Abstract
Cyclooxygenase (COX)-2 is involved in several physiologic and pathologic processes. COX-2 is overexpressed in human and canine prostate cancer, but little is known about COX-2 inducers in the prostate. Our objective was to investigate the effect of sex steroid hormones on COX-2 expression in the canine prostate in vivo. COX-2 expression was evaluated by immunohistochemistry in intact and castrated dogs treated with exogenous androgen or estrogen. Results showed that no COX-2 staining was observed in prostates of untreated or androgen-treated castrated or intact dogs. However, treatment of intact and castrated dogs with estrogen resulted in squamous metaplasia with intense COX-2 expression observed in both squamous epithelial cells and in cells of acini without metaplasia. This is the first report to demonstrate the induction of COX-2 by estrogen in the prostate in vivo.
The enzyme cyclooxygenase (COX) is responsible for the conversion of arachidonic acid into prostaglandin H2, a common precursor for the synthesis of all prostanoids. 15 COX-derived prostaglandins have been shown to play a key role in several physiologic and pathologic processes, including gastrointestinal and reproductive functions, bone metabolism, glomerular filtration, inflammation, and oncogenesis. 11 , 15 COX enzymes represent the target of nonsteroidal anti-inflammatory drugs. Two isoforms of COX (COX-1 and −2) have been characterized. 11 , 15 COX-1 is present in a wide variety of tissues and is known as the constitutive form. COX-2 is generally undetectable in most tissues, but can be induced by various agonists, and is referred to as the inducible form. Several inducers of COX-2 expression have been identified, including endotoxins, activated oncogenes, cytokines, tumor promoters, and hormones. 10 , 11 Recent studies have revealed that an alternative spliced variant of the COX-1 gene is expressed in the canine cerebral cortex. 4
COX-2 is not present in the normal prostate, but overexpression of the enzyme has been reported in human and canine prostatic adenocarcinomas. 7 , 14 On the basis of this finding and on other experimental evidence, a role for COX-2 and prostaglandins in prostate carcinogenesis has been proposed. 11 However, the signals leading to COX-2 expression in the prostate are currently unknown. Considering that the prostate is under the influence of sex steroid hormones, the objective of this study was to study the effect of these hormones on COX-2 expression in the canine prostate in vivo.
Prostatic tissues were collected from castrated (n = 42) or intact (n = 39) adult mongrel dogs that were subjected to hormonal manipulations or left untreated. Hormonal treatments consisted of intramuscular injections (three times a week for 3 weeks) of 25 mg of the androgen 5α-androstane-3α, 17β-diol (3,17β-diol [Steraloids Inc., Wilton, NHcb; n = 9 and n = 8 castrated and intact dogs, respectively) or 2.5 mg of the estrogen 17β-estradiol 17-cyclopentyl proprionate (ECP, [Steraloids Inc.]; n = 16 and n = 4 castrated and intact dogs, respectively) diluted in 1 ml of propylene glycol, as described. 12 Hormonal treatments in castrated animals were initiated 4 weeks after castration, and animals not receiving hormonal treatment included 17 castrated and 27 intact dogs.
Immunohistochemical staining was performed using the Vectastain ABC kit (Vector Laboratories, Burlingame, CA) and the anti–COX-2 antibody MF243, as described previously. 14 In brief, formalin-fixed tissues were paraffin embedded, and 3-µm-thick sections were prepared and deparaffined through graded alcohol series. Endogenous peroxidase was quenched by incubating the slides in 0.3% hydrogen peroxide in methanol for 30 minutes. After rinsing in phosphate-buffered saline (PBS) for 15 minutes, sections were incubated with diluted normal goat serum for 20 minutes at room temperature. The antibody MF243 (kindly provided by Dr. Stacia Kargman, Merck Frosst Centre for Therapeutic Research, Pointe-Claire-Dorval, Québec, Canada) diluted in PBS (1 : 7,500) was applied, and sections were incubated overnight at 4 C. Control sections were incubated with PBS or with nonimmune rabbit serum. After rinsing in PBS for 10 minutes, a biotinylated goat anti-rabbit antibody (1 : 222 dilution; Vector Laboratories, Burlingame, CA) was applied, and sections were incubated for 45 minutes at room temperature. Sections were washed in PBS for 10 minutes and incubated with the avidin DH–biotinylated horseradish peroxidase H reagents for 45 minutes at room temperature. After PBS wash for 10 minutes, the reaction was revealed using diaminobenzidine tetrahydrochloride (DAB) as the peroxidase substrate. Sections were counterstained with Gill's hematoxylin stain and mounted.
Chi-square and Pearson's tests were used to analyze frequencies of COX-2 expression between intact and castrated animals that received either 3,17β-diol or ECP. Statistical analyses were performed using the JMP Software (SAS Institute Inc., Cary, NC).
Results showed that prostatic tissues of untreated intact animals had a well-developed secretory epithelium, but none expressed COX-2 (Figs. 1, 7). Castration led to the disappearance of the secretory epithelium, with only the basal cell layer remaining (Fig. 2), whereas treatment of castrated dogs with the androgen 3, 17β-diol restored the secretory epithelium (Fig. 3). However, no COX-2 staining was observed in untreated castrated animals and in 3,17β-diol–treated castrated dogs (Figs. 2, 3, 7). Likewise, no COX-2 immunoreactivity was detected in prostates of intact dogs treated with 3,17β-diol (Fig. 7). In contrast, a clear pattern of COX-2 induction was observed in prostatic tissues of intact and castrated dogs treated with the estrogen ECP (Figs. 4–7). Treatment of castrated dogs with estrogen resulted in the metaplasia of basal cells into a squamous epithelium, and intense COX-2 expression was observed in both the squamous epithelial cells (Fig. 5) and in the basal cells of acini without metaplasia. Similar results were observed in prostates of intact ECP-treated dogs (Fig. 4).
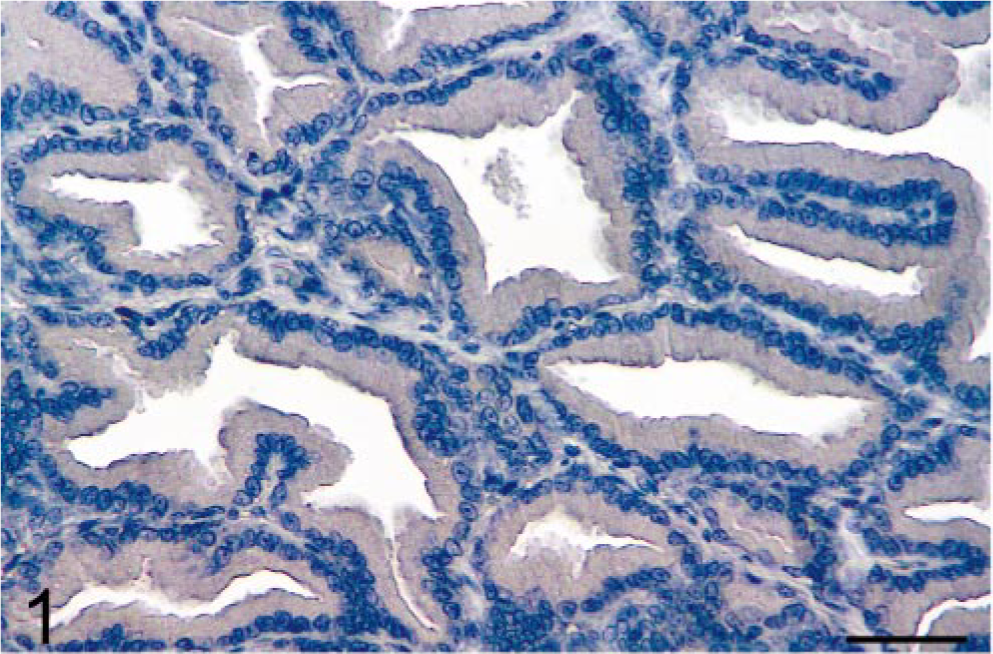
Prostate; intact dog. Normal prostatic tissue from an intact dog is negative for COX-2 immunoreactivity. Avidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain. Bar = 35 µm.
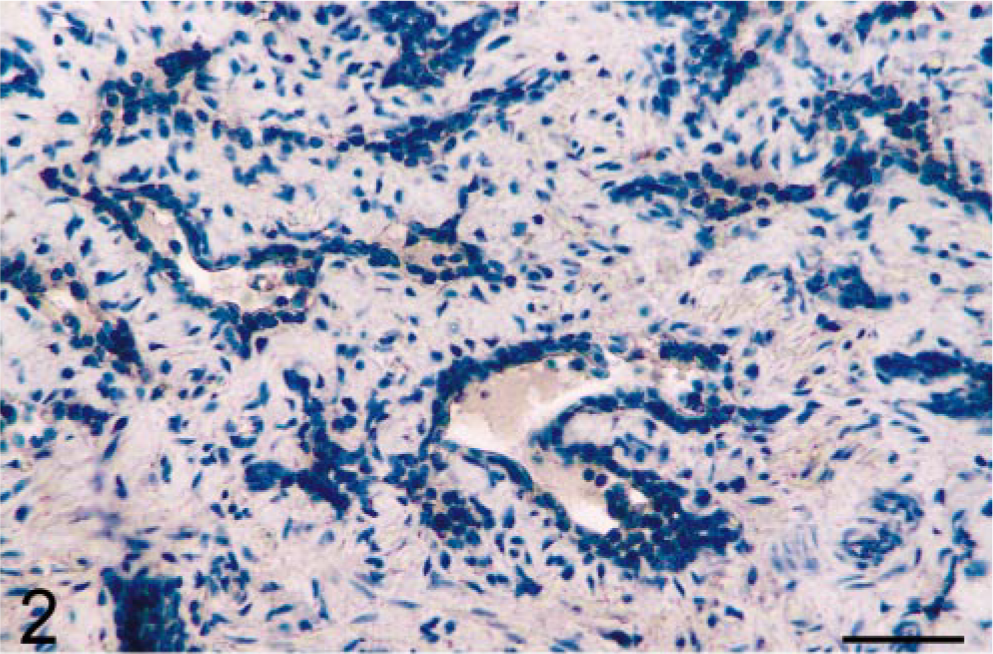
Prostate; castrated dog. Castration results in the disappearance of luminal cells, leaving only the basal cells that are COX-2 negative. Avidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain. Bar = 35 µm.
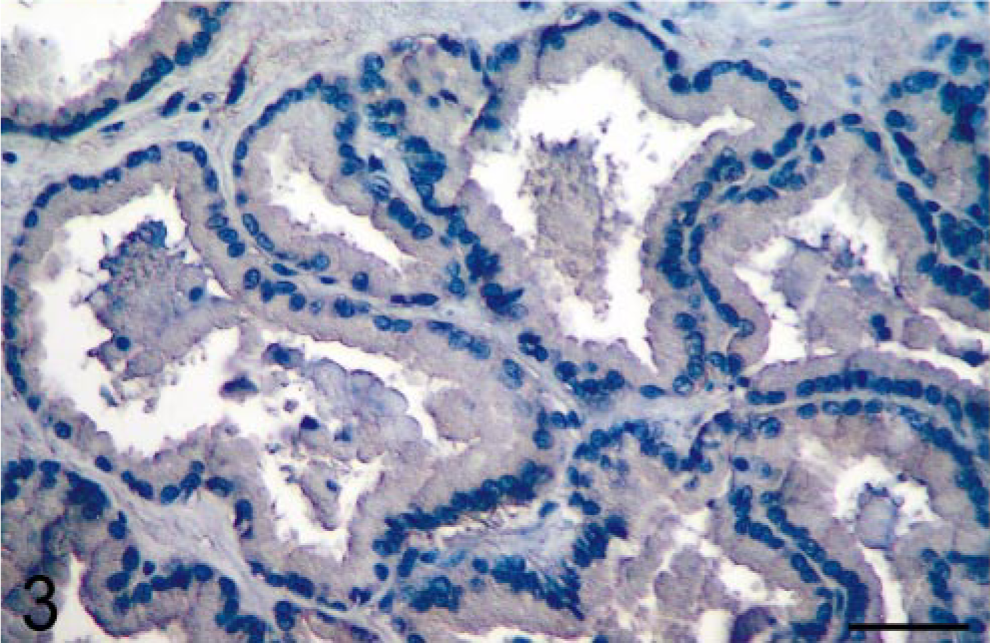
Prostate; castrated dog treated with the androgen 5α-androstane-3α, 17β-diol. Administration of androgen after castration restores a luminal epithelium, but COX-2 expression remains negative. Avidin–biotin–peroxidase complex method, Gill's hematoxylin counter-stain. Bar = 35 µm.
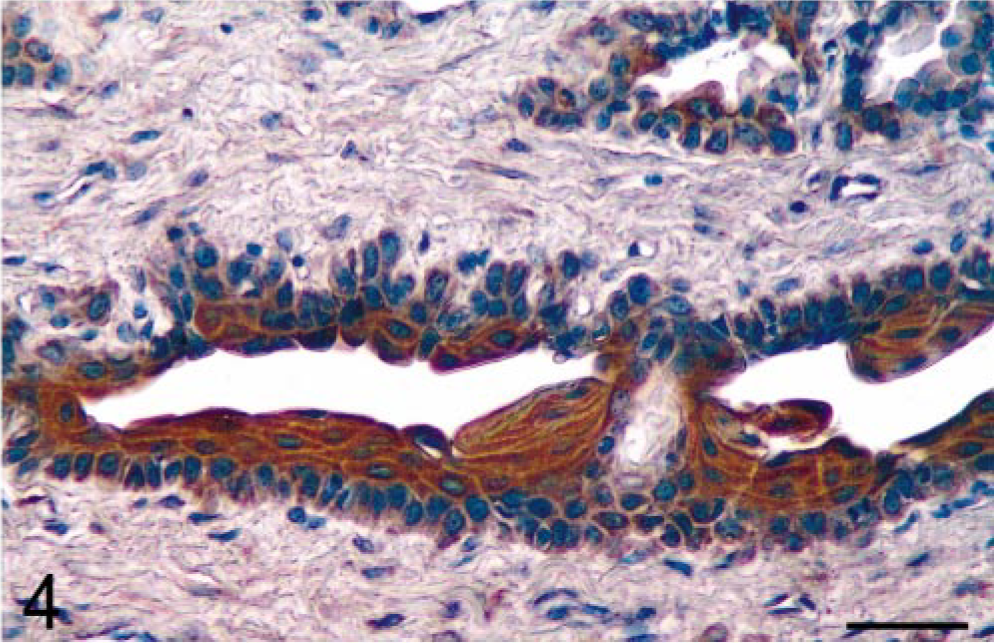
Prostate; intact dog treated with the estrogen 17β-estradiol 17-cyclopentyl proprionate. Administration of estrogen to an intact dog results in abundant COX-2 expression in the squamous epithelium. Avidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain. Bar = 35 µm.
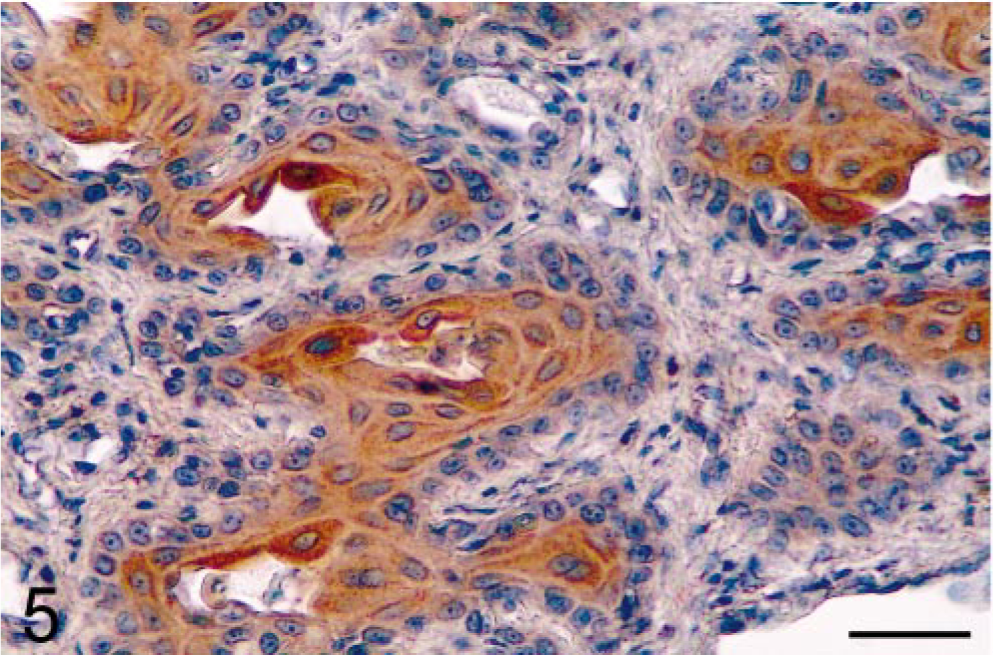
Prostate; castrated dog treated with the estrogen 17β-estradiol 17-cyclopentyl proprionate. Administration of estrogen after castration results in abundant COX-2 expression in the squamous epithelium. Avidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain. Bar = 35 µm.
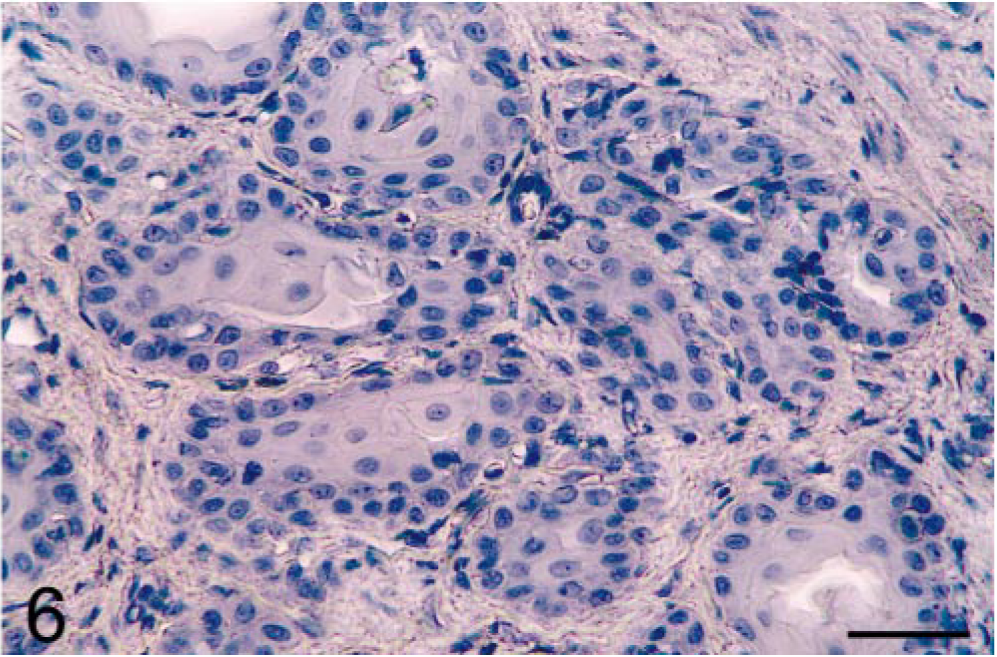
Prostate; castrated dog treated with the estrogen 17β-estradiol 17-cyclopentyl proprionate. Control staining was negative when the primary antibody was replaced with PBS. Avidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain. Bar = 35 µm.
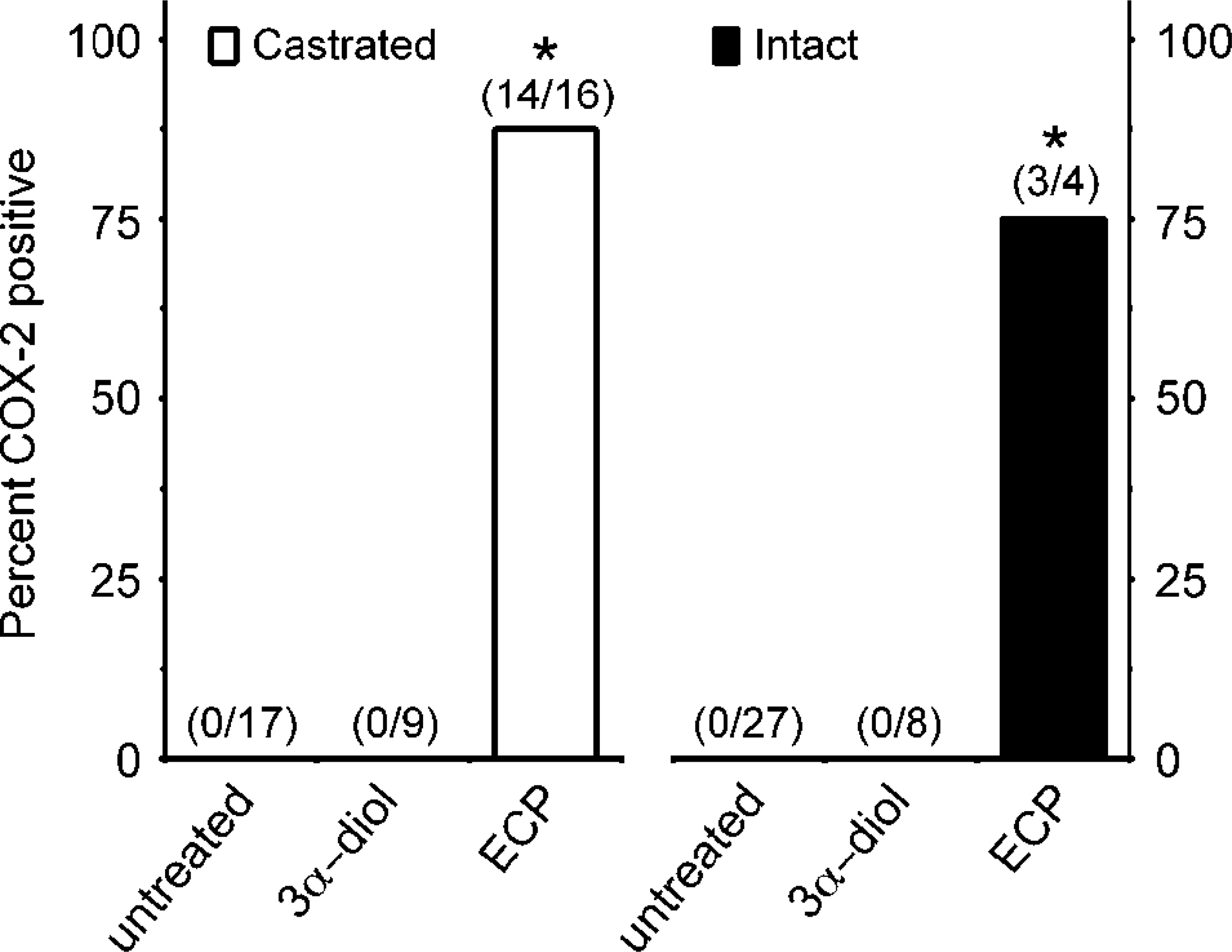
Induction of COX-2 expression in prostates of estrogen-treated dogs. The expression of COX-2 was detected by immunohistochemistry on prostatic tissues of castrated (open bars) or intact (close bars) adult dogs that were subjected to hormonal manipulations or left untreated. Hormonal treatments included the administration of the androgen 5α-androstane-3α, 17β-diol (3,17β-diol) or the estrogen 17β-estradiol 17-cyclopentyl proprionate (ECP). Numbers in parentheses indicated number of COX-2–positive animals over total number of dogs in each group. Bars with an asterisk are significantly different from the corresponding untreated group (two-sided chi-square and Pearson's tests; P < 0.05).
This report provides for the first time evidence that estrogen, but not androgen, induces COX-2 expression in prostatic tissues in vivo. The induction of squamous metaplasia is a well-documented effect of estrogen stimulation on prostatic tissues, and this study identifies the sites of COX-2 induction as epithelial cells that have undergone squamous metaplasia, as well as cells remaining in acini after estrogen treatment. In castrated dogs, these cells have previously been shown to be basal cells and to express increased levels of different protein tyrosine kinases after estrogen treatment. 1 , 12 Because these tyrosine kinases are thought to be involved in the transmission of the estrogen-dependent mitogenic signals, 1 it will be interesting to determine whether they also contribute to the transduction pathway leading to COX-2 expression. Also, it will be important to establish whether COX-2 induction results from a direct action of estrogens on the epithelial cells or involves indirect paracrine interactions through stromal cells. The presence of estrogen receptor (ER)α (ERα) and ERβ in stromal and epithelial compartments suggests that both pathways could be involved. 2 , 3
The precise significance of COX-2 expression in prostates of estrogen-treated animals is currently unknown. Overexpression of COX-2 has been demonstrated in cancerous prostatic tissues of humans and dogs, and a potential association between the enzyme and prostate carcinogenesis has been proposed. 7 , 11 , 14 Experimental studies suggest that estrogens could be involved in prostate cancer progression. 3 , 9 Indeed, it has been shown that sequential treatment with testosterone and estrogens is more effective than testosterone alone at inducing prostatic adenocarcinomas in Noble rats. 13 The expression of estrogen biosynthetic and metabolic enzymes has recently been demonstrated in microdissected prostatic tumor cells and malignant prostatic cell lines. 5 , 8 In a recent study, forced overexpression of COX-2 into the human cancerous prostatic cell line LNCaP cells, which express low levels of COX-2, resulted in increased proliferation in vitro and tumor growth rate in vivo. 6 These effects were independent of androgen receptors and led the authors to speculate that COX-2 could contribute to progression of prostate cancer from an androgen-dependent to an androgen-independent disease. Interestingly, elevated ERα expression was found in recurring human prostatic carcinomas after androgen ablation therapy, suggesting a role for ER in prostate cancer progression to an androgen-insensitive stage. 2 Results of this study indicate that estrogens, directly or indirectly, are able to induce the expression of COX-2 in the prostate. The consequences and/or implications of this estrogen-mediated COX-2 expression in the prostate deserves further investigation.
Footnotes
Acknowledgements
Supported in part by the Natural Sciences and Engineering Council of Canada grant 195188 (to M. Doré) and the Canadian Institutes for Health Research (CIHR) grant MT-13190 (to J. Sirois). J. Sirois is a recipient of CIHR Investigator Award.
