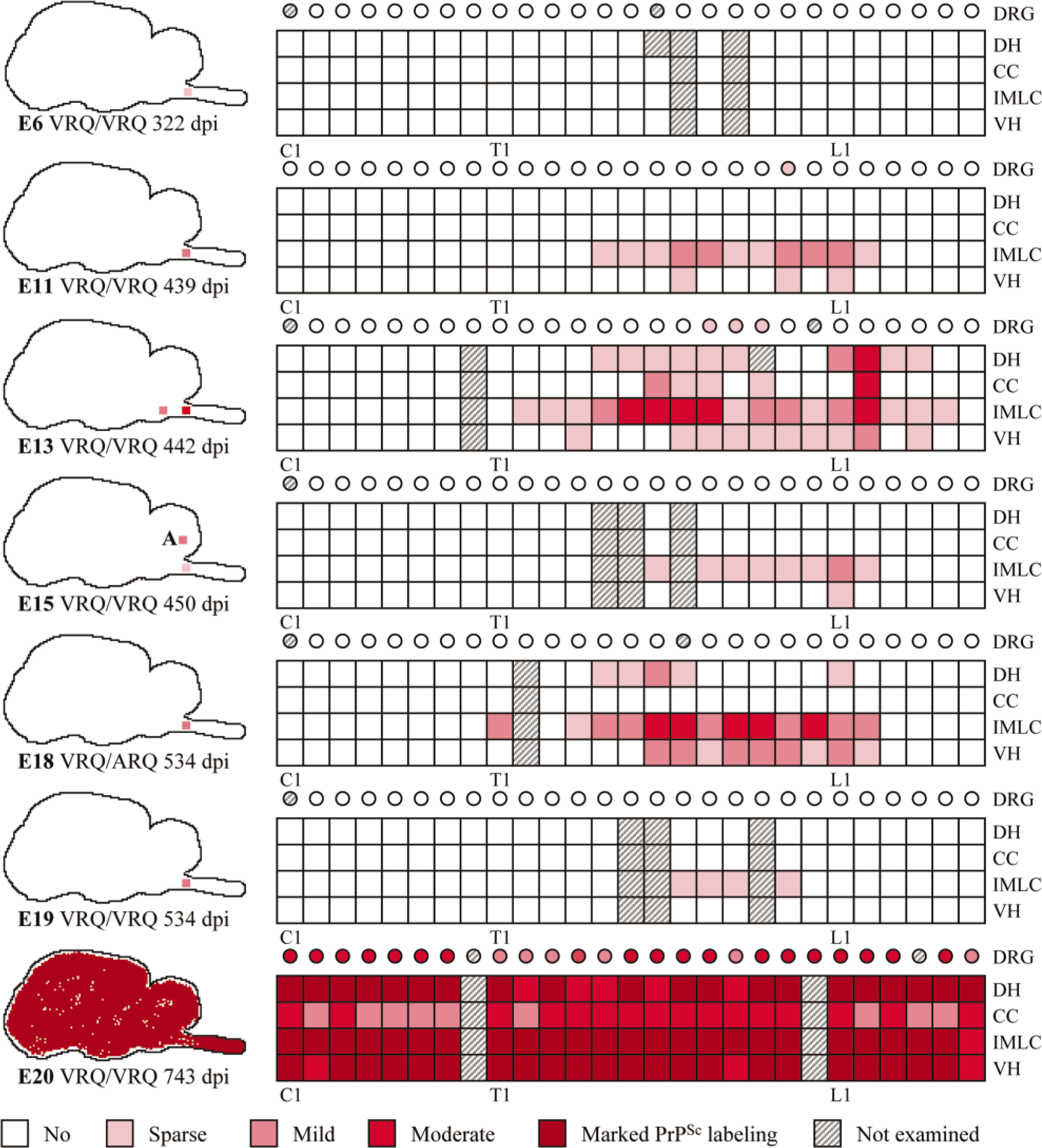
Diagrammatic representation of the distribution and severity of PrPSc in brain, spinal cord, and DRG of experimentally infected sheep. C = cervical segments; T = thoracic segments; L = lumbar segments; DRG = dorsal root ganglion; DH = dorsal horn; CC = central canal and surrounding tissue; IMLC = intermediolateral column; VH = ventral horn; A = amyloid; dpi = days postinoculation.
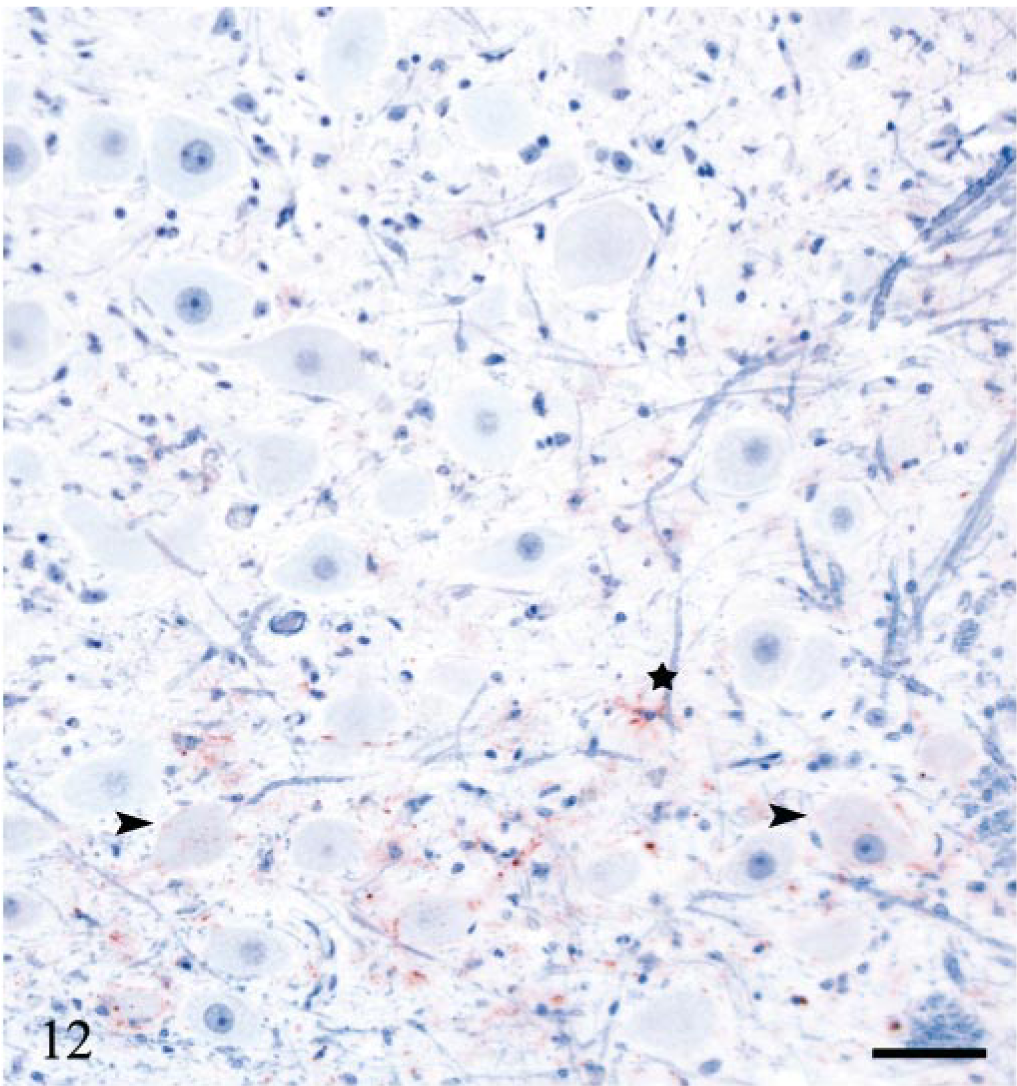
DMNV; sheep E11, 439 dpi, PrP genotype VRQ/VRQ. PrPSc labeling at the ventral margin of the nucleus. There are sparse perineuronal and intraneuronal deposits (arrowheads), stellate labeling of glial cells (star) and granular PrPSc in the neuropil. PrP antibody F89/160.1.5, Envision™ + kit, Mayer's hematoxylin counterstain. Bar = 53 μm.
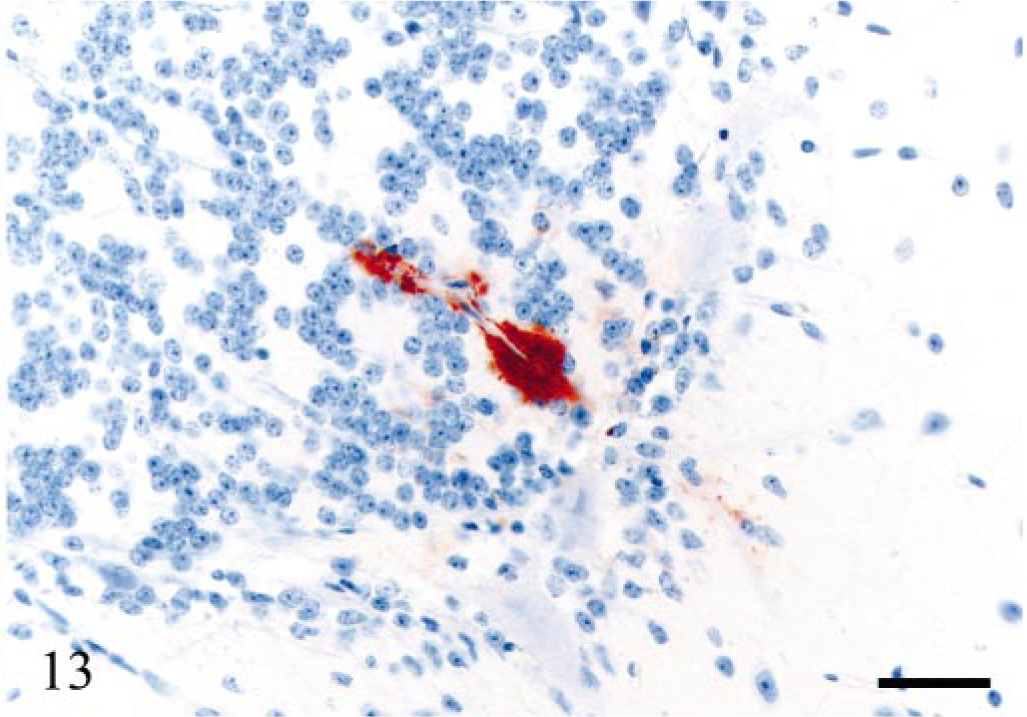
Cerebellum; sheep E15, 450 dpi, PrP genotype VRQ/VRQ. Perivascular and mural amyloid-like deposits of PrPSc in a small blood vessel of the granular cell layer. Weak immunolabeling of the surrounding neuropil. PrP antibody F89/160.1.5, Envision™ + kit, Mayer's hematoxylin counterstain. Bar = 40 μm.
All sheep, except one (E6), 322 dpi, that were PrPSc-positive in the brain had concomitant PrPSc labeling of variable areas and segments of the spinal cord. Granular intraneuronal and intraglial immunolabeling was observed through the whole spinal cord of sheep E20. Glial cells intimately associated with the ependymal cells of the central canal were immunolabeled in several areas. This sheep also had extensive labeling of white matter glial cells. In addition, dorsal root nerve fibers had peripheral granular PrPSc deposits in some segments. All DRG examined in this sheep were positive and had granular neuronal perikaryonal and satellite cell labeling (Fig. 14). In several DRG sections, PrPSc deposits were evident at the periphery of nerve fibers (Fig. 14). The other sheep (except E6) had variable PrPSc distribution in the spinal cord, and all were positive in thoracic (T) segments T8–T12. Accumulation of PrPSc were always present in the IMLC. In the least affected segments, this was the only site labeled. In more affected segments, the PrPSc deposits extended into the lateral margins of the dorsal and ventral horns (Fig. 15). In several animals, labeling was asymmetrical. At the cranial and caudal positive segments, labeling tended to be unilateral. Both neurons and glial cells tended to be targeted early. In the VRQ/VRQ sheep (E13), 442 dpi and VRQ/ARQ sheep (E18), 534 dpi, glial cells in white matter outside the IMLC were PrPSc labeled (Fig. 15). In contrast to the general description, the VRQ/VRQ sheep (E19), 534 dpi, had markedly less PrPSc in the spinal cord than the VRQ/ARQ animal (E18) at the same point in time. There was no labeling in the caudal part of the medulla oblongata or in the cervical spinal cord of those sheep that were positive in the obex and the thoracic and lumbar spinal cord. Two VRQ/VRQ sheep (E11 and E13) revealed sparse PrPSc labeling of some of the last thoracic DRG, having mainly granular deposits of satellite cells associated with a few weakly labeled neurons.
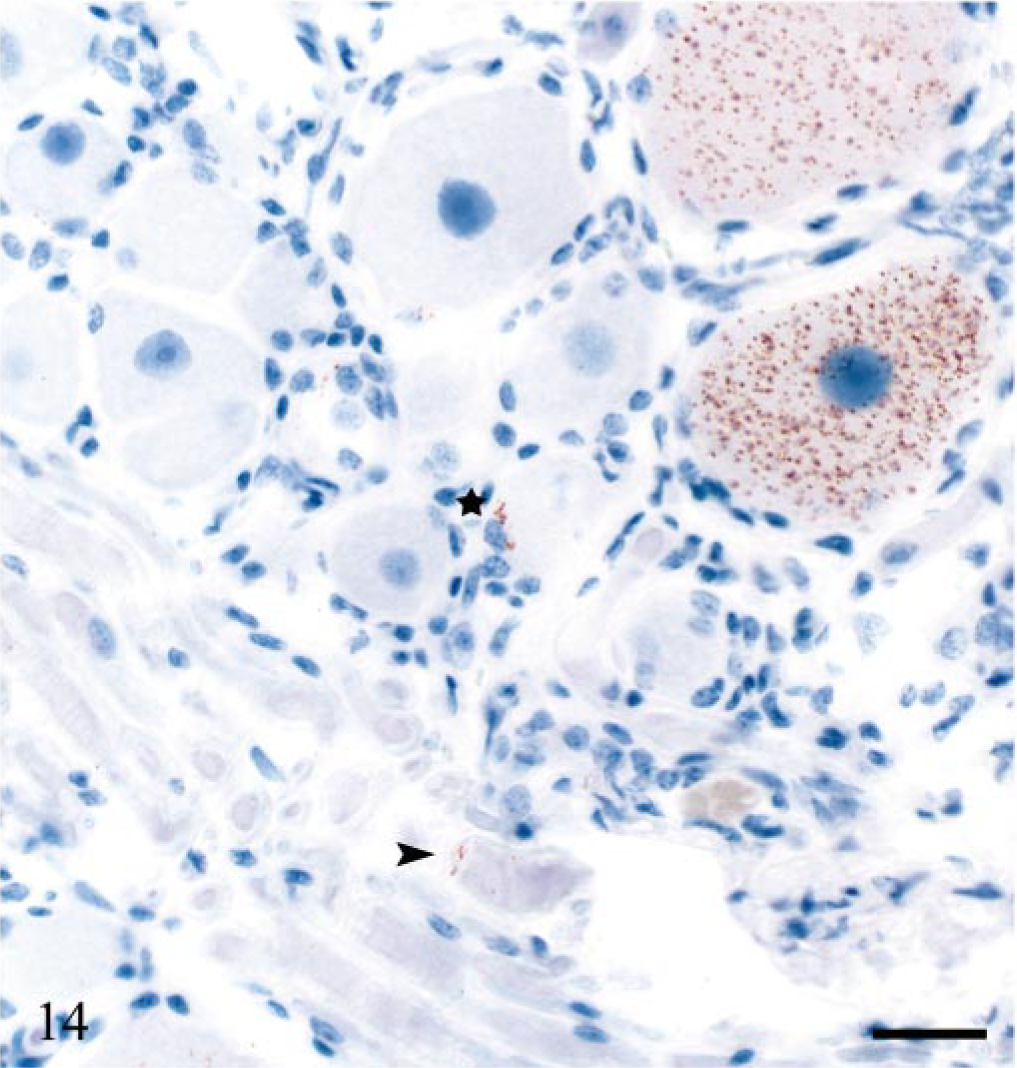
Dorsal root ganglion of cervical segment 2; sheep E20, 743 dpi, PrP genotype VRQ/VRQ. Intracellular PrPSc accumulation of neurons, sparse periaxonal labeling (arrowhead) and granular PrPSc of satellite cells (star). PrP antibody F89/160.1.5, Envision™ + kit, Mayer's hematoxylin counterstain. Bar = 27 μm.
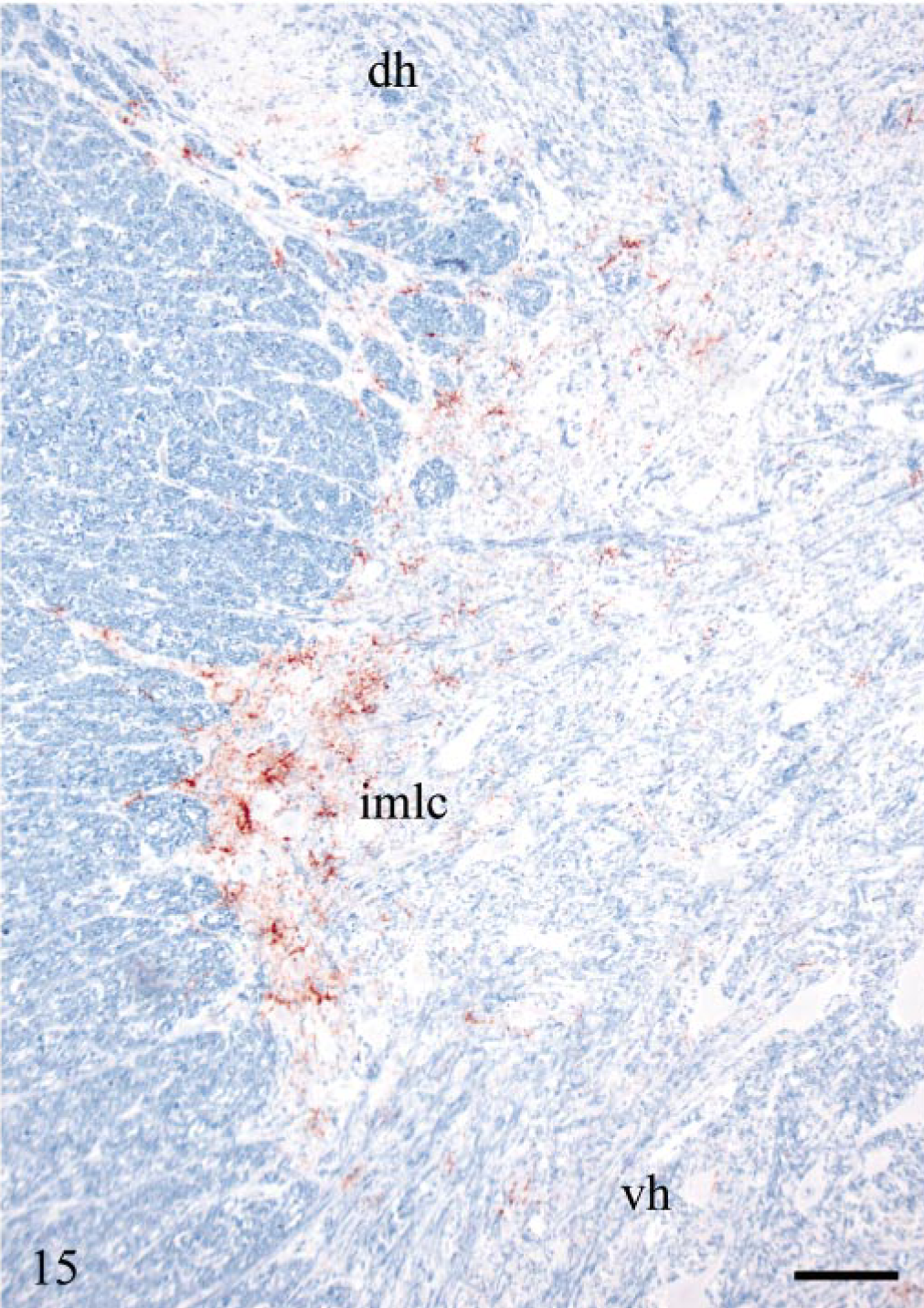
Lumbar segment 2 of the spinal cord; sheep E13, 442 dpi, PrP genotype VRQ/VRQ. Mainly stellate PrPSc labeling in the intermediolateral column (imlc) and less PrPSc in adjacent white matter. The labeling extends into the lateral margin of dorsal (dh) and ventral horns (vh). PrP antibody F89/160.1.5, Envision™ + kit, Mayer's hematoxylin counterstain. Bar = 110 μm.
Naturally infected sheep
The distribution of PrPSc accumulation in the brain, spinal cord, and DRG is illustrated in Fig. 16. All clinically affected sheep had marked PrPSc accumulation in the medulla oblongata, and the immunolabeling generally decreased in the rostral direction. Multifocal areas of PrPSc accumulation were observed in the cerebellar and cerebral cortices. Well-defined PrPSc patterns were found in these sheep, 20 and the immunolabeling was usually associated with neurodegenerative changes. All clinically affected sheep revealed vascular amyloid-like PrPSc labeling, varying in size and distribution, but restricted to the thalamus, the cerebral cortex with adjacent white matter, and the cerebral meninges. Plaques were both mural and perivascular, and they affected capillaries and larger vessels. The surrounding neuropil, glial cells, and neurons of larger lesions were also labeled. In the spinal cord of the clinically affected sheep, there was moderate or marked labeling in the dorsal horns, as well as generally moderate PrPSc accumulation around the central canal and in the ventral horns. Marked PrPSc deposits of the IMLC and around the central canal of segments T6 to T13 were observed in sheep N28. This sheep showed less labeling of the ventral horn in the cervical and first thoracic segments, and all DRG examined were positive. Subclinically affected sheep N23 had marked labeling in the medulla oblongata, moderate amounts of PrPSc in the cerebellum, the midbrain, and the thalamus and had sparse deposits in the internal capsule. There were PrPSc deposits in cervical, thoracic, and lumbar segments in the spinal cord of sheep N23. PrPSc accumulation of the subclinically affected sheep N24 was confined to the DMNV, a few other sites in the obex, and to the IMLC and ventral horn of the last thoracic spinal cord segments. Immunolabeling of the DMNV was moderate, whereas the labeling elsewhere in the obex and in the spinal cord was sparse. Sheep N22 was PrPSc-negative in the CNS and DRG.
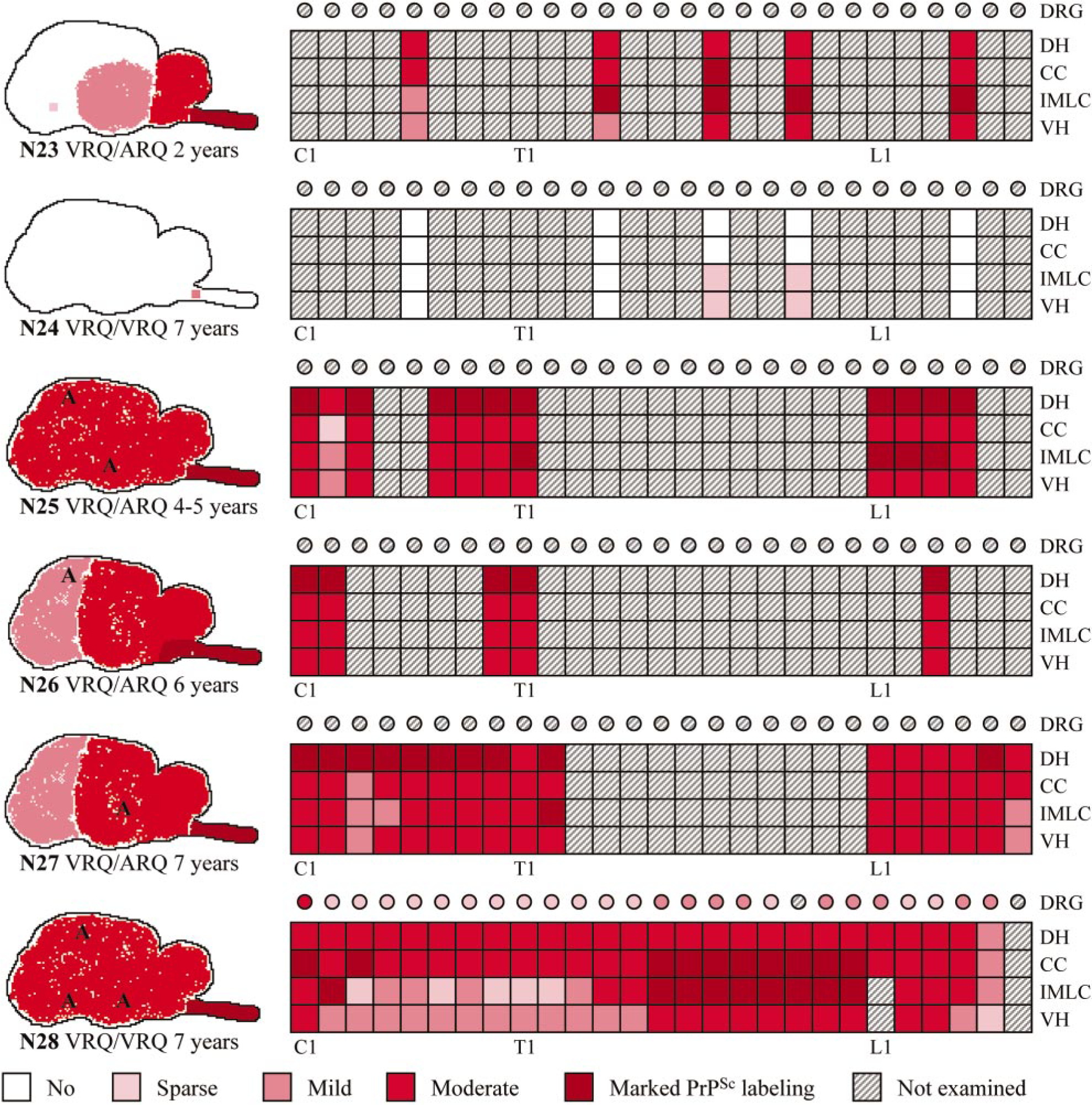
Diagrammatic representation of the distribution and severity of PrPSc in brain, spinal cord, and DRG of naturally infected sheep. C = cervical segments; T = thoracic segments; L = lumbar segments; DRG = dorsal root ganglion; DH = dorsal horn; CC = central canal and surrounding tissue; IMLC = intermediolateral column; VH = ventral horn; A = amyloid; dpi = days postinoculation.
Western blot and ELISA in experimentally infected sheep
The terminally ill VRQ/VRQ sheep inoculated with ARQ/ARR material was PrPSc-positive in all organs investigated, when analyzed using both western blot and ELISA. The VRQ/ARR, AHQ/AHQ, and ARQ/ARR sheep were PrPSc-negative in all tissues when using each of the two methods. The western blot and ELISA results correlated with the immunohistochemical detection for all animals.
Discussion
In this study, we examined sheep having different PrP genotypes, either inoculated by using a stomach tube or naturally infected with scrapie, for the presence of PrPSc in peripheral tissues and in the CNS. In the experimentally infected animals, we found differences between VRQ/VRQ and VRQ/ARQ sheep in their respective propagation of PrPSc. Accumulation of PrPSc in peripheral tissues was markedly lower, or even absent in some tissues, in the inoculated sheep carrying the A136 allele.
Mucosa-associated lymphoid tissue (MALT) is most probably involved in the initial uptake of orally ingested PrPSc. 1, 5, 26, 37 Judging from the limited PrPSc labeling of the mucosa-associated follicles, VRQ/ARQ sheep may be less efficient in their uptake of the infectious prions from the gastrointestinal tract or have lower accumulation of PrPSc in the MALT (or both), as compared with homozygous V136 sheep. There was also less PrPSc in other lymphoid and peripheral nervous tissues in the inoculated VRQ/ARQ heterozygotes as compared with the VRQ/VRQ sheep at all points in time that were studied. We therefore propose that the dissemination of PrPSc from the MALT to other tissues is slower or less effective (or both) in A136-allele–bearing animals. Transfer of PrPSc between FDCs and nerves has been demonstrated in the spleen of transgenic mice, 40 and the presence of nerves within lymphoid follicles in the gut has been demonstrated in sheep. 24 The postnatal expansion of the IPP in lambs reaches its peak development at approximately 3 months of age, after which the lymphoid tissue gradually starts to involute. 42 In contradistinction to VRQ/ARQ animals, scrapie-infected VRQ/VRQ lambs at this age may have abundant PrPSc in the IPP, facilitating a transfer of PrPSc from lymphoid cells to nerves.
PrPSc accumulated in the MALT may be disseminated through nerves, lymph or blood, or through a combination of these pathways. Afferent and efferent fibers of both the splanchnic nerves (sympathetic fibers) and the vagus nerve (parasympathetic fibers) are connected to the nerve plexus in the gut wall. In sheep, the efferent fibers of the major and minor splanchnic nerves arise from neurons in the IMLC of the sixth thoracic to the second lumbar spinal cord segments. 39 In this study, evidence for dissemination along autonomic nervous pathways was the sequential appearance of PrPSc in the ENS, the CMGC, the nodose ganglion, and in the CNS in the DMNV and the IMLC of the last thoracic spinal cord segments. Earlier studies have shown such a propagation both in sheep 49 and in rodents. 38 In the ENS, PrPSc labeling was mainly associated with satellite cells. Only satellite cells were found to be PrPSc-positive in the least affected CMGC of the inoculated VRQ/ARQ sheep. Furthermore, these cells outnumbered the positive neurons also in other sheep with PrPSc deposits in this site. This novel finding may indicate that PrPSc targets these cells earlier than the neurons in the peripheral nervous system. In the inoculated sheep that was terminally ill, PrPSc labeling was found in the periphery of nerve fibers associated with the CMGC, some DRG, and dorsal root fibers of the spinal cord. In the peripheral nervous system, nerve fibers are ensheathed in a series of Schwann cells. These cells share their origin and major cytologic characteristics with satellite cells that surround the neuronal perikarya in peripheral ganglia. 34 Labeling of neurons in ganglia, satellite cells, and along nerve fibers has been reported in clinically diseased sheep and rodents. 3, 22 Schwann cells of mice express PrPC, and replication of PrPSc has been shown in a peripheral glial cell line. 17 A recent study in transgenic mice overexpressing tau showed that prion neuroinvasion was not impaired, although the fast axonal transport was defective. 35 This finding suggests an alternative transport mechanism for PrPSc associated with nerves. In this study, early labeling of satellite cells, along with the weak, but evident PrPSc deposits associated with nerves in the later stages of the disease, may indicate that PrPSc is transported periaxonally through Schwann cells.
There was an early accumulation of PrPSc in thoracic DRG in two VRQ/VRQ sheep, both having limited immunolabeling in the CNS. This pattern of accumulation could indicate an early anterograde dissemination of PrPSc out of the CNS along an afferent nervous pathway. Alternatively, it could indicate the accumulation of PrPSc in these ganglia during inward spreading to the CNS. In a study of PrPSc dissemination in naturally infected VRQ/VRQ sheep, PrPSc was found in DRG at a later point in time and after a more widespread dissemination in the CNS. 49
In this study, we demonstrated that there were PrPSc-positive cells in efferent lymph vessels and lymphoid sinuses, of different lymph nodes, thereby, indicating spreading through the lymph. This mode of spreading is likely to take place between the MALT and regional lymph nodes such as the RPLN and the DJLN. Indeed, the early presence of PrPSc in the RPLN and the DJLN, in addition to the tonsil and the IPP, was demonstrated in lambs as early as 5 weeks after infection, using the same experimental procedure as described in this study. 25 Lower accumulation of PrPSc in the spleen and the SCLN as compared with the RPLN and the DJLN could be because of the close association with the MALT and the probable transport of PrPSc through lymph to the latter tissues. Dissemination of PrPSc by blood to the spleen and lymph nodes not associated with the digestive tract was suggested in a sequential study of scrapie in mice. 37 It is also likely in our sheep model.
Focal or multifocal vascular PrPSc-positive amyloid-like plaques located at several places in the brain, but never in the brainstem, were found in both inoculated and naturally infected sheep. The amyloid-like PrPSc deposits appeared as isolated changes that were not associated with areas having marked immunolabeling and thus indicate a possible additional spread of PrPSc from the periphery to the CNS through blood. Vascular PrPSc-positive amyloid in the same locations has been described previously in sheep. These authors suggested that PrPSc might accumulate in and later be released from endothelial cells. 30 In a recent study, infectivity was detected in blood from both scrapie-infected and BSE-infected sheep. 28 New research has shown that VRQ/VRQ and VRQ/ARQ animals have higher expression levels of PrPC on blood monocytes than sheep with less susceptible PrP genotypes. This may indicate that blood monocytes play a role in the propagation of the disease and that the difference in PrPC expression levels is significant in governing susceptibility. 15
In the placenta, PrPSc was detected in two morphologically different cell populations, interpreted as maternal epithelium and fetal trophoblasts, from a VRQ/VRQ sheep with a VRQ/ARQ fetus and from a VRQ/ARQ ewe, where the PrP genotypes of the fetuses were unknown. Placentas from susceptible sheep with at least one less susceptible fetus were negative. PrPSc-labeling patterns described are in accordance with the findings of Tuo et al., 46 who also used one of the same PrP antibodies as that used in this study. These results are in contrast to those of Andréoletti et al., 2 who reported PrPSc accumulation predominately in the fetal trophoblasts.
In this study, the incubation time was shorter in VRQ/VRQ sheep as compared with that in VRQ/ARQ sheep. This observation is in agreement with previous studies in both natural 9, 29 and experimental disease. 27 Generally, there was a sparse amount of PrPSc in the CNS of experimentally infected sheep with early clinical signs. However, in all except one of the VRQ/ARQ sheep, there was no detectable PrPSc nor were there degenerative lesions in the CNS. In contrast, one of the naturally infected VRQ/ARQ sheep had considerable amounts of PrPSc and vacuolation in both the brain and spinal cord, although showing no clinical signs. This sheep was clinically examined and monitored for 2 months in our isolation facility before an autopsy was performed. Therefore, there seems to be no clear association between clinical signs, neurode-generative changes, and the amount of detectable PrPSc in the CNS.
Previously, we showed that a VRQ/ARR lamb had PrPSc deposits in a few IPP follicles. 16 This finding indicates that sheep with this PrP genotype have a lymphoreticular phase, although markedly less than that present in VRQ/VRQ animals. VRQ/ARR sheep subcutaneously injected with the scrapie isolate SSBP/1 also have shown PrPSc in lymphoid tissue. 27 In this study, we were unable to detect PrPSc in any tissue sample from experimentally infected sheep with the PrP genotypes VRQ/ARR, ARQ/ARR, and AHQ/AHQ by using immunohistochemistry, ELISA, and western blot. However, it should be noted that these animals were inoculated with heterologous VRQ/VRQ brain material. In contrast, an ARQ/ARR inoculum was successfully transmitted to a VRQ/VRQ sheep, which developed terminal disease. This result illustrates the significance of the recipient PrP genotype. It also shows that heterologous transmissions are possible when the recipient has a susceptible PrP genotype. Possibly, an inoculum of the same PrP genotype as the recipient is important to achieve an efficient conversion of PrPC to PrPSc in recipient sheep of less susceptible PrP genotypes. Our homologous ARQ/ARR inoculation was unsuccessful, and this may indicate that the time needed to detect conversion and dissemination of PrPSc in less susceptible sheep may exceed the time frame of this experiment. In a cell free in vitro conversion system, ovine PrPSc(VQ/VQ) converted homologous PrPC(VQ) more effectively than heterogenous PrPC(AQ). In contrast, PrPSc(AQ/AQ) induced conversion of PrPC(AQ) and PrPC(VQ) with almost the same efficiency. 8 Because the amount of infectivity in each inoculum and the strain(s) of agent in this study are not known, we cannot exclude the fact that these factors have influenced the results.
The propagation of PrPSc in susceptible sheep in our experimental model was comparable with natural V136 homozygous cases reported by van Keulen et al. 49 and with the naturally infected sheep with scrapie included in this study. Thus, the experimental procedure of infecting lambs between the ages of 47 and 62 days using a stomach tube appears to mimic natural infection. In this study, we argue for a parallel dissemination of PrPSc in nerves, lymph, and blood, and we show that satellite cells are early targets for PrPSc in the peripheral nervous system of ovine scrapie. There were also apparent differences in the propagation of PrPSc, where VRQ/ARQ sheep showed a more limited distribution of PrPSc in peripheral tissues than V136 homozygotes in the early stages of disease.
Footnotes
Acknowledgements
We are grateful to Ingunn K. Wiig, Esben Seljeskog, Inger Rudshaug, and Laila Aune for preparing the histologic slides, Eli Brundtland and Truls Moum for doing the PrP genotyping and to Sandrine Melo for performing the western blots. This study was supported by grant 119688/110 funded by the Research Council of Norway.
