Abstract
Immunohistochemistry, the standard method for diagnosing amyloid A (AA) amyloidosis, is limited in animals because it requires a large array of animal-specific anti-AA antibodies, not commercially available. The Shtrasburg method (SH method) is a highly specific and sensitive technique, helping in the diagnosis and determination of AA amyloidosis in humans. The aim of this study is to determine whether the SH method is applicable in the diagnosis of AA amyloidosis in a variety of animals. Tissue samples were obtained from animals suffering from spontaneous or experimentally induced AA amyloidosis (mice, hamsters, guinea pigs, cheetahs, cats, cows, ducks, a dog, a goose, a chicken, and a turaco). Detection of the amyloid and quantitative evaluation were performed using Congo red staining, and specific AA typing was performed by the potassium permanganate technique. The studied tissues were subjected to the SH method, which confirmed the AA nature of the amyloid deposit, by displaying in polyacrylamide gel electrophoresis protein bands consistent with the molecular weight of the species-specific AA, in all the animals examined, except mice, hamsters, and guinea pigs. N-terminal analysis of these bands corroborated their AA origin. We conclude that the SH method may be used as an ancillary simple tool for the diagnosis of AA amyloidosis in a large number of domestic and wild animals. Moreover, our findings further increase the feasibility of applying this method in humans.
Keywords
Amyloidosis is a disease characterized by extracellular deposition of amyloid, a fibrillar material composed of a major protein and common elements. 15, 20 Amyloid fibrils are usually identified by their unique feature of displaying green birefringence under polarized light 37 after Congo red staining. The amyloid deposit is categorized by its major component, which is determined by the underlying disease. 4 Often, the diagnosis of the amyloid-inducing disease depends on the elucidation of the amyloid type, usually carried out histologically, using immunohistochemical techniques or by the potassium permanganate method. 8, 12, 29, 39, 48 When these methods give inconclusive results, caused by cross-reactivity of the antibodies with nonspecific epitopes, unavailability of specific anti-AA antibodies, or inconsistent results of the permanganate assay, complementary methods should be applied.
Reactive amyloidosis is a condition that complicates a long list of chronic inflammatory, chronic infectious, malignant, and hereditary disorders. 13, 16, 52 The main protein involved in this disease is amyloid A (AA), an N-terminal fragment of serum amyloid A (SAA), a serum protein with an explosive rise and a role in inflammation. 17, 24 The identification of AA is usually based on its reactivity with specific anti-AA antibodies and sensitivity to permanganate pretreatment. 12, 39, 48, 51
In 1982, we reported an additional method to identify AA and distinguish it from other amyloid types. 47 This method was based on the ability of AA, unlike other amyloid proteins, to undergo extraction from formalin-fixed, paraffin-embedded tissue and retain its electrophoretic and antigenic properties despite formalin masking. 25, 34, 50 Over the years, the Shtrasburg method (SH method) became well established 6, 9, 12, 28, 35, 38, 40 and was applied for AA identification, 1, 2, 10, 33, 53 isolation, and even purification for amino acid sequencing. 3, 26, 27, 34
Recently, the SH method has been reevaluated and found to be highly sensitive and specific. It even recognized a range of human AA proteins, which differ in length and composition from the classical 76 amino acid protein in humans, but did not detect various non-AA amyloid proteins, such as light chains, senile and familial transthyretin, β2-microglobulin, calcitonin of medullary carcinoma of the thyroid, and senile amyloid of the seminal vesicle. 21, 43
To further expand the spectrum of AA proteins detected by this assay and to determine whether it retains its sensitivity for AA proteins, we studied and report in this study the application of the SH method to amyloidotic tissues of a variety of animals. In this study, the SH method emerges as a novel tool, helpful in the diagnosis of AA amyloidosis in domestic and wild animals.
Materials and Methods
General experimental design
Animal tissues, containing amyloid deposits that were formed spontaneously or induced experimentally, were studied by the SH method.
Experimental animals
Seven- to 16-week-old male and female Swiss mice (Table 1, case Nos. 1–3), 12-week-old male C57Bl, C3H, and AKR mice (case Nos. 4–6), 5- to 6-week-old male golden hamsters (case Nos. 7 and 8), and 4-week-old male guinea pigs (case No. 10) were purchased from the Breeding Colony of Lewenstein, Yokneam, Israel, and subjected to amyloid induction. Care and use of all laboratory animals were in agreement with the criteria set by the animal experimentation committee of the Sheba Medical Center.
Tinctorial features of the amyloidotic tissues studied by Congo red staining and permanganate technique.
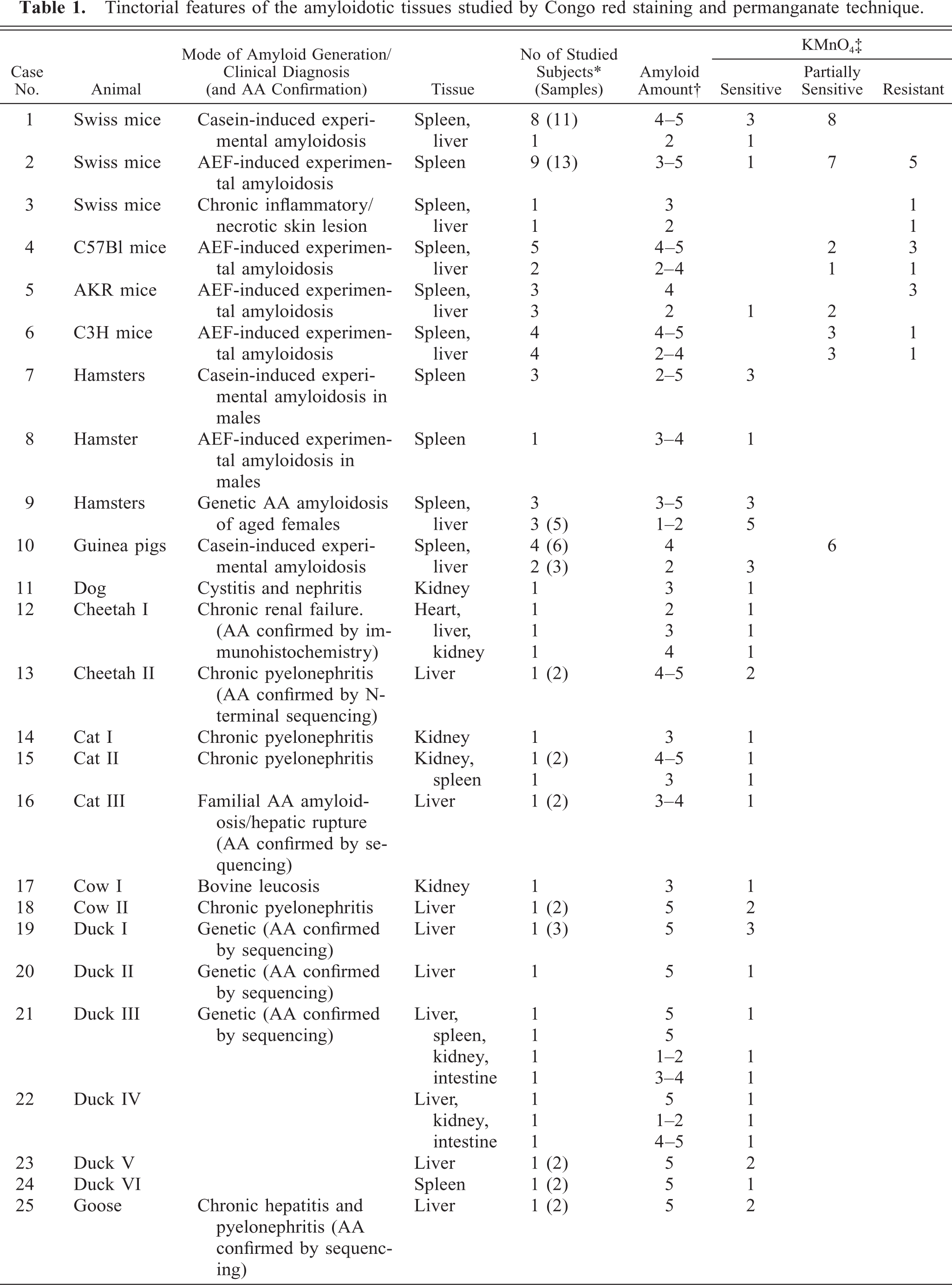

∗ The parentheses show the total number of samples studied (when larger than the number of the subjects).
† Evaluated by the Crush and Smear technique (unfixed tissues) or by histology (paraffin blocks).
‡ Displayed is the number of samples studied. Although not always consistent, the large majority of mice and guinea pig samples were insensitive or only partially sensitive to permanganate treatment.
Induction of amyloidosis by casein
Amyloidosis was induced by casein in male Swiss mice, hamsters, and guinea pigs (Table 1, case Nos. 1, 7, and 10) according to established protocols. 5, 15 In brief, mice were injected subcutaneously (s.c.) 5 days a week for 3 weeks with 0.5 ml/day of 15% (wt/vol) vitamin-free casein (NBC, Cleveland, OH) suspended in 0.02 mol/liter NaOH. Hamsters (case No. 7) were treated with 1 ml/day of the s.c. casein, three times a week for 8 weeks, and the guinea pigs (case No. 10) received 2 ml/day s.c. injections of the casein preparation two to three times a week for 8 weeks. After these treatments, the animals were euthanatized; one half of their spleen was evaluated for the presence and amount of amyloid, using the crush and smear (C&S) technique, 41 and the other half was fixed in 10% formalin (4% formaldehyde), processed, and embedded in paraffin for histologic evaluation. Congo red, with or without permanganate treatment, and the SH method were used for amyloid assessment.
Preparation of amyloid-enhancing factor
Amyloid-enhancing factor (AEF) was prepared as published earlier. 42, 44 In brief, splenic tissues from preamyloidotic mice were homogenized in 50% acetone/H2O and centrifuged. The sediment was dialyzed against H2O and lyophilized and was found to be extremely active and capable of inducing amyloidogenesis in mice. 42, 45
Induction of amyloidosis by AEF
Amyloidosis was induced in male and female mice and in a male hamster (Table 1, case Nos. 2, 4–6, 8), using a modification of our standard amyloid induction protocol. 42, 46 In brief, for mice, a single intravenous injection of 1 μg AEF, suspended in 0.5 ml phosphate-buffered saline, was given, followed by three daily s.c. injections of 0.5 ml AgNO3 2% (wt/vol). The mice were euthanatized on days 14–16, and the amyloid in their spleen was studied as described previously. The longer protocol (14–16 days, instead of 6 days) was applied to generate larger amounts of amyloid deposits. In the male hamster, AEF was injected intraperitoneally in a dose of 200 μg, and 2% (wt/vol) AgNO3 was given s.c., 1 ml every other day for 13 days. The hamster was euthanatized 3 weeks after the last AgNO3 injection, and its amyloid was studied as in the mice.
Tissues from animals with natural amyloidosis
Tissue samples containing amyloid deposits from a variety of animals with natural (spontaneous) amyloidosis were obtained (Table 1, case Nos. 3, 9, 11–27). In all, amyloidosis was diagnosed, and the amyloid quantity was evaluated using the regular Congo red (Bennhold's) method. 37, 41 All were classified as AA amyloidosis on the basis of clinical diagnosis and/or the potassium permanganate method. 51 In addition, the amyloid of cheetah I (Acinonyx jubatus, case No. 12) was diagnosed as AA using monoclonal anti-AA antibodies and immunohistochemistry. 33 Ducks I–IV (case Nos. 19–22), chicken (case No. 26), and cat III (case No. 16) amyloids were confirmed as AA by amino acid sequencing. 14, 23, 32 The amyloid of the aged female hamsters (case No. 9) was defined as AA on the basis of previous publications. 7, 30, 31 In this study, the AA nature of amyloid deposits in the tissues of the remaining animals was determined by N-terminal sequencing analysis (see below).
Quantification of amyloid deposits
Semiquantification of amyloid deposits was performed in unfixed tissues from the experimental animals using the C&S technique with a 5-grade scale. 41, 42 In the formalin-fixed, paraffin-embedded tissues, evaluation of the amyloid amount was based on Congo red staining and a 5-grade scale, comparable to that of the C&S method. 41–43
SH method
The SH method is intended to identify AA and distinguish it from other types of amyloid proteins. It was performed as published previously. 43, 47 In brief, formalin-fixed, paraffin-embedded tissue samples were deparaffinized by melting at 65°C and serial overnight washings with xylol, absolute and 95% ethanol, and saline. The treated specimens were then homogenized, washed in saline, and frozen and thawed several times. Sodium dodecyl sulfate (SDS) sample buffer was then added, and the samples were heated, centrifuged, and the supernatants were subjected to Sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE). Processing and running were repeated at least twice for each sample studied. Negative control samples consisted of organs from nonamyloidotic animals of the same species.
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis
SDS-PAGE was performed in 17% polyacrylamide gel, containing 0.1% SDS, in Tris-glycine running buffer. 11, 22 Human AA protein, purified from an amyloidotic spleen of a familial Mediterranean fever patient, 11, 43 recombinant human apo-SAA protein (PeproTech Inc., Rocky Hill, NJ), and Dalton Mark VII-L molecular weight (MW) standards, with an MW ladder of 14.2, 20.1, 24, 29, 36, 45, and 66 kd (Sigma Chemical Co., St. Louis, MO), were used as reference. 11, 42, 43
N-terminal sequence analysis
Samples obtained by the SH method were run on SDS-PAGE and electroblotted on Sequi-Blot polyvinylidene difluoride (PVDF) membranes using 10 mmol/liter 3-cyclo-hexyl-amino-1-propanesulphonic acid (Sigma) buffer (pH 11) containing 10% methanol and stained with 0.5% Coomassie Blue R250 (BioRad, Melville, NY) in 40% methanol/1% acetic acid. The protein bands were excised from the PVDF membrane, and the amino acid sequence analysis was performed using an Applied Biosystem Procise protein sequencer, run in pulsed liquid mode, according to the manufacturer's protocol. Phenylthiohydantoin (PTH)-amino acids were identified online from a 5-pmol PTH-amino acid standard. Database searches were done against the Swiss Prot Database.
Results
Table 1 shows the tinctorial features of the tissues that were studied by the Congo red and permanganate assays. All animals suffered from reactive (AA) amyloidosis with a moderate to large amount of amyloid deposits. This was verified by medical history, Congo red staining, sensitivity to permanganate treatment, and AA sequencing. Our findings demonstrated that in reactive amyloidosis of mice (case Nos. 1–6) and guinea pigs (case No. 10), the sensitivity of the AA for permanganate treatment is inconsistent, and in hamsters (case Nos. 7–9), although the Congo red staining is weak, the amyloid deposits remain permanganate sensitive. Table 2 shows the N-terminal sequences of the amyloid proteins extracted from the studied tissues. These include the amyloid proteins of cheetah II (case No. 13), cat III (case No. 16), ducks V and VI (case Nos. 23 and 24), a goose (case No. 25), and a turaco (a bird of the Musophagide family, case No. 27). As mentioned (see Materials and Methods), the amyloid proteins of cheetah I (case No. 12), cat III (case No. 16), ducks I–IV (case Nos. 19–22), and the chicken (case No. 26) were previously studied and published. 14, 23, 32, 33 The N-terminal of the AA protein of the dog (case No. 11) and the cow (case No. 18) was found to be blocked. The amino acid sequences obtained were homologous to published sequences of the appropriate AA protein subunits, with the exception of the turaco (case No. 27), which could not be found in any database but appeared to be consistent with a sequence of an AA protein.
N-terminal amino acid sequences of the amyloid protein bands.

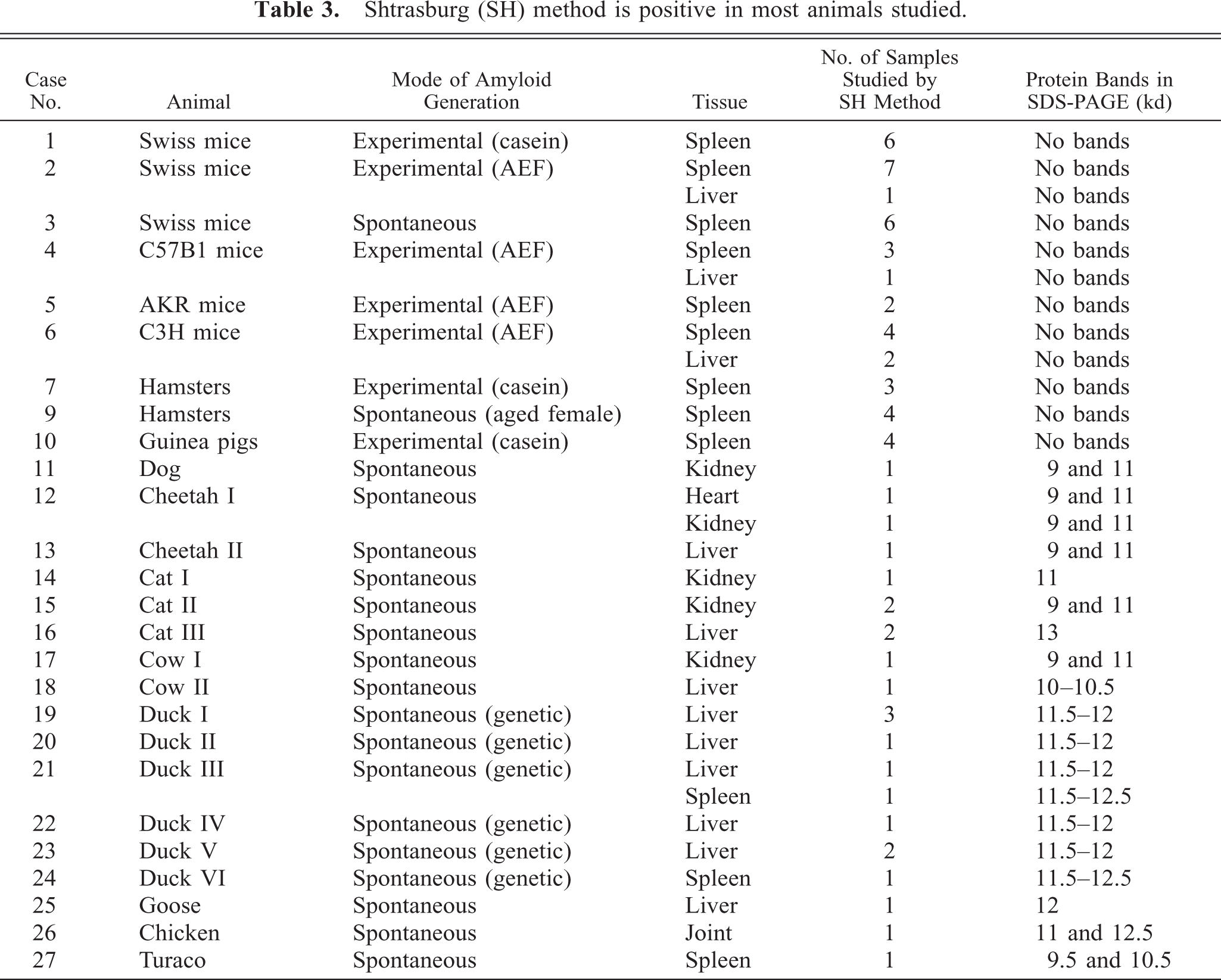
Samples of the formalin-fixed, paraffin-embedded amyloidotic tissues with an amyloid grade above +2, were subjected to amyloid evaluation by the SH method. Normal tissues from the same organ of nonamyloidotic subjects of the same species were studied as negative control. As seen in Table 3, bands consistent with the specific animal AA by its MW appeared in all specimens from the amyloidotic tissues, except those of the mice (case Nos. 1–6), hamsters (case Nos. 7 and 9), and guinea pigs (case No. 10). These are the same animals in which the results of permanganate or Congo red staining were inconsistent. Of interest, the amyloid deposits of the dog (case No. 11), the two cheetahs (case Nos. 12 and 13), cat II (case No. 15), cow I (case No. 17), the chicken (case No. 26), and the turaco (case No. 27) contained two fragment sizes of the AA protein (two bands, Table 3). In cheetah II (case No. 13), the N-terminal of the two protein bands was sequenced and showed, as expected, the same sequence. Similarly, the N-terminal sequences of the two protein bands of the turaco (case No. 27) were identical (Table 2). Figure 1 shows some examples of the SDS-PAGE of extracts of the tissues studied, which were positive by the SH method.
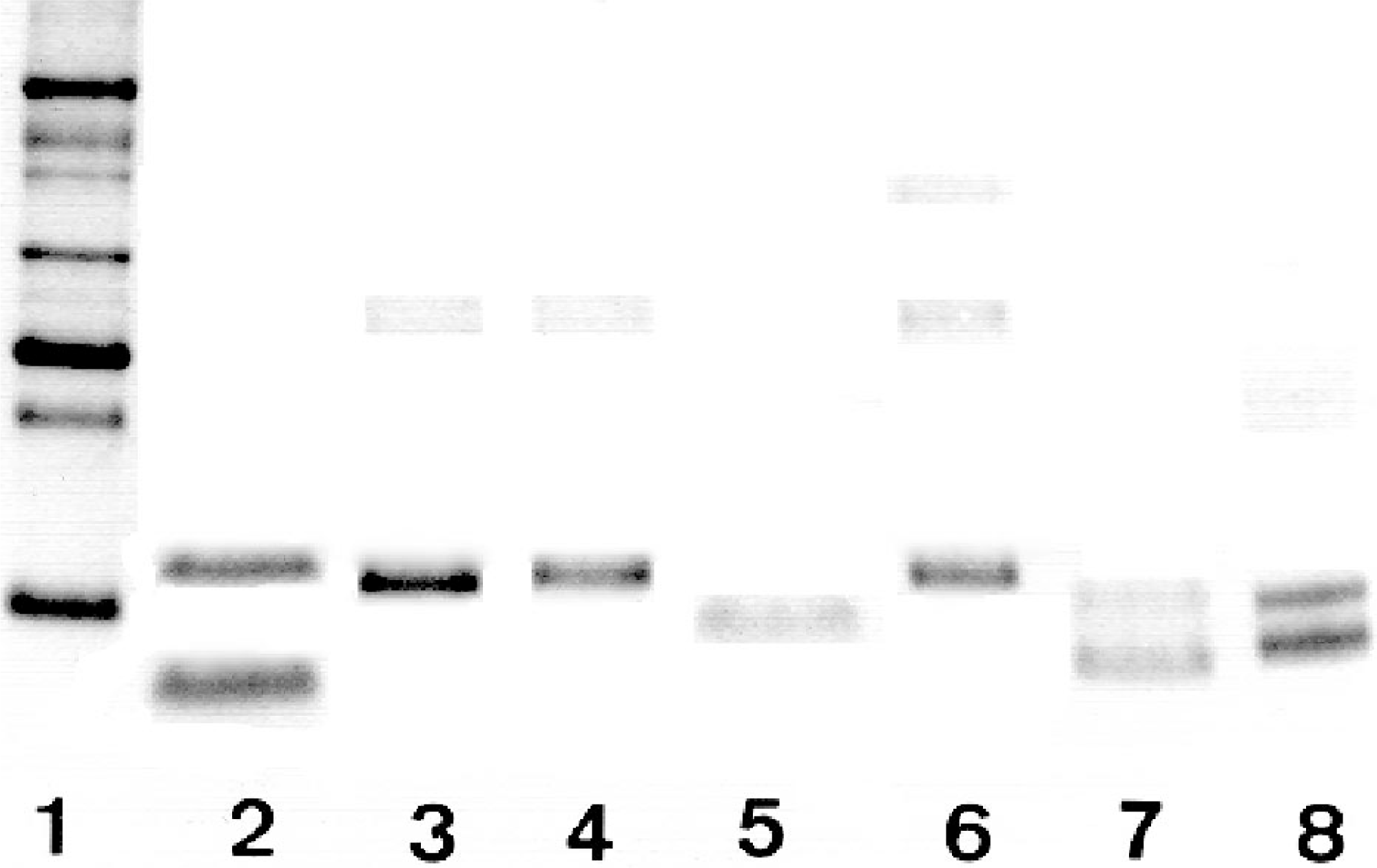
Polyacrylamide gel electrophoresis of extract samples from amyloid-containing tissues, using the SH method. Lane 1: MW markers from top to bottom—66, 45, 36, 29, 24, 20.1, and 14.2 kd. Lane 2: upper band, recombinant human apo SAA (11.7 kd); lower band, purified human AA protein (8.5 kd). Lanes 3–8, extracts from amyloidotic liver of a duck (case No. 23); liver of a goose (case No. 25); liver of a cow (case No. 18); spleen of a duck (case No. 24); liver of a cheetah (case No. 13); and spleen of a turaco (case No. 27), respectively. The bands appearing in lanes 3–8 present amyloid proteins. Note that samples 7 and 8 demonstrate two amyloid protein bands on the gel.
Shtrasburg (SH) method is positive in most animals studied.
Discussion
The usefulness of the SH method in the diagnosis of reactive amyloidosis in a number of animal species was evaluated. The diagnosis of amyloidosis of the AA type in these animals was based on clinical history, permanganate assay, and amino acid sequencing (Tables 1, 2). In cheetah I (case No. 12), it was confirmed by the cross-reactive monoclonal anti-human AA antibodies mc4 and mc29 (Table 1). 33 In all animals studied, except for the mouse, hamster, and guinea pig, the SH method gave positive results, namely, extracts of amyloid-containing tissues displayed distinct protein bands in SDS-PAGE (Table 3, Fig. 1). The decision that these protein bands are the amyloid proteins of the AA amyloidotic animals was based on the results of the N-terminal amino acid sequence analysis of the bands (Table 2), the absence of protein bands in negative control tissues, and the size of the specific animal AA protein. 18, 23, 32, 49, 52
In the mice (case Nos. 1–6), hamsters (case Nos. 7 and 9), and guinea pigs (case No. 10), the SH method failed to demonstrate protein bands and therefore cannot be applied for the diagnosis of reactive amyloidosis in these animals. The insensitivity of the SH method to AA in these animals is not related to the amount of amyloid deposition or to the mode of its generation (spontaneous or experimental). This is inferred from the finding that the SH method gave negative results both in experimental amyloidosis, (generated either by AEF or by casein), and in spontaneous (natural) amyloidosis with chronic inflammatory skin lesions (case No. 3) or of naive, aged, female hamsters (Table 3, case No. 9).
A high degree of structural homology of SAA, the precursor protein of AA, is known to exist between species, particularly in positions 36–58 and 94–105. 17, 19 An attempt to discern structural differences in animals' AA, separating mice and hamsters from all the other animals examined, revealed only two sites with such amino acid sequence variability (we could not find a published sequence of the AA of the guinea pig beyond position 20). One change is in serine in position 22, 32 which is substituted by threonine in the AA of the mouse and hamster. The other change is in position 65. This site is highly versatile in many animals, but only in the mouse and hamster does phenylalanine substitute for isoleucine, which is found in human AA. The difference in position 22 is unlikely to cause a significant change in the nature of the AA protein or to affect its binding to other tissue components because both serine and threonine are neutral and polar amino acids and closely resemble each other in their structure. The role of the change in position 65 is hard to predict, but it may be more important because the two amino acids are structurally different.
Additional factors that could have determined the inaccessibility of the AA protein of these three species to the SH method are the other constituents of the amyloid fibril, which in mice, hamsters, and guinea pigs may disrupt AA extractability. Moreover, it is possible that the nonsusceptibility of the amyloid of these animals to the SH method and the insensitivity to the potassium permanganate assay (Table 1) share a common mechanism. This hypothesis concurs with that of Mezza et al., 30 who demonstrated insusceptibility to permanganate treatment in hamster amyloidotic tissues as well. Because Congo red staining is structurally dependent, it could be inferred that structural changes in the β-pleated sheet configuration of amyloid may be involved in both occasional permanganate resistance and the SH method negativity.
The size of the AA protein bands in our study, ranging around 9–13 kd, is as expected for AA proteins, based on previous publications. 18, 23, 32, 49, 52 In the two cheetahs, the dog, cat II, cow I, the chicken, and the turaco (case Nos. 11–13, 15, 17, 26, 27), the two MW bands that were demonstrated on the SDS-PAGE gel are both probably fragments originating from the specific precursor SAA protein (Table 2 and published data 11, 33, 36 ). A similar phenomenon was widely described in reactive amyloidosis in humans. 11, 36, 43
The high homology between human and animal AA allows one to view the study as further extending the range of the AA proteins recognized by the SH method and thus expanding its validity and reliability as a clinical and investigative tool. Similarly, resting on the high homology of the other analogous human and animal amyloid proteins, resistance of animal non-AA amyloid proteins to the SH method is also conceivable. However, this assumption still remains to be experimentally corroborated. The nonavailability for us of other types of animal amyloidosis precluded the demonstration of the specificity of the SH method to AA in animal amyloidosis.
Our study adds a supplementary tool to the spectrum of techniques used in the diagnosis of animal amyloidosis. Its advantage is that it does not require a wide array of specific antibodies, which are not commercially available, and may be performed outside of a histological laboratory, thus facilitating clinical decisions.
