Abstract
Early abortion in ovine toxoplasmosis has had limited investigation. This study evaluated the immune response in the placenta of sheep orally infected with Toxoplasma gondii and euthanized between 2 and 4 weeks postinfection. Toxoplasma infection of the placenta was only found at 4 weeks after infection. Parasitic debris in foci of necrosis were immunolabeled in the maternal caruncle, whereas well-preserved intracellular parasitic vacuole-like structures were found in trophoblasts of fetal cotyledon. Early abortions had increased macrophages in caruncular septa, whereas in later abortions the placentas containing the parasite had an increase of T lymphocytes and macrophages mainly in the fetal cotyledons. This study suggests that the immune response in both the fetal and maternal compartments of the placenta may contribute to the pathogenesis of ovine toxoplasmosis and that these responses differ between early and late presentations of the disease.
Toxoplasma gondii typically causes abortions in sheep that usually occur 1 month after infection, 2 but recent studies have also described a clinical presentation of early abortions only 2 weeks after experimental infection. 1,4
We previously described that the time of gestation at which sheep are infected influences the severity of the lesions and the parasite distribution in the placenta 5 as well as the cytokines that can be found at the peripheral and local (placental) level. 3 Cellular immune responses are regarded as most crucial for controlling T. gondii, 8 particularly at the maternal-fetal interface where infection can disrupt the fine-tuned placental immune balance in both humans and mice, by activating the cellular immune response and secretion of interferon-γ (IFN-γ). 13 However, it is difficult to extrapolate results observed in humans and mice to sheep because of differences in placental morphology. The ovine placenta is synepitheliochorial, where the fetal cotyledon and the maternal caruncle are firmly connected within discrete placentomes formed by interdigitation of fetal chorionic villi and maternal crypts. 6 Furthering previous work in which primiparous ewes were infected with T. gondii, 3,5 the aim of the present study was to investigate the pathogenesis by characterizing distribution of the parasite and the inflammatory cell infiltrate in placentomes from 27 of these 36 sheep.
The experimental design was reported previously. 5 Briefly, 36 pregnant primiparous churra sheep aged 24 to 30 months were infected at different stages of gestation: Group 1 (G1; first stage) at day 40, Group 2 (G2; second stage) at day 90, and Group 3 (G3; third stage) at day 120 of gestation (Supplemental Table S1). Nine sheep from each group were orally infected with 50 sporulated oocysts of M4 T. gondii (Moredun Research Institute, Edinburgh, Scotland, UK), whereas uninfected sheep (n = 3 in each group) received 50 mL of PBS (phosphate-buffered saline). Three infected sheep and one control of each group were scheduled to be euthanized at days 12, 19, and 26 postinfection (dpi). However, due to early abortions during the acute phase of the disease (<12 days postinfection) or stillbirths in the group infected at the last stage of gestation, some sheep were euthanized when fetal death was detected by ultrasound, which altered the number of sheep euthanized at the planned dpi (Supplemental Table S1). Additionally, 5 early abortion and 4 stillbirth cases were excluded because of autolysis. Thus, placental samples from 27 sheep were used in the current study: 9 from control animals (3 mock-infected in each stage of gestation), 3 from early abortion cases (2 infected at day 40 of gestation and one infected at day 90 of gestation), and 15 sheep euthanized according to the original schedule (7, 5, and 3 animals infected at 40, 90, and 120 days of gestation, respectively; Supplemental Table S1). Because of the limited group size, the study could not evaluate the relationship between local placental immune response and the time-point of gestation at infection or the dpi when sheep were euthanized. Assuming that these variables had no effect on the inflammatory infiltrate at the placentomes, animals were grouped as follows: control (n = 9), early abortion (n = 3), and euthanized (n = 15).
For this study, 3 formalin-fixed paraffin-embedded placentome samples were randomly selected from each animal as described. 5 The presence and distribution of the parasite was evaluated in 3-μm-thick immunolabeled histologic sections using an in-house polyclonal antibody to T. gondii. 5 Sections were immunolabeled for CD3, CD20, CD163, CD335, calprotectin, and Iba1 (Supplemental Table S2). A single investigator counted the number of clearly labeled cells in 20 random 400× fields within the interdigitating area of each placentome. Pairwise comparisons between groups were analyzed using Mann-Whitney test (GraphPad Prism 5.0).
Toxoplasma gondii was only found in 5 sheep from the euthanized group, all of them euthanized on the fourth week postinfection (wpi), and these were the only ones with histologic lesions of multifocal mononuclear placentitis. 5 Parasite antigen was detected in 2 patterns (Fig. 1). In the caruncle, immunolabeling was always in areas with moderate numbers of lymphocytes and macrophages and/or necrosis, and it appeared as particulate antigen or parasitic debris. In the cotyledon, immunolabeling was mainly in the trophoblast layer of chorionic villi with no relation to histological lesions. In some cotyledons, immunolabeling was also present in the connective tissue of the villi associated with infiltration of inflammatory cells. In contrast to the caruncle, the cotyledon never had necrosis, parasite integrity was better preserved based on positive immunolabeling of structures resembling parasitophorous vacuoles, and no particulate antigen (parasitic debris) was found (Fig. 1). Based on this finding, the “euthanized” group was divided into 2 groups: “euthanized-positive” in which T. gondii antigen was found, and “euthanized-negative” in which no antigen was found.
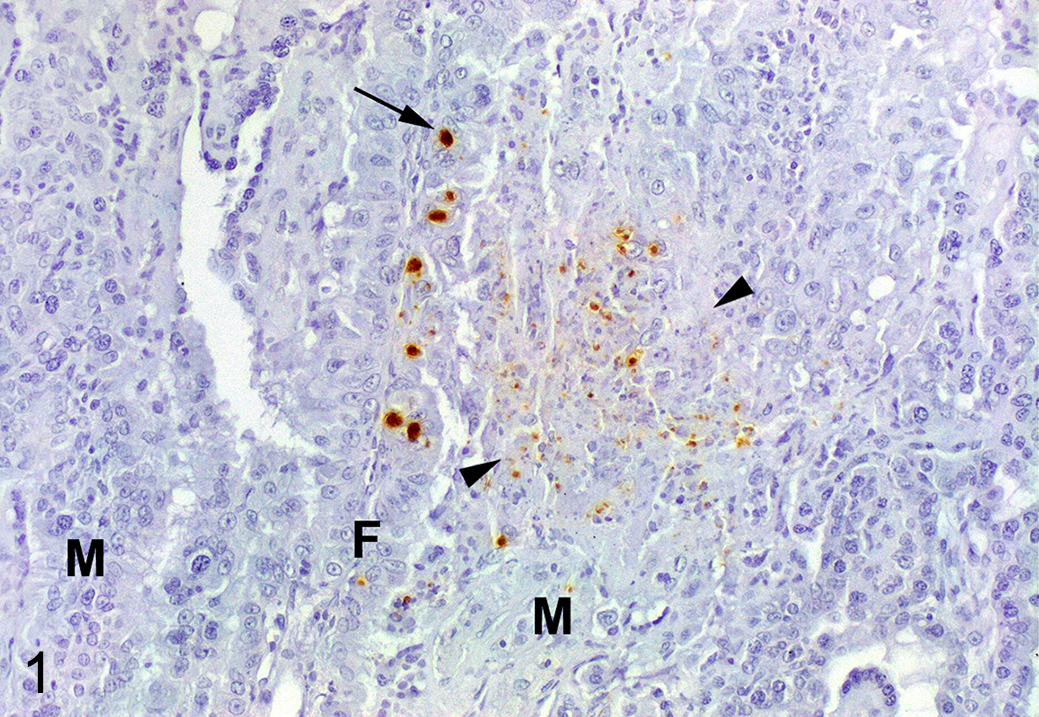
Toxoplasma gondii infection, placenta, sheep, euthanized-positive group. Immunolabeling for T. gondii is detected in small parasitophorous vacuoles or as particulate antigen within a focus of necrosis (arrowheads) located in the maternal septum (M). Strong immunolabeling is present in larger parasitophorous vacuoles (arrow) within trophoblasts located on the fetal cotyledonary villus (F).
As previously described for this experiment, 5 lesions were scarce and not prominent in the interdigitating area of the placentomes and mainly characterized by small foci of necrosis and small foci of infiltration by mononuclear inflammatory cells (possibly macrophages and lymphocytes). By immunohistochemistry, CD335+ (NK cells) or CD20+ (B cells) lymphocytes were infrequent in the placentomes of all groups (Table 1), and those present were randomly distributed throughout the placentome with no relation to histological lesions or parasite antigen. In contrast, the presence of T lymphocytes was related to the presence of the parasite and the clinical presentation. Control animals showed scattered CD3+ cells throughout the parenchyma of the caruncle crypts (Fig. 2). The distribution and number of these cells was similar in ewes affected by early abortion (Fig. 3) and in euthanized ewes where no parasite was detected (euthanized-negative group). In ewes euthanized at 4 wpi that showed parasite antigen (euthanized-positive group), the caruncle had only a mild increase of CD3+ cells that was related to foci of necrosis when these were present. In contrast, this group had increased number (P < .001) of CD3+ cells within the connective tissue of certain fetal villi (Fig. 4, Table 1).
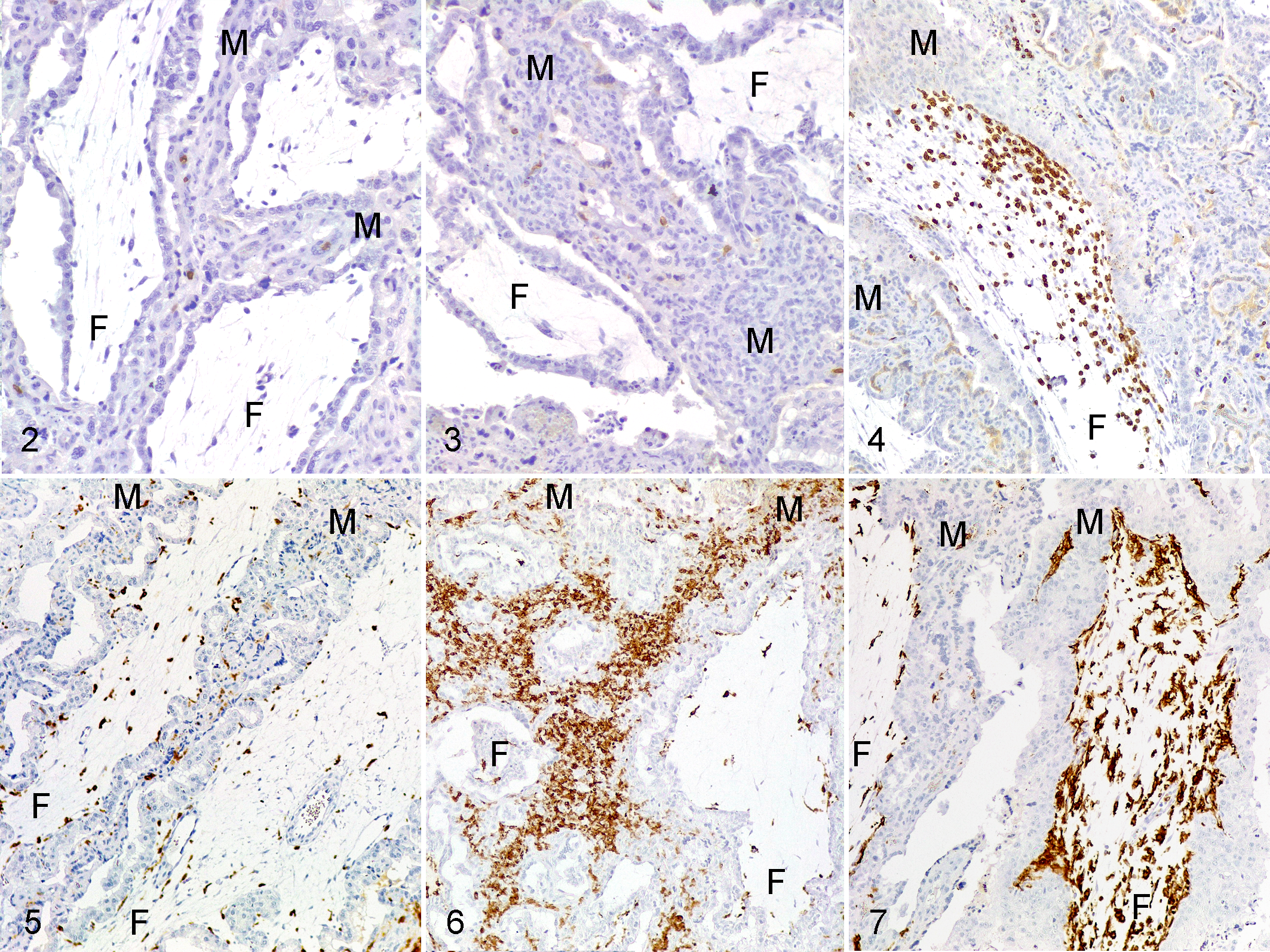
Toxoplasma gondii infection, placenta, sheep. Labeling of inflammatory cells in the cotyledonary villus (F) or maternal septum (M).
Immunohistochemistry Findings in the Interdigitating Area of Placentomes From Sheep With Experimental Toxoplasma gondii Infection and Uninfected Controls*.
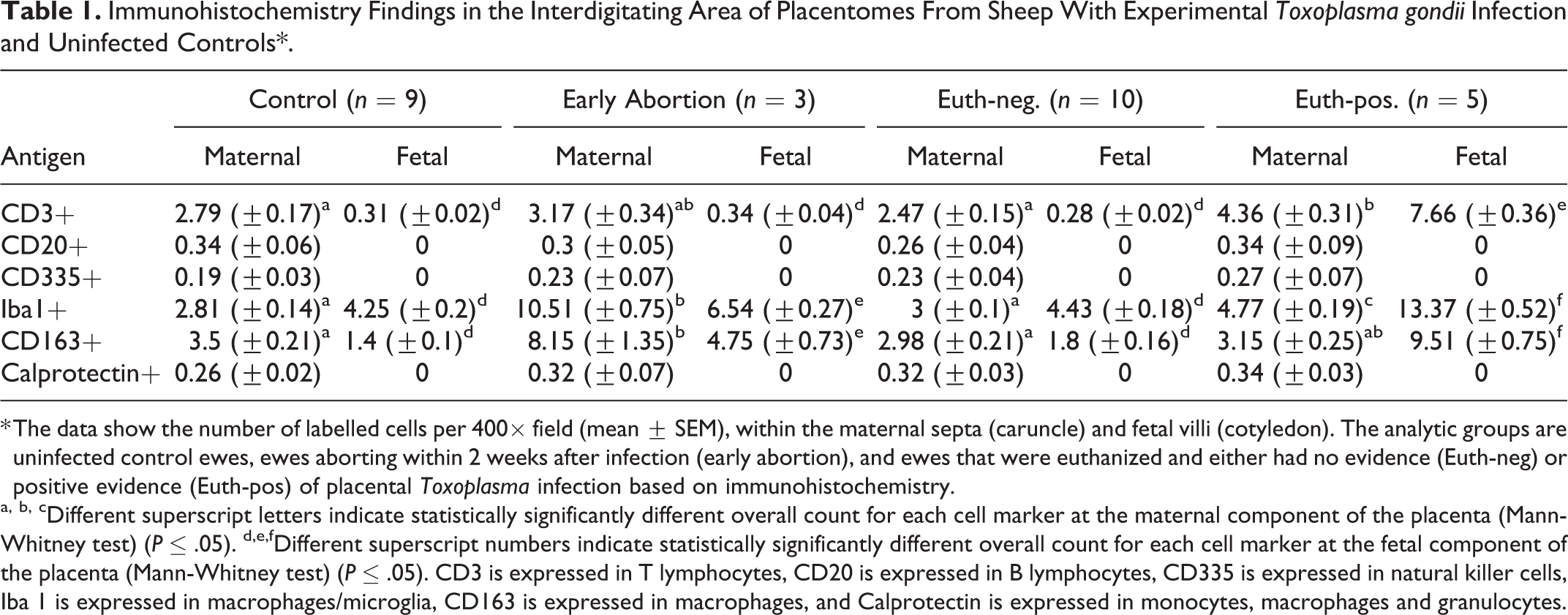
* The data show the number of labelled cells per 400× field (mean ± SEM), within the maternal septa (caruncle) and fetal villi (cotyledon). The analytic groups are uninfected control ewes, ewes aborting within 2 weeks after infection (early abortion), and ewes that were euthanized and either had no evidence (Euth-neg) or positive evidence (Euth-pos) of placental Toxoplasma infection based on immunohistochemistry.
a, b, cDifferent superscript letters indicate statistically significantly different overall count for each cell marker at the maternal component of the placenta (Mann-Whitney test) (P ≤ .05). d,e,fDifferent superscript numbers indicate statistically significantly different overall count for each cell marker at the fetal component of the placenta (Mann-Whitney test) (P ≤ .05). CD3 is expressed in T lymphocytes, CD20 is expressed in B lymphocytes, CD335 is expressed in natural killer cells, Iba 1 is expressed in macrophages/microglia, CD163 is expressed in macrophages, and Calprotectin is expressed in monocytes, macrophages and granulocytes.
Macrophage populations varied by group. Iba1+ and CD163+ cells were similarly distributed and more numerous than calprotectin+ cells. In control and euthanized-negative animals these cells were frequent and were distributed as isolated cells in maternal crypts and fetal villi (Fig. 5). These cells were more numerous in the early abortion and euthanized-positive sheep (Table 1). In early abortion cases, the increase of Iba1+ and CD163+ cells occurred in the caruncular septa (Fig. 6), whereas in euthanized-positive sheep, the cells infiltrated certain chorionic villi (Fig. 7) similar to that of CD3 lymphocytes.
This study of the pathogenesis of abortion in ovine toxoplasmosis showed differences between maternal and fetal placental compartments with respect to the clinical disease presentation and the host-parasite interaction. Lesions in the caruncle were characterized by necrosis 2 associated with small parasitic structures or parasite debris, suggesting an aggressive reaction causing damage to the tissue (ie, necrosis) and the destruction of the parasite. In contrast, the parasite appeared to proliferate in the fetal tissue, as fetal trophoblast cells had intact parasitic vacuole-like structures but no necrosis, suggesting the parasite remains viable in this location. The susceptibility of ovine trophoblast cells to Toxoplasma infection is not known, although a bovine trophoblast cell line is more susceptible to Neospora caninum than a bovine caruncular cell line, and this susceptibility was attributed to the phagocytic activity of the trophoblasts. 10 Furthermore, the studies of bovine neosporosis suggest that the maternal caruncle but not the fetal cotyledon elicited an active immune response against the parasite. 15 A similar mechanism could explain the presence of necrosis and parasite debris in the ovine caruncle but not the cotyledon.
T cells seem to be the only lymphocyte population in lesions of ovine toxoplasmosis once the parasite has reached the placenta. The lack of NK cells was surprising not only in infected sheep but also in the controls. Despite their importance for sustaining the gestation in human and mice, their absence in gravid ovine uterus has been previously reported. 17 Instead, gamma delta T lymphocytes have been proposed as a key lymphocyte population in the development of ruminant gestation, 11 and their role in ovine toxoplasmosis deserves further investigation.
Only the early abortion and euthanized-positive groups showed significant increases in placental inflammatory cells, but the distribution of these cells varied between these groups, suggesting a different pathogenesis. 5 The current study suggested that lymphocytes (ie, T, B, or NK cells) may not play a key role in early abortion. Instead, the early abortions seem related to the macrophage response in the caruncle, including macrophages expressing Iba1, a general marker for macrophages, 12 and CD163, a scavenger marker found in monocytes/macrophages associated with the resolution of inflammation. 7 It has been suggested in humans that increased number of macrophages could be associated with pregnancy complications such as preclampsia, 9 which in turn is associated with fetal leucomalacia, 16 the main fetal lesion found in early abortion in ovine toxoplasmosis. 1 This suggests a role of caruncular macrophages in the clinical presentation of early abortions.
On the other hand, the distribution of inflammatory cells differed in the euthanized-positive group (where parasite antigen was detected in the placenta). In this group, macrophages and T lymphocytes infiltrated the fetal chorionic villi. T lymphocytes are a source of IFN-γ in adult sheep with toxoplasmosis, 8 but their participation in the fetal response against the parasite is unknown. Fetal placental macrophages (Hofbauer cells) can differentiate from villous mesenchymal stem cells or from hematopoietic cells. 14
The current study shows a lymphocyte and macrophage reaction in the lesions of T. gondii, and differences in the distribution of these cells depending on the clinical presentation of ovine toxoplasmosis. Further characterization of macrophage and T cell subpopulations involved in this response would help understand the role of the placental immune response in the pathogenesis of ovine toxoplasmosis.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_0300985820923987 - Macrophages and T Lymphocytes in the Ovine Placenta After Experimental Infection With Toxoplasma gondii
Supplemental Material, sj-pdf-1-vet-10.1177_0300985820923987 for Macrophages and T Lymphocytes in the Ovine Placenta After Experimental Infection With Toxoplasma gondii by Pablo Castaño, Miguel Fuertes, Miguel Fernández, M. Carmen Ferreras, Ignacio Ferre, Luis Miguel Ortega-Mora, Valentín Pérez and Julio Benavides in Veterinary Pathology
Footnotes
Authors’ Note
The data and samples that support the findings of this study are available from the corresponding author on reasonable request.
Acknowledgements
The authors are in deep gratitude to Dr Frank Katzer, from Moredun Research Institute, for kindly providing the tachyzoites from M4 isolate; all the staff of the IGM (CSIC-ULE) responsible for handling the experimental animals; and the technical assistance of Mr Panero, Mrs Espiniella, Mrs González, and Mrs Agudín. P. Castaño is funded by a predoctoral contract from the “Junta de Castilla y León.”
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grants AGL2011-30205 and AGL2016-75935-C2-2-R of the Spanish Ministry of Science and Innovation and Grant LE253U13 from the Junta de Castilla y León, Spain.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
