Abstract
The incidence of AA amyloidosis is high in humans with rheumatoid arthritis and several animal species, including cats and cattle with prolonged inflammation. AA amyloidosis can be experimentally induced in mice using severe inflammatory stimuli and a coinjection of AA amyloid; however, difficulties have been associated with transmitting AA amyloidosis to a different animal species, and this has been attributed to the “species barrier.” The interleukin-1 receptor antagonist knockout (IL-1raKO) mouse, a rodent model of human rheumatoid arthritis, has been used in the transmission of AA amyloid. When IL-1raKO and BALB/c mice were intraperitoneally injected with mouse AA amyloid together with a subcutaneous pretreatment of 2% AgNO3, all mice from both strains that were injected with crude or purified murine AA amyloid developed AA amyloidosis. However, the amyloid index, which was determined by the intensity of AA amyloid deposition, was significantly higher in IL-1raKO mice than in BALB/c mice. When IL-1raKO and BALB/c mice were injected with crude or purified bovine AA amyloid together with the pretreatment, 83% (5/6 cases) and 38% (3/8 cases) of IL-1raKO mice and 17% (1/6 cases) and 0% (0/6 cases) of BALB/c mice, respectively, developed AA amyloidosis. Similarly, when IL-1raKO and BALB/c mice were injected with crude or purified feline AA amyloid, 33% (2/6 cases) and 88% (7/8 cases) of IL-1raKO mice and 0% (0/6 cases) and 29% (2/6 cases) of BALB/c mice, respectively, developed AA amyloidosis. These results indicated that IL-1raKO mice are a useful animal model for investigating AA amyloidogenesis.
Keywords
Amyloidoses, which are characterized by extracellular deposits of amyloid, β sheet-rich fibrils, have been classified into more than 30 types in the medical field based on the precursor proteins. 22 Of these, amyloid A (AA) amyloidosis is frequently reported in both domestic animals 11 and humans. 22 Since the precursor of AA is serum amyloid A (SAA), an acute-phase protein, 3 AA amyloidosis is a severe complication of several chronic inflammatory diseases, including rheumatoid arthritis (RA), Crohn’s disease, systemic lupus erythematosus, and tuberculosis. 6 Sasatomi et al 20 reported that 91% of patients with AA amyloidosis were affected by RA in Japan.
AA amyloidosis has been experimentally induced in mice by exposure to inflammatory stimuli, such as a subcutaneous (s.c.) injection of silver nitrate, 14 azocasein, 24 and Freund’s complete adjuvant. 19 The additional administration of an amyloid-enhancing factor (AEF) has been shown to markedly shorten the onset of AA amyloidosis, 17 and the amyloid fibril itself acts as an AEF. 14 AA amyloidosis has been experimentally induced in rodent models by inoculating AA from different animal species 4,27 ; therefore, a prion-like mechanism has been suggested in the transmission of AA amyloidosis. 14 Previous studies reported difficulties reproducing the transmission of AA amyloidosis between different animal species. 9 These findings suggested the presence of a “species barrier” to the transmission of AA amyloidosis, similar to prions. 21
Interleukin-1 (IL-1) is a cytokine that plays an important role in inflammatory and autoimmune diseases. Various cells, including macrophages, monocytes, and synovial cells, produce IL-1, and IL-1 itself induces various chemokines, cytokines, and inflammatory mediators. 5 The IL-1 signal is transduced into cells by the type 1 IL-1 receptor (IL-1R1), which is expressed on the surfaces of various cell types, including osteoclasts. 10 The IL-1 signal activates osteoclasts in the joint and produces metalloproteases and collagenases that cause erosion of bones in the joint. 16 The IL-1 receptor antagonist knockout (IL-1raKO) mouse was generated as a rodent model of human RA. 8 This mouse cannot produce the IL-1 receptor antagonist, a natural inhibitor for IL-1R1; therefore, these mice spontaneously develop chronic arthritis from 5 weeks of age in a conventional environment. Individual differences have been noted in the severity of lesions; however, most mice develop lesions by 12 weeks of age. Since IL-1 activates the production of SAA in the liver, 23 IL-1raKO mice continuously produce SAA. These findings indicate that IL-1raKO mice present with chronic arthritis, similar to human patients with RA, and may be able to maintain a high concentration of SAA.
In the present study, we attempted to induce AA amyloidosis in IL-1raKO mice using AEFs from 3 different animal species—mice, cats, and cattle—to establish reproducible rodent models for the transmission of AA amyloidosis.
Materials and Methods
Experimental Design
The experimental protocols used and constitution of experimental groups are summarized in Tables 1 and 2. We used a murine AEF to verify the function of IL-1 in the development of AA amyloidosis and set the prerequisite conditions for the induction of AA amyloidosis. We established 10 groups that were separated by the purification of AEFs and inflammatory stimuli (Table 1). We also used feline and bovine AA amyloid fibrils to examine the transmission of AA amyloidosis with the administration of AA amyloid fibrils from different animal species. In this experiment, we used 8 groups that differed in the source and purification of AEFs (Table 2). In each experiment, we established IL-1raKO and BALB/c groups for all conditions, the latter of which was the genetic background of the IL-1raKO mouse.
Transmission of AA Amyloidosis Using the Murine Amyloid-Enhancing Factor (AEF).
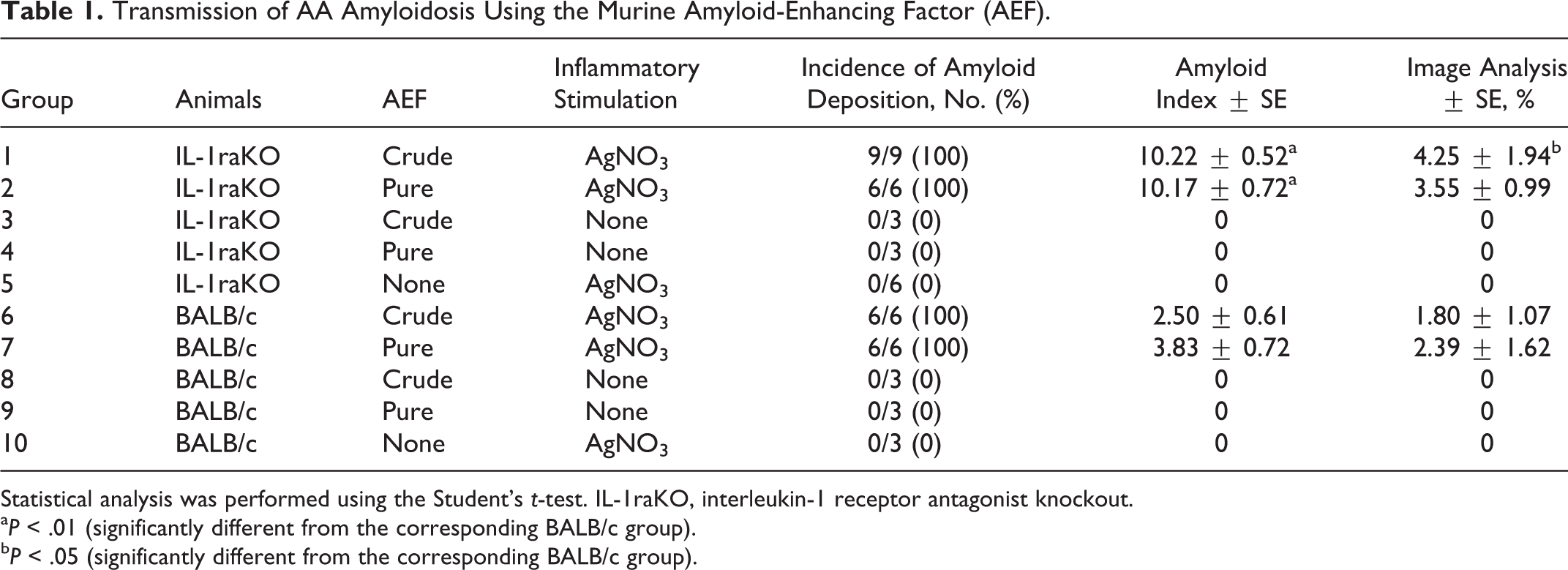
Statistical analysis was performed using the Student’s t-test. IL-1raKO, interleukin-1 receptor antagonist knockout.
a P < .01 (significantly different from the corresponding BALB/c group).
b P < .05 (significantly different from the corresponding BALB/c group).
Transmission of AA Amyloidosis Using Amyloid Extracts From Different Animal Species.
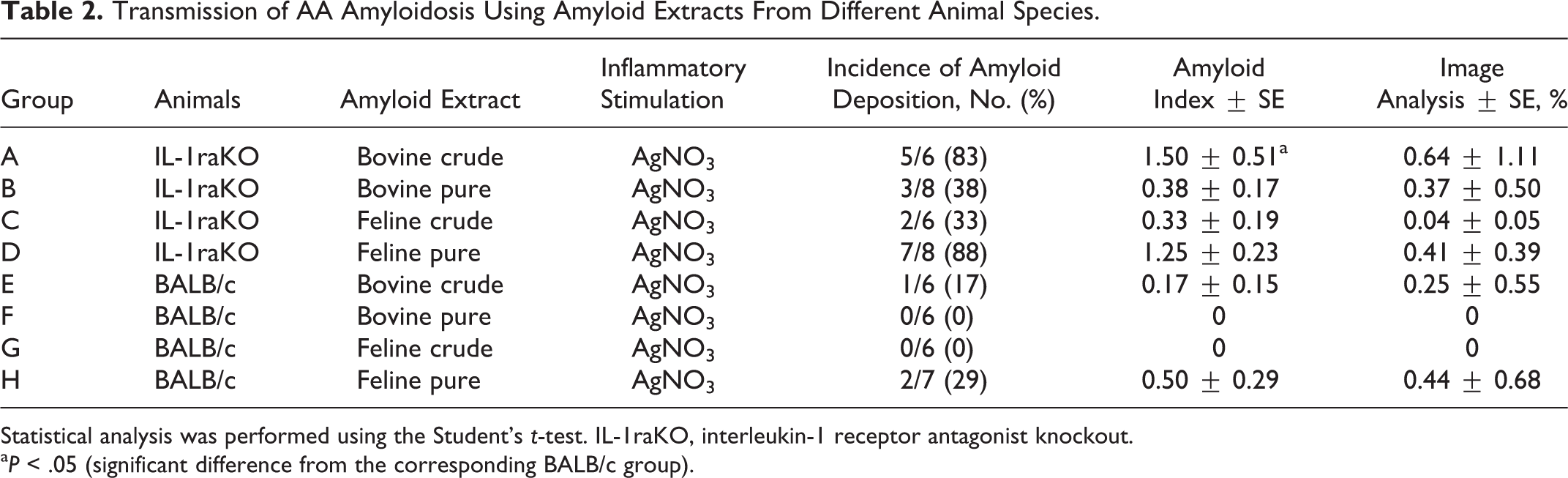
Statistical analysis was performed using the Student’s t-test. IL-1raKO, interleukin-1 receptor antagonist knockout.
a P < .05 (significant difference from the corresponding BALB/c group).
Animals
All procedures used in the present study were approved by the Animal Care Committee at the University of Tokyo. Twelve-week-old IL-1raKO and BALB/c mice were used in the present study. The genetic information and physiologic features of IL-1raKO mice have already been described. 8 Five to 10 mice were housed in a cage with free access to food (MS food; Oriental Yeast, Tokyo, Japan) and tap water.
Preparation of AEFs and Amyloid Extracts
AEFs and amyloid extracts were prepared from the livers of a mouse, cat, and cow with AA amyloidosis. Murine AA amyloidosis was experimentally induced by an intraperitoneal (i.p.) injection of an AEF in addition to a subcutaneous pretreatment of 500 μl of 2% AgNO3. AA amyloid extracts of cat and cow were extracted from the livers of animals with spontaneous AA amyloidosis. Two types of AEFs were prepared from the livers of mouse; one was a homogenate of an AA-laden tissue (crude AEF) while the other was a water-soluble AA amyloid fibril (pure AEF). To make a crude AEF, 1 g of tissue was homogenized with 10 ml phosphate-buffered saline (PBS) for 30 seconds on ice. A pure AEF was extracted according to the method of Pras et al. 18 Briefly, 1 g of tissue was homogenized with 10 ml saline for 30 seconds on ice. The homogenate was centrifuged (40 000 × g at 4°C for 20 minutes). The pellet was suspended in saline and centrifuged again to remove extra proteins. The washing steps were repeated 7 to 10 times until the absorbance of the supernatant was less than 0.075 at a wavelength of 280 nm. The pellet was then resuspended in cold distilled water and centrifuged (30 000 × g at 4°C for 1 hour) again to collect the supernatant. Centrifugation was repeated 4 times, and supernatants of the second to fourth centrifugations contained water-soluble AA amyloid fibril. These fractions were gathered and centrifuged (45 000 × g at 4°C for 1 hour) to concentrate. After the last centrifugation, the pellet was collected as pure AEF. We stained a smear of an aliquot of the AEFs with Congo red to confirm the presence of amyloid. The total protein concentrations of both crude and pure AEFs were measured using the DC protein assay kit (Bio-Rad, Hercules, CA) and adjusted to 1.0 mg/ml. As AEFs were prepared, crude and pure types of feline and bovine amyloid extracts were extracted by the same protocols. AEFs and amyloid extracts were stored at –80°C until use.
Immunoblotting of AEFs and Amyloid Extracts
AEFs and amyloid extracts (10 μg per well) were mixed with Laemmli sample buffer (Bio-Rad) containing 5% 2-mercapthethanol (Sigma-Aldrich, St Louis, MO) and boiled at 99°C for 5 minutes. Samples were separated on a 15% polyacrylamide gel (ATTO, Tokyo, Japan) for sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto a polyvinylidene membrane (PVDF) (Merck Millipore, Billerica, MA). The membrane was blocked with 1% w/v skim milk in Tris-buffered saline (TBS) buffer with Tween-20 (3% v/v). Primary antibodies used were goat anti–mouse SAA1.1 polyclonal antibody (R&D Systems, Minneapolis, MN) for the detection of murine AEFs and mouse anti–human AA monoclonal antibody (Kyowa, Tokyo, Japan) for the detection of bovine and feline amyloid extracts. Secondary antibodies were horseradish peroxidase (HRP)–conjugated sheep anti–mouse IgG (GE Healthcare UK Ltd, Little Chalfont, Buckinghamshire, UK) and HRP-conjugated donkey anti–goat IgG (Santa Cruz Biotechnology, Dallas, TX). Blots were developed using ECL Prime Western Blotting Detection Reagent (GE Healthcare UK Ltd). Immunoreactive bands were detected with the ChemiDoc XRS+ System (Bio-Rad).
Transmission of AA Amyloidosis Using AEFs and Amyloid Extracts
Twelve-week-old IL-1raKO (n = 27) and BALB/c (n = 21) mice were divided into 10 groups (groups 1–5: IL-1raKO; groups 6–10: BALB/c), with 3 to 9 mice in each group. Mice in groups 1 to 4 and 6 to 9 were i.p. injected with 500 μg of the mouse AEFs. At the same time, mice in groups 1, 2, 5, 6, 7, and 10 were s.c. injected with 500 μl of 2% AgNO3 (Table 1). Serum samples were collected at 0, 5, and 21 days in groups 1 and 6 to measure the concentration of SAA. To induce a transmission of AA amyloidosis using amyloid extracts from different animal species, we set 8 more groups. Twelve-week-old IL-1raKO (n = 28) and BALB/c (n = 25) mice were divided into 8 groups (groups A–D: IL-1raKO; groups E–H: BALB/c), with 6 to 8 mice in each group. Mice in groups A, B, E, and F were i.p. injected with 500 μg of the bovine amyloid extracts, and mice in groups C, D, G, and H were i.p. injected with 500 μg of the feline amyloid extracts. At the same time, mice in groups A to H were s.c. injected with 500 μl of 2% AgNO3 (Table 2). All mice were euthanized by the collection of whole blood under deep isoflurane anesthesia 3 weeks after the AEF and AgNO3 treatments, and the liver, spleen, kidneys, heart, adrenal glands, thyroid glands, and intestines were obtained at necropsy and fixed in 10% neutral buffered formalin.
Histopathology and Amyloid Index
Formalin-fixed paraffin sections were stained with Puchtler’s alkaline Congo red and counterstained with hematoxylin. The intensity of amyloid deposition in the spleen, liver, kidneys, heart, adrenal glands, thyroid glands, and ileum was semi-quantitatively determined under a polarized microscope. The histopathologic amyloid index was determined as follows: 0, none; 1+, minute; 2+, moderate; and 3+, severe amyloid deposition. Each score was calculated as the amyloid index.
Image Analysis of Amyloid Deposition
The intensity of splenic amyloid deposition was quantified using an image-processing method. After immunostaining with a goat anti–mouse SAA1.1 polyclonal antibody (R&D Systems), immunopositive areas were measured using ImageJ software (National Institutes of Health, Bethesda, MD). Briefly, immunopositive pixels were counted in 10 randomly selected fields in the spleen.
Measurement of Serum SAA Concentrations
We used an enzyme-linked immunosorbent assay (ELISA) kit (BioSource International, Camarillo, CA) to measure the concentration of SAA. Serum samples that had been stored at –80°C were thawed. ELISA was performed according to the manufacturer’s instructions.
Statistical Analysis
Regarding the amyloid index, image analysis, and SAA concentration, data were analyzed using the Student’s t-test, and P < .05 was accepted as significant.
Results
Immunoblotting of AEFs and Amyloid Extracts
AEFs and amyloid extracts consist of amyloid fractions and other minor molecules (Fig. 1a). Pure AEF and pure amyloid extracts largely contain water-soluble AA amyloid fibrils, which are approximately 14 to 16 kDa molecules in SDS-PAGE and immunopositive for mouse SAA1.1 or anti–human AA amyloid (Fig. 1b,c). Crude AEF and crude amyloid extracts also contain similar molecules, but the rate of that is lower. Other immunopositive bands were weakly detected in crude samples; the sharpest one, approximately 28 kDa, was common in all crude samples.
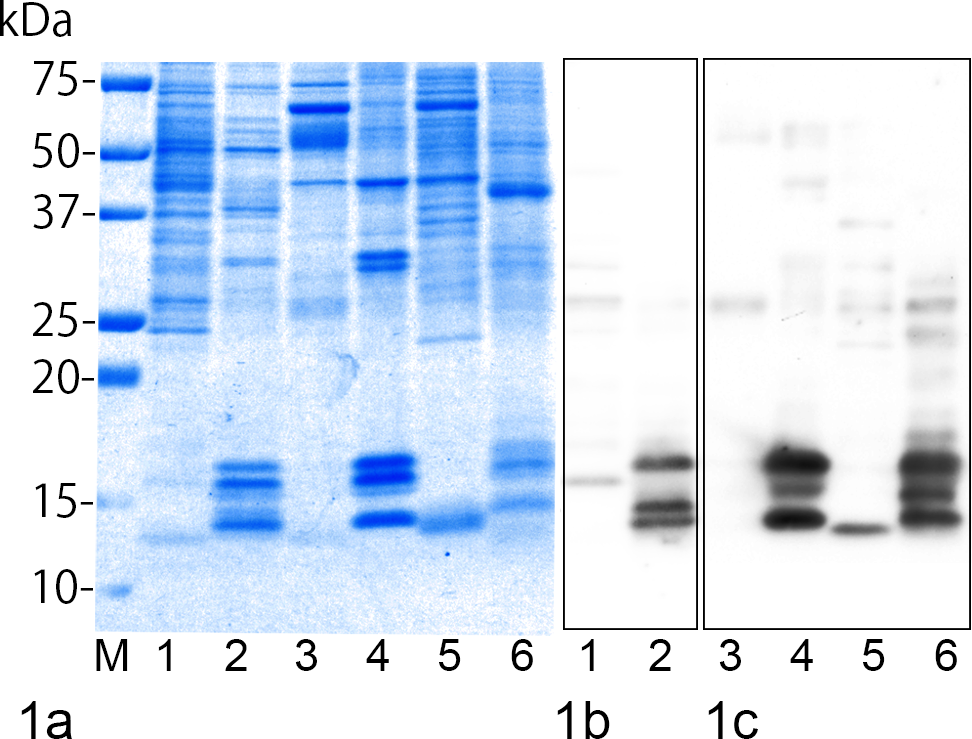
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and Western blot of full amyloid-enhancing factors (AEFs) and amyloid extracts. Lane 1, murine crude AEF; lane 2, murine pure AEF; lane 3, bovine crude amyloid extract; lane 4, bovine pure amyloid extract; lane 5, feline crude amyloid extract; lane 6, feline pure amyloid extract; M, molecular weight marker. (a) SDS-PAGE of AEFs and amyloid extracts. AEFs and amyloid extracts consist of amyloid fractions and other minor molecules. Coomassie Brilliant Blue stain. (b) Western blot of murine AEFs using anti–mouse SAA1.1 antibody. (c) Western blot of bovine and feline amyloid extracts using antibody for anti–human AA amyloid.
Transmission of AA Amyloidosis Using Murine AEF
Amyloid was deposited in the spleen, liver, kidneys, heart, thyroid glands, intestines, and adrenal glands of IL-1raKO mice (Figs. 2–10, 11) and in the spleen, liver, kidneys, heart, thyroid glands, and intestines of BALB/c mice (Fig. 11). Histopathologically, the deposition of amyloid was observed around the splenic follicles (Figs. 2–4) and in the space of Disse in the liver (Fig. 5), papillae of the kidney (Fig. 6), thyroid interstitium (Fig. 7), lamina propria of the intestines (Fig. 8), myocardial interstitium (Fig. 9), and marginal zone of the adrenal cortex and medulla. Amyloid exhibited a typical green birefringence on a Congo red–stained section by polarized microscopy and was immunopositive for mouse SAA1.1 (Fig. 10).
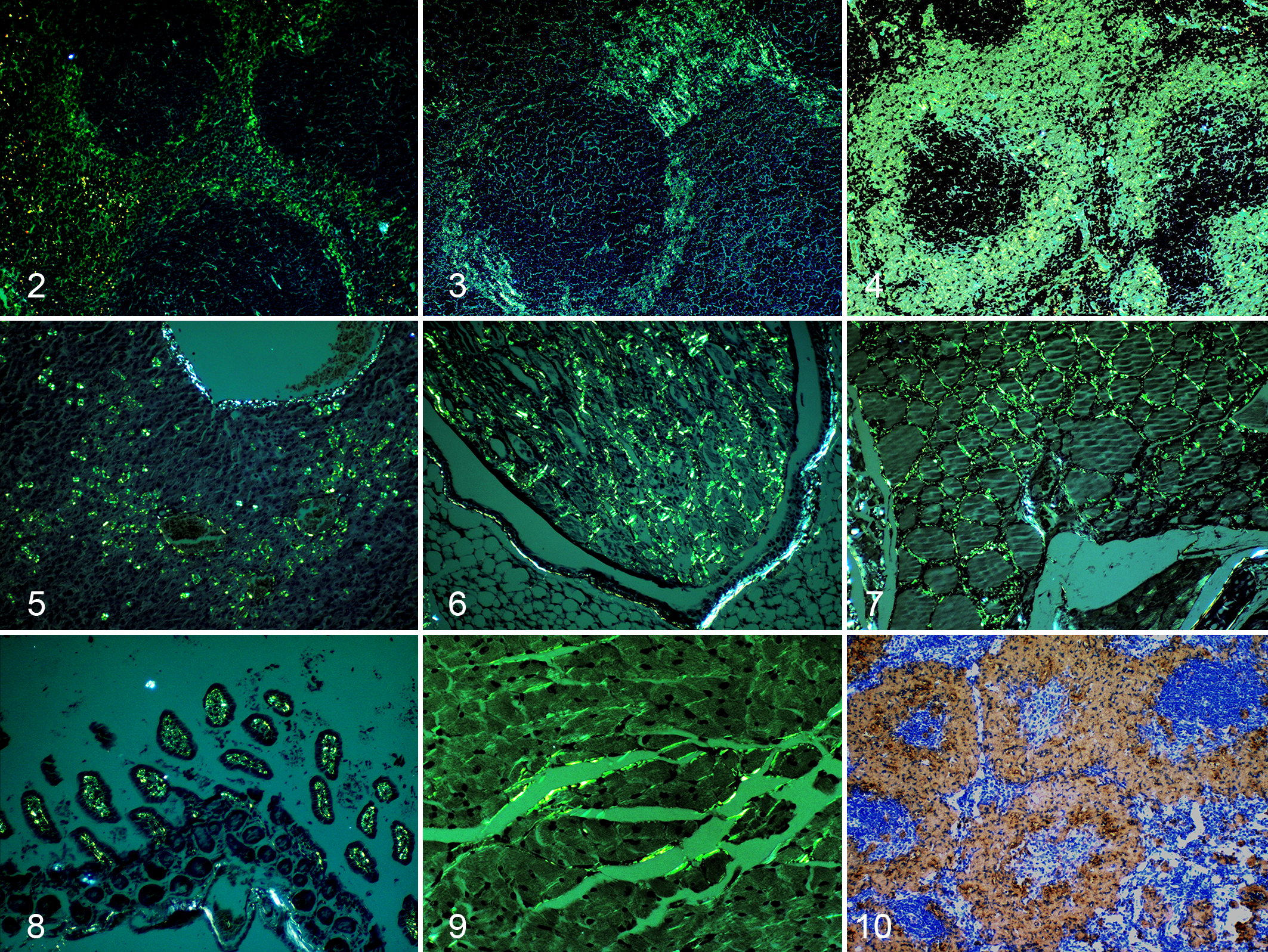
Amyloid A (AA) amyloidosis, mouse. Group 1: interleukin-1 receptor antagonist knockout (IL-1raKO) mice injected with 500 μg of murine crude amyloid-enhancing factor (AEF) and 500 μl of 2% AgNO3; group 6: BALB/c mice injected with 500 μg of murine crude AEF and 500 μl of 2% AgNO3.

Distribution and intensity of amyloid A amyloid deposition shown as a heatmap. The rows respectively show the amyloid index in each organ. Each group contains 6 to 9 mice. The intensity of amyloid deposition was represented as the amyloid index: 3+, black; 2+, dark gray; 1+, gray; and 0+, white.
Quantitative results are shown in Table 1. Amyloid deposits were observed in mice in groups 1, 2, 6, and 7, which had been treated with both AEF and AgNO3, but were absent in mice in groups 3, 4, 8, and 9, which had not been treated with AgNO3, as well as in those in groups 5 and 10, treated only with AgNO3. No significant difference was observed in the incidence of amyloidosis between IL-1raKO and BALB/c mice, whereas the amount of amyloid deposited was higher in IL-1raKO mice than in BALB/c mice. The averages of the amyloid index in groups 1 and 2 were 10.22 and 10.17, respectively, which were significantly higher than those of BALB/c mice under the same conditions (P = .0000007, P = .0002). In addition, a significant difference was observed in AA-positive areas between groups 1 and 6 (P = .02).
SAA Concentration
The concentration of SAA on day 0, prior to the administration of the murine AEF and AgNO3 inflammatory stimulation, was already higher in IL-1raKO mice (276.3 μg/ml) than in BALB/c mice (10.1 μg/ml). SAA concentrations were markedly elevated 5 days after the murine AEF and AgNO3 treatment (IL-1raKO, 828.4 μg/ml; BALB/c, 275.2 μg/ml) and subsequently decreased to the concentrations observed on day 21 (IL-1raKO, 299.3 μg/ml; BALB/c, 31.4 μg/ml). A significant difference was observed in SAA concentrations between IL-1raKO and BALB/c mice on day 5 and day 21 (P = .02, P = .00007, respectively) (Fig. 12).
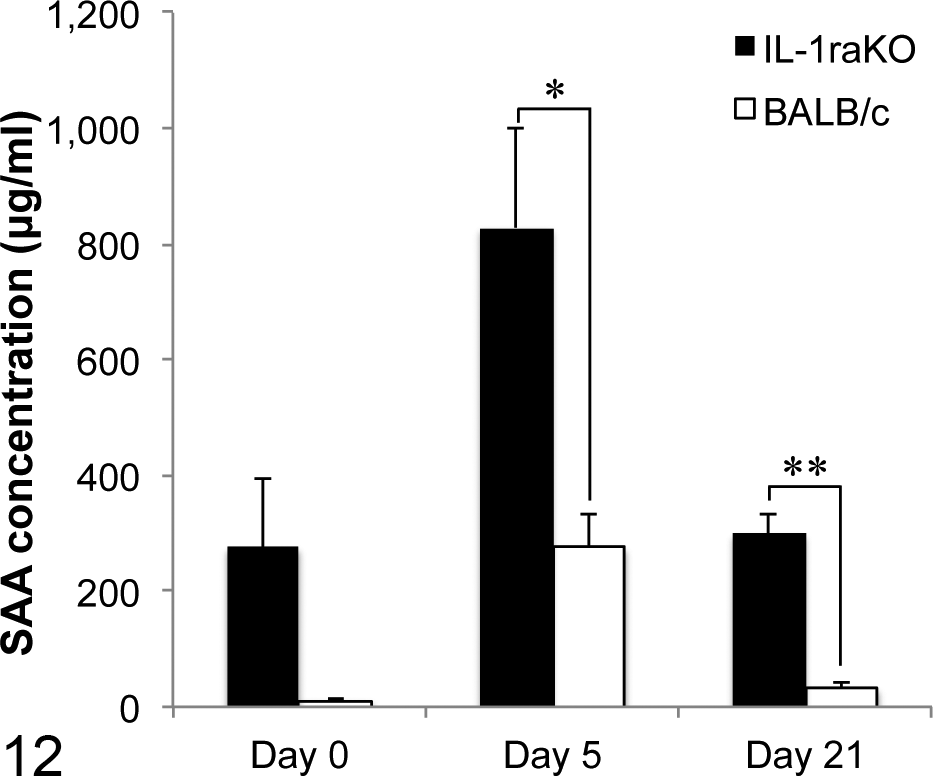
Serum amyloid A (SAA) concentrations. SAA concentrations were elevated on day 5 and decreased on day 21. SAA concentrations were higher in interleukin-1 receptor antagonist knockout (IL-1raKO) mice than in BALB/c mice. Bar, standard error. *P < .05. **P < .01.
Transmission of AA Amyloidosis Using Amyloid Extracts From the Cow and Cat
All IL-1raKO (15 cases) and BALB/c (12 cases) mice inoculated with the murine AEF together with the AgNO3 inflammatory stimulation developed AA amyloidosis (Table 1). In contrast, 83% (5/6 cases) and 38% (3/8 cases) of IL-1raKO mice and 17% (1/6 cases) and 0% (0/6 cases) of BALB/c mice inoculated with the crude or purified bovine AA amyloid together with the inflammatory stimulation developed AA amyloidosis, respectively (Table 2). Similarly, 33% (2/6 cases) and 88% (7/8 cases) of IL-1raKO mice and 0% (0/6 cases) and 29% (2/6 cases) of BALB/c mice inoculated with the crude or purified feline AA amyloid developed AA amyloidosis, respectively (Table 2). The deposition of amyloid was significantly less in mice that developed AA amyloidosis after being injected with the bovine or feline amyloid extracts than in mice injected with the murine AEF (P < .01, respectively) (Table 2; Figs. 13, 14). The deposition of amyloid was observed only around the splenic follicles, except for 1 mouse in group A in which mild deposits were noted in the liver (Table 2, Fig. 11). Amyloid in the spleen was immunopositive for mouse SAA1.1.
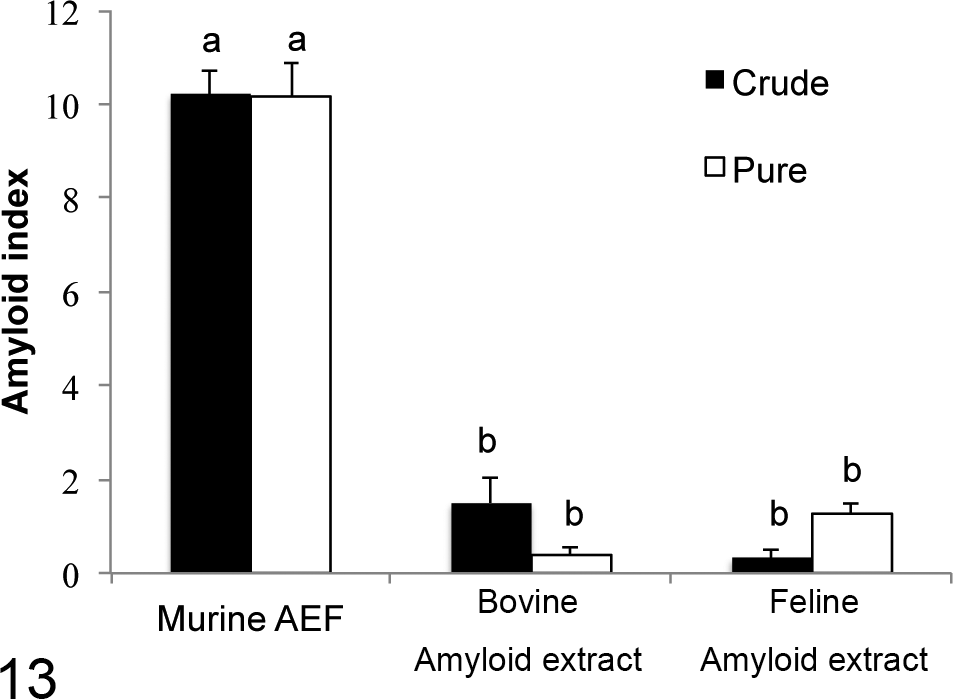
Amyloid index of interleukin-1 receptor antagonist knockout (IL-1raKO) mice. The intensity of amyloid deposition was semi-quantitatively determined in 7 organs as follows: 0, none; 1+, minute; 2+, moderate; and 3+, severe amyloid deposition. Each score was calculated as the amyloid index. The index of mice injected with the murine amyloid-enhancing factor was significantly higher than that of mice injected with the bovine and feline amyloid extracts. Bar, standard error. Between (a) and (b): P < .01.
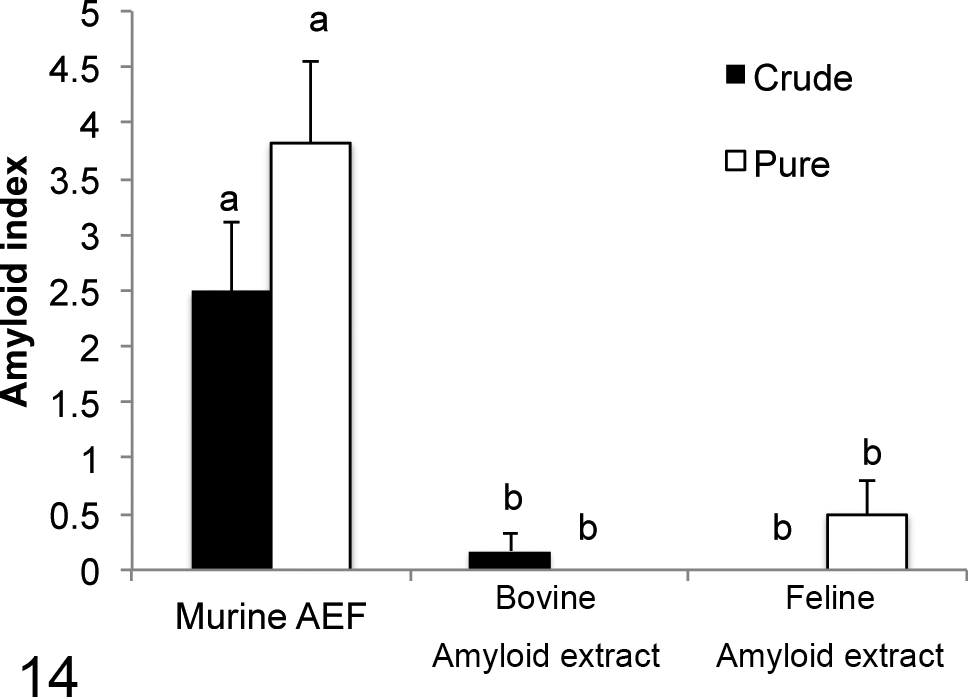
Amyloid index of BALB/c mice. The index of mice injected with the murine amyloid-enhancing factor was significantly higher than that of mice injected with the bovine and feline amyloid extracts. Bar, standard error. Between (a) and (b): P < .01.
Discussion
The classic rodent model for amyloidosis is an experimental AA amyloidosis model that has been improved to obtain a reproducible intensity of amyloid deposition. 14,19,24 Two popular mouse models have recently been proposed to investigate AA amyloidosis: an acute inflammation model and a transgenic mouse model. 26 Both use the same strategy: the maintenance of a high concentration of SAA to induce the greater deposition of AA amyloid. In the acute inflammation model, an s.c. injection of AgNO3 has been shown to induce transient elevations in SAA concentrations, thereby resulting in the deposition of AA amyloid. 14 Although this model is very simple, it is difficult to maintain high concentrations of SAA; therefore, the amount of AA amyloid deposited is inconstant. 14 In contrast, the constitutive expression of the human IL-6 transgene in transgenic mouse models, such as human IL-6 transgenic mice, maintained a higher SAA concentration (400–4000 μg/ml) and resulted in the marked deposition of AA amyloid. Furthermore, these mice died at about 9 months of age due to renal failure. 26 The present IL-1raKO mouse model unites the advantages of these 2 models. IL-1raKO mice maintain a higher concentration of SAA, and acute inflammation persists for longer than that in BALB/c mice. An inflammatory stimulation by a single injection of AgNO3 itself also induced a transient elevation in SAA. The concentration of SAA had already decreased to normal levels (<10 μg/ml) at necropsy in BALB/c mice but was still maintained at approximately 200 μg/ml in IL-1raKO mice. An SAA concentration of 200 μg/ml is approximately 20 times higher than that in BALB/c mice. IL-1 signaling is also known to stimulate the production of SAA in the liver but is less effective than IL-6 signaling. 7
The results of the present study indicate that the combined treatment with both AgNO3 and an AEF is necessary for the continuous induction of AA amyloidosis. Although IL-1raKO mice maintained an SAA concentration that was approximately 20 times higher than that in BALB/c mice, they did not develop AA amyloidosis with an AEF only. Similarly, IL-1raKO and BALB/c mice treated with AgNO3 only did not develop amyloid deposits. The transient elevation in SAA concentrations following the treatment with AgNO3 may not have been adequate for the development of AA amyloidosis; however, it acted as an important trigger. The presence of an AEF during the formation of amyloid fibrils appears to be necessary for the deposition of AA amyloid to be consistent. Furthermore, IL-1raKO mice naturally maintain high concentrations of SAA for a period suitable for the formation of amyloid fibrils following an injection with AgNO3. IL-1raKO mice possess a dysfunctional negative feedback system for IL-1 signaling; therefore, they continue to produce SAA. 10 We have cared for IL-1raKO mice for more than 2 years, and no mouse spontaneously has developed AA amyloidosis (data not shown). The results of the present study reveal that IL-1raKO mice do not spontaneously develop AA amyloidosis but have larger AA amyloid deposits following the treatment with both AgNO3 and the murine AEF than do BALB/c mice.
Although previous studies examined the amyloidosis-inducing activity of AEFs, 17 the underlying mechanism has not yet been elucidated. 2 In experimental AA amyloidosis, some molecular chaperons, such as apolipoprotein E 12 and amyloid P component, 25 and some substances, such as heparan-sulfate, are co-localized with deposited amyloid. These substances will play an important role for the formation of amyloid fibrils. The AEF activity of these substances is not clarified, whereas several constituents of AEFs such as ubiquitin have been shown to accelerate the formation of amyloid fibrils. 1 In addition to AEFs purified from amyloid-laden tissues, serum or peripheral blood monocytes from mice with AA amyloidosis also exhibit AEF activity. 28 In the present study, we compared AEF activities between 2 types of purification. We expected that pure AEF mainly consists of water-soluble AA amyloid, while crude AEF may contain more factors involved in the pathogenicity of AA amyloidosis than the pure AEF, so it induces higher incidence than pure AEF; however, we found no significant difference between the crude AEF and pure AEF.
The incidence and degree of AA amyloid deposition were significantly lower with feline and bovine amyloid extracts than with the murine AEF. This difference has been referred to as the “species barrier.” 4 The mechanisms involved in the species barrier currently remain unknown but may be closely related to the molecular mechanism of cross-seeding. 14 Therefore, we suggest that the species barrier to the transmission of AA amyloidosis may be determined by differences in the amino acid sequences between the injected amyloid extract and host SAA. A 1– or 2–amino acid difference may lead to differences in amyloidogenicity. Of the 3 isoforms of SAA with single amino acid variations at 2 sites, SAA1.3 was the only amyloidogenic isoform that could aggregate and produce straight fibrils in vitro. 29 In the present study, different activities were observed in the polymerization of murine SAA following the injection of AEFs or amyloid extracts, which include AA amyloid fibrils from 3 species. On the other hand, proteins such as Sap35, 15 silk-fibroin, 13 and human AL amyloid, 4 the amino acid sequences of which differ from that of murine SAA, also exhibited AEF activity in the acute inflammation model. These findings suggest that AEF activity is based not only on the primary structure, so further studies on the AEF activity and the 3-dimensional (3D) conformation of the protein are needed. If AEF activity is based on the 3D conformation of AEFs, a comparison of AEF activities and 3D conformations will be a key factor for future research. Further biochemical studies are needed to identify the mechanisms underlying the formation of AA fibrils and transmission of AA amyloidosis.
In conclusion, IL-1raKO mice represent a better tool for investigating the etiology of AA amyloidosis.
Footnotes
Acknowledgements
We thank Dr. T. Yamada at the Department of Clinical Laboratory Medicine, Jichi Medical University, for providing the murine AEF and technical support.
Author Contribution
Conception or design: KW, KU, JKC. Data acquisition, analysis, or interpretation: KW, JKC, MT, AS. Drafting the manuscript: KW. All authors participated in critically revising the manuscript, gave final approval, and agree to be accountable for all aspects of work to ensure integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
