Abstract
Amyloidoses are a group of protein-misfolding disorders that are characterized by the deposition of amyloid fibrils in organs and/or tissues. In reactive amyloid A (AA) amyloidosis, serum AA (SAA) protein forms deposits in mice, domestic and wild animals, and humans that experience chronic inflammation. AA amyloid fibrils are abnormal β-sheet–rich forms of the serum precursor SAA, with conformational changes that promote fibril formation. Extracellular deposition of amyloid fibrils causes disease in affected animals. Recent findings suggest that AA amyloidosis could be transmissible. Similar to the pathogenesis of transmissible prion diseases, amyloid fibrils induce a seeding-nucleation process that may lead to development of AA amyloidosis. We review studies of possible transmission in bovine, avian, mouse, and cheetah AA amyloidosis.
Keywords
Amyloidosis refers to a group of protein-misfolding disorders. Various proteins that are soluble under physiological conditions can undergo conformational changes to the β-sheet–rich structure and subsequently self-assemble into highly insoluble amyloid fibrils. At present, at least 28 different proteins have been shown to be amyloidogenic in humans and animals and are associated with disorders such as Alzheimer disease, prion diseases, type II diabetes mellitus, familial amyloid polyneuropathy, and reactive amyloid A (AA) amyloidosis. 61 Amyloidoses can be divided into 2 major classes: localized and systemic. In localized amyloidoses, amyloid fibrils are deposited in organs such as the brain and pancreas, in which precursor proteins are synthesized. On the other hand, in systemic amyloidoses, serum precursor proteins, such as immunoglobulin light chain in amyloid light chain (AL) amyloidosis, transthyretin in familial amyloid polyneuropathy, and β2-microglobulin in dialysis-related amyloidosis, circulate in the blood and polymerize to form amyloid fibrils that are then deposited throughout the body. 61 AA amyloidosis is a systemic type of amyloidosis and occurs in domestic, laboratory, and wild animals that experience chronic inflammation. 8,26,34 In humans, systemic AA amyloidosis can develops in patients with chronic inflammatory diseases, including rheumatoid arthritis. 21,50 Several recent studies have shown that susceptibility to AA amyloidosis in mice, cheetahs, chickens, and rabbits can be increased via the peripheral route, as well as orally through ingestion of the amyloid fibrils themselves. 47,64,82
The lag phase of disease onset is markedly shortened by the injection of AA amyloid fibrils isolated from amyloid-laden organs, suggesting that prion diseases and AA amyloidosis share a common pathogenic mechanism and may be transmitted by a prion-like infectious process that involves a seeding-nucleation–dependent mechanism. 39 Once nuclei or amyloid fibrils are added to a solution containing the normal form of amyloid protein monomers, the recruitment and attachment of proteins to the ends of existing fibrils become energetically favorable and result in the rapid growth of amyloid fibrils. 28,48 Here, we discuss important topical studies on the transmission of AA amyloidosis in domestic, laboratory, and wild animals.
Transmission of Bovine AA Amyloidosis
Clinical Signs and Histological Findings
Bovine AA amyloidosis is a relatively rare fatal disease in cattle that is characterized by nephrosis accompanied by diarrhea and edema. 26 Bovine AA amyloidosis occurs in adult cattle with chronic inflammatory disorders, such as chronic mastitis or arthritis, but amyloidoses without concurrent disorders are also occasionally observed. 16,58,59,77
Massive edema and enlargement of the kidneys are typically observed at autopsy in animals affected by AA amyloidosis. 58,59 Kidneys with amyloid deposits are grossly irregular and enlarged, waxy-yellow in color, with widened cortices. On the cut surface, amyloid deposits in glomeruli and medulla are stained brown by an iodine reaction test. In extra-renal organs, however, macroscopic changes may be subtle or absent, even if the amyloid deposition in tissues is extensive. 45
In bovine AA amyloidosis, amyloid deposits in systemic organs, including edible tissues such as skeletal muscles and intestines, can be observed by histological analysis. 45,77 Severe amyloid deposits are typically found in the kidney, particularly in the renal glomeruli and renal medulla, 16,58,59 and can cause clinical episodes of renal disease. However, since not all affected cattle exhibit clinical signs, 45 histopathologic analysis by autopsy or biopsy is essential for a definitive diagnosis of bovine AA amyloidosis.
Bovine Serum Amyloid A
The precursor protein of AA is serum AA (SAA), which is an acute-phase protein that is synthesized mainly in the liver in response to inflammation. 11 Sustained high SAA concentrations in serum can lead to aggregation of AA fibrils by seeding-dependent extension. In cattle affected by AA amyloidosis, serum SAA concentrations are significantly higher than those in unaffected cattle. 67 Seven SAA isoforms are known in cattle, and those with isoelectric points of 5.2 and 8.6 are thought to have amyloidogenic potential. 68 However, the amino acid sequence of either isoform has not been fully elucidated, and homology with amino acid sequences of AA proteins deposited in organs has not yet been clarified. On the other hand, antibodies specific for the bovine SAA isoforms are now available and are expected to be useful in the diagnosis and investigation of bovine AA amyloidosis. 66
Transmission of Bovine AA Amyloidosis to Other Species
In experimental animal systems, AA amyloidosis can be induced by repeated long-term inflammatory stimulation. 27,41,53,62 In these systems, the period required for inducing AA amyloidosis can be markedly shortened by injecting an amyloid-enhancing factor (AEF) at the same time as delivery of the inflammatory stimulus. 1,29 The essential mechanism of action of AEFs is thought to involve amyloid fibrils that may work through a seeding-nucleation mechanism. AEFs can sometimes act as cross-seeding factors among different animal species. 9,10,25,38,44,63,78
Bovine amyloid fibrils also exhibit AEF activity in experimental animal systems. Cui et al 9,10 reported the development of AA amyloidosis in mice following the simultaneous oral administration of semi-purified bovine AA fibrils and exposure to an inflammatory stimulus. In these studies, bovine AA fibrils appeared to behave solely as seeds for amyloid formation, because all of the deposited AA fibrils were derived from murine SAA. Furthermore, the incidence, transmission rate, and severity in the extent of amyloid deposits were dependent on the injected dose of AA fibrils, although the extent of amyloid deposits was lower than when mice were administered mouse AA fibrils. These results indicated that, as in most prion diseases, there is a barrier to cross-species transmission of AA amyloidosis. This species barrier is considered to depend on the primary amino acid structure of the N-terminus of SAA. 38 Bovine AA amyloidosis can be transmitted to mice by intravenous, intraperitoneal, and oral routes, with oral transmission being the least virulent of these modes of transmission. The AEF activity of amyloid fibrils is thus considered to be both dose and route dependent.
Similar to transmission in the murine system, we previously investigated the development of AA amyloidosis in rabbits after exposure to bovine AA amyloid fibrils. 25,44 In those previous studies, rabbits were subjected to intravenous administration of bovine amyloid fibrils accompanied by exposure to an inflammatory stimulus. While few rabbits developed AA amyloidosis, those that did had preexisting ulcerative pododermatitis, a condition known as “sore hocks.” Further investigation revealed that the bacterial infection responsible for sore hocks played an important role in the development of AA amyloidosis, indicating that transmission of bovine AA amyloidosis was also possible in animals other than mice. In the animals used in those studies, the combined effect of bacterial infection and other diseases was considered to enhance cross-species transmission of AA amyloidosis.
Transmission of Avian AA Amyloidosis
Spontaneous Avian Amyloidosis
Avian AA amyloidosis occurs in adult birds as a consequence of inflammatory disorders such as tuberculosis. 8,34 AA amyloidosis is not a rare disease in captive birds and occurs most frequently in waterfowl, which are known to develop AA amyloidosis secondary to bumblefoot (ulcerative pododermatitis) caused by Staphylococcus spp infection. 4,13,71 Furthermore, Pekin ducks also develop age-associated AA amyloidosis. 55 Chicken AA amyloidosis is referred to as amyloid arthropathy and is associated with Enterococcus faecalis infection, while spontaneous chicken systemic AA amyloidosis is less common. 33,65,80
Induced Avian Amyloidosis
Birds raised for human consumption may be subject to stressful environmental conditions and occasionally develop AA amyloidosis. Commercial foie gras, which is produced by force feeding geese, has been reported to contain AA fibrils at a high incidence. 63 Furthermore, we observed that chickens develop AA amyloidosis after inoculation with multiple vaccines (Figure 1). 46 Using the same principle, avian AA amyloidosis can be induced experimentally. In ducks and chickens, AA amyloidosis can be induced by repeated inflammatory stimulation, such as by inoculation with crude bacterial extracts or vaccination with oil-emulsified bacterins. 14,36,37 Antigenic stimulation with bacteria is thus considered to promote the development of avian AA amyloidosis.
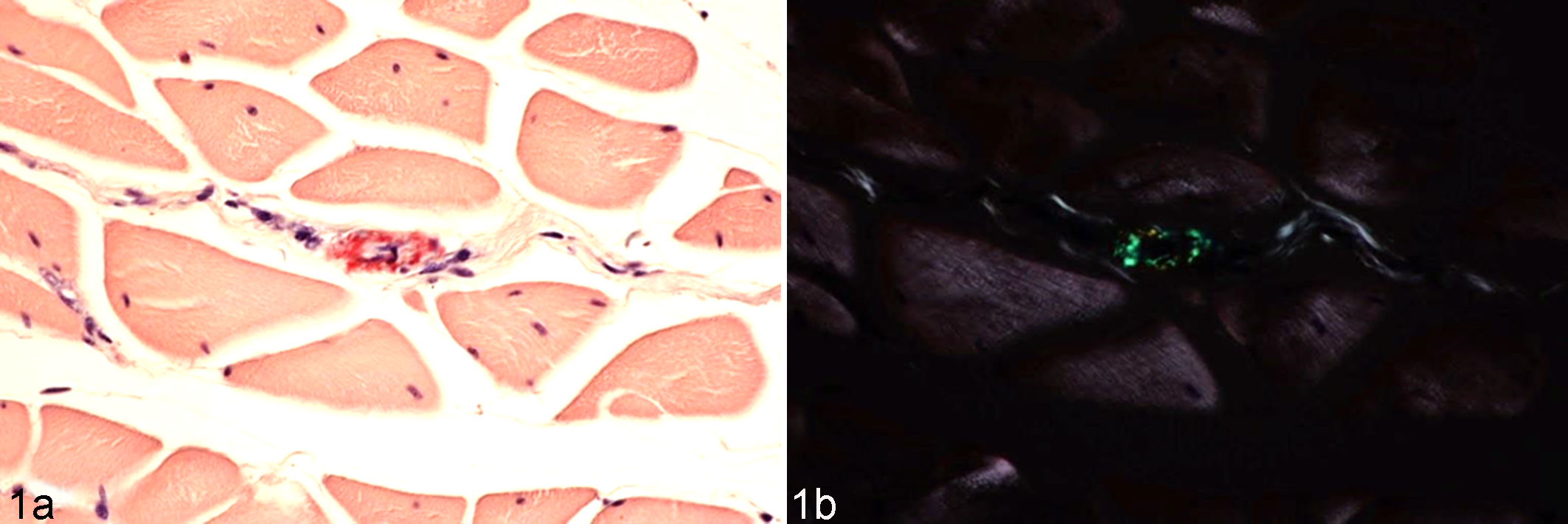
Histological images of amyloid deposits in pectoral muscle; amyloid A (AA) amyloidosis-affected chicken. Congo red staining. (a) Amyloid deposits in small vessel wall are stained red. (b) Amyloid deposits observed as green birefringence under polarized light.
Transmission of Avian AA Amyloidosis
Avian AA amyloidoses frequently manifest as epidemic outbreaks in wild and captive fowl populations. 51,56,60,71 When considering the transmissible properties of AA amyloidosis, the horizontal transfer of amyloidosis by ingestion of amyloid-contaminated feed or feces is suspected to occur among avian species. Indeed, several recent studies have suggested that avian AA amyloidosis is transmissible.
We investigated whether chicken AA amyloidosis can be transmitted within a species following either intravenous or oral delivery. 47 Chickens were administered a single intramuscular dose of inactivated Salmonella enteritidis vaccine before being inoculated with chicken amyloid extracts either intravenously or orally. Interestingly, while chicken AA amyloidosis was induced in the intravenous inoculation group as well as the oral inoculation group, the patterns of organ involvement differed between both groups. In the intravenous inoculation group, amyloid deposits were distributed homogeneously in all organs, while in the oral inoculation group, severe amyloid deposits were observed only in the spleen. This finding suggested that AA fibrils in the intestine might be taken up by lymphoid tissues, such as Peyer’s patches, and then initially transported to the spleen.
The transmission of avian AA amyloidosis is not restricted to the same species; it can also occur between species. Liu et al 38 showed that swan AA fibrils exhibit AEF activity in mice and that the induction of murine AA amyloidosis can be enhanced by intravenous administration of swan AA fibrils. Furthermore, AA fibrils extracted from duck or goose foie gras exhibit potent AEF activity following oral administration in transgenic mice engineered to produce human interleukin (IL)–6. 63 These experiments demonstrated that avian AA amyloidosis can be transmitted orally between different species.
Transmission of Mouse AA Amyloidosis
Spontaneous and Experimental AA Amyloidosis in Mice
Numerous reports have examined spontaneous or senile amyloidosis in mice since the first description of renal glomerular lesions by Gorer in 1940. 19 To date, 2 amyloid proteins have been found to form spontaneous deposits in mice. The first amyloid fibril protein that was demonstrated biochemically and immunochemically to be an AA protein was found in non-inbred male General Purpose White Swiss (GP) mice 18 and obese hyperglycemic mice. 74 AA amyloidosis in mice has been associated with chronic inflammation. For example, AA amyloid deposition can be induced experimentally in mice by repeated injection of silver nitrate, casein, or lipopolysaccharide, which greatly increases the concentration of circulating SAA. 24 These spontaneous and experimental AA amyloidoses are characterized by the systemic deposition of extracellular fibrils composed of SAA protein or proteolytic fragments, primarily in the spleen, kidney, small intestine, liver, and heart.
The other amyloid protein deposited spontaneously in mice is apolipoprotein A-II (apoA-II), which circulates in the blood associated with serum high-density lipoprotein (HDL) and forms amyloid fibrils (AApoAII) in age-associated systemic amyloidosis. 22 Mouse AApoAII amyloidosis is characterized by the systemic and extracellular deposition of amyloid fibrils composed of apoA-II, primarily in the liver, tongue, heart, stomach, small intestine, spleen, kidney, and blood vessel walls in aged mice. 69
The lag phase of AA amyloidogenesis can be markedly shortened by coinjection of amyloid materials extracted from mouse tissues, such as the spleen and liver, together with an acute or chronic inflammatory stimulus. 9,39,40 While it typically takes several weeks and repeated administration of inflammatory stimulus to induce the development of amyloid deposits, AA amyloidogenesis is detectable within 48 hours after coadministration of inflammatory stimulus plus amyloid extracts. 30 These findings suggested that experimental mouse AA amyloidosis may be transmitted by a prion-like infectious process. 73 Indeed, prion-like transmission was recently demonstrated for mouse AApoAII amyloidosis. 23,75,76
Mouse Serum Amyloid A
Four SAA genes have been identified in the mouse genome. The Saa1 and Saa3 genes are located with an interval of ∼30 kb on chromosome 7 with Saa4 located within this interval. 12 The Saa2 gene is located in a head-to-head orientation with the Saa1 gene with an interval of ∼20 kb. SAA1 and SAA2 are acute-phase proteins and are synthesized mainly in the liver in response to cytokines, such as IL-1, IL-6, and tumor necrosis factor–α (TNF-α). Only SAA1 is deposited as amyloid fibrils in mice. 79 The plasma concentration of SAA proteins increases rapidly from 1 to 5 μg to ∼1000 μg/ml in less than 24 hours during the acute-phase response to tissue injury and inflammation. SAA proteins circulate in the blood as apolipoproteins associated with HDL. Recently, SAA was reported to act as a signal for macrophages to secrete cytokines, which would in turn induce hepatic SAA synthesis. Once this cycle is established, higher SAA levels may be maintained during inflammation, and amyloid deposition can be accelerated. 3,5 However, further studies should be undertaken to clarify whether the use of delipidated or aggregated forms of SAA stimulates cytokine secretion by macrophages. 31,35
Transmission of Cheetah AA Amyloidosis
AA Amyloidosis in Captive Cheetahs
The cheetah (Acinonyx jubatus) is in danger of extinction. Consequently, considerable efforts have been made to breed cheetahs in zoos to increase their numbers and prevent extinction. However, at group breeding facilities with high rearing densities, the prevalence of AA amyloidosis associated with a high incidence of chronic lymphoplasmacytic gastroenteritis is a major hindrance to increasing the population. 52
We examined the possibility of horizontal transfer or transmission of AA amyloidosis in a group of captive cheetah with severe AA amyloid deposition in the kidney, liver, and spleen, as well as in other organs. 82 The AA fibril protein isolated from the liver was equivalent to the N-terminal part of the precursor protein SAA (the longest AA peptides consisted of amino acids 1–93 of the 111–amino acid, full-length precursor protein). While AA amyloid fibrils were not detected in cheetah urine, cheetah feces unexpectedly contained AA amyloid fibrils that differed from those seen in the liver. Compared with liver amyloid fibrils, fecal amyloid fibrils had a smaller molecular weight due to deletion of both N- and C-terminal sequences, a shorter and finer fibrillar shape on electron microscopy, 82 and increased sensitivity to denaturation treatments, such as incubation in 6 M guanidine hydrochloride solution. Interestingly, comparison of the transmissibility of fecal and liver AA amyloid fibrils using the mouse experimental AA amyloidosis system showed that the fecal amyloid fibrils from the cheetah exhibited significantly greater transmissibility. 82 In yeast, transmission of the amyloid fibril form of the Sup35 protein produces a [PSI+] phenotype. The metabolic phenotype [PSI+] results from conversion of soluble active monomers to an inactive amyloid of the yeast Sup35 translation termination factor. 57 It has been reported that distinct strains of the yeast prion [PSI+] exist and that strains exhibiting stronger transmission activity form less stable fibrils compared with strains exhibiting weaker transmissibility in general. 32,70 Amyloid fibrils were also detected in the feces of mice with AApoAII amyloidosis, and the injection of these fibrils was observed to induce amyloidosis in mice (Fig. 2). 75
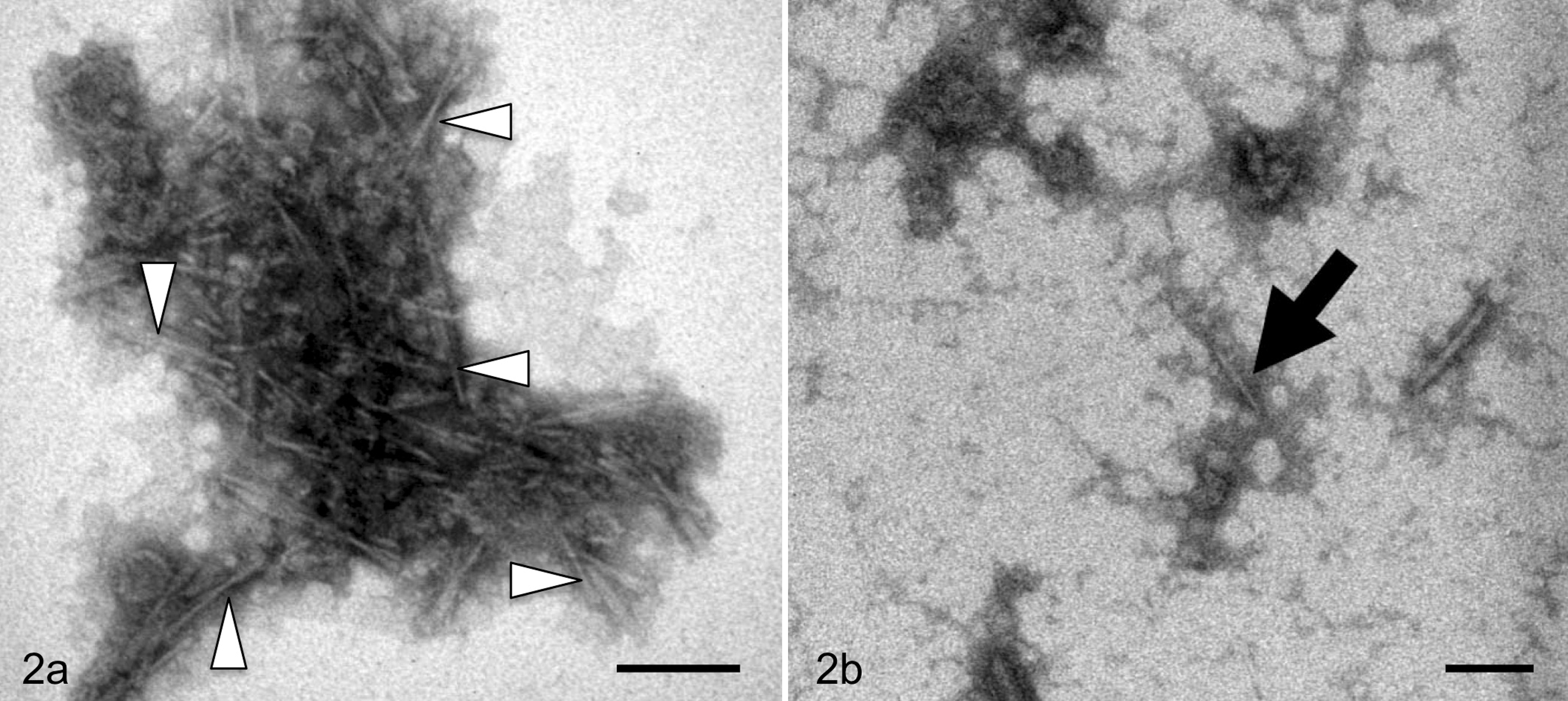
Analysis of amyloid protein in feces from mice with amyloidosis. The AApoAII amyloid fibrils from the (a) liver and (b) feces were observed by transmission electron microscopy. Open triangles indicate characteristic amyloid fibrils. The arrow shows fecal-specific fibrils. Scale bar, 100 nm.
Therefore, we propose that feces are a potential transmission vehicle and may be ingested by licking, consumption together with food, or inhalation. Ingested amyloid fibrils with higher transmissibility may accelerate AA amyloidosis in captive cheetah populations. The finding of possible transmission of cheetah AA amyloidosis is important for understanding the propagation and pathogenesis of AA amyloidosis, because it suggests that the transmission of AA amyloidosis, which has been observed only in the laboratory to date, may also occur in wild or captive populations of animals. Why is the incidence and transmission of AA amyloidosis high in cheetahs? Although chronic higher blood concentrations of SAA due to chronic gastritis are a necessary prerequisite for cheetah AA amyloidosis, genetic factors such as the structure of precursor SAA may also play a role.
Cheetah Serum Amyloid A
Given that the high incidence of AA amyloidosis in cheetahs may be due to unique features of the SAA gene in this species, we cloned and characterized the cheetah SAA1 gene. 6,81 Genome walking and polymerase chain reaction (PCR) cloning analysis revealed that cheetahs have 4 SAA genes on the same chromosome: SAA1A, SAA1B, SAA3A, and SAA3B. Of these, SAA1A and SAA1B genes show a high degree of nucleotide sequence identity, and the SAA1A and SAA1B proteins have the same amino acid sequences. Similarly, the SAA3A and SAA3B genes are homologous, but the SAA3A and SAA3B proteins have several amino acid substitutions. Notably, only the SAA1A protein associates to form AA amyloid fibrils, and SAA1B messenger RNA (mRNA) is not expressed in the cheetah. 6
These findings suggested a model for SAA gene evolution in cheetahs. Specifically, the ancestral SAA gene first duplicated and gave rise to the SAA1 and SAA3 genes. Subsequently, the SAA1 and SAA3 genes were then duplicated one more time, giving rise to 4 genes (SAA1A, SAA1B, SAA3A, and SAA3B) in the cheetah genome.
Screening of 51 cheetahs autopsied in Japan revealed nucleotide polymorphisms only in noncoding regions in the SAA1A gene, implying that the cheetah SAA1A proteins that formed AA amyloid fibrils were monomorphic among cheetahs. In contrast, single-nucleotide polymorphisms (SNPs) in the 5' flanking region (–13T/C) and in the coding region (2295 C/T and 3010 C/T) of the SAA1 gene constitute risk factors for development of AA amyloidosis in humans. 2,42,43 The finding that all cheetahs have the same SAA1A protein sequence may be an important factor underlying the very high incidence of AA amyloidosis in the cheetah. Further studies are required to characterize the amyloidogenicity of the cheetah SAA1 protein.
Discussion
Risk of Amyloid in Slaughtered Cattle and Birds
Among food animals, AA amyloidosis is observed mainly in cattle and birds. Although bovine AA amyloidosis is a rare disease in clinical practice, amyloid deposits in slaughtered cattle were observed at a rate of 0.8% to 5%. 17,72 In bovine AA amyloidosis, amyloid deposits were observed in edible parts of the animal, including the liver, digestive organs, and skeletal muscle. 77 Furthermore, some cattle with amyloidosis do not have clinical signs, nephrosis, or amyloid deposits in the glomeruli, despite the presence of severe amyloid deposits in their extrarenal organs. 45
Broiler chickens are considered resistant to amyloidosis, since they are slaughtered at an early age. However, egg-laying hens have longer life spans than broilers and may therefore develop AA amyloidosis due to repeated vaccinations. 46,49 After culling of these hens, certain body parts are also consumed as processed meat products. Our surveillance, albeit limited, revealed that amyloid deposits were rarely observed in commercial broiler chicken livers. However, we detected minute amyloid deposits in culled egg-laying chickens that had been administered multiple vaccinations during the rearing period, and these animals developed asymptomatic amyloidosis (unpublished data). In these birds, amyloid deposits were observed mainly in the muscles where the inoculations were delivered (Fig. 1). As described previously, amyloid deposits were observed in commercial foie gras, and the susceptibility of Pekin duck to AA amyloidosis has been observed to increase with age. 55,63 Thus, amyloid fibril deposits in animals intended for human consumption appear to be more insidious than initially thought.
Since bovine and avian AA amyloidosis can be transmitted between different species, 9,10,25,38,44,63,78 the presence of amyloid in edible meat may prove to be a public health hazard. Although there is a barrier to cross-species transmission of AA amyloidosis, 9,10 and the low levels of amyloid fibrils in muscle may indeed lessen the risk of transmission, the precise details of the transmission mechanism remain unclear. To minimize the risk of food-borne amyloidosis, further investigations of interspecies AA amyloidosis transmission are therefore required.
Horizontal Transmission of Avian AA Amyloidosis
During epidemic outbreaks of AA amyloidosis in wild and captive animals, horizontal transfer of AA amyloidosis among the affected groups has been suspected. In chicken AA amyloidosis, severe amyloid deposits have even been observed in the lamina propria of the gut. 46 Furthermore, the high frequency with which hemorrhagic enteritis is observed in amyloidosis-affected chickens suggests that the associated mucosal damage might enhance shedding of amyloid in the feces. As avian and cheetah AA amyloidosis can be transmitted orally, 47,82 it is possible that the horizontal transfer of amyloidosis by ingestion of amyloid-contaminated feed or feces also occurs among avian species.
Perspectives
The possibility of interindividual infectivity or transmissibility of nonprion amyloidosis was revealed experimentally in 2 systemic AA and AApoAII amyloidoses in mice. More recently, the possibility of prion-like transmission of AA amyloidosis was reported in domestic and wild animals, including cattle, birds, and cheetahs. A diagram of amyloid fibril formation and potential transmission of AA amyloidosis is presented in Fig. 3. The following major questions remain: (1) how are pathological agents such as amyloid fibrils ingested, and how do they enter into recipient animals? (2) How do these agents spread throughout the body? (3) How does transmission occur across the species barrier? (4) How can we obtain epidemiological evidence that confirms whether transmission occurs between animals and humans? While our studies showed that feces appear to be involved in amyloid transmission between animals, further studies confirming these observations are necessary.
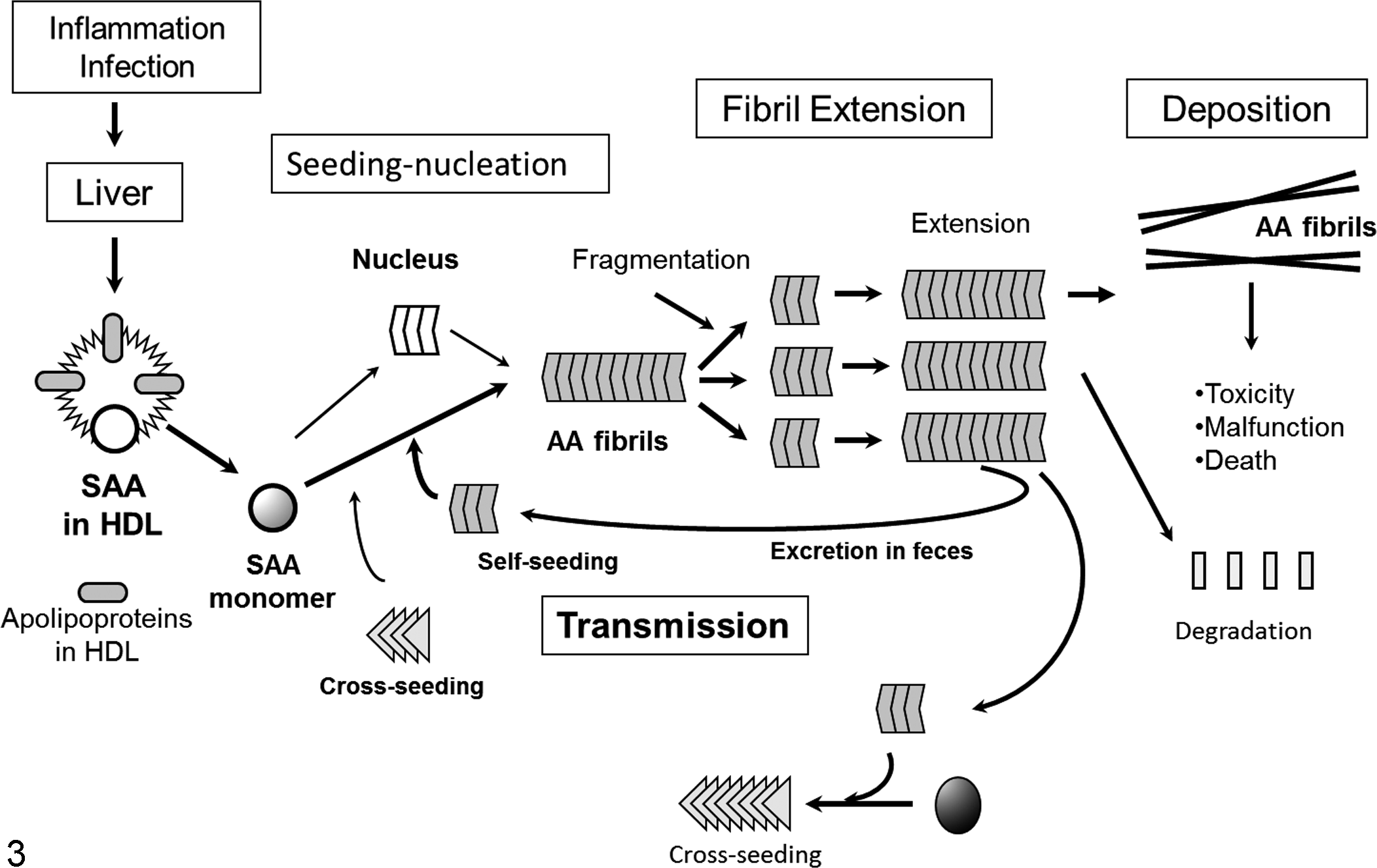
Diagram of amyloid fibril formation and potential transmission of amyloid A (AA) amyloidosis. Chronic inflammation induces serum AA (SAA) synthesis in the liver, resulting in a dramatic increase of high-density lipoprotein (HDL)–associated SAA circulating in the blood. Although spontaneous formation of the nucleus is slow, homologous or heterologous amyloid fibrils act as seeds and facilitate the formation of amyloid fibrils (self- and cross-seeding). Fragmentation of extended amyloid fibrils makes new seeds. Amyloid fibrils are possibly excreted from the body in feces, which may subsequently be ingested by other animals.
The concept of transmission via self-propagating protein structures is considered important for understanding the pathogenesis of protein-misfolding diseases, including amyloidoses. Recently, many groups have reported that neurodegenerative diseases, such as Alzheimer disease, 15 Huntington disease, 54 Parkinson disease, 20 and tauopathies, 7 can spread by cell-to-cell transmission of pathological and self-propagating amyloid-like protein structures. The findings reported here suggest that AA amyloidoses may be similarly transmitted.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Our studies were supported by Grants-in-Aid for Scientific Research (B) 23390093, Science Research on Priority Areas 22020015, and Challenging Exploratory Research (23659150) from the Ministry of Education, Culture, Sports, Science, and Technology of Japan, as well as by grants to the Research Committees for Amyloidosis from the Intractable Disease Division of the Ministry of Health, Labor, and Welfare.
