Abstract
Tumors of the adrenal cortex account for 10–20% of the naturally occurring Cushing's syndrome diagnosed in dogs. Differentiating between adrenocortical adenoma and carcinomas is often difficult. The purposes of this study were to determine which histopathologic criteria can be used as markers for malignancy in canine adrenocortical tumors and the relevance of the proliferation marker, Ki-67, for differentiation between cortical adenomas and carcinomas. Twenty-six adrenocortical carcinomas, 23 adenomas, and 11 normal adrenal glands were examined. Morphologic criteria significantly associated with adrenocortical carcinomas included a size larger than 2 cm in diameter, peripheral fibrosis, capsular invasion, trabecular growth pattern, hemorrhage, necrosis, and single-cell necrosis, whereas hematopoiesis, fibrin thombi, and cytoplasmic vacuolation were significantly associated with adrenocortical adenomas. The mean (± SD) proliferation index, measured by immunohistochemistry for the Ki-67 antigen, was 9.3 ± 6.3% in carcinomas, 0.76 ± 0.83% in adenomas, and 0.58 ± 0.57% in normal adrenal glands. The Ki-67 proliferation index was significantly higher in carcinomas compared with adenomas and normal adrenal glands. A threshold value of the proliferation index of 2.4% reliably separated carcinomas from adenomas. Based on these results, it appears that thorough evaluation of morphologic features combined with immunohistochemical assessment of the proliferation index is extremely useful for differentiating between adrenocortical adenomas and carcinomas in dogs.
Keywords
Tumors of the adrenal cortex account for 10–20% of the naturally occurring Cushing's syndrome diagnosed in dogs. 7,22 In the absence of metastases or obvious invasion, differentiating between adrenocortical adenoma and carcinomas is often difficult. Adrenal-dependent hyperadrenocorticism can be diagnosed with blood tests; however, both adenomas and carcinomas can be functional. 4,7,22 Radiography, abdominal ultrasonography, computed tomography, and magnetic resonance imaging can be useful in identifying, localizing, and characterizing an adrenal mass, but none of these tests consistently differentiates between benign and malignant tumors. 7,22 The correct diagnosis, therefore, relies on integrating the above information with the histologic evaluation of the tumor. Pleomorphism, hemorrhage, necrosis, and increased mitotic rate have been associated with adrenocortical carcinomas in dogs. 4,13,14,35 However, to the authors' knowledge, there are no studies in the veterinary literature that rigorously evaluate morphologic features of adrenocortical neoplasms in dogs.
In humans, systems using histologic and clinical criteria have been developed to differentiate between cortical adenomas and carcinomas. 6,11,17,29,33,34 These systems use scores and predictive values or rely on the presence of a determined number of criteria to evaluate malignancy in adrenocortical neoplasms. Tumor size and tumor weight, although not absolute, are helpful in predicting clinical behavior because larger masses are more often malignant. 6,11,17,29 Other nonhistologic criteria include clinical manifestations of adrenocortical disease, urinary 17-ketosteroids concentrations, response to adrenocorticotropic hormone, and weight loss. 11,17,26
A number of histologic criteria have been used to diagnose malignancy in adrenocortical tumors, including vascular and capsular invasion, growth pattern, necrosis, intratumoral fibrosis, pleomorphism, hemorrhage, calcification, mitotic index, nuclear grade, atypical mitotic figures, abnormal nuclei, and cytoplasmic eosinophilia. 6,11,17,29,33,34 A diffuse growth pattern, vascular invasion, single-cell necrosis, and the presence of fibrous bands within the tumors are associated with an increased risk of metastasis. 11 A high mitotic rate has been associated with a shorter disease-free period. 6,17
In addition, the diagnostic value of a number of immunohistochemical stains has been studied in adrenocortical neoplasms in humans. 5,10,12,16,18,26,30 Ki-67 has been validated as a simple reliable and repeatable marker that is useful in the differential diagnosis of adrenocortical tumors in humans. 2,5,9,10,12,16,18,25–28,30,32 Ki-67 (MIB-1) is a nuclear protein expressed in cells that are in the non-G0 phases of the cell cycle. In human patients with an adrenocortical carcinoma, a higher Ki-67 labeling index was associated with a reduced life expectancy. 16,27,28 Ki-67 has already been shown to be prognostically relevant in other types of tumors, such as mammary tumors, melanomas, and mast cell tumors, in dogs. 1,15,21,23,24
The purposes of this study were to document the morphological characteristics of canine adrenocortical tumors, determine which histopathologic criteria can be used as markers for malignancy, and determine the relevance of the Ki-67 proliferation marker for differentiation between adrenocortical adenomas and carcinomas in dogs.
Materials and Methods
Morphology
Neoplastic canine adrenal glands were retrieved from the archives of the Pathology Service of the Veterinary Medical Teaching Hospital (University of California, Davis, CA). Cases diagnosed with an adrenocortical tumor between 1993 and 2002 were reviewed for eligibility. Adrenocortical tumor tissue obtained at either surgery or necropsy was included. Tumors were selected irrespective of their functional status.
Tumors were diagnosed as carcinomas when there was histologically confirmed metastases or vascular invasion. Tumors were diagnosed as adenomas when there was disruption of the normal adrenal architecture, compression of the adjacent parenchyma, and presence of a fibrous capsule or fibrovascular stroma surrounding at least part of the tumor in the absence of metastases or vascular invasion. Normal canine adrenal glands were retrieved from dogs that died or were euthanatized for unrelated causes.
Only samples that allowed proper histologic examination and were suitable for immunohistochemistry were used. Samples were excluded when 1) paraffin blocks did not contain sufficient material for serial sectioning, 2) samples were autolyzed, 3) follow-up to verify the presence of metastases was not available, and 4) animals were treated with lysodren before surgery or postmortem examination. Twenty-six carcinomas, 23 adenomas, and 11 normal adrenal glands were included in the study.
Clinical records were consulted to document breed, age, sex, and weight of the animal, presence and sites of metastases, and size of the tumors. Tumors were grouped into those with less than or equal to 2 cm in diameter or greater than 2 cm in diameter. All tissues used in this study were immediately fixed in formalin and routinely processed for histology. From each specimen, a 5-µm hematoxylin and eosin–stained section was made for histologic examination.
The following criteria were evaluated for each adrenal gland (Table 1): growth pattern, vascular invasion, peripheral fibrosis and capsular invasion, necrosis, single-cell necrosis, hemorrhage, fibrin thrombi, intratumoral fibrosis, presence of hematopoiesis, mitotic index, cellular pleomorphism, nuclear size and atypia, nuclear chromasia, and cytoplasmic vacuolation and eosinophilia. Growth patterns were recorded as either diffuse or nesting and trabecular. Tumors were considered to have a trabecular pattern when at least one third of the tumor consisted of this pattern. Vascular invasion was determined using the surgical report as well as histologic examination of the tumor. All the tumors examined were at least partially surrounded by fibrous stroma. However, a tumor was considered to have peripheral fibrosis when at least part of the tumor was surrounded by a prominent multilayered band of fibrous tissue. Capsular invasion was noted when neoplastic cells infiltrated or perforated the capsule. Small nests of adrenocortical cells within the capsule were not interpreted as capsular invasion. Necrosis was evaluated by the presence or absence of lakes of coagulative necrosis. Cells undergoing single-cell necrosis (bright eosinophilic cytoplasm and pyknotic nucleus) were counted in 10 high-power fields (HPF, 400×), and a threshold was set at an average of three cells undergoing single-cell necrosis per HPF based on initial review of biopsy and necropsy material. Presence or absence of hemorrhage within the tumor was noted. Large dilated and congested vessels did not constitute hemorrhage. Fibrin thrombi within tumor vasculature, fibrous bands dissecting the tumor, and clusters of hematopoietic cells were noted as present or absent. The mitotic index was determined for each tumor by counting mitotic figures in 10 HPF. Cases were separated using a threshold of an average of 1 mitosis per HPF, which was established after initial review of biopsy and necropsy material. Cellular pleomorphism, nuclear size, nuclear chromasia, and cytoplasmic eosinophilia were noted as mild or severe. Tumors were interpreted to have cytoplasmic vacuolation when a minimum of two thirds of the neoplastic cells had prominent cytoplasmic vacuoles.
Comparison of selected criteria in canine adrenocortical adenomas and carcinomas.
Statistically significant (P < 0.05).
Using Fisher's exact test.
NA = not applicable.
Proliferation index
The proliferation index was determined for the 49 tumors and 11 normal adrenal glands by means of immunohistochemistry for Ki-67 antigen (MIB-1). Serial 4-µm sections were cut and mounted on “lplus” coated slides (Fisher Scientific, Pittsburgh, PA). Immunohistochemical staining was performed using a standard streptavidin–biotin–horseradish peroxidase technique as previously described with the following modifications for paraffin. 19 Sections were deparaffinized in two 10-minute changes of xylene followed by gradual rehydration through alcohol to phosphate-buffered saline, pH 7.2. Tissue sections for primary antibody mouse monoclonal anti-human Ki-67 antigen, clone MIB-1 (1 : 75, Dako Corp., Carpenteria, CA), were heat retrieved in citrate buffer, pH 6 (Dako Corp.), for 30 minutes using steam followed by cooling for 20 minutes. The secondary biotinylated reagent and the tertiary streptavidin reagent were provided as a predilute kit (Biocare Medical, Walnut Creek, CA) and applied for 10 minutes each at room temperature. A positive reaction was visualized using an aminoethyl carbozole chromogen (Zymed Labs, South San Franscisco, CA) followed by a light counterstain with Mayer's hematoxylin (Sigma, St. Louis, MO). Normal canine lymph nodes were used as the positive control, and negative controls were prepared by omitting the primary antibody and substituting a mouse myeloma IgG correlate. A minimum of 1,000 nuclei were counted per sample in the areas of highest labeling density. The Ki-67 proliferation index was defined as the number of Ki-67–positive nuclei (adrenocortical cells) divided by the total number of adrenocortical cells counted and was expressed as a percentage.
Statistical analyses
The statistical software used for the analyses were SPSS (Release 10) (SPSS Inc., Chicago, IL) and MINITAB (Release 13) (Minitab Inc., State College, PA). For each criterion (Table 1), the chi-square test was used to assess homogeneity between normal adrenal glands, adrenocortical adenomas, and adrenocortical carcinomas. Multiple paired analyses were performed to identify the significant differences between the three groups for each criterion that lacked homogeneity. Significant differences between adrenocortical adenomas and carcinomas were evaluated for each criterion. A Fisher's exact test was used to compare nuclear atypia between adenomas and carcinomas. A chi-square test was used to assess statistical differences between adrenocortical carcinomas with metastases (with or without vascular invasion) and carcinomas with vascular invasion without metastases for each criterion. A chi-square test for homogeneity was used to compare sex distribution of dogs with normal adrenals, adenomas, and carcinomas. A one-way analysis of variance (ANOVA) was used 1) to compare age between dogs with normal adrenals, adrenocortical adenomas, and carcinomas and 2) to compare weights between dogs with normal adrenals, adrenocortical adenomas, and carcinomas. A one-way ANOVA was also used to assess homogeneity between the proliferation index of normal adrenal glands, adrenocortical adenomas, and adrenocortical carcinomas; paired analyses using the same test were performed to identify the significant differences between the three groups and between carcinomas with metastases and carcinomas with vascular invasion alone. The threshold distinguishing carcinomas from adenomas was determined using the methods of logistic regression analysis. A level of significance of 0.05 (P < 0.05) was used for all tests.
Results
The 49 adrenocortical tumors were obtained from dogs of 26 different breeds. Nine breeds of dogs with tumors presented more than once were Labrador Retriever (six carcinomas, five adenomas), Golden Retriever (three carcinomas, three adenomas), German Shepherd Dog (three carcinomas, two adenomas), Dachshund (two carcinomas, one adenoma), Shih Tzu (two carcinomas, one adenoma), terrier breeds (one carcinoma, one adenoma), Doberman (one carcinoma, one adenoma), Shetland Sheepdog (one carcinoma, one adenoma), and Australian Shepherd (two carcinomas). The normal adrenal glands were obtained from breeds represented at least once in the tumor group. Dogs with carcinomas had a mean age of 10.6 years (SD, 2.2 years; range, 5–15 years), dogs with adenomas had a mean age of 11.4 years (SD, 1.8 years; range, 8–15 years), and controls with normal adrenal glands had a mean age of 9.0 years (SD, 4.0 years; range, 1–15 years). Age was not significantly different between the dogs with carcinomas, adenomas, and normal adrenal glands. Dogs with carcinomas had a mean body weight of 27.7 kg (SD, 13.1 kg; range, 5.2–50.0 kg), dogs with adenomas had a mean body weight of 32.0 kg (SD, 19.1 kg; range, 4.4–82.0 kg), and controls with normal adrenal glands had a mean body weight of 29.5 kg (SD, 16.8 kg; range, 4.0–50.0 kg). Seventy-four percent (34/46) of the dogs with tumors weighed 20 kg or more. Weight was not available for two dogs with carcinomas and one dog with an adenoma. Weight was not significantly different between the dogs with carcinomas, adenomas, and normal adrenal glands. Of the dogs with a carcinoma, five of 10 male dogs were castrated and all 16 female dogs were spayed, and of the dogs with an adenoma, seven of 11 male dogs were castrated and 10 of 12 female dogs were spayed. Four of the five male dogs with normal adrenal glands were castrated, and the six female dogs with normal adrenals were spayed. There was no statistical evidence that these sex groups lacked homogeneity.
Fourteen of the 26 dogs with carcinomas had metastases, 12 of which had both metastases and vascular invasion. Twelve of the 26 dogs diagnosed with carcinomas had vascular invasion alone, without metastases. Sites of metastasis included liver (9/14), lungs (7/14), kidneys (2/14), ovary (1/14), and mesenteric lymph node (1/14).
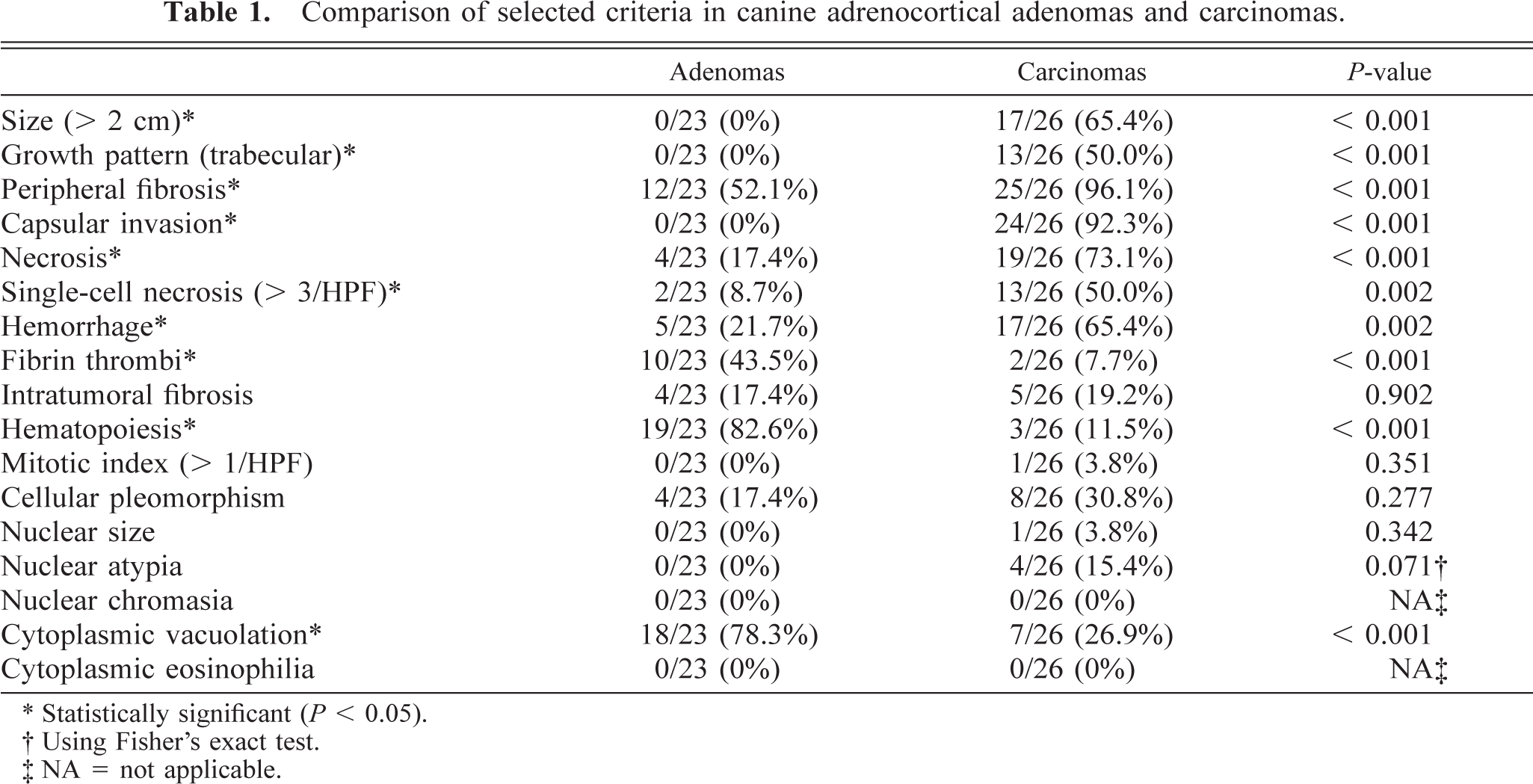
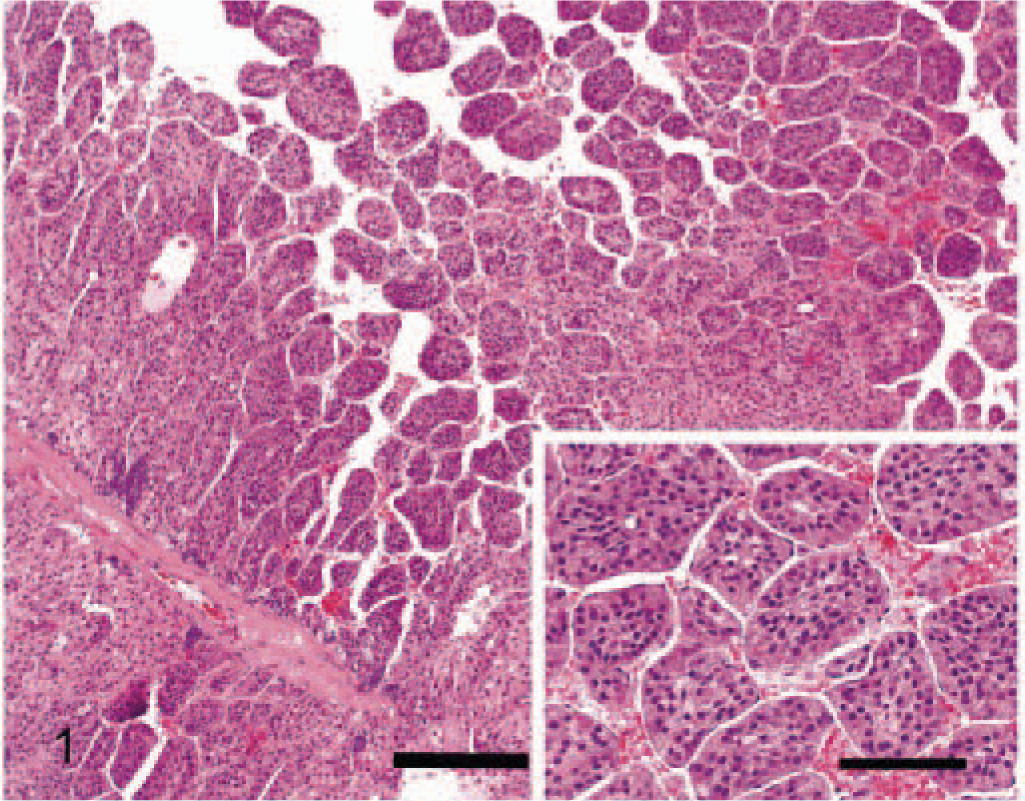
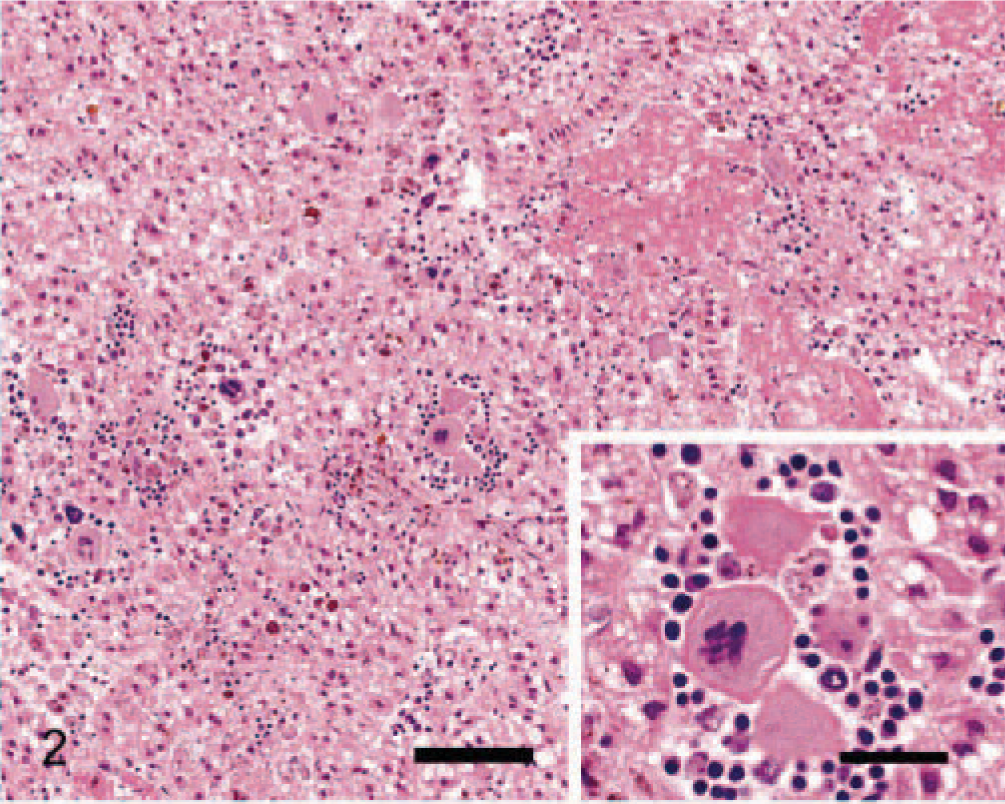
None of the criteria evaluated were present in normal adrenal glands. The following gross and morphologic criteria differed significantly between adenomas and carcinomas (Table 1). The 26 carcinomas ranged from 0.5 cm to more than 15 cm in diameter with 17 larger than 2 cm. All the adenomas were smaller than 2 cm in diameter, ranging from 0.4 to 1.8 cm in diameter. A significantly greater number of carcinomas were larger than 2 cm in diameter compared with adenomas. Most carcinomas (96%, 25/26) and 52% of the adenomas (12/23) had peripheral fibrosis (partially surrounded by a prominent multilayered band of fibrous tissue). Peripheral fibrosis was more prominent and significantly more frequent in carcinomas. Ninety-two percent of carcinomas (24/26) had one or more foci of capsular invasion, whereas none of the 23 adenomas had capsular invasion. The growth pattern within individual carcinomas was variable. Half the carcinomas showed a trabecular growth pattern for a minimum of one third of the tumor, and the remaining carcinomas and all adenomas had a predominately diffuse or nesting growth pattern (Fig. 1). Areas of hemorrhage and lakes of coagulative necrosis were significantly more frequent in carcinomas. These features were often present in the same tumors and were often, but not exclusively, associated with larger tumors. Half the carcinomas had an average of at least three cells undergoing single-cell necrosis per HPF, whereas increased single-cell necrosis was a feature of only two adenomas. Carcinomas with single-cell necrosis frequently also had lakes of coagulative necrosis. Clusters of hematopoietic cells were observed in a large proportion (83%, 19/23) of adenomas and a small number of carcinomas (12%, 3/26) (Fig. 2). Large fibrin thrombi in dilated vessels were more common in adenomas and were found in tumors with numerous dilated, congested but intact vessels (Fig. 2). Forty-four percent of adenomas (10/23) and 8% of carcinomas (2/26) showed this feature, which was often present in association with hematopoiesis. Cytoplasmic vacuolation was variable within individual tumors; however, most adenomas (78%, 18/23) and fewer carcinomas (27%, 7/26) predominately comprised cells with numerous prominent cytoplasmic vacuoles (Fig. 3).
Adrenocortical carcinoma; dog. The artifactual separation highlights the trabecular growth pattern. HE. Bar = 200 µm. Inset: HE. Bar = 80 µm.
Adrenocortical adenoma; dog. Tumor with numerous fibrin thrombi and prominent hematopoiesis. HE. Bar = 100 µm. Inset: Hematopoietic elements often formed aggregates. HE. Bar = 30 µm.
Adrenocortical adenoma; dog. Neoplastic cells have markedly vacuolated cytoplasm. HE. Bar = 30 µm.
A summary of the criteria that were not significantly different between adrenocortical adenomas and carcinomas is presented in Table 1. Although more carcinomas (31%, 8/26) showed severe pleomorphism than adenomas (17%, 4/23), there was no statistical difference between these groups. Fibrous bands dissecting through the tumors were present in a small number of both carcinomas (19%, 5/26) and adenomas (17%, 4/23). The mitotic index was less than 1 mitosis per HPF in all tumors, except one carcinoma. Changes in nuclear size, atypia, and chromasia were only present in a few carcinomas. Cytoplasmic eosinophilia was not a feature of these tumors.
The only significant difference between carcinomas with metastases and carcinomas with vascular invasion alone was the growth pattern. The presence of a trabecular growth pattern in a minimum of one third of the tumor was significantly more frequent (P = 0.018) in carcinomas with metastases (69%, 10/14) compared with carcinomas with vascular invasion alone (25%, 3/12) (Fig. 1).
Immunoreactivity for the Ki-67 antigen was consistent between cases and limited to the nucleus. The distribution of positively staining nuclei was different between carcinomas, adenomas, and normal adrenal glands. In carcinomas, immunoreactivity was almost uniformly present throughout the tumor (Fig. 4). In adenomas, positive cells were scattered, occasionally forming small clusters (Fig. 5). In normal adrenal glands, staining was most often present at the junction between the zona glomerulosa and the zona fasciculata (Fig. 6). The mean proliferation index was 9.3% (SD, 6.3%; range, 1.1–28.5%) in carcinomas, 0.76% (SD, 0.83%; range, 0.10–3.80%) in adenomas, and 0.58% (SD, 0.57%; range, 0.10–1.98%) in normal adrenal glands (Fig. 7). The proliferation index was significantly higher in carcinomas compared with adenomas and normal adrenal glands. There was no significant difference in the proliferation index between adenomas and normal adrenal glands. Based on the proliferation index alone, a threshold value of the proliferation index of 2.4% separated adenomas from carcinomas in 96% of the dogs. All but one carcinoma had proliferation indices greater than 2.4%, and most (21/26, 81%) had a proliferation index greater than 5%. All but one adenoma had proliferation indices less than 2.4%, and most (19/23, 83%) had a proliferation index equal or less than 1%. There was no significant difference in the proliferation indices of carcinomas with metastases and carcinomas with vascular invasion alone.
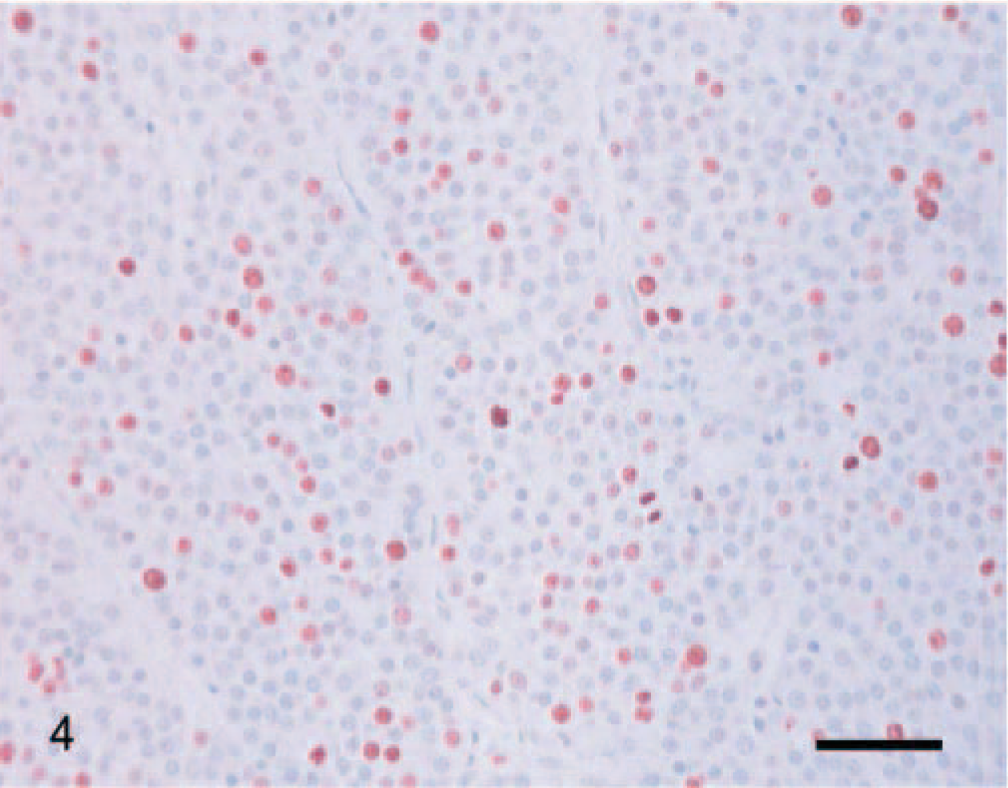
Adrenocortical carcinoma; dog. Ki-67–immunoreactive cells are diffusely distributed throughout the tumor. Ki-67 immunostain, streptavidin–biotin complex method. Mayer's hematoxylin counterstain. Bar = 60 µm.
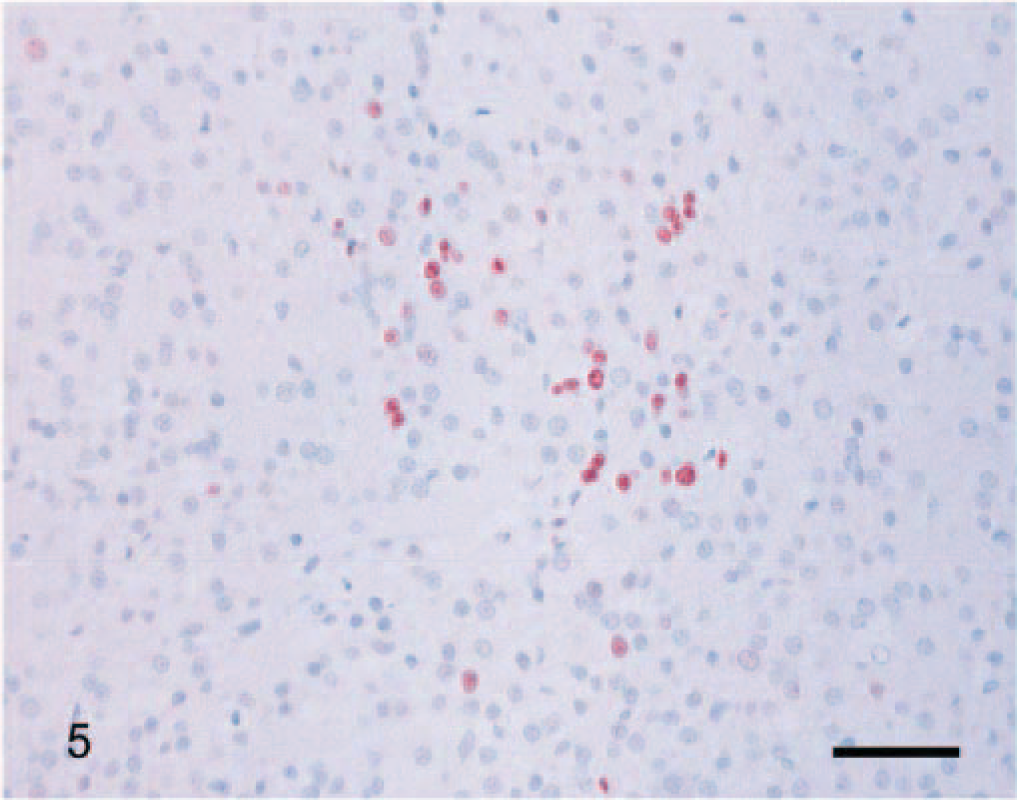
Adrenocortical adenoma; dog. A cluster of Ki-67–immunoreactive cells in a mostly immunonegative tumor. Ki-67 immunostain, streptavidin–biotin complex method. Mayer's hematoxylin counterstain. Bar = 60 µm.
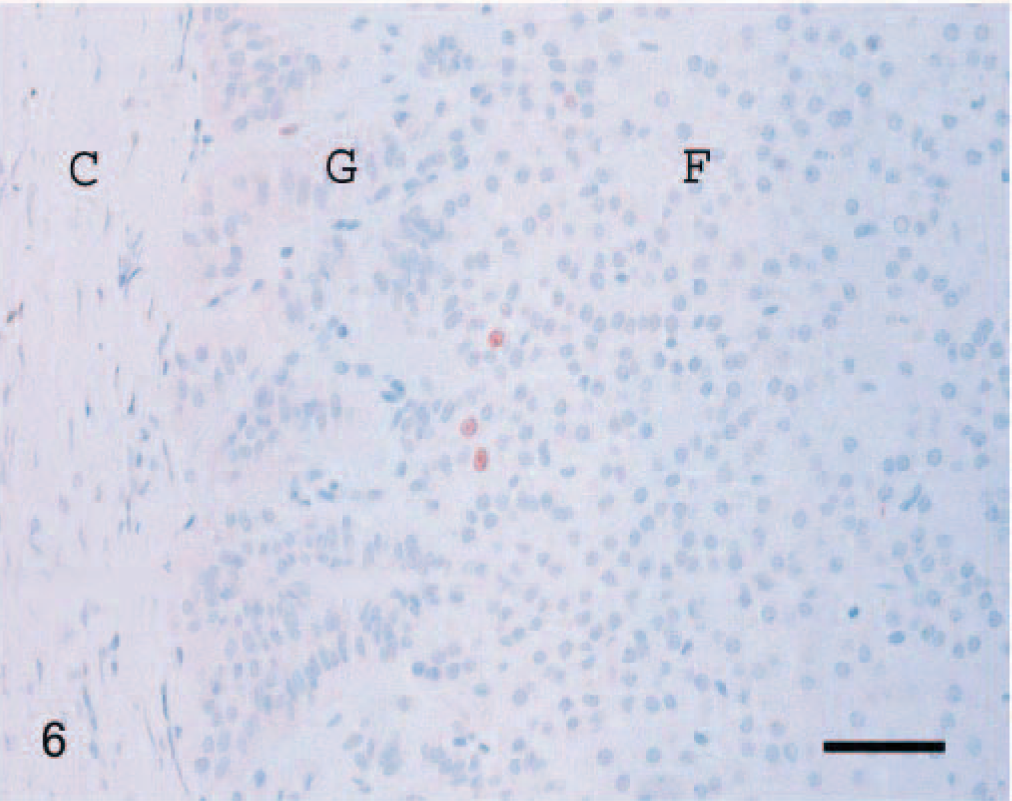
Normal adrenal gland; dog. A few Ki-67–immunoreactive cells are present at the junction between the zona glomerulosa (G) and the zona fasciculata (F). Capsule (C). Ki-67 immunostain, streptavidin–biotin complex method. Mayer's hematoxylin counterstain. Bar = 60 µm.
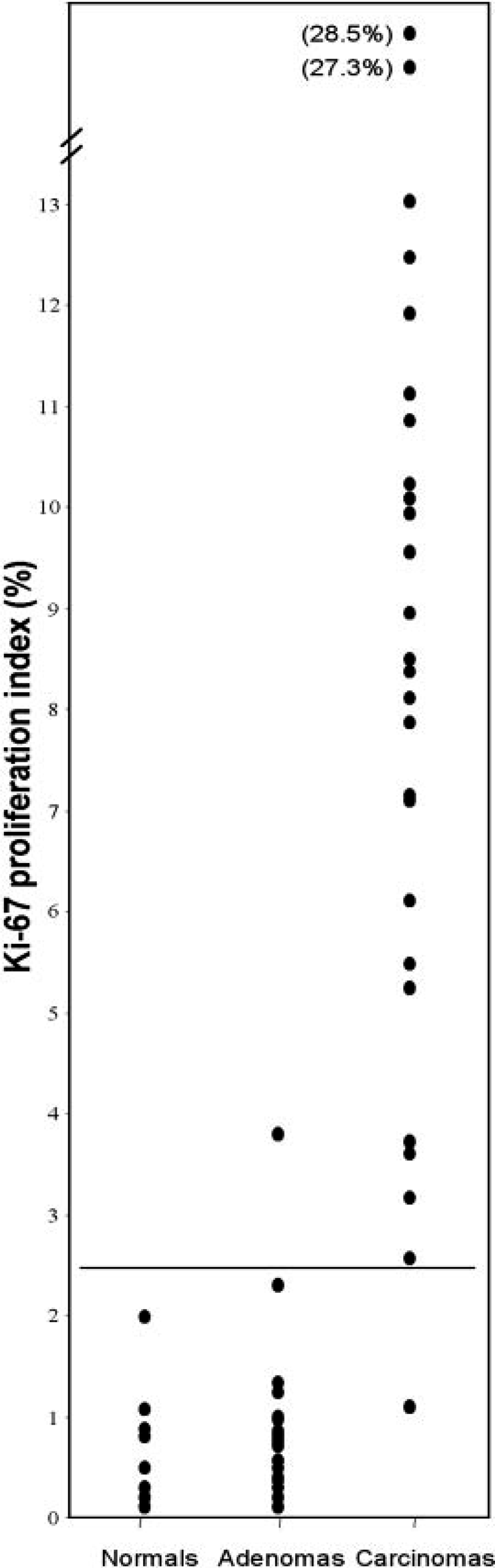
Ki-67 proliferation index in normal adrenal glands (n = 11), adrenocortical adenomas (n = 23), and adrenocortical carcinomas (n = 26). Bar is set at 2.4%.
Discussion
Diagnosing malignancy in adrenocortical neoplasms can be very challenging for the pathologist. In human medicine, a combination of clinical, histologic, and immunohistochemical criteria are used to differentiate between adenomas and carcinomas of the adrenal cortex. 2,3,6,9–12,16–18,25,26,28–30,32–34 In our study, 10 histopathologic criteria were determined to be diagnostically useful in differentiating between adrenocortical adenomas and carcinomas in dogs. These included size, peripheral fibrosis, capsular invasion, trabecular growth pattern, hemorrhage, necrosis, single-cell necrosis, hematopoiesis, fibrin thombi, and cytoplasmic vacuolation (Table 1).
As in humans, carcinomas were significantly larger than adenomas. 6,11,17,29 The difference in tumor size may serve as an indicator of malignancy in diagnostic imaging studies or at surgery. However, some caution is required because adrenocortical adenomas in dogs have been reported to measure up to 6 cm in diameter. 7
The importance of evaluating growth pattern is manifold. The trabecular growth pattern is easily identifiable because artifactual separation often highlights the trabecular, almost papillary, pattern. This pattern was significantly associated with carcinomas. Moreover, most of the carcinomas with metastases had a trabecular growth pattern, suggesting that this growth pattern may be associated with a more aggressive biological behavior.
The fibrin thrombi, a feature of adenomas, were usually located in large dilated veins and, therefore, thought to be due to blood stasis. Hematopoiesis, another easily identifiable feature, was present in 83% (19/23) of the adenomas and only 12% (3/26) of the carcinomas. The origin of the hematopoiesis is uncertain, but as with adrenal myelolipomas, it may develop from metaplastic transformation of cells in the adrenal cortex or cells lining the adrenal sinusoids, embryonic rests, or embolism of bone marrow cells. 4,20 Cytoplasmic vacuolation of neoplastic cells was significantly associated with adenomas (78%, 18/23) but has been associated with canine adrenocortical carcinomas in other reports. 4,14 Cellular pleomorphism, mitotic index, nuclear size, nuclear atypia, nuclear chromasia, cytoplasmic eosinophilia, and fibrosis were not significantly different between adenomas and carcinomas (Table 1).
Some histologic factors that differentiate cortical adenomas from carcinomas in dogs differ from those reported in human adrenal tumors. Presence of hematopoiesis has rarely been described as a feature of human adrenocortical tumors, although there are reports of myelolipomas arising within adrenocortical tumors. 8,31 Pleomorphism, mitotic index, and the nuclear changes used in human grading systems were not significantly associated with carcinomas in this study. 6,11,16,17,26,29,33,34 Interestingly, a trabecular growth pattern was associated with malignancy in dogs, whereas a diffuse growth pattern indicates malignant potential in humans. 11,33,34 Also, intratumoral fibrosis, which is an important diagnostic criterion in humans, was only occasionally present in both adenomas and carcinomas. 11,17,29
A key point to emphasize is that most of the significant criteria (peripheral fibrosis, capsular invasion, hemorrhage, necrosis, single-cell necrosis, hematopoiesis, and fibrin thombi) were evaluated as either present or absent. This facilitates reproducibility and decreases interobserver variability. Growth pattern and cytoplasmic vacuolation were variable within individual tumors and thus warranted setting a requirement of one third of the tumor to comprise the trabecular growth pattern and two thirds of the tumor to be composed of cells with cytoplasmic vacuolation for these criteria. It should, however, be noted that some of these histologic features were present in both benign and malignant neoplasms and that all significant criteria should be considered when evaluating adrenocortical tumors.
In the second part of this study, the Ki-67 proliferation index was evaluated in canine adrenocortical adenomas and carcinomas. As in humans, the proliferation index was significantly higher in carcinomas compared with adenomas, but there was no significant difference between adenomas and the normal adrenal cortex. 3,9,12,16,18,25–28,30,32 There was some overlap between the proliferation indices of adenomas and carcinomas (1.1–3.8%). However, 83% (19/23) of the adenomas had a proliferation index equal to or less than 1%, whereas 81% (21/26) of the carcinomas had a proliferation index greater than 5%. A threshold value of the Ki-67 proliferation index of 2.4% accurately predicted malignancy in 96% of the dogs. Various thresholds have been proposed in humans ranging from 2.2% to 9.0%. 12,16,26,28,32
Positive immunohistochemical staining for the Ki-67 antigen at the junction between the zona glomerulosa and the zona fasciculata in the normal adrenal cortex has been reported in humans and is consistent with the proposed mechanism of cell migration or centripetal cell turnover. 25 The distribution of the staining was more diffuse in carcinomas than adenomas. Positive cells in adenomas were occasionally clustered in one area of the tumor, and this should be considered when examining small samples.
In conclusion, we determined that evaluation of a set of histologic criteria in combination with the immunohistochemical assessment of the Ki-67 proliferation index is helpful in the differentiation of adrenocortical adenomas and carcinomas in dogs.
Footnotes
Acknowledgements
We thank Esi Djan, Alyssa Lee, Diane Naydan, Barry Puget, Sarah Schwendinger, and Marty Ynostroza for skillful technical assistance. This study was supported by the Center for Companion Animal Health, School of Veterinary Medicine, University of California, Davis, California.
