Abstract
The common marmoset (Callithrix jacchus) is a small New World primate native to Brazil that has been used extensively in biomedical research. A retrospective analysis of archived hematoxylin and eosinstained tissue sections and clinical records was conducted at the New England Primate Research Center on 86 marmosets more than 1 year of age that were euthanized during the past decade because of morbidity and failure to thrive. Approximately 17% (15 of 86) were found to have amyloid deposits in one or more organs, including the liver, adrenal glands, kidneys, and intestine. This material was shown by amino acid sequence analysis to be composed of serum amyloid A (SAA)-related protein. This type of amyloidosis, designated AA or “secondary,” is associated typically with an inflammatory process that induces elevated levels of the SAA amyloidogenic precursor molecule. Notably, there were no significant pathologic differences or other distinguishing features in animals with amyloid versus those without; furthermore, on the basis of the limited number of serum specimens available for analysis, the SAA concentrations in the two groups were comparable, thus suggesting the possible inheritable nature of the disorder. In this respect, the common marmoset provides a unique experimental model for study of the pathogenesis and treatment of AA and other forms of systemic amyloidosis.
AA or reactive “secondary” amyloidosis is the most clinically relevant systemic amyloid-associated disease found in both mammals and nonmammalian species 14 and results from the pathologic deposition of an ≈76-residue N-terminal fragment of the serum amyloid A (SAA) protein. 2, 10, 19 This precursor molecule is an acute-phase apolipoprotein reactant synthesized mainly by hepatocytes under control of certain cytokines, including interleukin (IL)–1 and IL-6, as well as tumor necrosis factor α. 10, 22, 23 In humans, the plasma concentration of SAA normally is ≈0.1 mg/ml but can increase over 1,000-fold in response to an inflammatory stimulus. As part of this process, the SAA molecule undergoes partial proteolysis and the N-terminal cleavage product is deposited systemically as amyloid fibrils in vital organs, including the liver, spleen, kidneys, and adrenal glands. 19 With rare exception, AA amyloidosis develops in patients with chronic inflammatory or infectious diseases (rheumatoid arthritis, tuberculosis, etc) or other conditions that are associated with sustained elevation of SAA levels (e.g., certain forms of cancer, Castleman disease, and familial Mediterranean fever). 2, 10, 19 AA-related amyloid also has been found in cats, sheep, goats, cattle, dogs, horses, rabbits, hamsters, and birds, 6, 8, 12, 13, 16 as well as in primates such as rhesus and pigtailed macaques 1, 4, 21 and chimpanzees. 9 Although the SAA levels in these non-humans were not provided, postmortem examination invariably revealed chronic inflammation or infection.
We now report the relatively frequent occurrence of systemic amyloidosis among common marmosets (Callithrix jacchus) housed at the New England Primate Research Center (NEPRC). The pathologic deposits, which had an anatomical distribution similar to that found in humans affected with this disorder, were identified immunohistochemically and, unequivocally, by amino acid sequence analysis as AA. Notably, the mean SAA levels or extent of inflammatory-related pathology in the affected animals did not differ from marmosets without amyloid. On the basis of these findings, we posit that other factors, perhaps hereditary, are responsible for the development of AA amyloidosis in this primate. In this regard, the common marmoset could serve as a unique experimental model for study of the pathogenesis and therapy of AA and other systemic amyloid disorders.
Materials and Methods
Animals and veterinary care
Common marmosets (≈ 250) were housed at the NEPRC in accordance with standards of the American Association for Accreditation of Laboratory Animal Care and Harvard Medical School's Animal Care and Use Committee. As described previously, 7 all animals were pair-housed or grouped in small extended families consisting of one adult breeding male and female and two to six siblings less than 18 months of age. The marmosets were fed a standard commercial New World Primate diet (Harlan Teklad, Madison, WI) supplemented with a variety of fresh fruit, mealworms, and hardboiled eggs.
Histopathology
For light and polarizing microscopy, 4- to 6-μm-thick tissue sections were cut and stained with hematoxylin and eosin (HE) and a freshly prepared alkaline Congo red solution, respectively. For electron microscopy, sections were embedded in Epon (Ted Pella, Redding, CA), sectioned, and examined with a JEOL 100S transmission electron microscope.
Immunohistochemistry
Paraffin-embedded tissue sections (6-μm-thick) were cut on a microtome, mounted on poly-
SAA quantitation
Antemortem blood specimens were obtained by vena-puncture, and the separated serum was frozen at −80 C. SAA concentrations were measured by an enzyme-linked immunosorbent assay (ELISA) using the Multispecies SAA ELISA kit according to directions supplied by the manufacturer (Biosource, Camarillo, CA). Standard curves were prepared using known amounts of human SAA protein and absorbance was measured at 405 nm with a model 4450 BioRad plate reader (Fullerton, CA).
Amyloid extraction and purification
The methods used to extract amyloid from tissue were as described by Pras et al. 18 In brief, a portion of marmoset liver obtained at necropsy and maintained at −80 C was homogenized with cold saline in an ice bath using an Omni-Mixer (Omni International, Waterbury, CT). The extract was centrifuged at 10,000 rpm for 30 minutes at 4 C and the pellet reextracted twice more with cold saline, once with 0.1 M sodium citrate Tris-buffered saline, pH 8.0, and then again with saline until the A280 of the supernatant was < 0.10. The resultant pellet was homogenized with cold distilled water, and the mixture centrifuged at 35,000 rpm for 3 hours at 4 C. The pellet obtained from the water extract was then lyophilized.
Electrophoretic and sequence analyses
The amyloid protein extract was dissolved in 0.1% sodium dodecyl sulfate (SDS) buffer containing 0.1 M dithiothreitol and 8 M urea and electrophoresed on 10% SDS–polyacrylamide gel electrophoresis (PAGE) gels (NuPage kit, Invitrogen, Carlsbad, CA). For automated sequence analysis by Edman degradation, the protein band was transferred onto a polyvinylidine difluoride (PVDF) membrane and placed in the blot cartridge of an ABI Model 494 Procise gasphase sequencer (Applied Biosystems, Foster City, CA). 17
Statistical analysis
For statistical comparisons, Wilcoxon, Mann-Whitney, and Fisher's exact tests were used with INSTAT 2.01 software.
Results
Index case
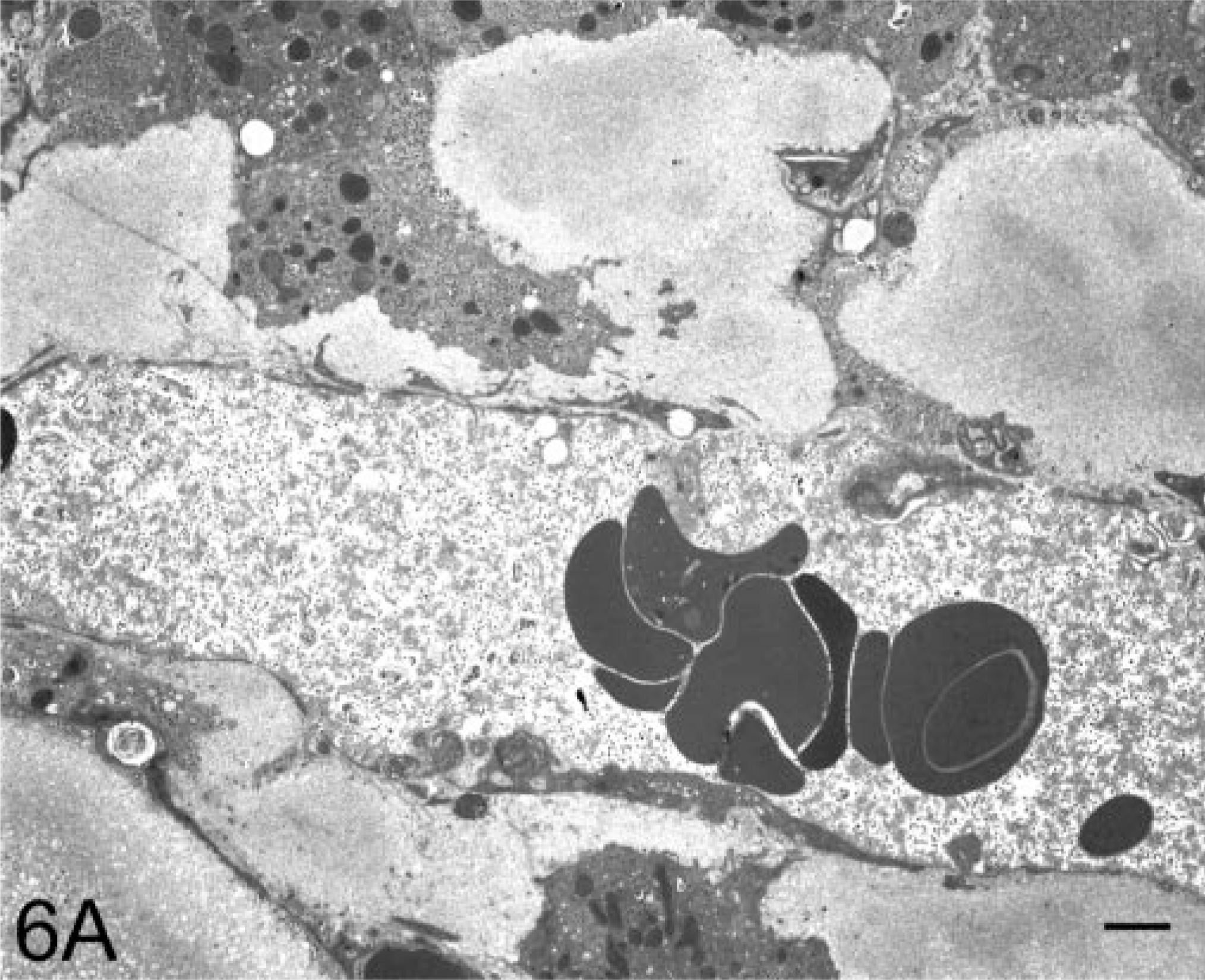
Case No. 408–89 was a 13-year-old female common marmoset with progressive weight loss that was found to have hepatomegaly; normocytic, normochromic anemia; and hypoalbuminemia (3.1 g/dl). A liver biopsy was performed and examination of an HE-stained section revealed the presence of a homogeneous, eosinophilic, extracellular substance suggestive of amyloid (Fig. 1). After Congo red staining, this material exhibited green birefringence when viewed under polarized light (Fig. 2). The animal was euthanatized, and at postmortem examination, the liver was found to be markedly enlarged (Fig. 3). Microscopically, similar deposits were seen in specimens of small intestine (Fig. 4), kidneys (Fig. 5), and adrenal glands. The material consisted of ≈ 10-nm-diameter unbranched fibrils, as evidenced by electron microscopy (Fig. 6).
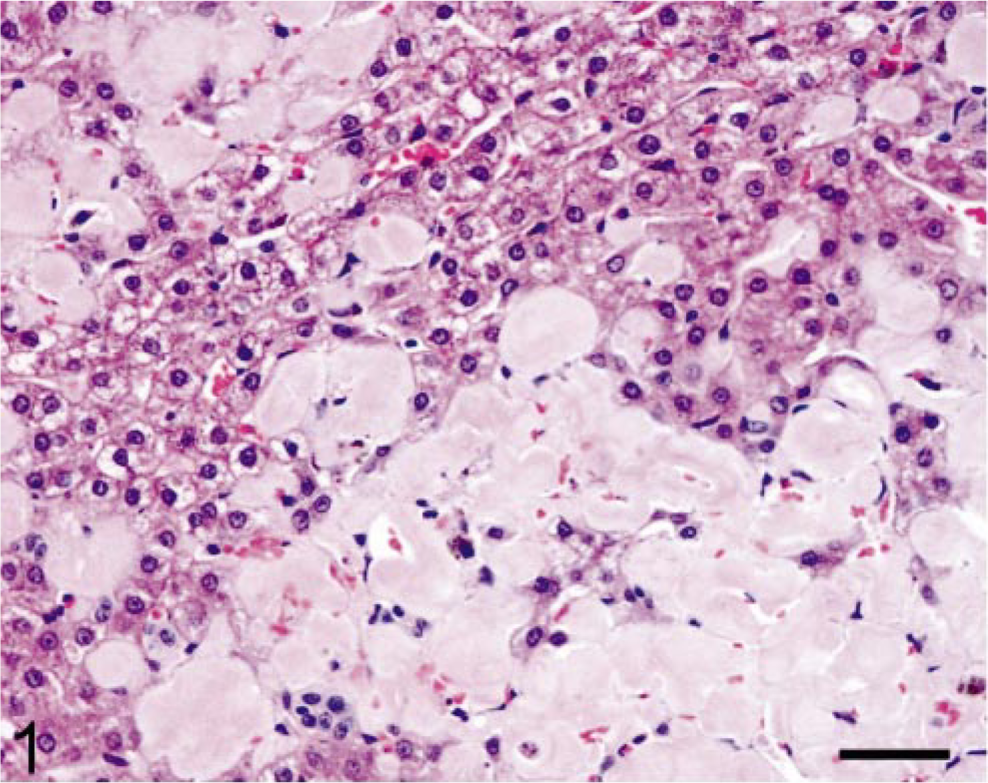
Liver; marmoset. Extensive deposition of homogeneous, eosinophilic material (amyloid) within the hepatic parenchyma. HE. Bar = 95 μm.

Liver; marmoset. Green birefringement material evidenced in Congo red–stained section as viewed under polarized light. Bar = 100 μm.
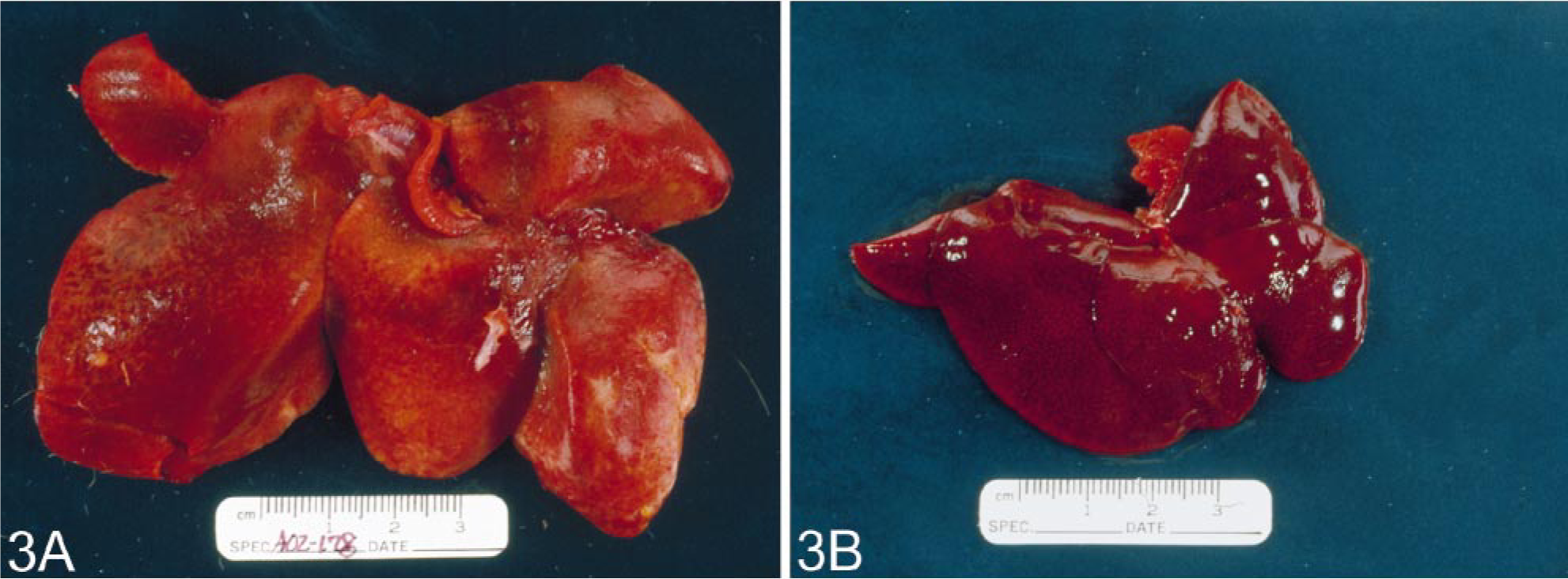
Gross appearance of livers from two age-matched common marmosets.
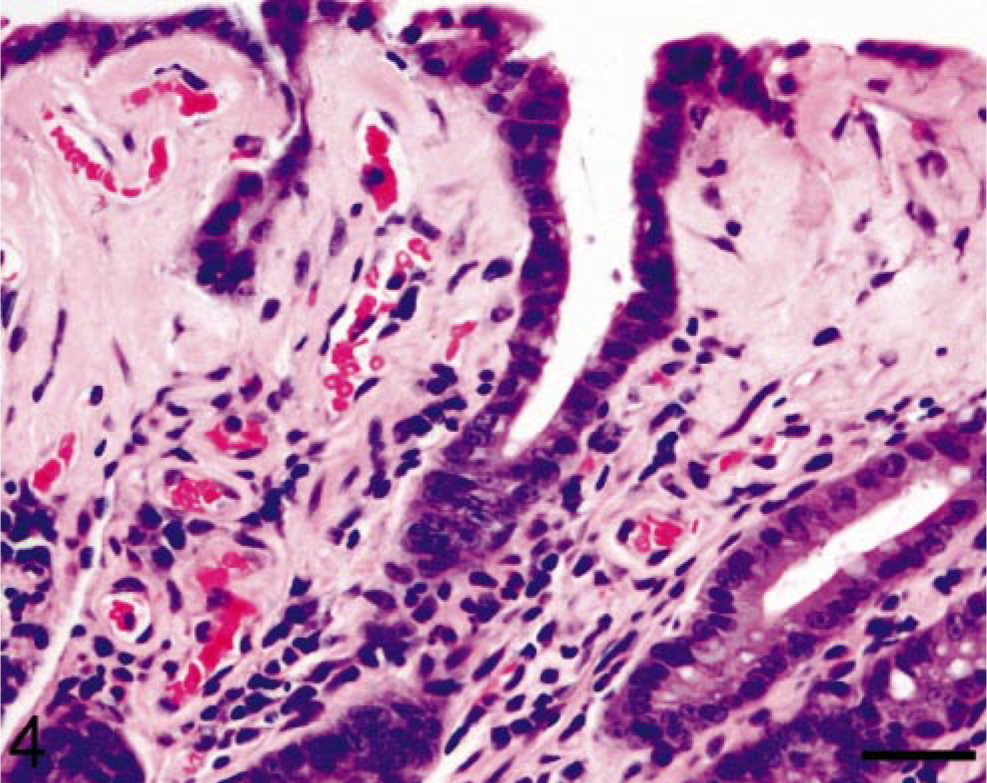
Small intestine; marmoset. Amyloid deposition in the small intestine occurs primarily within the villus tips. HE. Bar = 95 μm.
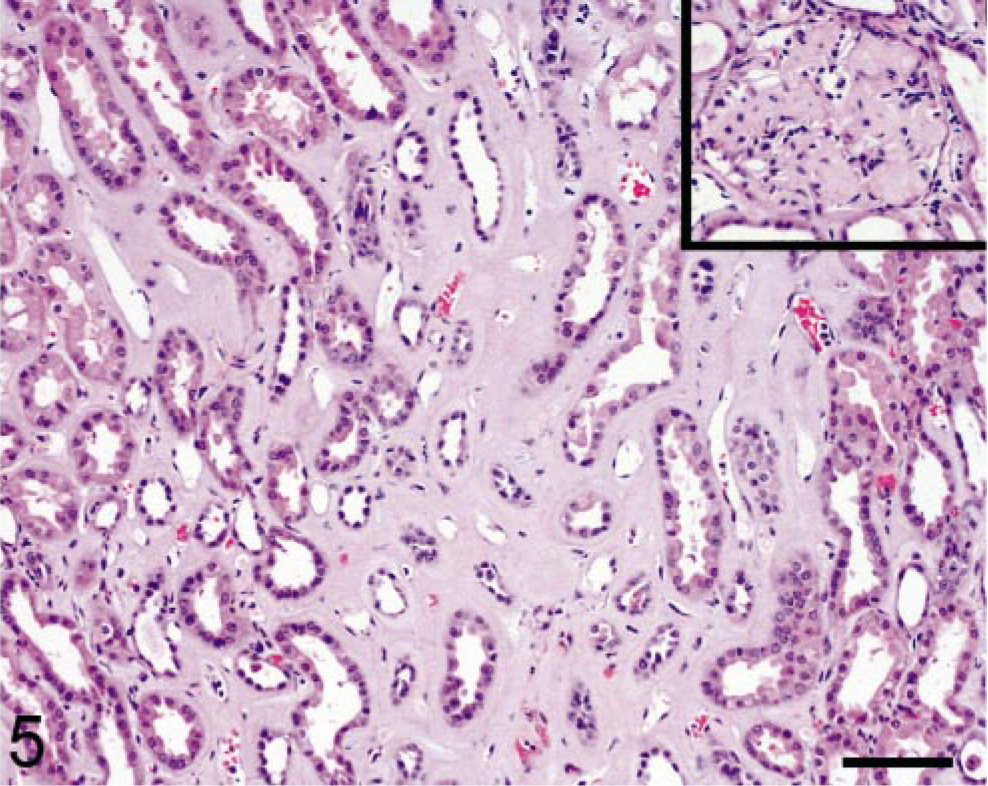
Kidney; marmoset. Marked amyloid accumulation within the renal interstitium and glomeruli (inset). HE. Bar = 95 μm.
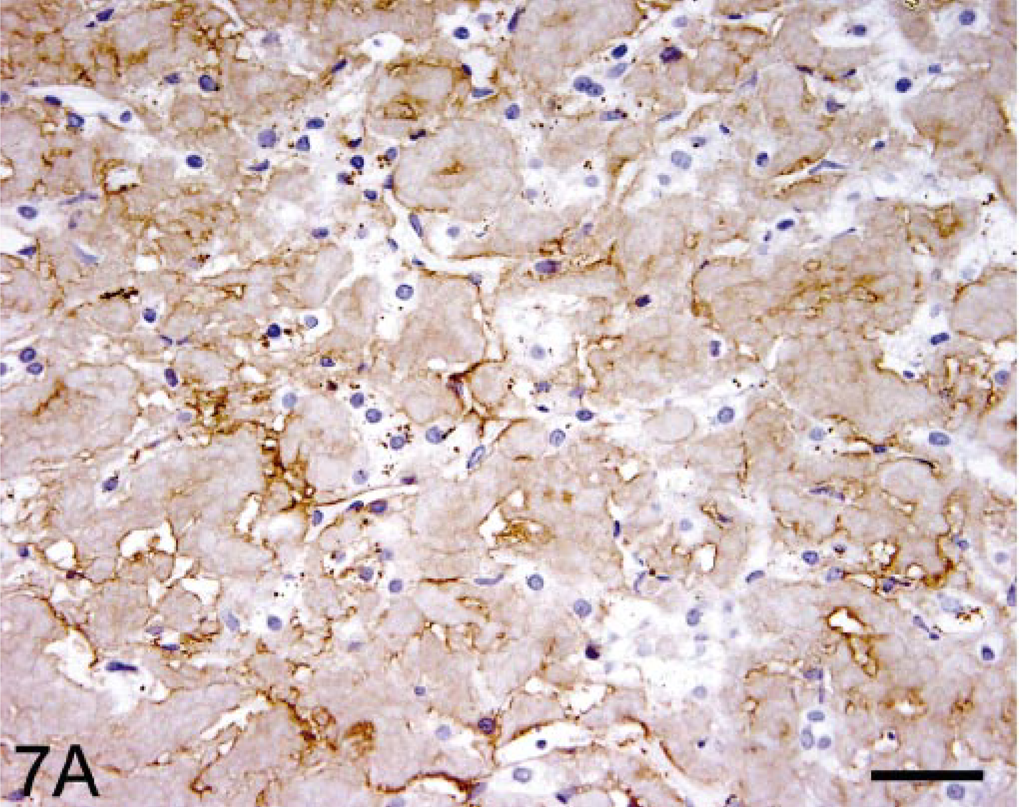
Immunohistochemical studies of hepatic tissue revealed that the amyloid deposits were recognized by the anti-human SAA mAb (Fig. 7) but reacted only weakly with the anti-mouse SAA polyclonal antiserum. 26 Immunostaining was most intense using the mAb prepared against the AA protein extracted from the liver of the index case (not illustrated). In other experiments, this reagent also was shown to bind human, murine, and bovine AA-related amyloid.
To verify chemically that the hepatic amyloid was indeed AA in nature, fibrils were extracted from 0.36 g (wet weight) of fresh-frozen tissue, where ≈ 20 mg of water-soluble protein was recovered. When this material was examined by SDS-PAGE, the major band had a molecular mass (Mr) of ≈ 8 kd (Fig. 8). After transfer onto a PVDF membrane, the sequence of the first 37 residues of this component was determined by Edman degradation. As shown in Fig. 9, the protein was highly homologous in primary structure to that of rhesus monkey and human AA, differing only by six and four amino acids, respectively (the absence of the N-terminal arginine [R] and serine [S] residues has been noted in AA proteins of other species, e.g., cats, cows, dogs, horses 12 ).
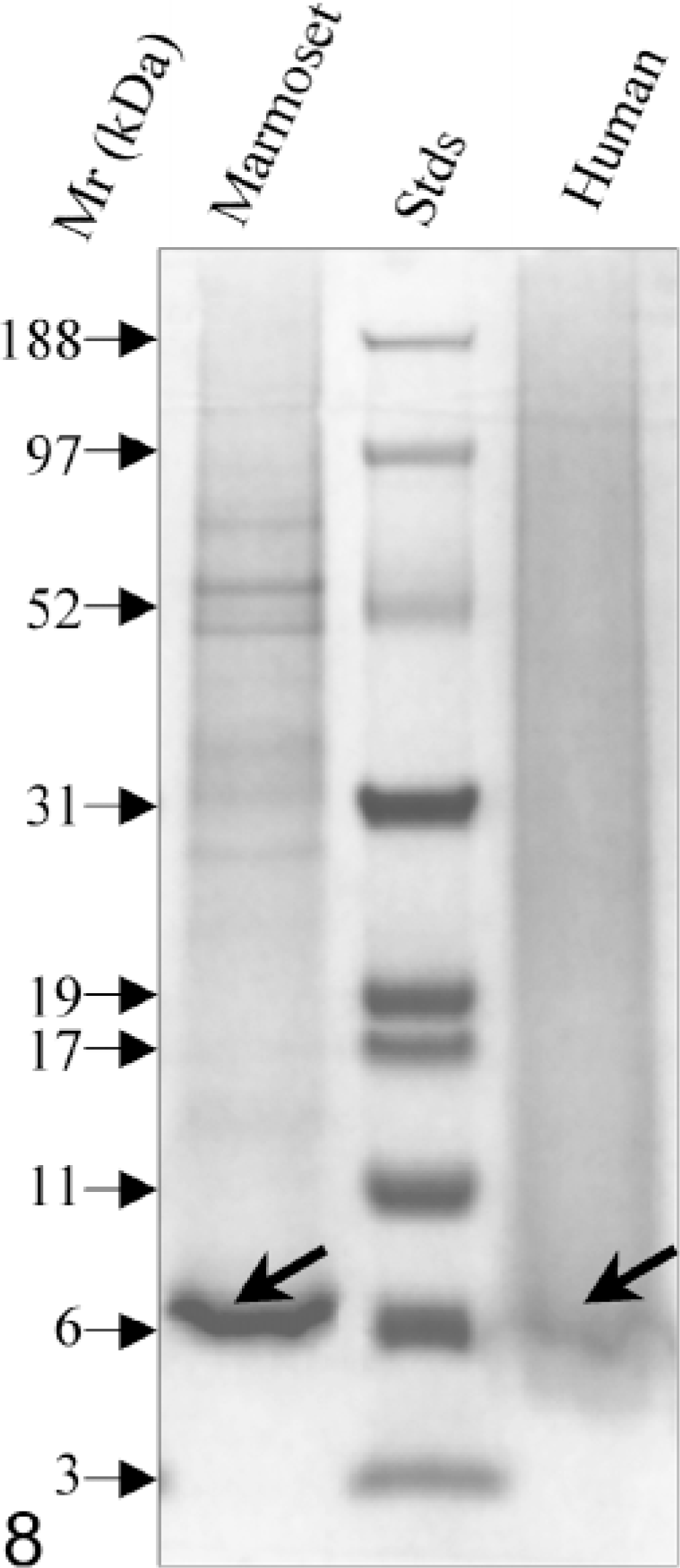
SDS-PAGE of amyloid fibrils extracted from the liver of a common marmoset and from a human with AA amyloidosis. As evident from the location of the reference standard proteins, the molecular mass of the amyloid proteins are ≈ 8 kd. Mr = molecular mass.
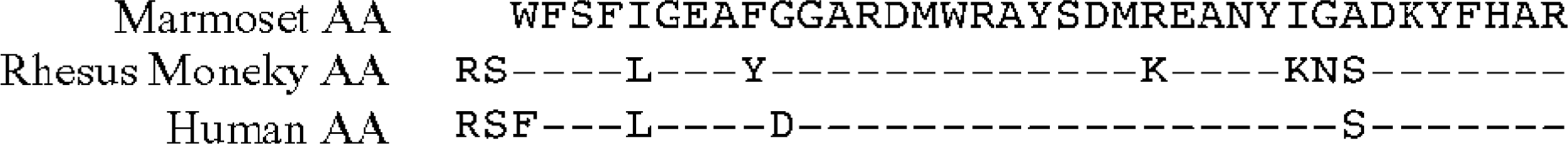
Comparison of the N-terminal amino acid sequence of marmoset, rhesus monkey, and human AA proteins. The residues are indicated by the one-letter code; (−); sequence identity.
Retrospective analyses
The diagnosis of systemic AA amyloidosis in the index case led to our review of tissue sections and clinical records of the 86 common marmosets in our colony greater than 1 year of age that had been euthanatized because of morbidity (weight loss not responsive to treatment) and consequent failure to thrive during the ≈ 9-year period between 1 February 1991 and 1 June 2000. On the basis of histochemistry, 15 of the 86 animals (17%) had amyloid in one or more tissues with the following distribution: small intestine, 12 of 15 (80%); liver, 11 of 15 (73%); adrenal glands, 11 of 15 (73%); renal glomeruli, 9 of 15 (60%); stomach, 9 of 15 (60%); colon, 9 of 15 (60%); renal interstitium, 7 of 15 (47%); and spleen, 7 of 15 (47%). In the case of hepatic amyloid, the deposits appeared first in the space of Disse and, in severe cases, progressed to involve more than half of the organ, diffusely disrupting and compressing hepatic cords. Gastric and colonic deposits were located predominantly in the villus tips (Fig. 4). Both adrenal cortex and medulla were affected and, in one case, less than 5% of normal tissue remained. Three animals had involvement of the renal glomeruli (Fig. 5, inset), as well as interstitium. Amyloid accumulated in the red and white pulp of the spleen, causing atrophy of the lymphoid tissue. Depending on the principal organ involved, the clinical manifestations of disease included hepatomegaly, chronic renal disease, diarrhea, and weight loss. Seven of the affected marmosets had a normochromic, normocytic anemia; an elevated alkaline phosphatase level and hypoalbuminemia were documented in another three.
The mean age at death of the amyloidotic C. jacchi differed significantly from unaffected animals (6.75 versus 4.58 years; Wilcoxon Mann-Whitney test, Z = 3.54, P < 0.0002). The youngest that was found to have amyloid was 2.73 years old. Although 10 of the 15 affected marmosets were female, this predominance was not statistically significant since 57 of the 86 in the sample population (66%) were of this sex. In one family, both parents had systemic amyloidosis; however, only one of five offspring that survived to at least age 4 years developed the disease.
To determine whether the 15 diseased marmosets had other significant pathologic findings, as compared with the 71 nonamyloidotic animals, tissue was obtained postmortem from all and subjected to detailed microscopic examination. Notably, one affected animal had no inflammatory-related pathology and, in two others, the only abnormality noted was extramed-ullary hematopoiesis. The frequency of chronic lymphocytic enteritis (CLE) did not significantly differ in amyloidotic versus nonamyloidotic animals. Although there was a higher incidence of interstitial nephritis, cholecystitis, hemosiderosis, etc. in the amyloid-bearing marmosets, this difference was not statistically significant (Table 1). Enteropathic Escherichia coli was found in 14 animals, 13 of which were amyloid free.
Concurrent disease in marmosets with and without systemic amyloidosis.∗
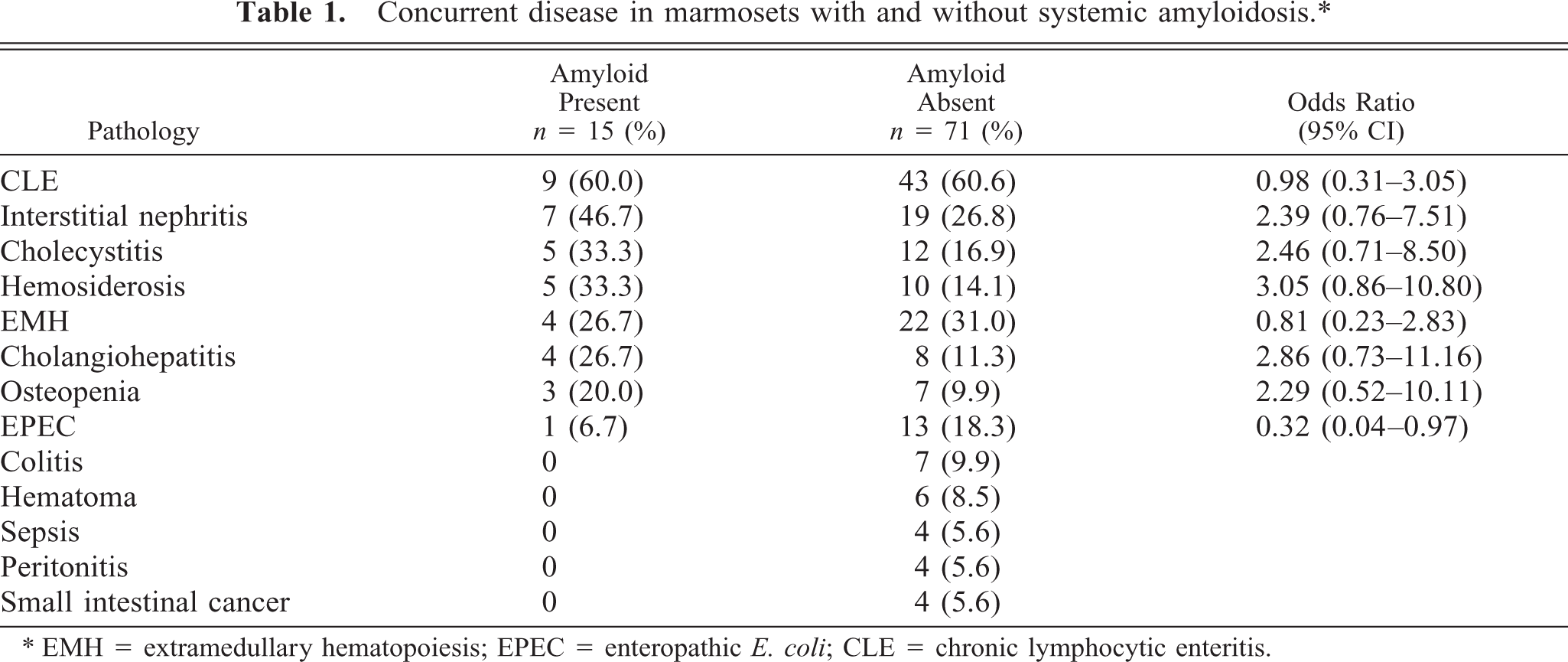
∗ EMH = extramedullary hematopoiesis; EPEC = enteropathic E. coli; CLE = chronic lymphocytic enteritis.
Serial serum specimens were available for quantitative SAA assays from only the index case and 2 of the 15 amyloidotic marmosets. Because of this small number and the lack of material from nonaffected animals, we analyzed sera from 16 and 31 additional amyloid-bearing and amyloid-free marmosets, respectively. As determined by ELISA, the SAA concentrations in 17 of the 19 amyloidotic group ranged from 3 to 173 μg/ml (mean ± SEM, 37.9 ± 5.9) and in the 31 without amyloid, from 3 to 131 μg/ml (mean ± SEM, 30.6 ± 4.6). This difference was not statistically significant (unpaired t-test, P = 0.36). Of the two amyloidotic animals with SAA levels > 400 μg/ml, only in one were multiple samples available for analysis. In this animal, the initial value was 530 μg/ml and then 32 and 54 months later, 13.3 and 48.6 μg/ml, respectively. Notably, when SAA levels from all 19 affected and 31 nonaffected primates were analyzed statistically, there were no significant differences between the groups (unpaired t-test, P = 0.14).
According to the manufacturer, the anti-SAA antibody provided in the kit is designated “multispecies” because of its reactivity with at least five different sources of this apolipoprotein, e.g., human, canine, bovine, equine, and ovine AA amyloid. To demonstrate that this reagent also recognized marmoset SAA, this component was isolated from marmoset serum by differential ultracentrifugation using a sodium bromide gradient. The SAA nature of the resultant product was evidenced by its ability to inhibit totally the interaction of the human SAA standard furnished in the kit. Furthermore, SDS-PAGE revealed a major protein band of ≈ 12 kd that, on western blotting, reacted with the manufacturer's anti-SAA antibody and, by sequence analysis, was found to be identical to the N-terminal portion of the marmoset AA protein.
Discussion
We have determined that ≈ 17% of common marmosets in our colony had pathologic deposits in various body tissues that possessed the characteristic tinctoral and ultrastructural features of amyloid; namely, they were congophilic and exhibited green birefringence when examined under polarized light and, by electronmicroscopy, were composed of ≈ 10-nm-diameter fibrils. Immunohistochemical studies revealed that this material reacted with both the specific anti-human SAA mAb and anti-marmoset AA mAb. Chemical analyses of amyloid extracted from the liver of an affected animal unequivocally established its AA nature and, furthermore, that the fibrils were composed of an N-terminal fragment of SAA. This C. jacchus component had identical amino acids at 29 of 37 positions when compared with human and rhesus monkey AA and a Mr (≈ 8 kd) similar to that found for the ≈ 76-residue AA-related protein isolated from human or animal tissues. 10, 12, 13, 16, 19 These eight variations, as well as other sequence differences occurring in the remaining ≈ 39 residues of the marmoset AA, may have accounted for the limited reactivity of the anti-mouse antiserum. In contrast, this material stained intensely with the monoclonal anti-human SAA reagent and the anti-marmoset AA mAb. The development of systemic AA amyloidosis clinically or that induced experimentally 20 is typically associated with a chronic inflammation or infection and attendant elevation of the amyloidogenic SAA precursor molecule. 10, 19, 22, 23 Among the 15 amyloidotic marmosets, 3 had no histologic evidence of inflammatory disease, and the pathologies seen in the 12 others (e.g., CLE) also were present in the 71 unaffected animals. On the basis of the limited number of specimens available for analysis, the SAA concentrations in the two groups were not significantly different; however, to confirm this finding will require that multiple (i.e., monthly) serial specimens be collected from marmosets with and without amyloid. The absence of an identifiable inflammatory process that would distinguish affected from nonaffected animals and the relatively high frequency of amyloidosis in the colony in this study suggest an alternative explanation for the development of seemingly spontaneous AA amyloidosis in the common marmoset; namely, a genetic factor, as occurs in Abyssinian cat siblings. 6 Given the polymorphic nature of SAA in humans and other species, 2, 10, 12, 13 and the fact that certain isotypes are preferentially deposited as amyloid, 3, 5, 24, 25 it is possible that inheritable variations in the primary structure of this molecule among marmosets may be responsible for its amyloidogenicity, e.g., that animals carrying a particular SAA allele would be prone to develop AA amyloidosis. In this regard, we plan to clone the SAA-encoding DNA from the livers of affected and nonaffected animals and compare the predicted protein sequences with that of the deposited protein. If indeed, there are putative differences in the apolipoprotein SAA isotype distribution between the two marmoset populations, this (or possibly other) element(s) could account for the 2 year longer mean age at death of those with amyloidosis.
It also should be noted that the rate of amyloid formation by amyloidogenic isotypes of SAA can be accelerated by an agent designated “amyloid enhancing factor” (AEF). 11, 19 This material, which consists of fibrils extracted from amyloidotic organs, serves as a seed or nucleus to promote fibrillogenesis and is biologically active in fentamolor doses when injected parenterally or administered orally. 11 Thus, it is possible that amyloid development in the marmoset families housed together in the colony in this study may have been initiated through ingestion of AEF. 14
The discovery that common marmosets can spontaneously develop AA amyloidosis provides a unique opportunity to study in a primate model the pathogenesis of this disease and to develop therapies that can ameliorate or prevent this or other systemic amyloid-related disorders.
Footnotes
Acknowledgements
We thank Sallie D. Macy, Shuching Wang, Teresa K. Williams, Dennis A. Wolfenbarger, and Craig Wooliver for technical assistance; Ronda L. Reed for manuscript preparation; Kristen Toohey for photographic assistance; M. S. Kindy for the anti-AA reagents. This work was supported by NIH/ NCRR Grant P51RR000168–41 (KGM) and Research Grant CA10056 from the National Cancer Institute and the Aslan Foundation (AS). AS is an American Cancer Society Clinical Research Professor.
