Abstract
All sheep older than 1 year of age from a flock of the Rygja breed in which clinical scrapie was detected for the first time in two animals (4%) were examined for accumulation of pathogenic prion protein (PrPSc) by immunohistochemistry in the obex, the cerebellum, and the medial retrophayngeal lymph node. In addition, six lambs, 2–3 months old, all offspring of PrPSc-positive dams, were examined for PrPSc in the ileal Peyers' patch (IPP), the distal jejunal lymph node, the spleen, and the medial retropharyngeal lymph node (RPLN). In this flock, 35% (17/48) of the adult sheep showed accumulation of PrPSc, an eightfold increase compared with clinical disease. All positives carried susceptible PrP genotypes. Three sheep had deposits of PrPSc in the RPLN and not in the brain, suggesting that this organ, easily accessible at slaughter, is suitable for screening purposes. Two 7-year-old clinically healthy homozygous V136Q171 ewes showed sparse immunostaining in the central nervous system and may have been infected as adults. Further, two littermates, 86-days-old, showed PrPSc in the IPP. Interestingly, one of these lambs had the intermediate susceptible PrP genotype, VA136QR171. In addition to early immunolabeling in the dorsal motor nucleus of the vagal nerve, a few of the sheep had early involvement of the cerebellum. In fact, a 2-year-old sheep had sparse deposits of PrPSc in the cerebellum only. Because experimental bovine spongiform encephalopathy (BSE) in sheep seems to behave in a similar manner as natural scrapie, these results, particularly regarding spread of infectivity, may have implications for the handling of BSE should it be diagnosed in sheep.
Scrapie is a transmissible spongiform encephalopathy (TSE) in sheep and goats. At the molecular level the TSEs, or prion diseases, are characterized by accumulation of the pathogenic prion protein (PrPSc), a modified form of the host-encoded prion protein (PrPC). 8 Ultimately, scrapie is a neurodegenerative disease with vacuolation of neurons and neuropil, neuronal loss, and gliosis. 12 According to the prion hypothesis, the transmissible agent of all prion diseases, the prion, is composed largely, if not solely, of PrPSc. 39 However, verified disease has also been described without detection of PrPSc, 31 or with detection of very small amounts of it. 34 The normal form of the prion protein, PrPC, is found in a wide range of tissues, 6 and expression of PrPC in peripheral nervous and lymphoid tissue seems to be a prerequisite for transfer of infectivity and spread of the disease to the central nervous system (CNS). 7 , 40
In natural scrapie the gastrointestinal tract is considered to be the major route of infection. 17 , 38 The earliest accumulations of PrPSc are found in the ileal Peyer's patch (IPP), as well as in the draining mesenteric lymph node, 1 and this gut-associated lymphoid tissue is a probable port of entry for the scrapie agent in sheep. 20 Accumulation of PrPSc in the enteric nervous tissue has been reported to appear later but still in an early phase of the disease. 1 , 50 PrPSc also aggregates in other lymphoid tissues such as spleen, tonsils, and retropharyngeal lymph nodes in sheep with both clinical and nonclinical disease, and the primary sites of replication of the scrapie agent are likely to be the tissues mentioned. 1 , 25 , 26 , 44 , 48
Transport of prion infectivity along the splanchnic nerves and the vagal nerve has been indicated in several studies. 3 , 28 , 29 , 35 , 51 The earliest traces of PrPSc in the brain have been found in the dorsal motor nucleus of the vagal nerve (DMNV) when naturally infected lambs are 9 months old, and at this stage it was associated with few morphologic changes. 1 The scrapie agent seems to reach the spinal cord at approximately the same time, because deposits were found in the DMNV and the intermediolateral column of the thoracic segments T8–T10 in naturally infected lambs at the age of 10 months in another study. 51
Polymorphisms in the PrP gene at codons 136, 154, and 171 are strongly correlated to susceptibility and disease development in scrapie. 14 In several breeds, such as Cheviot, Romanov, or Rygja, the presence of valine (V) at codon 136 enhances susceptibility, 24 , 30 , 46 and a change from glutamine (Q) to arginine (R) at codon 171 renders the animal more resistant to the disease irrespective of breed. 15
Morphologic changes and accumulation of PrPSc in the brain of sheep with clinical scrapie have been described. 16 , 36 , 42 , 49 , 53 Recently, however, focus has turned to studies of PrPSc accumulations and early pathologic events occurring in the clinically healthy animal. Such studies are of great importance, not only for understanding the pathogenesis of sheep scrapie, but also for targeting tissues for screening purposes to obtain the earliest possible diagnosis. Because scrapie behaves differently between breeds and according to disease-linked polymorphisms in the PrP gene, we have undertaken a comprehensive immunohistochemical and morphologic study of a flock of the Rygja breed in which scrapie was diagnosed. We have previously characterized the PrP genetics of this breed in relation to natural scrapie, 46 and we are pursuing studies of this breed by using a model of experimental scrapie. 20 Lately, there have been a few reports on preclinical PrPSc accumulation and early lesions in the CNS in sheep with natural infection, 18 , 27 , 42 and a low level of preclinical infection and scrapie-like pathology in sheep flocks has been described. 5 , 33 This is the first report describing whole-flock distribution of PrPSc accumulations in nervous and lymphoid tissues, and scrapielike pathology in sheep with various PrP genotypes during an outbreak of natural scrapie in a Rygja flock. In Norway the whole flock is culled when a scrapie diagnosis is confirmed, offering favorable conditions for such investigations. As experimental bovine spongiform encephalopathy (BSE) in sheep seems to behave very much like natural scrapie, 11 , 26 the results, particularly regarding spread of infectivity, also may have implications for the handling of BSE should it be diagnosed in sheep.
Materials and Methods
Sheep
In 1999, scrapie was diagnosed in a pair of twin sheep (Nos. 1 and 2) in a flock of 49 Rygja sheep, both showing typical clinical signs of scrapie. 47 Shortly after, the whole flock was culled, except the dam (sheep No. 3) of the twins and her granddaughter (sheep No. 7), and six 2- to 3-month-old lambs, all offspring of the aforementioned ewes. These animals were brought to an isolation facility for observation and subsequent autopsy. One ewe died of unknown cause between the time of scrapie diagnosis and the culling of the flock and was not examined. The table of descent of the diseased sheep is shown in Fig. 1.

Descent of diseased sheep. Only animals examined by histopathology and IHC, sheep with suspect clinical signs, and dams of PrPSc-positive sheep are included. S = sheep; L = lamb. The number in parentheses indicates age. PrP genotypes are given if known. Bold text: PrPSc positive in the brain and the lymphoid tissue. Italic text: No obtained material, no history of disease. ∗Clinical cases. †Dubious clinic. ‡PrPSc positive in the RPLN only. §PrPSc positive in the IPP only. ‖Sheep investigated with negative result. #Sheep with suspect clinical signs but no obtained material.
The flock was established in 1992, when 20 ewes were recruited from two different farms. In 1993, 10 additional sheep were purchased from one of these farmers. The flock formerly held at the premises and all recruited animals were classified as scrapie contacts before 1999, according to a national scrapie surveillance program.
Sampling of material
From 48 adult sheep, the medulla oblongata, the cerebellum, and the medial retropharyngeal lymph node (RPLN) were collected. In addition, ileum was taken from the two ewes received in the isolation facility. From the six lambs, ileum with the IPP, the distal jejunal lymph node (DJLN), the spleen, and the RPLN were taken for the study. Tissue samples for histology and immunohistochemistry (IHC) were immersion fixed in 4% formaldehyde immediately after death and were subsequently dehydrated and paraffin embedded. For PrP genotyping, blood samples were taken.
PrP genotyping
PrP genotyping was performed by direct sequencing as described by Tranulis et al. 46
Immunohistochemistry
Tissue sections were mounted on 3-aminoalkyltriethoxysilane–coated glass slides and dried for 48 hours at 37 C. IHC was performed using the polyclonal anti-PrP antibodies R505 and R521 13 (kindly provided by J. Langeveld), the monoclonal anti-PrP antibodies L42 19 (kindly provided by M. Groschup), and F89/160.1.5 37 (Table 1). On the sections stained with R505 and R521, a streptavidin–biotin–peroxidase IHC was performed as described by van Keulen et al. 49 After rehydration, tissue sections immunolabeled with F89/160.1.5 and L42 were immersed in formic acid and autoclaved at 121 C in 0.01 M citric acid (pH 6.1) or distilled water. Endogenous peroxidase was blocked by use of 0.3% H2O2 in methanol for 30 minutes at room temperature. Tissue sections were then incubated with 20% normal goat serum in phosphate-buffered saline (PBS), pH 7.2, for 20 minutes, to block nonspecific binding sites. Proteinase K (Dako, S3020, California, USA) digestion for 2 minutes was included after the endogenous peroxidase–blocking step on tissue sections from the lambs, in addition to parallel runs without digestion. Sections were incubated with primary antibodies at room temperature for 15 minutes (F89/160.1.5, dilution 1:200) or overnight at 4 C (F89/160.1.5, dilution 1:2,500, and L42). Further steps were performed with the Envision™+ kit (Dako, K4005). Digital images of sections from the brain and the ileum were captured using a digital camera (Spot RT230-2 Slider, Diagnostic Instruments, Inc., Michigan, USA) mounted on a microscope (Leica DM RA/RXA, Leica Microsystems Wetzlar GmbH, Wetzlar, Germany).
Anti-PrP antibodies and tissues included in the study.
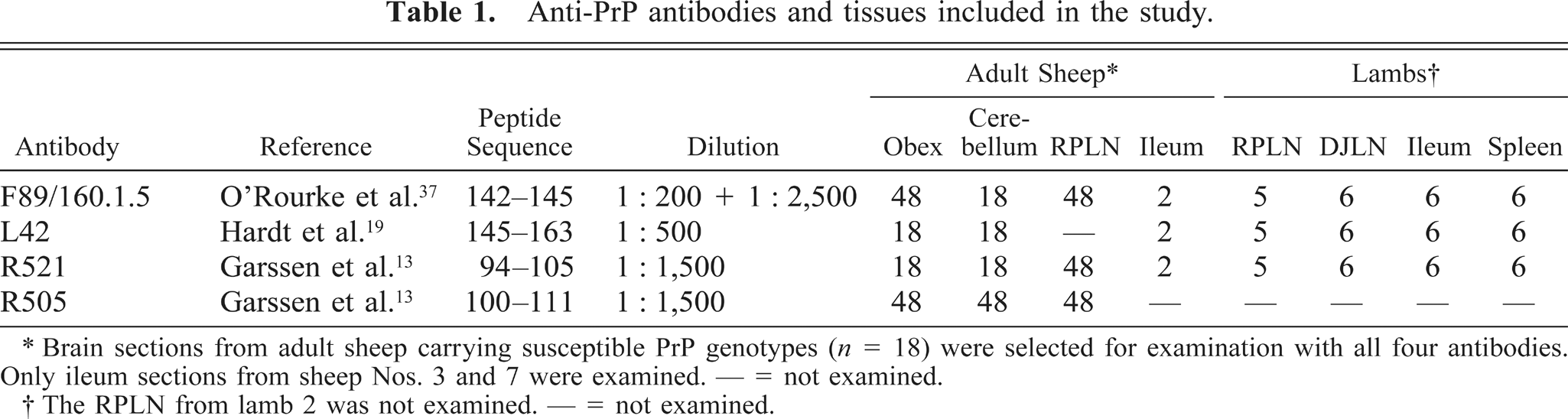
∗ Brain sections from adult sheep carrying susceptible PrP genotypes (
† The RPLN from lamb 2 was not examined. — = not examined.
Controls
All runs included at least one positive and one negative control section. The positive controls used were confirmed clinical scrapie cases. Negative control tissue was selected from lambs (lymphoid tissue) and sheep (brain) raised in scrapie-free flocks. To characterize nonspecific immunolabeling, PBS replaced the primary antibody on one section in each run.
Histopathology and lesion profiling in CNS
One section from the obex and the cerebellum was stained with hematoxylin and eosin. In the obex, nine areas (DMNV, Nucleus tractus solitarii [NTS], Nucleus n. hypoglossi, Nucleus cuneatus accessorius, Nucleus tractus spinalis n. trigemini [NTS-5], reticular formation, midline raphe, Nucleus ambiguus, and Nucleus olivaris) were examined. Each area was given a score for intraneuronal vacuolation, by counting the number of vacuolated neurons (score = number of vacuolated neurons), vacuolation in neuropil (0–4: score 0 representing six vacuoles in neuropil; score 1, 6–10 vacuoles; score 2, 11–20 vacuoles; score 3, 21–40 vacuoles; and score 4, > 40 vacuoles), and astrocytosis (graded 0–3: 0, nothing; 1, little; 2, moderate; 3, prominent). Caudal parts of the cerebellum were sampled. The whole cerebellum section and each of the nine areas in the obex were given a semiquantitative visual intensity score (0, negative; 1, little; 2, moderate; 3, widespread) of PrPSc deposition on serial sections.
Results
PrP genotypes
Scrapie susceptibility was graded basically as described by Dawson et al. 9 Altogether 18 (38%, mean age 2.7 years) of the adult sheep had highly susceptible, 15 animals (31%, mean age 3.7 years) had intermediate, and 15 (31%, mean age 3.7 years) had low-susceptibility PrP genotypes. Three lambs had PrP genotype VA136QQ171, one lamb had the genotype VA136QH171, and two lambs had the genotype VA136QR171. The distribution of PrP genotypes is shown in Fig. 2.
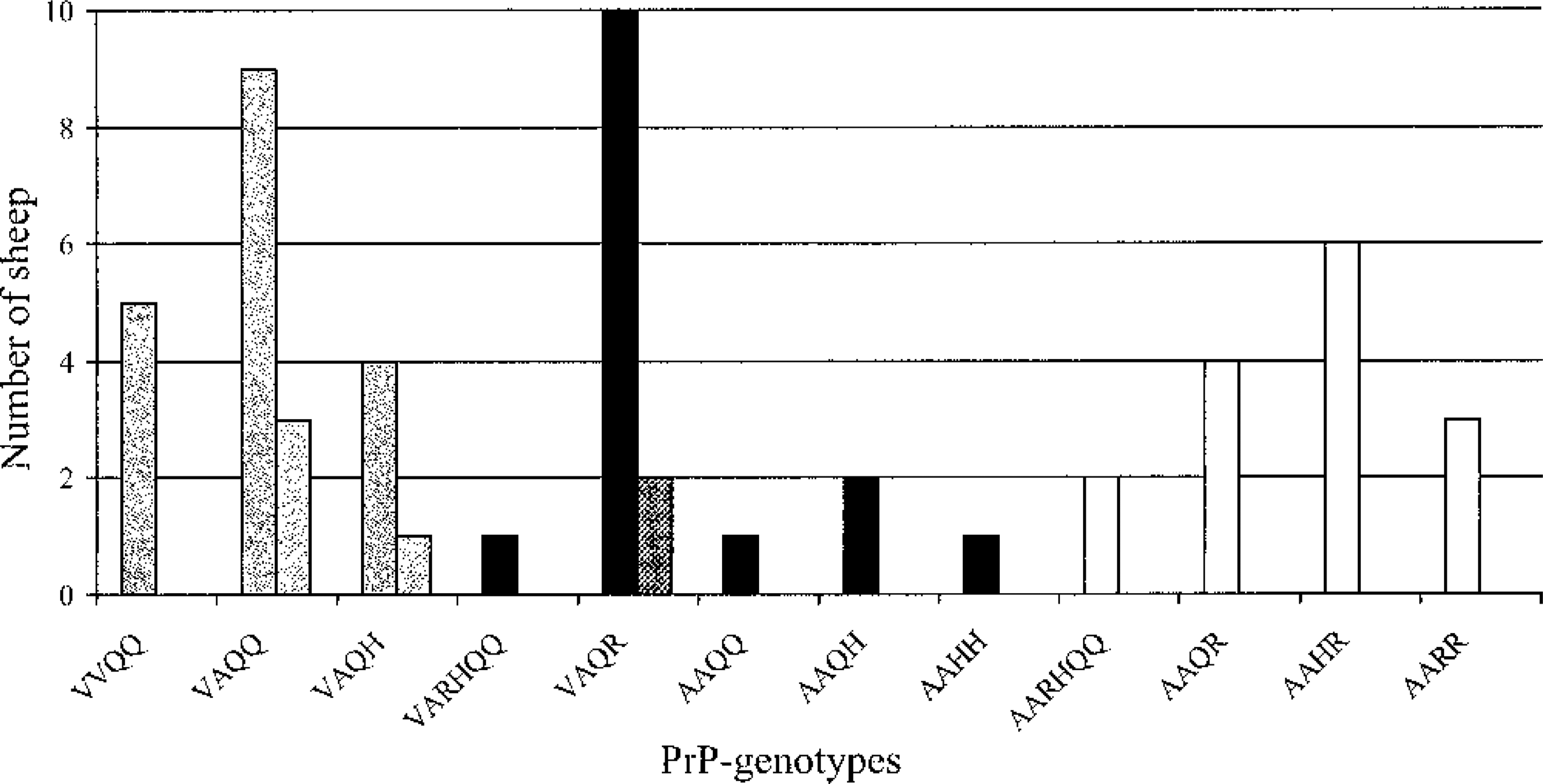
PrP genotypes in the flock. PrP-genotypes are given with alleles 136 and 171, and in addition allele 154 only when it differs from RR. Dotted bars show the PrP-genotypes of the lambs. Grey bars: Susceptible PrP-genotypes. Black bars: Intermediate susceptible PrP-genotypes. White bars: Low susceptibility PrP-genotypes.
Control slides
Lymphoid tissue
Without proteinase K in the IHC protocol, some of the anti-PrP antibodies produced weak immunolabeling of a few Goblet cells, lymphoid cells, and ganglion cells (Fig. 3) in ileum sections of the negative controls. When proteinase K was included in the protocol, the immunolabeling was completely removed from the negative controls, indicating successful destruction of residual PrPC or possible cross-reacting epitopes. Because proteinase K treatment was used to confirm disease specificity of labeling, the deposits in the positive controls and flock animals were designated as PrPSc. All four antibodies detected PrPSc in lymphoid tissues in the positive controls.

Enteric nervous system; negative control lamb. Weak and diffuse labeling of ganglion cells in Auerbach's plexus. Anti-PrP antibody L42, Envision™+ kit. Mayer's hematoxylin counterstain. Bar = 30 μm.
Brain
None of the anti-PrP antibodies used in this study produced immunolabeling in the negative controls. The positive control tissue showed reproducible patterns of PrPSc accumulations with all the anti-PrP sera used.
PrPSc in lymphoid tissue and enteric nervous system
Lambs
Two littermates carrying the PrP genotypes VA136QQ171 and VA136QR171 had deposits of PrPSc in the IPP (Table 2). Immunolabeling of several lymphoid follicles in each section was evident with intense and granular staining of macrophage-like cells (Figs. 4, 5). The L42 antibody without proteinase K pretreatment gave diffuse immunolabeling of some of the ganglion cells in ileum in all the six lambs, presumably representing PrPC and not disease specific PrPC (Fig. 6). The DJLN, the spleen, and the RPLN were negative in all six lambs.
Lambs: Individual data and immunohistochemical results.
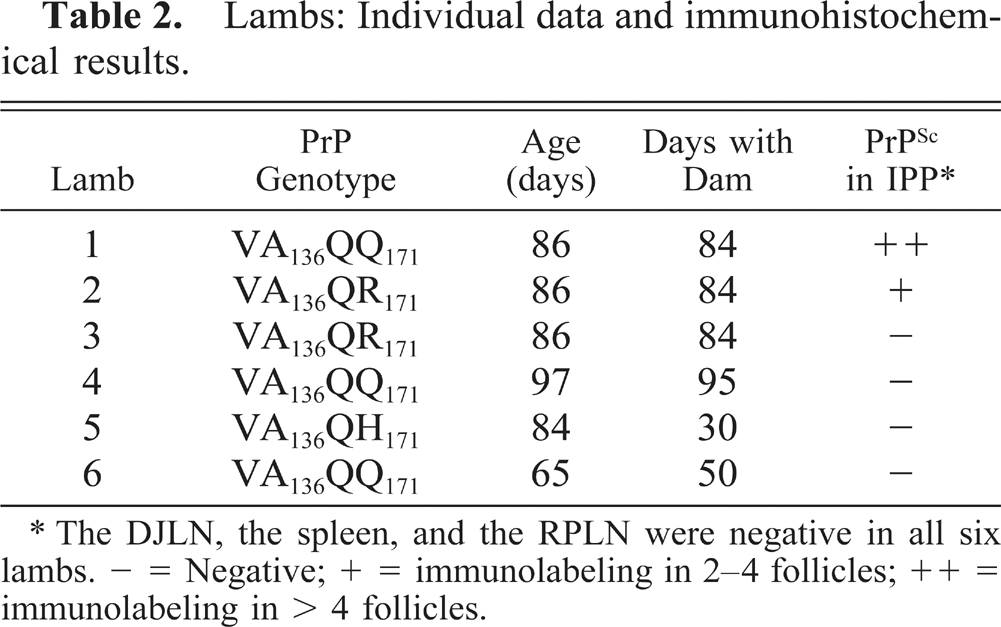
∗ The DJLN, the spleen, and the RPLN were negative in all six lambs. − = Negative; + = immunolabeling in 2–4 follicles; ++ = immunolabeling in > 4 follicles.
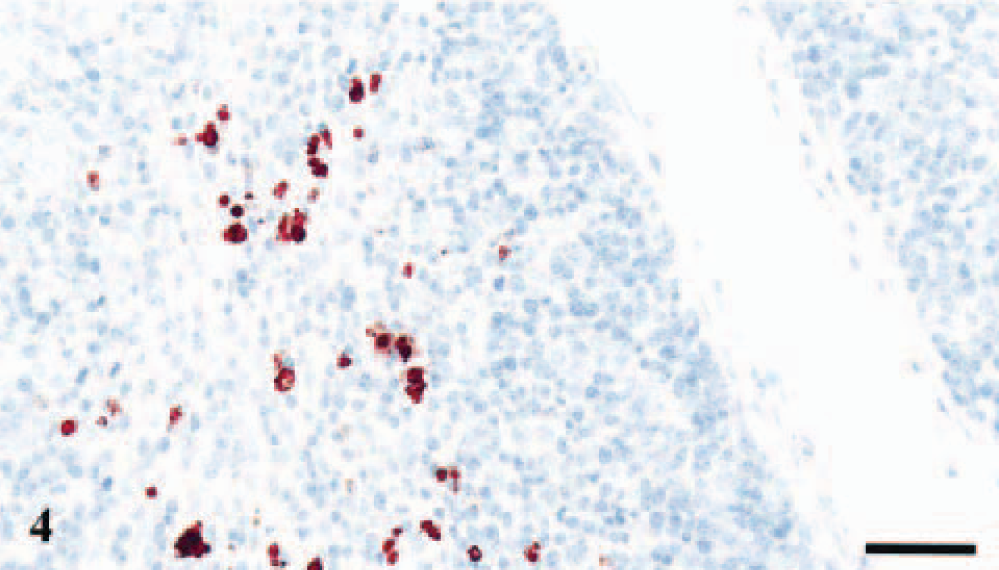
IPP; lamb No. 1, PrP genotype VA136QQ171. Strong PrPSc immunolabeling of tingible body macrophages in a lymphoid follicle. Anti-PrP antibody L42, Envision™+ kit. Mayer's hematoxylin counterstain. Bar = 40 μm.
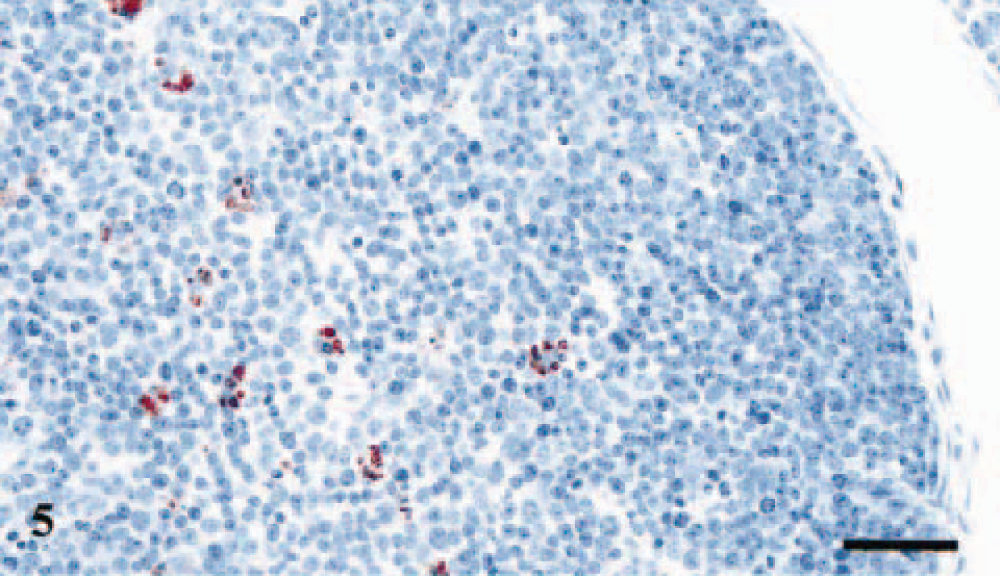
IPP; lamb No. 2, PrP genotype VA136QR171. Evident PrPSc immunolabeling of tingible body macrophages in a lymphoid follicle in the IPP. Anti-PrP antibody L42, Envision™+ kit. Mayer's hematoxylin counterstain. Bar = 40 μm.
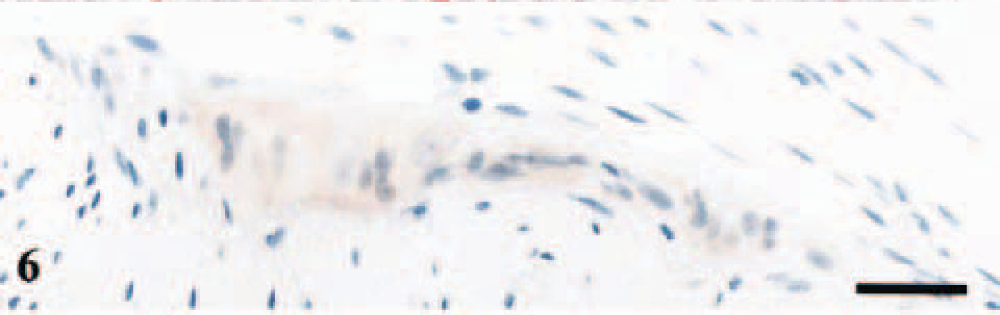
Enteric nervous system; lamb No. 3, PrP genotype VA136QR171. Diffuse labeling of ganglion cells in Auerbach's plexus. Anti-PrP antibody L42, Envision™+ kit. Mayer's hematoxylin counterstain. Bar = 30 μm.
Adult sheep
Altogether 17 (35%) of the adult sheep had PrPSc deposits in the RPLN; all positives had susceptible PrP genotypes. One sheep with a susceptible PrP genotype was found negative. Interestingly, two of the subclinical PrPSc-positive sheep with the highly susceptible VV136QQ171 PrP genotype were as old as 7 years. In three animals, 15 months (sheep No. 17) and 2 years old (sheep Nos. 15 and 16), PrPSc was found in the RPLN only. Ten of the PrPSc-positive sheep were from the same 2-year-old cohort.
In the RPLN, PrPSc was found mainly in the germinal centers; labeling involved presumed tingible macrophages and follicular dendritic cells. Single positive cells were occasionally found in the interfollicular and paracortical areas.
Sheep Nos. 3 and 7 showed granular immunolabeling of satellite and ganglion cells in Meissner's and Auerbach's plexa in ileum (Fig. 7), and in sheep No. 7 a residual ileal lymphoid follicle showed strong immunolabeling.
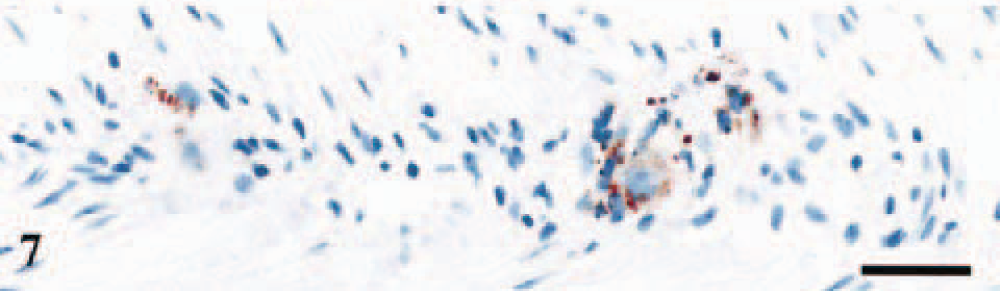
Enteric nervous system; sheep No. 3, PrP genotype VV136QQ171. Granular labeling of ganglion cells and satellite cells in Auerbach's plexus. Anti-PrP antibody F89/160.1.5, Envision™+ kit. Mayer's hematoxylin counterstain. Bar = 30 μm.
Detection of PrPSc in the brain
Of the 17 adult sheep that were positive for PrPSc in the RPLN, 14 also showed PrPSc deposits in the brain. All sheep with PrPSc deposits in the obex had immunolabeling in DMNV and NTS-5. One sheep (No. 10) was negative in the obex but had consistently punctuate deposits of PrPSc in the cerebellum when immunolabeled with the F89/160.1.5 and L42 antibodies. Six sheep, including the two clinical cases, were PrPSc positive in all the nine areas examined in the obex (Table 3).
Semiquantitative estimation of HP and IHC in the brain of all sheep with positive CNS findings.∗
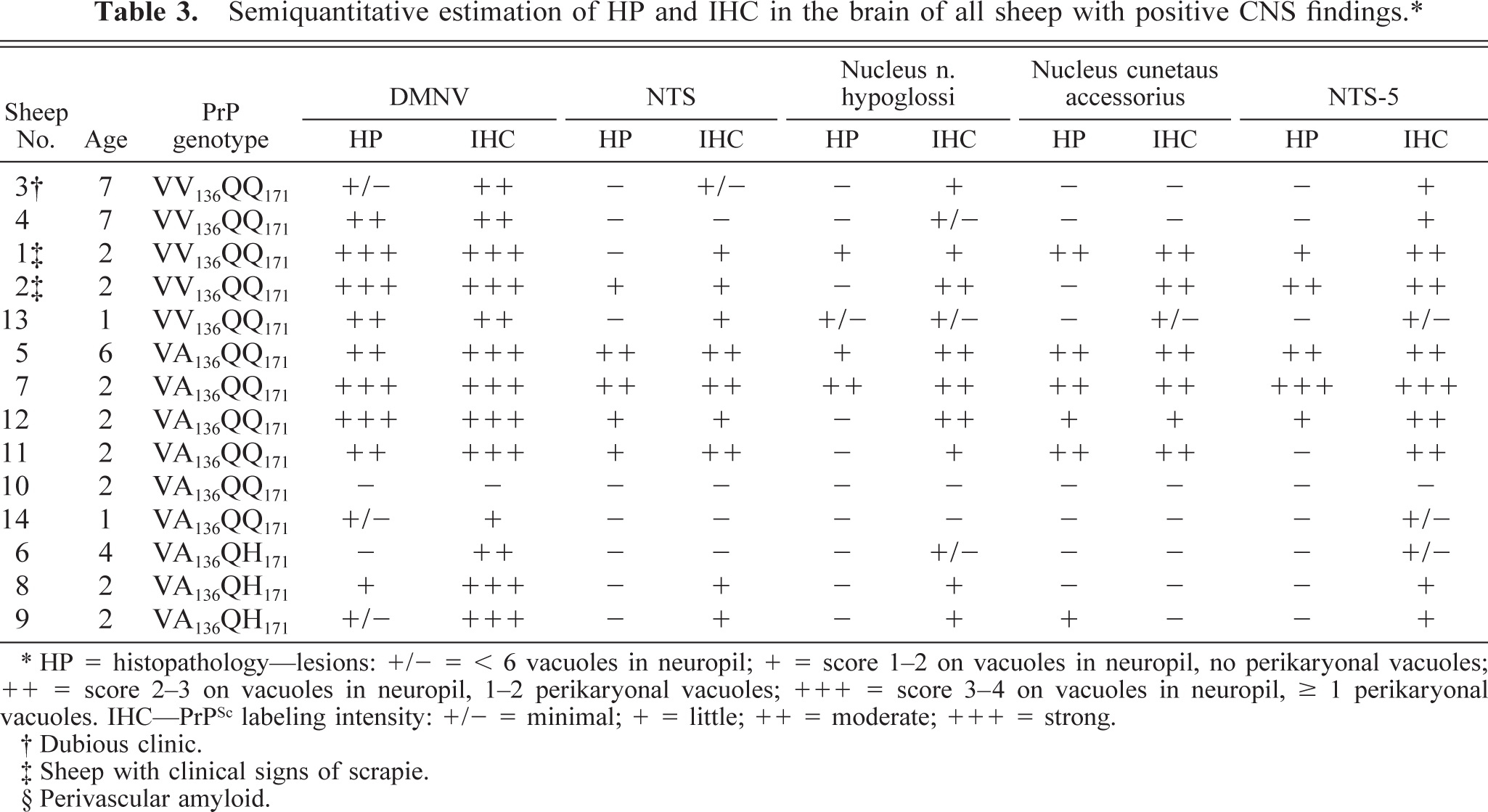
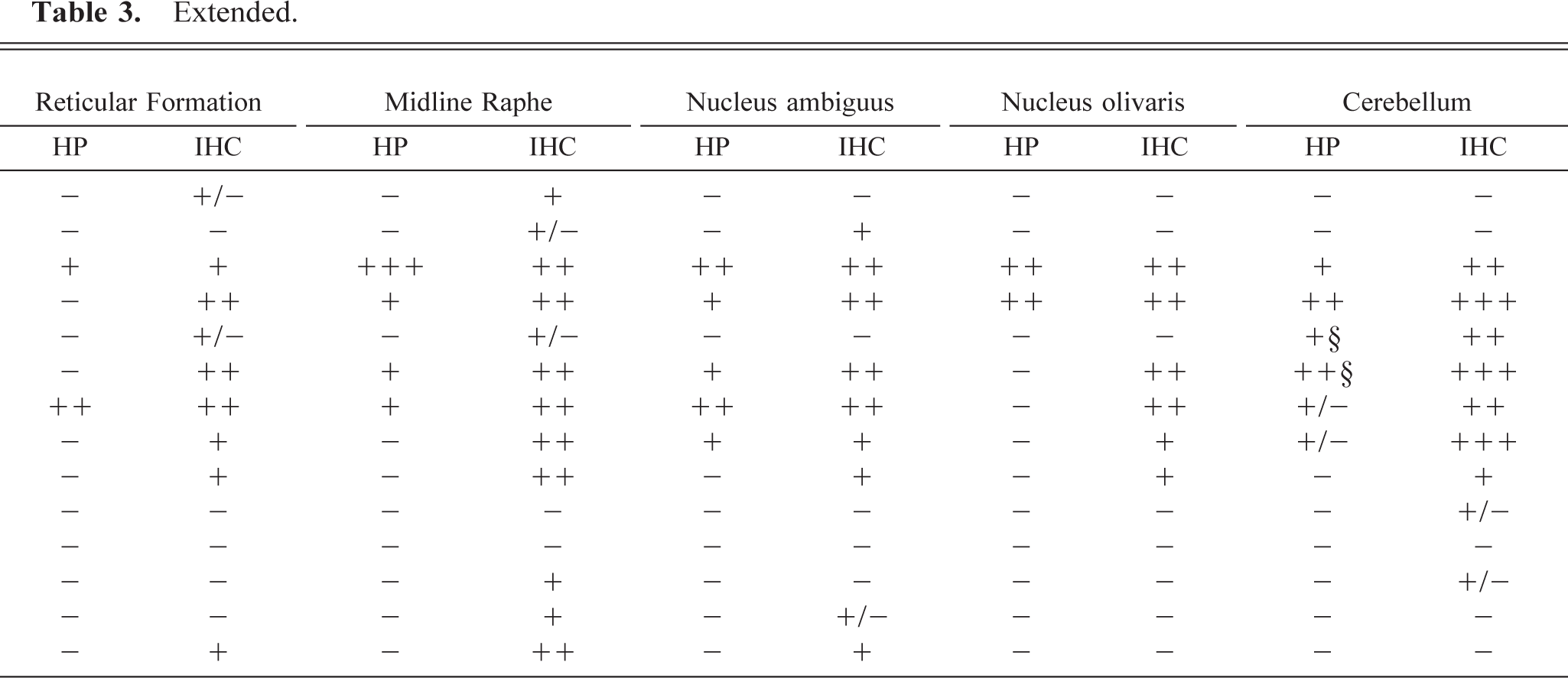
∗ HP = histopathology—lesions: +/− = < 6 vacuoles in neuropil; + = score 1–2 on vacuoles in neuropil, no perikaryonal vacuoles; ++ = score 2–3 on vacuoles in neuropil, 1–2 perikaryonal vacuoles; +++ = score 3–4 on vacuoles in neuropil, ≥ 1 perikaryonal vacuoles. IHC—PrPSc labeling intensity: +/− = minimal; + = little; ++ = moderate; +++ = strong.
† Dubious clinic.
‡ Sheep with clinical signs of scrapie.
§ Perivascular amyloid.
The PrPSc deposits were most intense in the DMNV. Sheep presumed to be in an early CNS phase showed deposits of PrPSc at the ventral border in this nucleus (Fig. 8). These animals had sparse immunolabeling in other areas in the obex. Other sheep, which were in a more advanced stage of disease, had a crescent distribution of staining, reaching dorsolaterally (Fig. 9). A third type of pattern was a diffuse distribution of staining comprising the whole nucleus (Figs. 10, 11). Sheep with the diffuse and the crescent type of staining had widespread PrPSc deposits in the obex. In sheep with ventral staining in DMNV, all but one had dorsal, sparse staining in N. n. hypoglossi and a low intensity of immunolabeling in midline raphe. The immunostaining in NTS-5 was confined mostly to the dorsolateral perimeter of the nucleus.
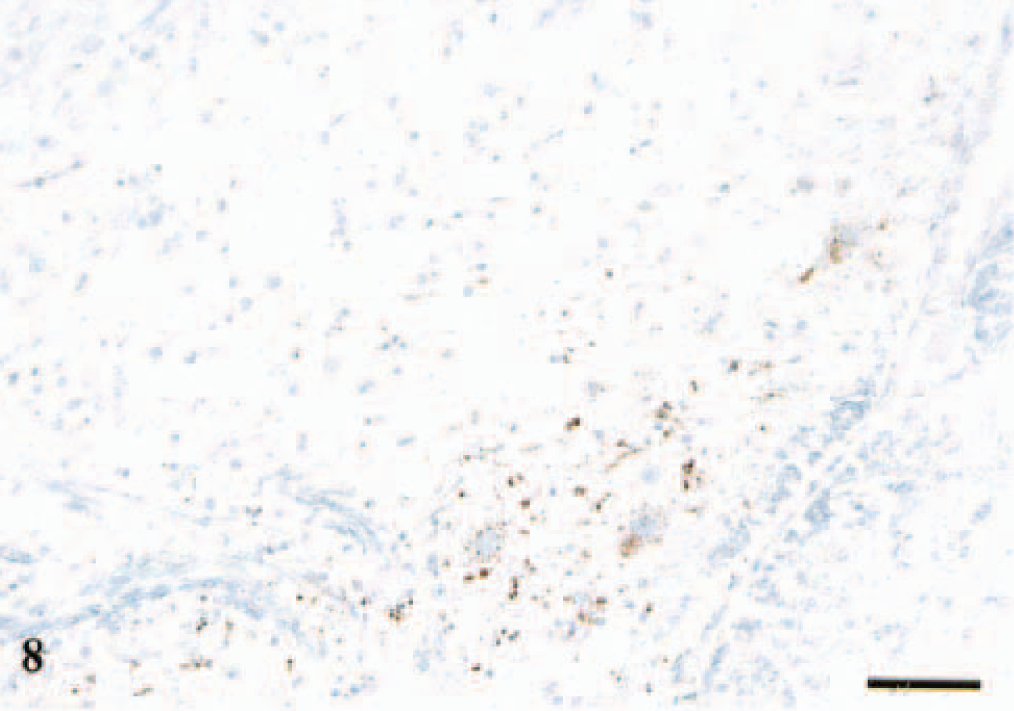
DMNV; sheep No. 3, PrP genotype VV136QQ171, 7 years old. Section shows intraneuronal, intraglial, and granular neuropil PrPSc labeling at the ventral margin of the nucleus. Anti-PrP antibody F89/160.1.5, Envision™+ kit. Mayer's hematoxylin counterstain. Bar = 80 μm.

DMNV; sheep No. 8, PrP genotype VA136QH171, 2 years old. Sections show a predominantly stellate and granular PrPSc labeling and some intraneuronal and intraglial (arrow) immunostaining in a crescent distribution. Anti-PrP antibody F89/160.1.5, Envision™+ kit. Mayer's hematoxylin counterstain. Bar = 80 μm.

DMNV; sheep No. 12, PrP genotype VA136QQ171, 2 years old. Section shows stellate (arrow) and neuropil PrPSc deposits and some intraneuronal immunostaining diffusely in the nucleus. Anti-PrP antibody F89/160.1.5, Envision™+ kit. Mayer's hematoxylin counterstain. Bar = 80 μm.
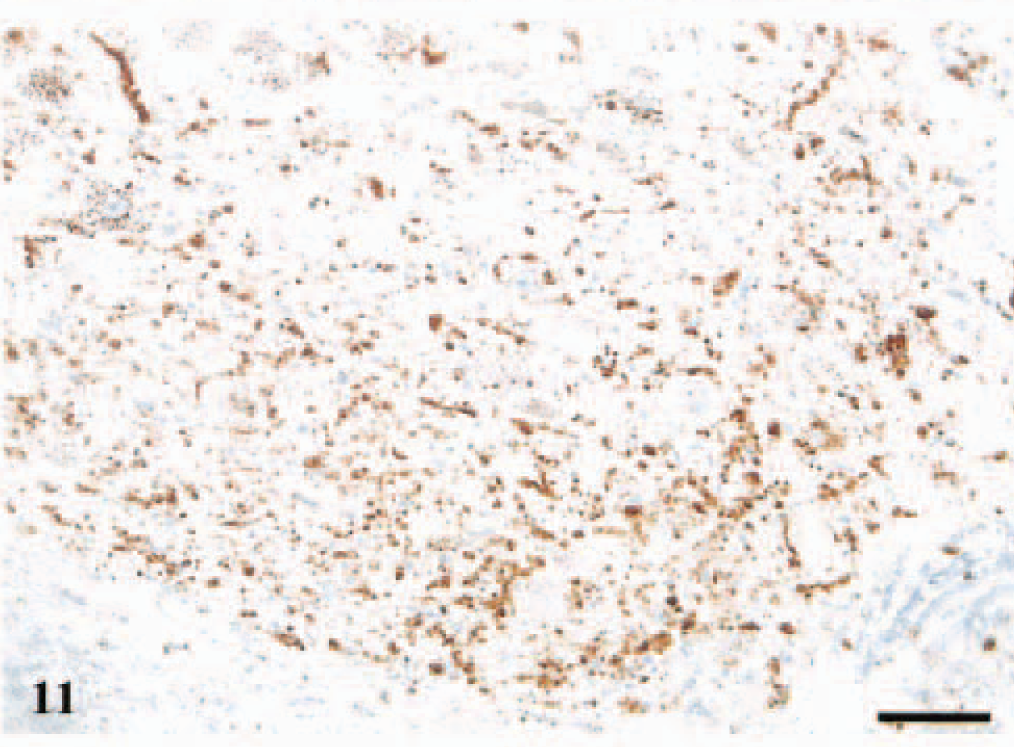
DMNV; sheep No. 2, PrP genotype VV136QQ171, 2 years old. Section shows a diffuse distribution of intraneuronal and intraglial granular PrPSc deposits and also granular deposits in the neuropil, linear and perineuronal immunostaining mainly ventrally in the nucleus. Anti-PrP antibody F89/160.1.5, Envision™+ kit. Mayer's hematoxylin counterstain. Bar = 80 μm.
PrPSc was associated with glial cells interspersed in neuropil, perivascularly and along the ependyme at the ventral margin of the fourth ventricle. Two glia-associated patterns were recognized: a stellate labeling (Fig. 10, arrow) and granular deposits near the glial nuclei (Fig. 9, arrow), the latter assumed to be intracytoplasmic. Both patterns were seen in all nine areas in the obex.
A number of different neuropil-labeling patterns were recognized. Granular deposits of PrPSc in the neuropil were regularly found in DMNV (Figs. 9–11), dorsally in NTS-5, in N. cuneatus accessorius, and sparsely in N. olivaris. Perineuronal staining (Fig. 11) was common, and in several of the sheep with widespread PrPSc deposits, there was a linear pattern (Fig. 11) of immunolabeling, likely the accumulation of PrPSc around neurites. These neuropil-staining patterns were frequently seen ventrally in DMNV, N. ambiguus, reticular formation, and midline raphe. All the animals had granular PrPSc deposits to some extent in the neuronal perikarya.
Nine sheep had PrPSc deposits in the cerebellum, and one sheep had sparse immunolabeling in the cerebellum only, indicating an early involvement of this part of the brain. PrPSc deposits were detected in all three gray matter layers and in the deep cerebellar nuclei. Two sheep had vascular amyloid-like deposits of PrPSc in the molecular layer. Subpial staining was quite prominent in several cerebellum sections. A stellate pattern was seen in the molecular layer and the deep cerebellar nuclei. Granular PrPSc deposits were found in all gray matter layers. Perineuronal and intraneuronal labeling was found in the Purkinje cell layer and the deep cerebellar nuclei. Sheep with sparse immunolabeling had granular deposits restricted to the granular and Purkinje cell layers.
There were no obvious differences in labeling patterns between the three groups of PrP genotypes that were positive for PrPSc, but the VA136QH171 animals had less PrPSc deposits than did the other two PrP genotypes (Table 3).
Histopathology in the brain
Vacuolar pathology in the obex and the cerebellum was usually associated with accumulation of PrPSc, but areas with PrPSc deposits were frequently without light microscopic pathology (Table 3). Five sheep with immunolabeling in the obex or the cerebellum had very few or no lesions at all in the obex (Fig. 12); three of these had the PrP genotype VA136QH171.
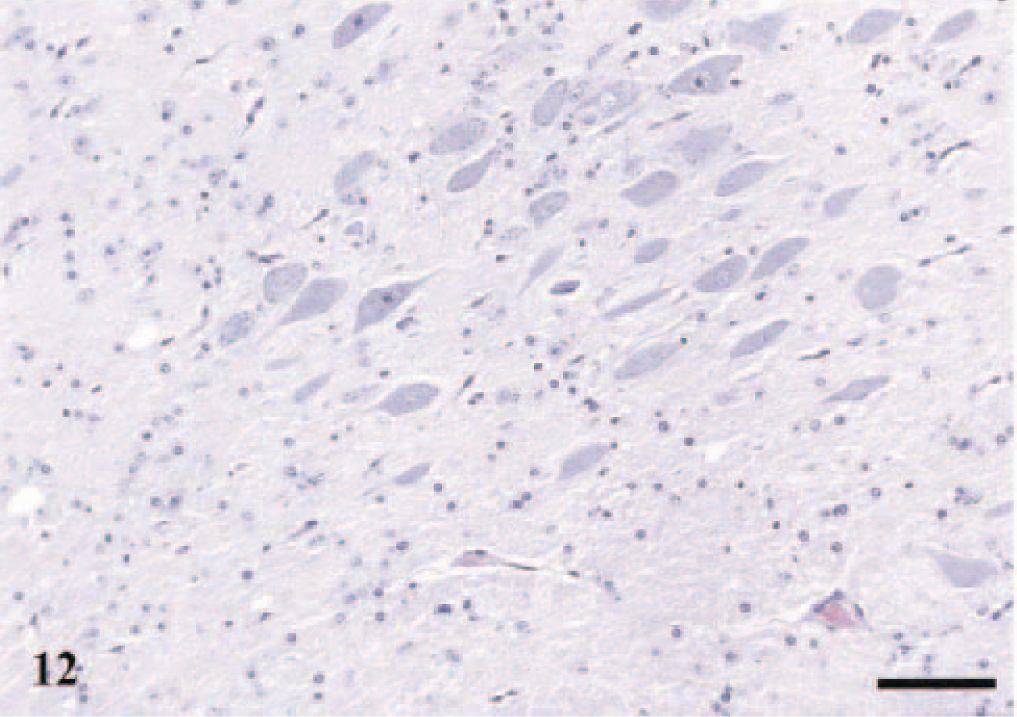
DMNV; sheep No. 3, PrP genotype VV136QQ171, 7 years old. Section shows a few vacuoles in the neuropil at the perimeter of the nucleus. HE. Bar = 80 μm.
The most extensive lesions were found in the DMNV, and the second most affected area was N. cuneatus accessorius. Vacuolation in the DMNV showed the same distribution as that described for PrPSc: ventral vacuolation, vacuoles spreading dorsally, and scattered vacuoles in the whole nucleus (Fig. 13, arrow shows intraneuronal vacuoles). Astrocytosis correlated well with the vacuolar changes in all areas.
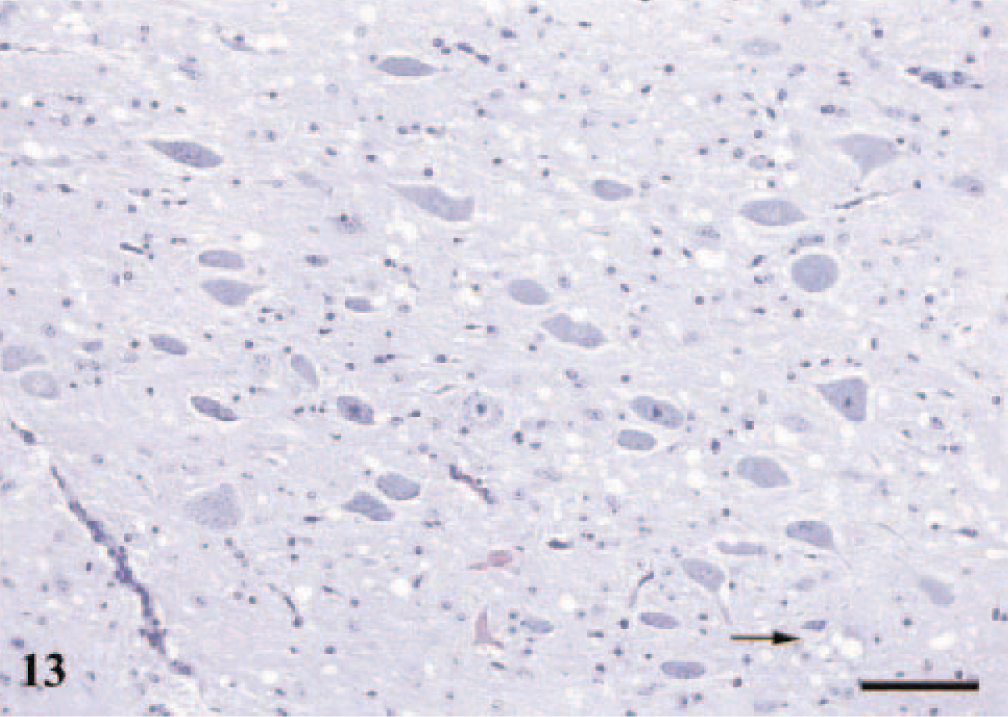
DMNV; sheep No. 7, PrP genotype VA136QQ171, 2 years old. Section shows widespread vacuoles predominantly in the neuropil; arrow points to a neuron with perikaryonal vacuoles. An increased number of glial cells are also present. HE. Bar = 80 μm.
Six animals had vacuolar pathology in the cerebellum. Two sheep (Nos. 5 and 13) also had vascular amyloid-like changes in the molecular layer.
The three sheep with immunolabeling in lymphoid tissue only had no histopathologic changes in the brain. Those animals negative for PrPSc in any of the organs examined generally had minor or no lesions in the obex.
Discussion
To our knowledge, this is the first report describing the distribution of PrPSc in nervous and lymphoid tissues in a naturally infected scrapie flock at a given time. Elsen et al. 10 studied a flock in which the animals were kept alive until the appearance of clinical signs. Our data show that the prevalence of PrPSc-positive sheep in a sheep flock with few clinical scrapie cases (4%) may still be considerably high. In this case, 35% of the sheep greater than 1 year of age showed accumulation of PrPSc, all contained within the group of animals carrying susceptible PrP genotypes. Only one sheep with a susceptible PrP genotype came out as PrPSc negative. It is of particular interest that two of the PrPSc-positive sheep with a highly susceptible PrP genotype were 7 years old and clinically healthy when culled. A possible explanation for the apparently slow development of disease in these animals, as opposed to younger flock-mates of similar PrP genotype, is that the older animals were infected as adults. Alternatively, the older sheep may have been exposed to a lower infective dose initially and, therefore, experienced a slower propagation of PrPSc. Newly born lambs in an infected environment are believed to receive a high dose of infectivity and may have a more rapid disease development. A previous study reported that infection with the scrapie agent occurred with higher incidence in lambs exposed from birth than in lambs moved to an infected environment when 3 and 9 months of age. 22 Scrapie susceptibility also may be influenced by flanking regions of the PrP gene 23 or by other still unknown genes. 32
Three sheep had deposits of PrPSc in the RPLN, and not in the brain, suggesting that this organ, easily accessible at slaughter, is suitable for screening purposes. Ten of the positive sheep (59%) were 2 years old, and these account for 67% of the 2-year-old cohorts still alive at the time the flock was culled. Seven PrPSc-positive animals, lambs included, were the first or second generation after the diseased dams. This could not be verified in the other 10 sheep, including a 15-month-old sheep (No. 13). This indicates the importance of an intense infection pressure in the environment combined with a susceptible PrP genotype in the transmission of the disease.
In accordance with recent studies 1 , 20 our data highlights the importance of the IPP as an organ involved in early uptake of the scrapie agent. In lambs the IPP reaches its peak development at the age of 3 months, after which it gradually involutes until 18 months of age. 41 In the present work two 86-day-old littermates had PrPSc deposits in the IPP. Surprisingly, one of these lambs had the intermediate susceptible PrP genotype VA136QR171, whereas the second positive lamb and the dam had highly susceptible PrP genotypes. To the best of our knowledge, this is the first observation of PrPSc deposits in lymphoid tissue in sheep with the VA136QR171 PrP genotype. Andréoletti et al. 1 studied lymphoid tissue from 2-, 4-, 6-, and 9-month-old, naturally infected VA136QR171 lambs, and in contrast to VV136QQ171 lambs, none of these showed any immunolabeling. Orally challenged VA136QR171 lambs seem to take up PrP rapidly at a young age, as Heggeb⊘ et al. 20 showed that some follicles in the IPP had strong immunolabeling 1 and 5 weeks after exposure. Based on the putative role of the IPP in absorption and dissemination of the scrapie agent, infection after involution of this tissue also may be less effective and may lead to a slower development of clinical disease.
In the brain, all sheep with PrPSc accumulation in DMNV had concomitant, but generally weaker, labeling in NTS-5. Ten sheep had PrPSc deposits in NTS. Early immunolabeling in DMNV suggests a retrograde spreading of PrPSc along efferent fibers of N. vagus and that this nucleus may be the site of entry of the scrapie agent into the brain. 5 , 42 , 51 However, DMNV is also a target for vagal afferents in sheep, and a spread along these fibers cannot be excluded. Further dissemination of the agent through neuroanatomic connections between DMNV, NTS, N. n. hypoglossi, and NTS-5 is possible because such connections are established in the lamb. 52 An additional port of entry of the agent may be any of the cranial nerves associated with the oro-pharyngeal cavity. However, studies on natural scrapie in sheep revealed PrPSc in Ganglion trigeminale subsequent to accumulation in the brain, suggesting an anterograde spread along afferent nerve fibers. 51
In the present study nine sheep showed accumulation of PrPSc in the cerebellum, and one sheep had deposits in the cerebellum only. Two of these animals were assumed to be in a very early phase of CNS involvement. Secondary trigemino-cerebellar connections have been established in sheep, and NTS-5 projects to all lobules of the cerebellar hemisphere. 43 In addition, cerebellar afferent projections from DMNV have been described in the cat. 54 The early involvement of the cerebellum of sheep in this study could be explained by these neuroanatomic connections.
This study also suggests that clinically normal VA136QQ171 sheep may accumulate more PrPSc than do homozygous V136Q171 animals before developing clinical disease. Our data indicate that the dissemination rate of PrPSc to the brain varies with the PrP genotype, being fastest in the V136Q171 homozygous sheep. However, a rapid spread to the brain also was observed in heterozygous VA136QQ171 sheep. Among adult sheep, VA136QH171 animals appear to have a slower dissemination of the scrapie agent to the brain than do the other two PrPSc-positive V136Q171 groups. The three sheep with the A136H171 allele had markedly reduced accumulation of PrPSc in the brain compared with the other two groups.
The most likely mode of introduction of the disease in this flock may be by recruitment of already infected sheep, as suggested in an epidemiologic study. 21 The age distribution of susceptible sheep in the present study indicates introduction of the disease in the near past. 4 As already mentioned, two of the sheep purchased as lambs in 1992 had sparse accumulation of PrPSc in the brain 7 years later and probably had not been infected as lambs. A third sheep (No. 5) was purchased as a lamb in 1993. This ewe had widespread immunolabeling in the obex and the cerebellum 6 years later and may have been infected as a lamb, thus introducing the infectivity. Second, there is a possibility that the environment was already contaminated when the sheep were stocked up, although there was no previous history of scrapie-like signs at the farm. A third possible entry of the disease is through feeds. Feeding of meat and bone meal to ruminants has been banned since 1990. 45 Such feeds, however, have been used for pigs and poultry until 2000, and cross-contamination at the mill, during transport, or at the farm cannot be entirely ruled out. 2
This study has shown that most of the susceptible animals in a scrapie-affected flock were infected, but only a small number showed clinical signs at a given time. PrPSc may be detected in lymphoid and nervous tissues of old, clinically normal, but highly scrapie-susceptible, sheep. VA136QR171 lambs are capable of accumulating PrPSc in gut-associated lymphoid tissue, but no adult sheep of this PrP genotype had any detectable accumulation of PrPSc in the RPLN or the brain.
Footnotes
Acknowledgements
We are grateful to Esben Seljeskog and Beate Rindal for preparing the histologic slides and to Eli Brundtland for doing the PrP genotyping. This study was supported by grants 119688/110 (C. Ersdal, M. J. Ulvund) and 133449/111 (S. L. Benestad) from the Norwegian Research Council.
