Abstract
A Chlamydophila abortus-induced abortion model was carried out on the basis of the experimental infection of ewes at day 75 of gestation. The infection induced abortions and the birth of weak lambs during the last 3 weeks of pregnancy. To study the kinetics of the infection in the placenta and in other organs, infected ewes were killed at 105, 120, and 130 days of gestation and also several days after abortion or parturition. Infected ewes developed a systemic infection that caused a mild and transient pneumonia and focal hepatitis. Pathologic changes were observed in placentas at 120 day of gestation, although the lesions varied between animals and even between placentomes of the same placenta. The first placental area infected was the maternal stroma and epithelium next to the intercaruncular areas, where neutrophilic response seemed to control the infection. A substantial degree of multiplication of C. abortus was then observed in the trophoblast cells of the placentome, periplacentomal choriallantoic membranes, and hilius, with an inflammatory exudate composed mainly of neutrophils, some macrophages, and very scarce lymphocytes. After abortion, the lesions affected the intercotyledonary areas of the aborted placentas, whereas in the uterus significant lymphocyte infiltration was observed, together with a rapid decrease of the C. abortus antigen in the degenerated caruncular tissues.
Chlamydophyla abortus (formerly Chlamydia psittaci serotype 1) is a gram-negative obligate intracellular bacterium, whose main target organ is the placenta. 16 This bacterium is the etiologic agent of enzootic abortion in small ruminants, a disease that has spread worldwide and which causes abortion during the last third of gestation. 23,27 Besides the severe economic losses it causes, it also represents an emerging zoonotic risk for pregnant women, and abortions induced by C. abortus have been reported in recent years in USA 19, The Netherlands 20, Switzerland, 26 and UK. 17
Adult ewes are infected as a result of contamination of lambing pens or of pasture by fetal membranes and discharges. Few or no clinical signs are observed in nonpregnant animals until, during a subsequent pregnancy, ewes abort. C. abortus has proved impossible to isolate from tissues of infected animals 7 days after infection. 1 Abortion always appears in the last weeks of gestation regardless of the moment of infection. 13 Extensive pathologic changes have been observed in the placenta 4 weeks after infection and just before abortion in a previous report of an experimental infection. 8 The data as a whole point to the existence of a latent stage of C. abortus, the localization of which remains unclear. A study of the presence of C. abortus in local lymph nodes after subcutaneous inoculation provided positive polymerase chain reaction results but no bacteria were isolated, 8 suggesting that lymphoid tissue is a candidate for the site of C. abortus latency. However, nonpregnant ewes were used in the study, and so, the relationship between latency, pregnancy, and abortion was not investigated. Similarly, the mechanisms that interrupt the stage of latency are poorly understood.
Our laboratory has described previously the kinetics of C. abortus colonization of placenta in an experimental mouse model, 7 extending from the first cells infected to the massive multiplication that leads to abortion. The aim of this work was to study the kinetics of C. abortus infection in the natural host and also to characterize the immune response induced by the microorganism using serologic, immunohistochemic, and cultural techniques, with special attention paid to the placenta to deepen our knowledge of the pathogenesis of ovine enzootic abortion.
Materials and Methods
Animals and experimental design
Fourteen naive Castellana ewes, aged 12 months, were obtained from a flock free of clinical ovine enzootic abortion for the previous 5 years. Ewes were seronegative to C. abortus antigen according to the complement fixation test (CFT) and enzyme linked immunosorbent assay (ELISA) using techniques described previously. 5 Additional analysis by CFT showed that ewes were also seronegative to Brucella melitensis and Coxiella burnetii. Ewes fed on grass and alfalfa, straw and cereal (50% oat and 50% barley) and drank water ad libitum. The ewes were synchronized for estrous using vaginal sponges with medroxyprogesterone acetate (Repromap, Syva, Spain) and served by rams with the same serologic status. Gestation was confirmed by ultrasound scanning 40 days after breeding. Confirmed pregnant animals were hosted in a facility with a biosecurity level P2. At 75 days of gestation (dg) 10 ewes were inoculated subcutaneously in the left shoulder with 4 ml of an inoculum containing 107 inclusion-forming unit (IFU) of C. abortus AB7 strain 29 in phosphate-buffered saline (PBS). Two infected ewes were killed at 105 dg (ewe Nos. 1 and 2), 120 dg (ewe Nos. 3 and 4), and 130 dg (ewe Nos. 5 and 6). The other four infected ewes were maintained until abortion or parturition, before being killed 3 (ewe Nos. 7 and 8) or 7 (ewe Nos. 9 and 10) days after abortion or parturition. Four noninfected ewes, maintained in a separate room, were used as control uninfected group, two being killed at 130 dg and two 3 days after parturition. The tissues of these uninfected ewes served as negative controls of the different techniques used in this study and allowed comparision to the histopathologic changes induced by C. abortus with normal tissues.
Rectal temperatures were recorded daily from the 3 days before the inoculation to the 10 days after inoculation. Ewes were bled the day before inoculation and subsequently every 15 days until abortion or parturition, and then at day 7 post-abortion or parturition: sera were stored at −20 C until used. Vaginal swabs were taken at 1, 2, 3, 5, and 7 days post-abortion or parturition. All fetuses or lambs were weighed and the crown-rump length was measured according to a method described previously. 15
Serology
An ELISA method described previously 5 was used to evaluate the changes in the C. abortus–specific antibody titer. Sera diluted at 1/100 were tested in triplicate, and the mean value of optical density was used to compare infected and noninfected values.
Necropsy and sampling
The ewes were euthanatized with an overdose of sodium pentobarbital administered intravenously and necropsied. In pregnant ewes, the uterus was opened and, after extraction of the fetuses, five placentomes and five interplacentomal areas adjacent to the placentomes were collected randomly from each ewe. Samples were observed with a lens (Leica, Wetzlar, Germany) to determine the gross pathology. Subsequently, each sample was subdivided into two blocks, one of which was fixed in 10% formaldehyde in PBS and the other snap frozen in 2-methylbutane cooled in liquid nitrogen and stored at −70 C until use for C. abortus isolation. Samples of the abomasum, liver, spleen, and lung from the fetuses and of liver, spleen, lung, and subscapular lymph node from ewes were fixed in 10% formaldehyde. In addition, from the ewes killed after abortion or parturition, caruncular and intercaruncular areas from uterus were taken and fixed in 10% formaldehyde. Additionally, aborted placentas and samples of abomasum, liver, spleen, and lung from dead lamb were fixed in formaldehyde and frozen at −70 C for C. abortus isolation.
Histopathology and immunohistochemistry
Samples fixed in formalin were embedded in paraffin wax and sectioned (4 µm). The sections were then stained with hematoxylin and eosin or the avidin–biotin–peroxidase complex (ABC) technique was applied with a monoclonal antichlamydial lipopolysaccharide (LPS) antibody 28 as follows. After hydration, the sections were washed in Tris-buffered saline solution 0.05 M (TBS), pH 7.6, treated with hydrogen peroxide 0.5% in methanol for 30 minutes, washed in TBS and pretreated with normal rabbit serum (Dakopatts, Glos-trup, Denmark) diluted 1 : 100 in TBS to block nonspecific binding sites. The slides were then incubated with the anti-LPS monoclonal antibody diluted 1 : 500 in TBS in a moist chamber for 60 minutes; they were again washed in TBS and incubated with biotinylated rabbit anti-mouse IgG (Dakopatts), diluted 1 : 250, for 45 minutes. Finally, the slides were washed in TBS and incubated with the ABC (Vector Laboratories, Burlingame, CA) for 45 minutes. Peroxidase activity was detected by incubation in a solution of 3,3′-diaminobenzidine tetrahydrochloride (Sigma, St. Louis, MO, USA) (0.05%) and hydrogen peroxide (0.03%) in TBS, pH 7.6; the reaction was stopped after 5 minutes by rinsing the slides in tap water. The slides were then counterstained in Mayer's hematoxylin for 2 minutes, rinsed in tap water, and covered with a coverslip. Every slide was analyzed together with an adjacent section labeled without the primary antibody to evaluate the background. Tissues from the uninfected ewes served also as negative control of the technique.
Immunophenotypic study of the placenta
Frozen placental sections (5 µm) were cut with a cryostat, mounted on slides, and fixed in acetone for 10 minutes. Sections were immunolabeled with the following monoclonal antibodies purchased from VMRD Pullman Inc. (Pullman, WA) that recognize surface markers of ovine lymphocytes: H58A (anti-MHC I), H42A (anti-MHC II), BAQ155A (anti-IgM), 36F18 (anti-CD2), GC50A1 (anti-CD4), CACT80C (anti-CD8), and CACTB6A (anti-TCR-1). The same ABC technique as above was followed but, after acetone fixation, the sections were rehydrated for 5 minutes in TBS and treated with phenylhydrazine (Sigma) 0.05% in TBS for 40 minutes to inhibit the endogenous peroxidase, as described previously. 22 The same controls described above were carried out with these samples. Tissue from the uninfected ewes served in this case to discern the normal distribution of the leukocyte subsets.
Isolation of C. abortus
Frozen samples from placentomes and material from abortions were thawed and homogenized with a Stomacher (Seward Medical, Norfolk, UK) in sterile saline buffer. The C. abortus burden in these tissues was evaluated by counting the number of IFUs after isolation on McCoy cell monolayers according to a method described previously. 12 The results are represented as the number of IFUs per gram of tissue. Samples from vaginal swabs were processed similarly, although the results are expressed as the number of IFUs per vaginal swab. The detection limit of the technique was 400 IFU/g of tissue or 80 IFU/vaginal swab. Additional cultures were carried out in Columbia agar to discard bacterial contamination. No signs of viral contamination were observed in the McCoy cell monolayers after infection.
Results
Clinical signs
The subcutaneous infection of ewes with C. abortus, was not associated with abnormal clinical signs except for a slight increase in the rectal temperature at days 1 and 2 after infection. Uninfected ewes showed a rectal temperature of 39.4 ± 0.2 C and 39.2 ± 0.3 C, respectively, whereas the temperature of the infected ewes was 40.6 ± 0.1 C and 40.4 ± 0.2 C during the first two days after infection. Among the infected ewes maintained until abortion or parturition, three aborted at 128 dg (ewe No. 7), 131 dg (ewe No. 8), and 133 dg (ewe No. 9), whereas ewe No. 10 gave birth at 140 dg to a weak lamb (2.8 kg), which died after 3 days, of respiratory distress. The noninfected ewes gave birth to lambs weighing 4.6 and 4.8 kg at 148 and 150 dg, respectively.
Serology
C. abortus infection resulted in a systemic antibody response to the microorganism (Fig. 1). The response reached a maximum level 15 days after infection, after which it decreased slightly until just before abortion or parturition, when a second increase in the antibody levels to C. abortus was observed.
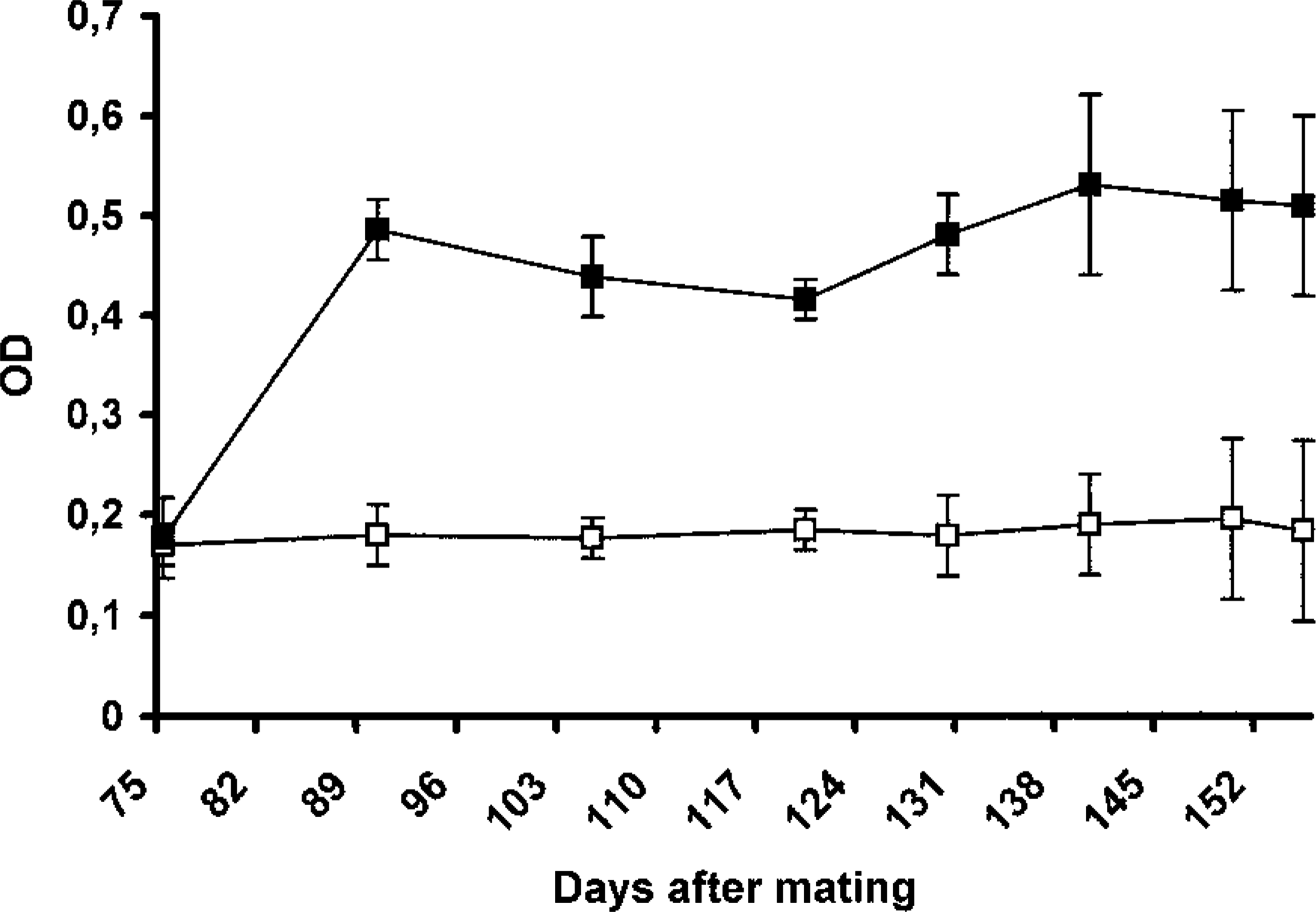
Level of C. abortus–specific antibodies in the serum of infected (black) and uninfected (white) ewes. Results are expressed as the mean±sd of the values of optical density obtained using an ELISA. The number of ewes per group was between 10 and two (in the last days) in the infected group and between four and two in the uninfected group.
Gross pathology
At necropsy, the fetus from ewes killed at 105 and 120 dg showed the correct size corresponding to their gestation day. Furthermore, the size and weight of the 130 dg fetus were similar to the fetus from noninfected control ewes (data not shown). No gross lesions were observed in any organ of the ewes. Detailed study of each placentome using a lens showed that one placentome of a ewe killed at 120 dg (ewe No. 4) and two placentomes of a ewe killed at 130 dg (ewe No. 6) showed a scarce adherent white exudate between the placentome and the periplacentomal chorioallantoic membrane.
The placentas recovered after abortion or parturition showed dark red cotyledons, which were soft and matted with a dirty reddish exudate, some of which was partially detached. Intercotyledonary areas and fetal membranes were thickened and contained a yellowish, gelatinous fluid. The surface of some of these areas was covered with a yellowish-white exudate attached to the cotyledons.
In the uterus of the ewes sacrificed 3 days after abortion or parturition, caruncules were soft, yellow-red in color, and covered with a brown exudate, which was also observed over the surface of the adjacent intercaruncular areas. In some caruncules, cotyledon remains were still attached. Seven days after abortion or parturition, the caruncules were smaller, and some were still partially covered with a brown exudate. No lesions were observed in the placenta at the moment of parturition or in the uterus at 3 days postpartum in the control ewes.
Aborted fetuses or lambs showed no pathologic changes, except the lamb born at 140 dg with respiratory distress. This animal displayed craneoventral dark areas of consolidation in the lung after death.
Histopathology and immunohistochemistry
In both ewes sacrificed at 105 dg, a mild bronchointersticial pneumonia with a mixed mononuclear infiltrate and few neutrophils was observed. Only in the lung of one of these ewes (ewe No. 1) was intracellular C. abortus antigen detected in mononuclear cells in the alveolar septa (Fig. 2). A moderate periportal and focal mononuclear infiltration was also observed in the liver of both ewes. At 120 dg, similar but slighter lesions were found in the lung and liver, although no C. abortus antigen was detected. These lesions were not observed from 130 dg onward. Prominent germinal centers were observed in subscapular lymph nodes and spleens with numerous neutrophils in the marginal area of the spleen of all infected ewes.
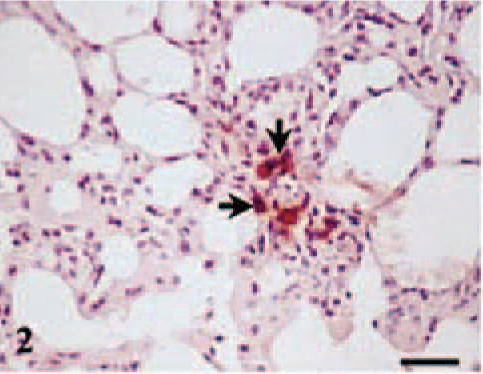
Lung, ewe No. 1 killed at 105 dg. Intracellular C. abortus antigen in mononuclear cells (arrows) located in alveolar septa. Antichlamydial LPS, ABC, Mayer's hematoxylin. Bar = 30 µm.
Placental lesions were present at 120 and 130 dg but only in the ewes showing gross lesions in the placentomes (ewe Nos. 4 and 6). These placentomes showed a suppurative placentitis associated with the presence of C. abortus antigen in the trophoblast cells of the periplacentomal chorioallantoic membrane, the base of the chorionic villi, and peripheral areas of the placentome (Fig. 3), and in the limbus of the placentomal hilius. Trophoblast cells were always negative to MHC I labeling, both in infected and uninfected ewes. In some areas, many trophoblasts cells were necrotic and the chorioallantoic membranes were ulcerated and covered with cell debris. The underlying mesenchyme showed significant neutrophil infiltration with areas of leukocyte degeneration and a great amount of both intracellular and extracelular C. abortus antigen. The rest of the chorioallantoic stroma showed edema, mild cellular infiltration, which was largely composed of mononuclear MHC II+ cells (Fig. 4) with some neutrophils, and vasculitis with fibrinoid necrosis of the wall and mononuclear infiltration around the adventitia. Very few mononuclear cells were CD2+ lymphocytes. Both CD4+ and CD8+ cells were found among this scarce T lymphocyte population. TCR-1+ cells were not found. Occasionally, extracellular and intracellular C. abortus antigen was observed in the chorioallantoic stroma.
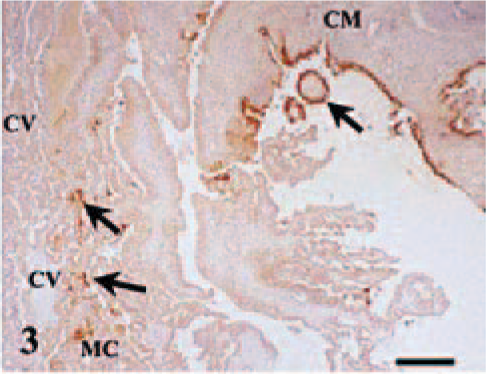
Placentome; ewe No. 4 killed at 120 dg. C. abortus antigen (arrows) in trophoblast cells of the periplacentomal chorioallantoic membrane (CM), peripheral cotyledonary villi (CV), adjacent syncytial epithelium of the maternal crypts (MC), and maternofetal space. Antichlamydial LPS, ABC, Mayer's hematoxylin. Bar = 380 µm.
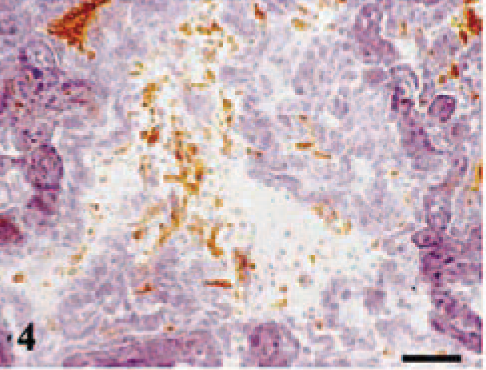
Placentome; ewe No. 4 killed at 120 dg. MHC II+ cells in the diffuse infiltration of the chorioallantoic stroma. H42A MoAb (anti-MHC II), ABC, Mayer's hematoxylin. Bar = 60 µm.
The maternal placenta adjacent to the affected chorionic villi showed focal necrosis of the syncytial epithelium, low amounts of C. abortus antigen, and a leukocyte infiltration formed mainly of neutrophils in the connective tissue beneath this epithelium (Fig. 5). Leukocytes also infiltrated the adjacent intercaruncular stroma. Immunophenotypic characterization of the leukocyte subpopulations on frozen sections showed scarce MHC II+ cells and no CD2+ T lymphocytes. An exudate composed of degenerated leukocytes and cell debris with intra- and extracelular C. abortus antigen was present in the maternofetal space between the placentome and periplacentomal chorioallantoic membrane (Fig. 3). This exudate was also observed between the affected maternal crypts and chorionic villi and in the hematomas between the tips of the maternal septa and the erythrophagocytic trophoblast at the base of chorionic villi.
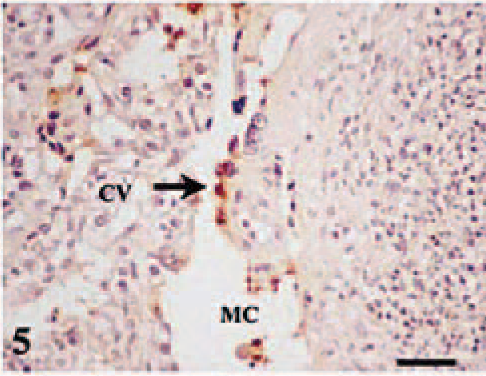
Placentome; ewe No. 6 killed at 130 dg. Necrosis of the trophoblast cells of cotyledonary villi (CV) and of the syncytial epithelium of the maternal crypts (MC), the underlying mesenchyme shows neutrophil infiltration. C. abortus antigen is located in necrotic cells (arrow). Antichlamydial LPS, ABC, Mayer's hematoxylin. Bar = 80 µm.
In ewe No. 4 (killed at 120 dg), the other four placentomes showed no histopathologic changes. In ewe No. 6 (killed at 130 dg), of the three placentomes showing no gross lesions, one placentome was unaffected, one only showed mononuclear cells containing C. abortus antigen in the endometrial stroma of the adjacent intercotyledonary area (Fig. 6), whereas the third placentome showed, besides these infected cells in the endometrial stroma, a focal necrosis of the maternal epithelium with intracellular C. abortus antigen (Fig. 7), although no associated inflammatory response was observed. Neither lesions nor C. abortus antigen was observed in the interplacentomal areas in any ewe. No evident lesions were observed in the fetus of the ewes killed during pregnancy, except in the fetus of ewe No. 6, which showed a necrotic multifocal hepatitis with C. abortus antigen and neutrophils in the foci.
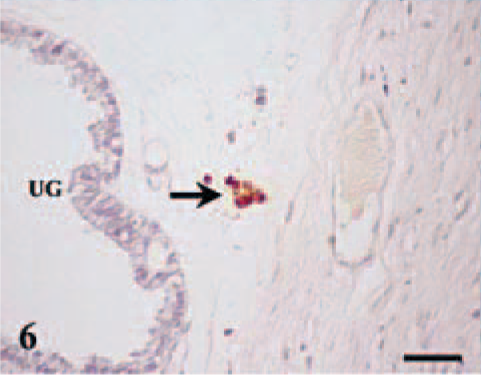
Placentome; ewe No. 6 killed at 130 dg. Intracellular C. abortus antigen in mononuclear cells (arrow) located in the periplacentomal stroma between the uterine glands (UG). Antichlamydial LPS, ABC, Mayer's hematoxylin. Bar = 60 µm.
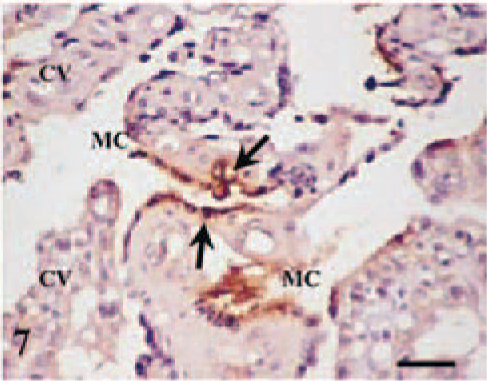
Placentome; ewe No. 6 killed at 130 dg. C. abortus antigen in the syncytial epithelium (arrows) of the maternal crypts (MC) without inflammatory reaction neither affection of the adjacent cotyledonary villi (CV). Antichlamydial LPS, ABC, Mayer's hematoxylin. Bar = 75 µm.
After abortion or parturition, the placentas showed necrosis of the trophoblast with a suppurative inflammatory exudate containing large amounts of C. abortus antigen on the surface of the cotyledonary and intercotyledonary areas. Severe leukocyte infiltration, thrombosis, and vasculitis were present in the chorioallantoic mesenchyma. Aborted fetuses showed mononuclear cells containing C. abortus antigen but no inflammatory response in the septa of the lung. The lamb born at 140 dg with gross lesions in the lung showed severe catarrhal bronchopneumonia with large amounts of extra-and intracellular C. abortus antigen.
Three days after parturition, the uterus of the infected ewes showed large amounts of intra- and extracellular C. abortus antigen over the surface and within of the crypts of the caruncular degenerated tissues, and in contrast to uninfected ewes, the stroma immediately beneath showed a strong leukocyte infiltration with a predominance of lymphocytes. The stroma of the intercaruncular areas showed stronger leukocyte infiltration than the control, consisting of the lymphocytes and plasma cells with some neutrophils.
In the uterus of infected ewes 7 days after abortion, the caruncules showed no degenerated remains of placental tissue. The surface was partially reepithelized and the central areas were formed mainly of connective tissue. The stroma of caruncular and intercaruncular areas showed a strong infiltration of lymphocytes and plasma cells. Only sporadic C. abortus antigen was observed in the uterine epithelial cells.
C. abortus isolation
C. abortus was recovered (in low number) from the placentomes of the ewe killed at 105 dg (ewe No. 1) (Table 1), from one ewe killed at 120 dg (ewe No. 4), and from one ewe killed at 130 dg (ewe No. 6). The last ewe shows a very high burden of C. abortus in its placentomes. Regarding the immunohistochemic detection of C. abortus antigen, both techniques showed similar results when the C. abortus burden was high, i.e., >106 IFU/g of tissue.
Clamydophila abortus isolation from five randomly selected placentomes of killed ewes at different days of gestation (dg).

IHC = Immunohistochemistry.
Below the limit of detection of the technique.
A very high burden of C. abortus (>106 IFU/g of tissue) was recovered from the cotyledons from all the abortions. The three aborted fetuses showed positive isolation in the abomasum (1.2 ± 0.4 × 104 IFU/g of tissue) and spleen (0.8 ± 0.2 × 104 IFU/g of tissue) but only in the liver of one of the fetuses (4 × 103 IFU/g of tissue). The weak lamb that died after parturition showed a very high burden of C. abortus in the lung (>106 IFU/g of tissue) and also positive isolation in the abomasum (2.2 × 104 IFU/g of tissue), spleen (4 × 103 IFU/g of tissue), and liver (3 × 103 IFU/g of tissue).
The aborted ewes excreted C. abortus with their vaginal discharges. The amount of infective C. abortus found in the vaginal swabs varied, although all the four ewes studied excreted during days 1–3 (Table 2). In the two surviving ewes, excretion decreased or disappeared between days 5 and 7 after abortion.
Clamydophila abortus isolation from vaginal swabs of ewes at different days after abortion.

Animals were killed at day 3 after abortion.
Below the limit of detection of the technique.
Discussion
Experimental C. abortus infection at day 75 of gestation induced a disease whose main clinical sign, abortion, appeared during the last 3 weeks of pregnancy. The first microscopic pathologic changes were observed in the placenta at day 120 of gestation. These findings agree with previous studies, in which lesions were observed in the third term of pregnancy, regardless of the day of inoculation. 2,9,25
In our study, no clinical signs of the disease were observed in the first month after infection, except a slight increase in body temperature during the first few days after infection. However, detectable levels of specific antibodies in the serum were found 15 days after infection in all the infected ewes. When the first animals were killed at 105 dg, mild interstitial pneumonia associated with C. abortus antigen and a very mild focal hepatitis were observed. These lesions decreased in the following days, which suggests that systemic dissemination of C. abortus was controlled apparently by the immune response of the hosts. The only tissue that showed an increase in the severity of the lesions was the placenta. An immune response that forces C. abortus into to a latent state has been reported previously, 1,10,24 although the exact location where latency is established until the placenta is colonized remains unknown.
When the uterus and placenta of infected pregnant ewes were analyzed, the first location of C. abortus antigen was within mononuclear cells in the endometrial stroma. This location was previous to the the first pathologic changes described by other authors 9 in the placenta, i.e., colonization and necrosis of the trophoblast cells, with a subsequent suppurative necrotic placentitis. Furthermore, focal necrosis and C. abortus colonization of the maternal epithelial cells of the placentome was also observed without trophoblast damage. Both results point to the fact that the placenta infection starts in the maternal side. When the fetal placenta colonization was established, we observed C. abortus antigen only in the trophoblast of the placentome and periplacentomal choriollantoic membrane but not in the intercotyledonary trophoblast until abortion or parturition, as has been reported by other authors. 9
In our study, C. abortus colonization of the placentome induces an inflammatory response composed mainly of neutrophils, which seems to be sufficient to control C. abortus dissemination in the maternal placenta but not in the fetal side. In mouse infection models, neutrophils have been shown to play a very important role in the early response to chlamydial infection. 3,6 Neutrophils also play an important role as a bridge between the innate and specific immune response during C. abortus infection; in fact, neutrophils are crucial for the recruitment of other leukocyte sub-populations, especially CD8+ T cells. 21 However, in the present study, the extensive infiltration of neutrophils was not followed by a subsequent recruitment of T lymphocytes as a specific response to C. abortus infection. This lack of recruitment of lymphocytes could be related to the immunologic suppressor mechanisms that prevent the attack of the maternal immune response on the fetal placenta 18 and which favors the development of intracellular pathogens, such as Toxoplasma or Chlamydophila. 13 An increase in the Th2 response might also act locally in the placenta. 11,30
Although colonization of the maternal placenta seemed to be efficiently controlled, this did not occur in the fetal placenta, in which very extended infection of the trophoblast cells was observed, suggesting that these cells are one of the main targets for the multiplication and dissemination of C. abortus. Two observations might explain the importance of these cells in C. abortus infection. Firstly, trophoblast cells did not express the MHC I antigen, suggesting that they are very resistant to CD8 T cell–dependent cytolytic mechanisms. Secondly, trophoblast cells did not induce the expression of the tryptophane-degrading enzyme indoleamine 2,3-dioxygenase after activation by interferon-γ (IFN-γ), 14 so that these cells provided an optimal tryptophane-rich environment for the growth of C. abortus because this is an essential amino acid in the development of the bacterium. 4
The inflammatory response in the chorioallantoic stroma was composed mainly of neutrophils and macrophages, with a very scarce number of lymphocytes, reflecting the findings of a previous study. 8 The same study also reported the low expression of IFN-γ and the absence of interleukin-4. In our study, C. abortus antigen was found in the chorioallantoic stroma and in the lesions of the lung and liver of fetuses or aborted lambs; this fact could be due to the immaturity of the immune system of these animals which would be unable to prevent dissemination of the infection.
Our experiments showed an irregular pattern of C. abortus colonization of the placenta because although all the placentas after abortion showed necrotic lesions and a high burden of C. abortus, the beginning of this colonization was not synchronized. Some ewes aborted at 128 dg, whereas one ewe showed neither pathologic changes nor positive isolation at 130 dg, despite the fact that serum analysis confirmed that the ewe had been infected. The optimal environment for the massive multiplication of C. abortus probably occurs at a specific moment that may be vary between individuals. Whatever the moment, the characteristic growth cycle of C. abortus saw the bacteria develop a logarithmic burst within very few days. Such variations would explain the clinical signs of the disease. 25 The early colonization of a great number of placentomes could lead to the rapid destruction of trophoblast cells and to the lack of placenta hormonal control of gestation, abortion occurring despite the restricted colonization of the fetus. Slower and nontotal colonization of the placentomes would mean defective fetus growth and lead to the birth of weak and infected lambs.
After abortion or parturition, C. abortus antigen was only found on the surface of glandular uterine crypts. These areas were surrounded by a substantial lymphocyte infiltration, explaining the strong specific immune response against subsequent C. abortus infections. If, as we suggest above, the placental infection starts in the maternal side, the specific response established after primary infection will be able to avoid the placental colonization. Thus, the multiplication in the immunologically susceptible fetal trophoblast will be prevented.
Although the C. abortus infection in organs such as the lung and the liver was controlled by the host, this fact did not occur in the placenta, where after an early colonization, a rapid and massive multiplication was observed from 120 dg onward, leading to the abortion. However, the factors driving this multiplication or burst during the last weeks of gestation remain unknown and need to be studied, if we are to better understand C. abortus colonization of the placenta.
Footnotes
Acknowledgements
This work was partially supported by the European Commission (FEDER) and Ministerio de Ciencia y Tecnologia grant 1FD97-1242-CO2-01 and 02. C.M. Martínez and N. Ortega are the recipients of predoctoral grants from Ministerio de Ciencia y Tecnologia and University of Murcia, respectively.
