Abstract
Eighty-nine canine mammary tumors and dysplasias of 66 bitches were investigated to determine the immunohistochemical expression of classical estrogen receptor (ER-α) and its clinical and pathologic associations and prognostic value. A complete clinical examination was performed and reproductive history was evaluated. After surgery, all animals were followed-up for 18 months, with clinical examinations every 3–4 months. ER-α expression was higher in tumors of genitally intact and young bitches (P < 0.01, P < 0.01) and in animals with regular estrous periods (P = 0.03). Malignant tumors of the bitches with a previous clinical history of pseudopregnancy expressed significantly more ER-α (P = 0.04). Immunoexpression of ER-α decreased significantly with tumor size (P = 0.05) and skin ulceration (P = 0.01). Low levels of ER-α were significantly associated with lymph node involvement (P < 0.01). Malignant tumors had lower ER-α expression than did benign tumors (P < 0.01). Proliferation index measured by proliferating cell nuclear antigen immunostaining was inversely correlated with ER-α scores (P = 0.05) in all tumors. Low ER-α levels in primary malignant tumors were significantly associated with the occurrence of metastases in the follow-up (P = 0.03). Multivariate analyses were performed to determine the prognostic significance of some follow-up variables. ER-α value, Ki-67 index, and age were independent factors that could predict disease-free survival. Lymph node status, age, and ER-α index were independent prognostic factors for the overall survival. The immunohistochemical detection of ER-α in canine mammary tumors is a simple technique with prognostic value that could be useful in selecting appropriate hormonal therapy.
Canine mammary tumors (CMT) are heterogeneous from their pathologic aspects to their clinical behavior. The demonstration of clinical and pathologic parameters with prognostic and/or therapeutic significance in these neoplasms is a valuable field of study in veterinary medicine. Several studies have been done on the prognosis of CMT, 3 , 20 , 23 , 28 , 30 , 32 , 38 but some areas, especially those concerning the role of estrogen receptors (ER), remain uncertain.
Analyses of classical ER (ER-α) and progesterone receptors (PR) have become accepted and useful tools in prognosis and prediction of hormonal therapy response in human breast cancer. 45 , 46 The initial biochemical methods, such as dextran-coated charcoal (DCC), have been replaced in the last decades by immunohistochemistry, which needs less tissue and is less expensive. In addition, immunohistochemical detection of ER-α is a more accurate in situ technique; these receptors are preferentially located within the nuclear compartment, 35 and their determination in tissue homogenates has been questioned. Several monoclonal antibodies against human ER have been developed to avoid the disadvantages of biochemical methods. 13 , 21 , 35 Recently, a novel estrogen receptor, ER-β, has been detected in several human breast biopsy samples and several human breast epithelial cell lines using reverse transcription polymerase chain reaction analysis, suggesting a possible role for ER-β in human breast cancer. 10
In human mammary tumors, the role of ER-α in prognosis and therapy is well known. The expression of ER-α is related to disease-free survival, overall survival, and proliferation index measured with anti-Ki-67 and anti-proliferating cell nuclear antigen (PCNA) antibodies. 13–16, 21 , 36 , 40 , 49 , 50 Antiestrogen therapy with drugs such as Tamoxifen™ has been shown to improve survival in postmenopausal women. Patients with breast cancer tumors containing ER-α have higher survival rates and better response to hormonal treatment than patients without ER-α. 5 , 34
In the last decade, several studies using biochemical methods have revealed the presence of ER-α in CMT. 4 , 9 , 11 , 17 , 25 , 27 , 38 , 42 , 43 These results indicate a role for sexual steroid hormones in CMT growth promotion; ER-α and PR are present in more than 50% of mammary neoplasms in dogs. 11 , 17 , 25 , 38 It has been postulated that the biochemical methods used in these published reports produce an ER-α overexpression and false-positive results in canine malignant tumors. 41 There are no previous reports on immunohistochemical demonstration of ER-α in canine tissues, but a relationship between the ER-α content and histologic type of CMT has been indicated. Normal mammary glands, mammary dysplasias, and benign tumors have higher ER-α levels than do malignant tumors. 22 , 26 , 27 Metastases frequently are negative for both receptors. 42 Also, some correlation between epidemiologic and clinical parameters and ER-α content have been found, 9 , 27 , 38 , 42 but many aspects are still unconfirmed.
In this study, the presence of ER-α in formalin-fixed, paraffin-embedded samples of normal, dysplastic, and neoplastic canine mammary tissues was determined using immunohistochemical techniques. The possible association of ER-α levels with pathologic and clinical parameters was analyzed, and the prognostic value of ER-α immunohistochemical detection was evaluated and compared with other known prognostic factors in CMT.
Materials and Methods
Clinical assessment
Eighty-nine mammary tumors and dysplasias of 66 bitches were included in this study. Animals, ranging in age from 5 to 13 years, were clinically examined and surgically treated in the Veterinary Teaching Hospital of the Veterinary School of Madrid between January 1992 and July 1993. Clinical and reproductive histories were obtained from the owners. Anamnesis variables included age, breed, body size, and history of ovariohysterectomy, prevention of estrus with hormonal treatment, frequency, regularity, and duration of estrus, age at first estrus and at first parturition, number of full-term pregnancies, and presence of pseudopregnancy or pseudolactation. Complete physical examinations were done. Rate of tumor growth (defined as slow, medium, or fast) was based upon owner information, tumor size (the largest of three dimensions was used for the statistical analysis), adherence to underlying tissues, and skin ulceration.
Clinical stages were determined using the classification of the World Health Organization (WHO), 18 including radiologic evaluation of the thorax where tumor size was classified as T1 (3 cm), T2 (3–5 cm), or T3 (>5 cm), N indicated the condition of the regional lymph nodes, confirmed by histology or cytology, and M for distant metastases. Only dogs clinically and radiologically free from distant metastases were included in the study (M0).
Histologic examination
Two normal mammary gland tissue samples and 89 postsurgery tumor samples were fixed in 4% buffered formalin and embedded in paraffin. The neoplastic samples were histologically examined and classified according to the WHO classification. 18 Malignant tumors were graded according to histologic malignancy grade (HMG), using the system of human breast carcinomas 2 , 44 and considering the criteria of malignancy in CMT tumors as previously published: 23 , 32 HMG I = well-differentiated tumors; HMG II = moderately differentiated neoplasm without hematic or lymphatic invasion; and HMG III = poorly differentiated tumors with or without vascular invasion. The nuclear grade (NG) was established separately for each sample: 2 , 23 , 44 NG I = well differentiated; NG II = moderately differentiated; NG III = large and irregular nuclei.
Immunohistochemistry
Immunohistochemical determination of ER-α was performed on deparaffined sections using the streptavidin-biotin-peroxidase complex method, using as primary antibody a monoclonal anti-human ER-α after pretreatment of the slides in a stainless steel pressure cooker. Four-micrometer paraffin-embedded sections were placed in Vectabond™ (Vector Laboratory, SP1800)-covered slides and boiled in a pressure cooker containing 2 liters of 10 mM sodium citrate buffer (pH 6.0). When the pressure indicator valve rose to the highest position, the sections were incubated for 2 minutes. The cooled slides were washed in distilled water and Tris-buffered saline (TBS) (0.1 M Tris base, 0.9% NaCl, pH 7.4). Endogenous peroxidase was blocked in 1.5 ml H2O2/100 ml methanol for 15 minutes. After pretreatment, the sections were successively incubated at 37 C (2 hours) with an anti-ER-α antibody (Novocastra NCL-ER-LH2, clone CC4-5) at a dilution of 1:40, anti-mouse biotinlated secondary antibody (DAKO E04233, 1:200, 45 minutes, 37 C), and streptavidin conjugated with peroxidase (Zymed P50242, 1:20, 45 minutes, 37 C) in a humidity chamber. Dilutions were made in TBS. The sections were then developed with 3-3′diaminobenzidine tetrachloride (Sigma Chemical Co. D5059) and H2O2 (0.01%) in TBS. After washing in distilled water for 10 minutes, slides were counterstained 45 seconds with Gill's hematoxylin (Sigma GH5-2-16), washed in tap water, dehydrated in three baths of alcohol, cleared in xylene, and mounted. Corresponding negative control sections were prepared by replacing only the primary antibody with TBS. Canine, goat, rabbit, and equine uterus were used as positive controls.
ER-α immunostaining was interpreted simultaneously by two observers counting 8–10 representative high-power fields (approximately 1,000 neoplastic cells). Brown nuclei were considered positive. Two human score systems were used: ERi 24 , 45 and ERp. The ERi score was calculated as P1 + (2 × P2) + (3 × P3) where P1, P2, and P3 are the estimated percentages of positive nuclei with low (P1), medium (P2), and high (P3) intensity of immunostaining. The ERp score considered only the percentage of positive cells independent of the grade of intensity.
Immunoexpression of Ki-67 and PCNA were studied using an indirect immunoperoxidase method on formalin-fixed, paraffin-embedded sections mounted on poly-
The percentage of Ki-67- and PCNA-positive cells was determined by counting the positive and negative nuclei of the tumor cells in 8–10 representative fields (minimum of 1,000 cells). Stromal nuclei were not considered.
Follow-up study
Dogs were clinically examined every 3–4 months after surgical treatment for 18 months. The follow-up included a radiologic evaluation of the thorax. Recurrences and metastases were recorded, and disease-free survival (DFS) and overall survival (OS) times were noted for each animal (low = 0–6 months; medium = 7–17 months; high = 18 months). The cause of death (tumor or other cause) was confirmed at necropsy.
Statistical analysis
To study the associations of ERi and ERp compared with the other variables, statistical tests were done using the Biomedical Data Processing software. 8 Linear regression was calculated to establish the correlation coefficients of continuous variables. Analyses of variance (F-test, pooled t-tests if variances are assumed to be equal and Welch test or separate t-tests if variances are not equal) were used to evaluate the differences of the means of ER-α (ERi and ERp) in the groups of categorical variables. Multivariate logistic regression analyses were performed to evaluate the influence of sets of variables on dependent follow-up variables (recurrences, metastases, DFS, OS), calculating its odds ratio (OR) according to the formula OR = esp (a 0 + a 1 X 1 +… + a n X n ), where a values are the coefficients affecting the sets of related independent variables included in the model (X 1, X 2, …, X n ). The independent variables are selected according to their significance in several steps. Those variables that are significant at one step are then considered constants to evaluate the combined effect of the other variables on the dependent variables. Only animals with complete data sets for the variables studied were included.
Dogs were divided in two groups: DA = animals with only dysplasias and/or benign tumors, DB = animals with at least one malignant tumor. Tumors were also separated in two groups: TA = dysplasias and benign tumors, TB = malignant tumors. Dog-related and tumor-related variables were examined separately and in combination. To analyze animal and tumor data together in dogs with more than one mammary lesion, the most representative values of ERi and ERp were assigned to each dog. In patients with more than one malignant neoplasm, variables of the tumor with the most severe clinical and pathologic features were chosen. The minimum level of significance was set at a P value 0.05.
Results
Eighty-nine tumors (57 benign and 32 malignant) were immunohistochemically processed for ER-α analysis. Eighteen tumors (14 benign and 4 malignant) had lost ER-α antigenicity (after several attempts, they remained unstained with an unreliable negative reaction) and were excluded from the study. The immunohistochemical detection of ER-α was reliable in 71 tumors from 49 animals and in the 2 normal mammary gland samples. Forty-three dysplasias and benign tumors from 23 patients and 28 malignant tumors from 26 animals were successfully stained and were included in the study.
ER immunostaining was localized in the nuclei of normal, benign, and malignant epithelial and myoepithelial cells. Minimal cytoplasmic staining was also observed in some samples. Normal stromal and neoplastic cells, cartilaginous cells, and bone cells in mixed tumors were negative. In normal and hyperplastic canine mammary glands, the ER-α expression was homogeneous in the cells of the same lobule (Fig. 1), but the intensity of immunostaining varied from one lobule to another. In the negative controls, all nuclei were negative. In the majority of tumors, the intensity of staining had a marked heterogeneity. Tumors showed several cell populations with groups of strongly positive ER-α cells close to areas with little or no ER-α expression (Figs. 2, 3).
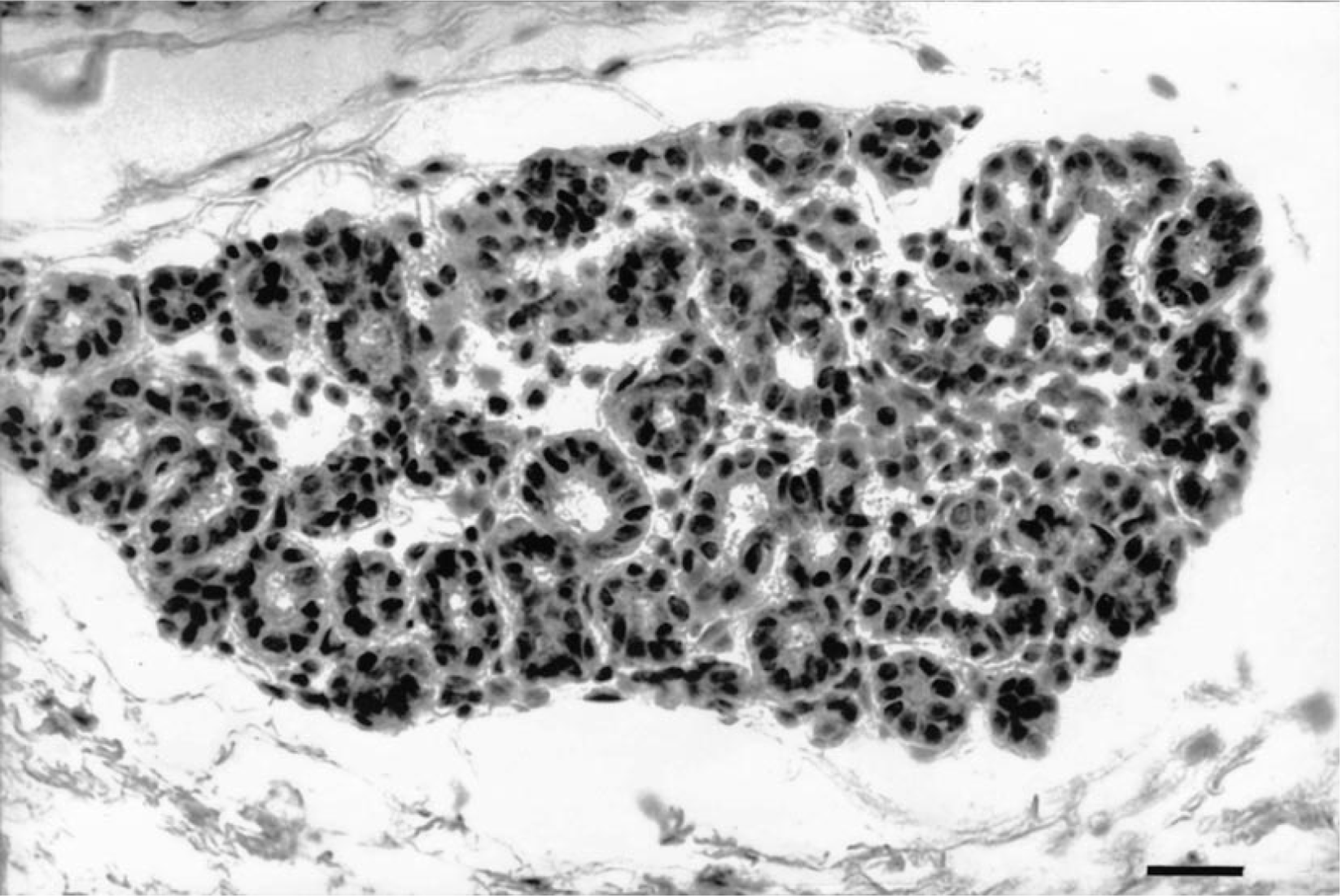
Mammary gland; dog 1. Intense nuclear ER-α immunostaining of hyperplasia (90% positive nuclei). Streptavidin-biotin-peroxidase complex method, monoclonal antibody anti-ER-α clone CC4-5, Gill's hematoxylin counterstain. Bar = 75 µm.
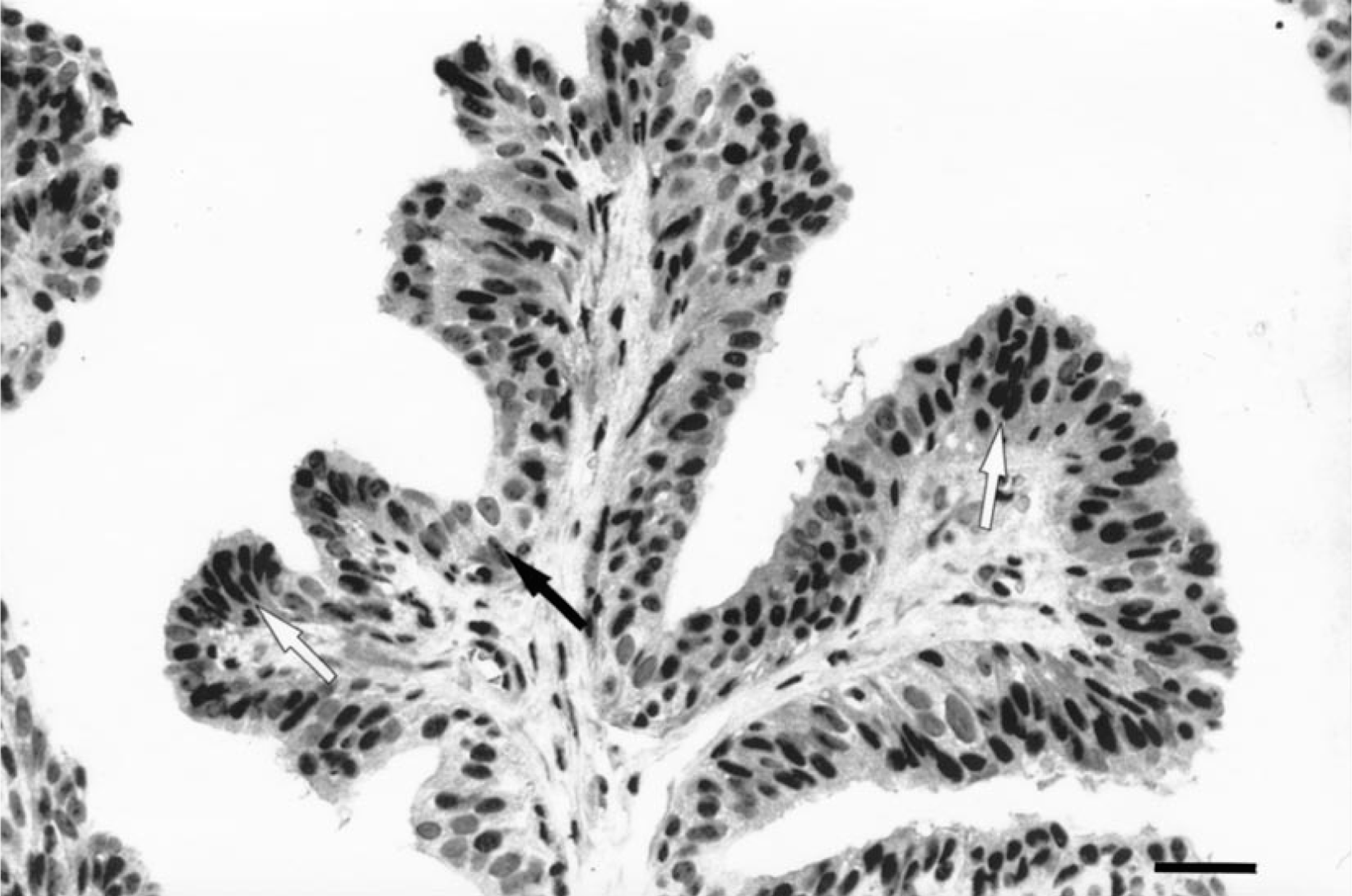
Mammary gland; dog 2. Papillomatous growth in a complex adenoma with ER-α immunostaining of 80%. Note variable intensity of nuclear staining. White and black arrows show, respectively, the highly stained and nonstained nuclei. Streptavidin-biotin-peroxidase complex method, monoclonal antibody anti-ER-α clone CC4-5, Gill's hematoxylin counterstain. Bar = 60 µm.
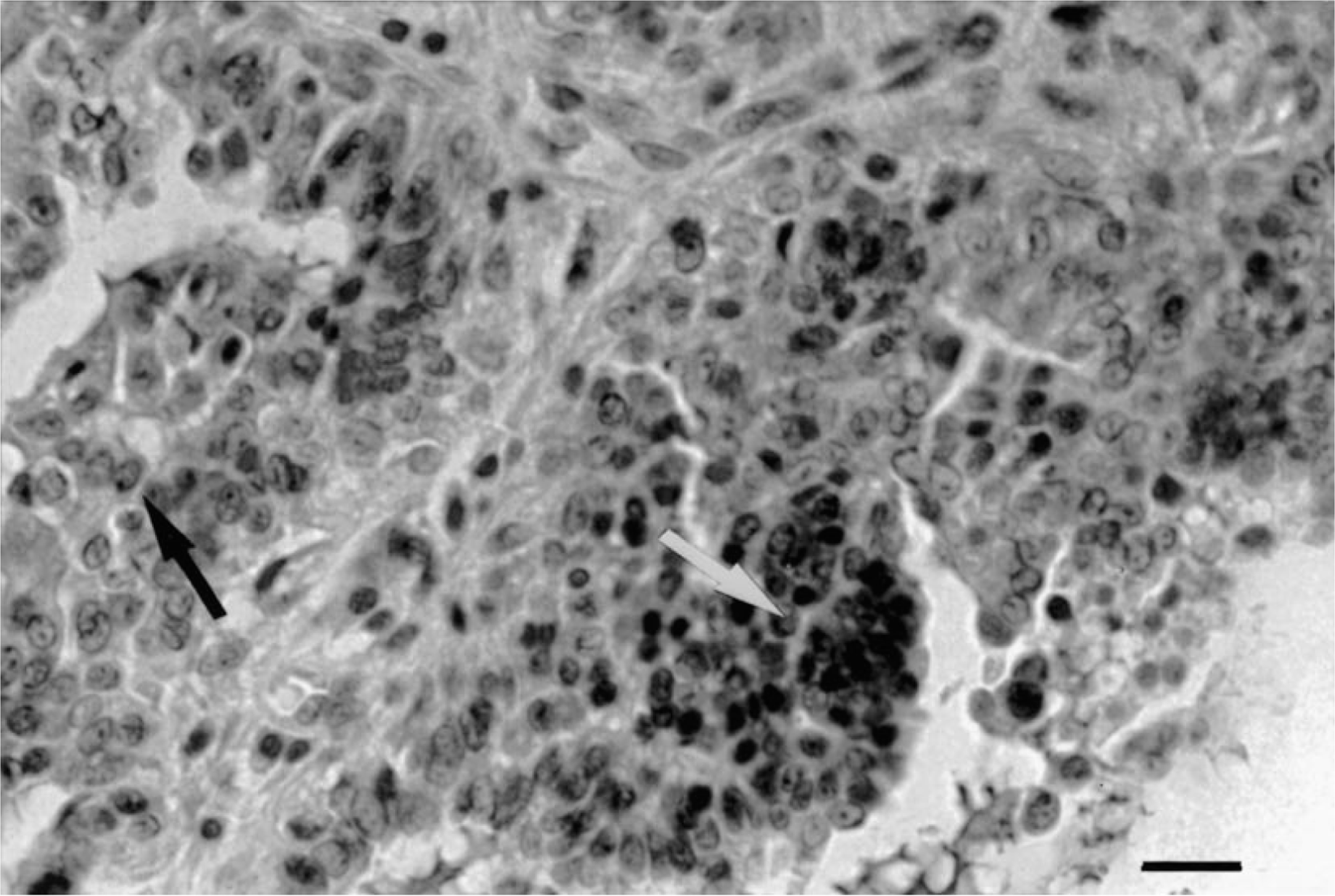
Mammary gland; dog 3. Epithelial area in a malignant mixed tumor with ER-α immunostaining of 40%. Note marked heterogeneity of staining. Some intensely positive nuclei appear grouped (white arrow) close to areas with little or no ER-α expression (black arrow). Streptavidin-biotin-peroxidase complex method, monoclonal antibody anti-ER-α clone CC4-5, Gill's hematoxylin counterstain. Bar = 75 µm.
Mean values of ERi and ERp were 54.98% (range = 0–240%, SD = 73.7%) and 24.59% (range = 0–90%, SD = 27.60%), respectively. Comparing both scores, a highly significant correlation was found between them (r = 0.87, P < 0.01). The relationship with the other variables included in the study was also similar for both indexes. To provide clearer understanding of the scores, in the univariate analysis only ERp scores (ER percentages) are shown.
Univariate analyses
Significant associations between epidemiologic and clinical data and ER-α expression are summarized in Table 1. ER-α expression was higher in young bitches (r = −0.33, P < 0.01). Significant increase in ER-α detection was observed in genitally intact animals (P < 0.01) and in patients with estrous cycle regularity (P = 0.03). Malignant tumors of bitches with a previous clinical history of pseudopregnancy (group DB) expressed significantly more ER-α. No relationship was found between breed, size of animal, age at first estrus or first parturition, number of estrus or full-term pregnancies, estrous cycle duration, pseudolactation, or previous estrus prevention treatment and ER-α content.
Significant associations between immunohistochemical detection of estrogen receptor alpha (ER-α) in canine mammary tumors and significant epidemiologic and clinical data.
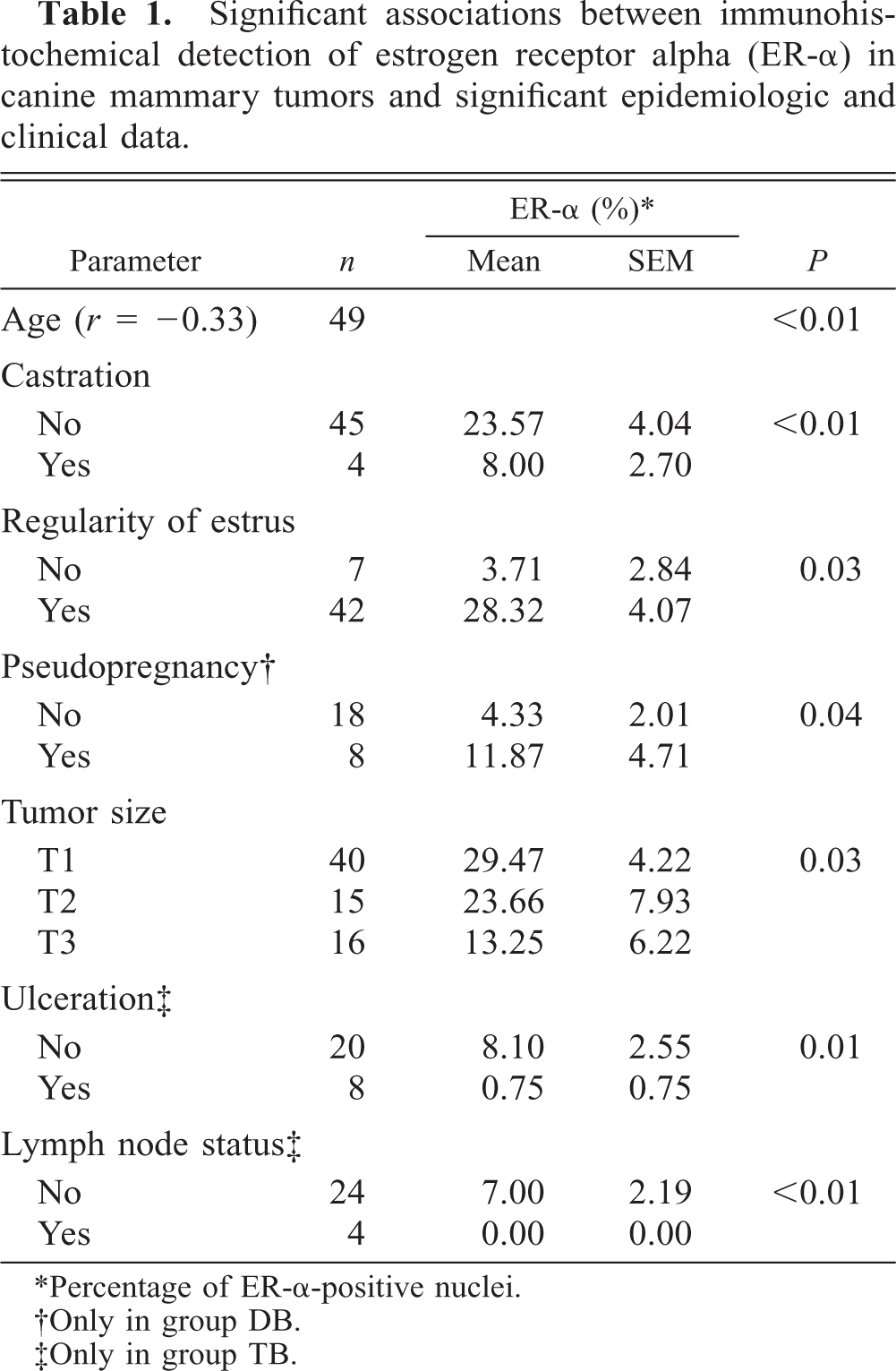
∗Percentage of ER-α-positive nuclei.
†Only in group DB.
‡Only in group TB.
Presence of ER-α decreased significantly with increasing tumor size (P = 0.03). However, considering the groups of benign (TA) or malignant (TB) tumors separately, rate of growth and tumor size were not significantly related to ER-α expression. For all tumors (groups TA and TB) and in malignant tumors (group TB), the presence of skin ulceration was associated with low expression of ER-α (P = 0.03 and 0.01, respectively). Considering the lymph node involvement at first presentation, low levels of ER-α were significantly associated with the regional stage (N1, with histologically confirmed lymph node involvement) (P < 0.01).
Regarding histologic features (Table 2), mean ER-α in the two normal mammary glands was 20% (range = 10–30%). Significant differences in ER-α immunostaining were observed between dysplasias (n = 11) and benign tumors (n = 32), and malignant tumors (n = 28) (P < 0.01). The ER-α mean was 42% (range = 0–80%) in dysplasias and 36.06% (range = 0–90%) in benign tumors. In malignant tumors, ER-α immunostaining was higher, but not significantly higher, in adenocarcinomas (7.76%, range = 0–40%) than in tumors without tubular formations such as solid carcinomas, squamous cells carcinomas, malignant mixed tumors, and sarcomas (4.46%, range = 0–30%). No significant differences were observed between dysplasias and benign tumors nor among the histopathologic malignant types. No significant variation of ER-α presence was noted in malignant tumors with different HMG or NG.
Significant associations between immunohistochemical detection of estrogen receptor alpha (ER-α) in canine mammary tumors and significant pathologic and follow-up variables.
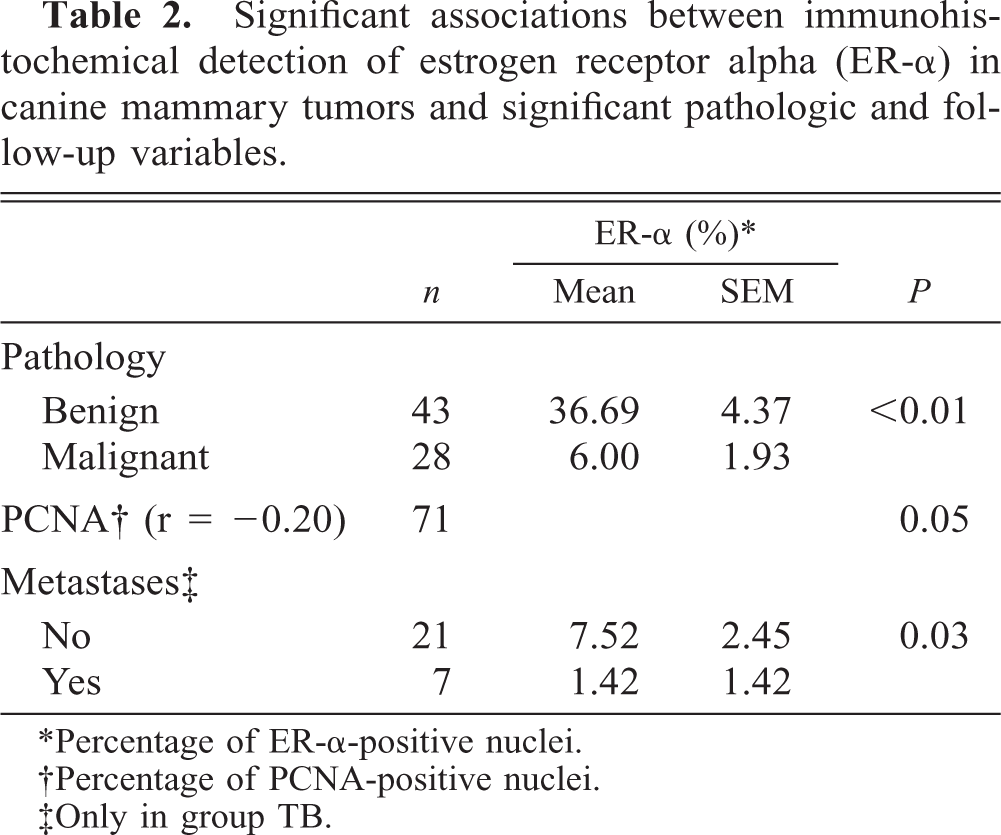
∗Percentage of ER-α-positive nuclei.
†Percentage of PCNA-positive nuclei.
‡Only in group TB.
ER-α score and Ki-67 index showed a nonsignificant inverse correlation (P = 0.09). ER-α immunoexpression decreased significantly with increased values of PCNA, considering all tumors as a group (P = 0.05).
Using the follow-up variables (Table 2) and taking only dogs with at least one malignant tumor (group DB), statistical association was noted between ER-α presence in the primary tumor and the appearance of metastases (P = 0.03) during the follow-up period. Recurrences were not related to ER-α values. No relationship was observed between detection and death by tumor, DFS, and OS in univariate analyses.
Multivariate analyses
To elucidate the prognostic value of ER-α and to understand the joint effect of other factors on follow-up variables (recurrences, metastases, DFS, and OS), several multivariate analyses were done using only animals of group DB with complete data for all the variables included (n = 23) (Table 3). Recurrences were not related to any variable included in the multivariate analysis. The Ki-67 index was the only independent prognostic indicator of metastases (P = 0.029).
Multivariate analyses of disease-free survival (DFS) and overall survival (OS) in dogs with malignant tumors, according to the formula: odds ratio = exp (a 0 + a 1 X 1 + … + an ), where a 0 is the constant of the dependent variable and a 1, a 2, …, an are the coefficients affecting the sets of related independent variables included in the model (X 1, X 2, …, Xn ).

∗The independent variables to predict DFS or OS are selected in several steps according to their significance. The variable selected in one step is then considered a constant to study the combined effect of the other variables on the dependent variables (DFS, OS).
†Percentage of ER-α-positive nuclei.
‡Proliferation index, percentage of Ki-67-positive nuclei.
§Because regional stage was found in most of the cases with low to medium OS, exponential coefficient and confidence interval were not calculated.
With DFS and OS as dependent variables, age, Ki-67 index, ERi, ERp, lymph node involvement, tumor size, ulceration, HMG, and NG were used as related variables. Taking DFS as a dependent variable (n = 23; DFS < 18 months, n = 16; DFS > 18 months, n = 7), age (P = 0.01) emerged as an independent and significant variable in step 1. In step 2 (age constant), ERp was the next variable affecting DFS (P = 0.04). In a third step (age and ERp constants), Ki-67 index was the only variable selected by the analysis near to a significant P value (0.08). All other variables were not independent prognostic factors of DFS. When OS was analyzed as a dependent variable (n = 23; OS < 18 months; n = 13; OS > 18 months, n = 10), clinical stage (P = 0.023) and age (P = 0.047) were independent prognostic factors selected in step 1 and 2, respectively. In step 3, ERp was selected (P = 0.012) as a prognostic variable. No other variables independently affected OS in the multivariate analyses.
Discussion
The presence of ER-α in CMT was detected using immunohistochemical methods. Immunostaining of ER-α was successful in the majority of the 89 tumors included in the study. Only a small number of neoplasms (n = 18, 14 benign and four malignant) showed no reactivity. The negative reaction observed was not reliable because the sample also contained some negative hyperplastic or normal mammary gland (which should have had positive reaction to ER-α). Similar results have been described in human tissues 6 , 45 and can be attributed to loss of tissue antigenicity. The content of ER-α in CMT has been analyzed previously by biochemical methods, especially DCC. 25 , 26 , 29 , 38 The immunohistochemical demonstration of estrogen receptors used in this study has several advantages over the biochemical measurement, the main one being the preservation of tissue structure. As opposed to biochemical methodology, the direct observation of the ER-α expression in tissue sections allows one to see the heterogeneity of staining and to eliminate the counting of positive normal mammary cells. 7 , 19 This difference in methodology may explain some of the different results obtained in this study in comparison with previous reports.
ER-α immunostaining in normal and neoplastic canine mammary glands was focused in the nuclear area and was heterogeneous regarding the intensity and the number of positive cells. Similar results have been found in human breast cancer 31 , 37 , 45 , 47 and can be attributed to differentiated cellular activity. Comparing the two counting methods to evaluate ER-α by immunohistochemistry (ERi and ERp), a highly significant correlation was seen between the two scores. ERp score (percentage of ER positive cells) was easier to calculate and to compare with other variables in CMT, as occurs in human breast tumors. 37 , 39 , 48
In this study, some epidemiologic, clinical, and histopathologic variables were associated with the presence of ER-α. Ovariectomized bitches and those with irregular estrous cycles had a lower ERp than did genitally intact bitches and those with regular estrous cycles, indicating neoplastic growth is influenced by factors other than estrogen in tumors with poor prognosis. Malignant mammary tumors of animals with previous histories of pseudopregnancy had a highly significant ER-α content, suggesting a possible beneficial effect of pseudopregnancy hormonal status in these animals. Nevertheless, in other studies, previous pregnancy, pseudolactation, or progestin administration were not related to ER-α levels. 9 , 27 , 42 Age, tumor size, skin ulceration, and clinical stage, four important clinical prognostic factors in CMT, 20 , 32 , 33 , 42 , 43 were associated with ER-α content. According to previous reports describing CMT, an inverse correlation was observed between age and ER-α presence. 9 , 38 , 42 In contrast, breast carcinomas of postmenopausal women contain more ER-α than do those of premenopausal women. 1 , 12 , 39 This coincidence between poor prognosis and age in bitches is not observed in women with mammary cancer. 1 , 12 , 39 Regarding the controversial association between tumor size and ER-α immunoexpression, 9 , 27 in this study a significant decrease in ER-α in larger tumors (group TA and TB) was observed. Skin ulceration was associated with lower ER-α expression in all tumors (group TA and TB) and in malignant tumors group (group TB). In contrast to other reports, 42 , 43 primary neoplasias of regional stage N1 (lymph node involvement histologically confirmed) have lower ER-α than those of local stage N0. ER-α content was also related to histopathology and tumor growth fraction. According to other authors who used biochemical assays, 17 , 22 , 25 , 42 ER-α values were significantly higher in benign tumors than in malignant tumors (P < 0.01). There were no significant differences among the different types of tumors, although simple and complex adenocarcinomas showed more ER-α expression than did undifferentiated, solid carcinomas or sarcomas. This fact indicates decreased values of ER-α with malignancy, although there was no significant association between ER-α and HMG nor NG, as has also been noted in previous reports regarding CMT. 42 Previous studies by our group have established the validity of the tumor growth fraction measured with Ki-67 and PCNA indexes as prognostic indicators. 32 In the present study, ER-α values were compared with proliferative activity. PCNA index was significantly correlated with ER-α, and the correlation with Ki-67 index was close to significant. This finding is in agreement with those of other studies of human breast cancer, indicating that well-differentiated tumors can maintain some hormonal regulatory mechanisms and have a low proliferation rate. The lack of correlation between Ki-67 or PCNA and ER-α values in some cases may be associated with the number of cells in G0 phase (negative to Ki-67 or PCNA). 13 , 14 , 48
One of the most important clinical events in cancer progression is the occurrence of metastases. We found significant differences in ER-α immunoreactivity with regard to the development of metastases at follow-up (P < 0.03). Lower rates of ER-α expression were observed in primary tumors that had given rise to distant metastases during the follow-up period. Multivariate analyses were performed to complement the univariate tests to study the joint effect and the real prognostic significance of some of the experimental variables over the follow-up variables (recurrences, apparition of distant metastases, DFS, and OS). ERp emerged as a strong independent predictor for DFS and OS, although the number of cases included in the multivariate analyses was low. Other factors such as age, Ki-67, and clinical stage were determined to be prognostic indicators, 32 in accordance with previous studies in human breast carcinomas. 36 , 40 , 49
Immunohistochemical detection of ER-α in CMT is easy and can be performed in a histopathology laboratory using an anti-ER-α monoclonal antibody. Moreover, Ki-67 and ER-α values could be used in the elaboration of future treatment protocols in CMT, as occurs in humans. 37 Endocrine therapy could be useful in carcinomas expressing ER-α and a low Ki-67, but not in positive ER-α carcinomas and tumors with a large growth fraction. 13 High Ki-67 counts and low ER-α content would suggest the need for chemotherapy and/or radiotherapy. 37
The results of this study indicate that hormonal status and proliferation of CMT are independent prognostic factors to predict DFS and OS. Knowledge of these parameters is necessary to select the correct therapy. Further studies including a great number of malignant tumors and appropriate therapy are needed to confirm the global validity of ER-α expression in this disease.
Footnotes
Acknowledgements
We thank Dr. Pedro Cuesta, the Processing Data Center of the Complutense University, for his assistance with the statistical work. We also appreciate the assistance of Mr. David Bruhn in preparing the English manuscript and Mr. Martin Cabrera for photography. This study was partially supported by the Complutense Research Project PR181/96-6800.
