Abstract
Pheochromocytoma is frequent in dogs and carries a guarded prognosis. Current histological criteria may not predict malignant behavior in dogs, similar to humans. In humans, characterization of tumors has been refined using the pheochromocytoma of the adrenal gland scaled score (PASS) and by immunohistochemistry. The study aim was to investigate PASS and immunohistochemical markers used in humans in 24 dogs with pheochromocytoma that underwent adrenalectomy. Dogs with pheochromocytomas were reviewed and tumors collected. Histological sections were evaluated to apply the PASS and were single-labeled for chromogranin A, Ki-67, COX-2, p53, BCL-2, c-erbB-2, vascular endothelial growth factor, and S100. Survival, age, and vascular and capsular invasion were compared for PASS and immunohistochemical markers; results of PASS were also compared for each marker. Associations between markers were tested. PASS and immunohistochemical markers did not differ for survival, age, and vascular and capsular invasion. Tumors showing BCL-2 expression in >50% cells had lower PASS than those with lower expression (PASS: 7 ± 2 vs 9 ± 2; P = .011). Tumors positive for S100 had higher PASS than those that were negative (PASS: 10 ± 2 vs 7 ± 2; P = .001). Results of the different markers were not associated. In conclusion, in the context of canine pheochromocytoma, PASS and the selected immunohistochemical markers are not associated with survival, age, or vascular or capsular invasion. The higher PASS in S100-positive tumors may indicate that pheochromocytomas developing morphologic changes acquire S100 expression. The significance of lower PASS in tumors with elevated BCL-2 expression is uncertain. Overall, the use of PASS and the present immunohistochemical markers may not be useful in dogs with pheochromocytoma.
Keywords
Pheochromocytoma is a frequent neoplasm of the adrenal gland in dogs that arises from chromaffin cells. 3,10,14 Most pheochromocytomas are unilateral and are usually identified as large masses occupying the entire gland and less frequently as small nodules surrounded by a compressed rim of normal adrenal parenchyma. 3,5,10 Presenting complaints associated with pheochromocytoma are vague in the majority of dogs; during physical examination, systemic arterial hypertension and cardiac arrhythmias may be identified. 3,5,10,14 Pheochromocytomas often invade the adrenal capsule and, through invasion of the adjacent tissues, can reach the caudal vena cava, promoting formation of large thrombi, which partially or totally occlude the vein leading to abdominal effusion. 20 Sudden weakness or death due to acute bleeding from the tumor or injured vessels has also been described in affected dogs. 25
Pheochromocytomas in dogs are usually considered malignant when invasion through the capsule into adjacent tissues is present or when metastasis to distant sites is documented; they are locally invasive in 39% to 50% of cases and lead to distant metastases in 13% to 24% of cases, including lymph nodes, liver, lungs, kidney, spleen, and bone. 3,10 With regard to prognosis, it is worth mentioning that survival time of dogs with pheochromocytoma undergoing adrenalectomy is mostly linked to surgical factors, while it is unclear if local invasion and metastases have any prognostic relevance. 3 Indeed, if dogs survive surgery and are discharged following hospitalization, median survival is longer than 3 years and most are lost to follow-up or die of another disease. 3 Further factors associated with a favorable outcome in dogs with pheochromocytoma are younger age and shorter duration of surgery. 11 Other investigations of the survival of dogs with pheochromocytoma have included both adrenal medullary and cortical tumors, and as a consequence, the results may be biased; 2,15 –17 invasion of the caudal vena cava was variably associated with poor outcomes in these publications.
From a histological standpoint, morphologic criteria of tumor cells as defined by the World Health Organization (WHO) classification scheme may not predict malignant behavior in dogs with pheochromocytoma. 14 Similarly, prediction of pheochromocytoma behavior in humans based on histological assessment is debated, and no morphologic criteria have been identified that reliably anticipate a malignant course. 22 Hence, the diagnostic approach in humans with pheochromocytoma has been refined by the use of a histological score that takes into consideration different microscopic features and the use of immunohistochemistry. Concerning the former, the pheochromocytoma of the adrenal gland scaled score (PASS), which is based on the combination of 12 different histologic parameters, has been developed and in different studies was higher in malignant lesions compared to those with a benign course. 21,22 With regard to immunohistochemistry, a number of markers have been investigated in pheochromocytoma in humans. Among these, a higher proliferation rate as assessed by Ki-67 and increased expression of cyclooxygenase-2 (COX-2), tumor suppressor gene product p53, antiapoptotic product BCL-2, proto-oncogene product c-erbB-2, and vascular endothelial growth factor (VEGF) in tumor cells, as well as a decreased expression of protein S100 in sustentacular cells of the tumor, have all been associated with worse prognosis. 7,19,22,23
Because of the limited data currently available in dogs, the aim of the present retrospective study was to apply PASS and immunohistochemical markers used in human pheochromocytoma in a series of dogs affected by pheochromocytoma and undergoing adrenalectomy. Surgical outcome, age, and frequency of vascular and capsular invasion caused by the tumor were compared together with the results of PASS and immunohistochemistry. In addition, PASS was assessed in relation to immunohistochemical findings.
Materials and Methods
Selection of Cases
From the pathology archive of the Department of Comparative Biomedicine and Food Science of the University of Padua, Italy and from 4 surgeons (S.Ni., V.M., F.M., G.R.), paraffin blocks of adrenal glands of dogs with a histological diagnosis of pheochromocytoma were retrieved. To be included in the study, pheochromocytoma had to be surgically excised by adrenalectomy and the tumor had to be chromogranin A positive or, if negative, had to show characteristic microscopic features of pheochromocytoma as described by the WHO Classification of Endocrine Tumors. 14
Data pertaining to signalment, including age, sex, breed, and affected adrenal gland, were retrieved from pathology records. Additional data were provided by the above surgeons or were requested from practitioners submitting adrenal samples for histologic examination. For the latter purpose, a questionnaire was devised to acquire information on clinical and laboratory findings; the presence of distant metastasis based on abdominal ultrasound, thoracic radiography, or computed tomography prior to surgery; tumor invasion of vessels such as caudal vena cava, renal vein, or phrenicoabdominal vein as identified by the radiologist with diagnostic imaging or by the surgeon during adrenalectomy; survival to discharge from hospitalization; and long-term survival (ie, >30 days).
Histology
From each formalin-fixed, paraffin-embedded specimen of canine pheochromocytoma, 4-μm-thick sections were obtained, stained with hematoxylin and eosin, and evaluated using light microscopy (Olympus BX-40; Olympus, Segrate, Italy). All tissue sections were assessed by 2 pathologists (L.C., S.F.) in a blinded fashion on 2 different sessions, and at each time, they compared their results. If there was no perfect agreement, each morphological criterion of PASS or result of the immunohistochemical marker was revised to explain the difference. If discrepancies were identified, the 2 pathologists resolved them by consensus. The microscopic criteria used by Thompson 22 in 2002 to develop the PASS for human pheochromocytoma were applied to all cases, as originally reported: (1) large nests (ie, 3-4 times the size of the so-called zellballen nest—namely, a cluster of cells enveloped by thin fibrovascular stroma, or the size of medullary paraganglia nests) or diffuse growth (ie, >10% of tumor area), (2) central (ie, in the middle of large nests) or confluent (ie, diffuse) tumor necrosis, (3) high cellularity (ie, cells with high nuclear-to-cytoplasmic ratio in the majority of the tumor), (4) cellular monotony (ie, monomorphic cell population), (5) tumor cell spindling (even if focal), (6) mitotic figures >3 in 10 high-power fields (HPF; 0.237 mm2 diameter), (7) atypical mitotic figure(s), (8) extension into adipose tissue, (9) vascular invasion, (10) capsular invasion (ie, neoplastic cells breaking through the capsule and invading the periglandular tissue), (11) profound nuclear pleomorphism (ie, enlarged nuclear size, irregular shape, or bizarre forms), and (12) nuclear hyperchromasia (ie, complete opacification and heavy nuclear deposition of chromatin in the majority of cells). The number of points assigned for the presence of each of the findings from (1) to (8) was 2 and, for each of the findings from (9) to (12), was 1; if findings were absent, 0 points were given. The PASS was calculated for each tumor as the sum of points to all 12 parameters.
Immunohistochemistry
Additional 4-μm-thick sections were obtained from each specimen to perform immunohistochemistry using monoclonal or polyclonal antibodies with the following specificities: chromogranin A (dilution 1:50, incubation 32 minutes, clone rabbit anti–chromogranin A; Thermo Fisher Scientific, Chelmsford, MA), Ki-67 (dilution 1:50, incubation 20 minutes, clone MIB-1; Dako, Carpinteria, CA), COX-2 (dilution 1:700, incubation 28 minutes, purified mouse anti–COX-2; BD Biosciences, Milano, Italy), p53 (dilution 1:200, incubation 12 minutes, NCL-p53-CM1; Novocastra, Newcastle, United Kingdom), BCL-2 (dilution 1:200, incubation 32 minutes, rabbit polyclonal IgG; Santa Cruz Biotechnology, Dallas, TX), c-erbB-2 (dilution 1:250, incubation 32 minutes, c-erbB-2 oncoprotein; Dako), VEGF (dilution 1:100, incubation 20 minutes, rabbit polyclonal IgG; Santa Cruz Biotechnology), and S100 (dilution 1:1000, incubation 32 minutes, rabbit polyclonal anti-S100; Dako).
Sections were mounted on Superfrost Plus microscope slides (Menzel, Braunschweig, Germany) and dried at 37°C for 30 minutes. All samples were tested with an automated immunostainer (BenchMark; Ventana Medical Systems, Tucson, AZ), which included dewaxing and rehydration, antigen retrieval with CC1 (Ventana Medical Systems) at 95°C for 8 minutes and at 100°C for 30 minutes, primary antibody incubation, antigen detection with an ultraView Universal diaminobenzidine (DAB) kit (Ventana Medical Systems) for 8 minutes, and counterstaining with Mayer’s hematoxylin for 8 minutes. Finally, slides were manually dehydrated through a graded series of alcohols and mounted (Eukitt mounting medium; Eukitt, Fort Washington, PA). Primary antibody dilutions were performed using a commercial antibody diluent (Ventana Medical Systems).
Different criteria were used to evaluate slides for each immunohistochemical marker. In particular, for chromogranin A, the cytoplasmic staining intensity of neoplastic cells was scored as negative, mild, and moderate to strong. 4 Normal canine adrenal medulla was used as positive control and normal canine adrenal cortical tissue as negative control.
For Ki-67, the percentage of neoplastic cells with nuclear positivity was counted in 1000 cells (counting 200 cells per 5 HPF); 4 to avoid overestimation of counts, neoplastic cells near or within vessels and necrotic and hemorrhagic areas were not considered. Normal canine haired skin was used as control.
For COX-2, cytoplasmic labeling of neoplastic cells was scored as negative (if 0%-10% were positive) or positive (>10%). 19 Normal juxtaglomerular cells from canine renal tissue was used as positive control and normal canine adrenal gland as negative control.
For p53, the percentage of neoplastic cells with nuclear immunolabeling was scored as negative (if 0%-10% were positive), mildly positive (11%-50%), and moderately to strongly positive (>50%). 7 Normal canine haired skin was used as control.
For BCL-2, cytoplasmic immunolabeling of neoplastic cells was scored as negative, positive in <50% cells, and positive in >50% cells. 7 Normal canine lymph node was used as positive control and normal canine adrenal cortical tissue as negative control.
For c-erbB-2, membrane immunolabeling of neoplastic cells was scored as negative or positive. Normal human mammary gland was used as positive control and normal canine adrenal gland as negative control.
For VEGF, the percentage of neoplastic cells with cytoplasmic immunolabeling was scored as negative (if 0%-10% cells were positive), mildly positive (11%-50%), and moderately to strongly positive (>50%). 20 An internal control was used, with vessels being the positive control and nonvascular structures being the negative control.
Expression of S100 was investigated in tumor cells and scored based on the staining intensity as negative, mild, and moderate to strong. 22 Stellate or fusiform cells with hyperchromatic oval nuclei and S100 immunolabeled cytoplasm were identified as sustentacular cells; in humans, they are individually localized between tumor cells or around the nests. 4,24 The tumors were then considered as sustentacular cell negative or sustentacular cell positive based on the absence or presence of these cells. If positivity was documented, the percentage of sustentacular cells was counted in 1000 cells (counting 200 cells per 5 HPF) and as an average number of sustentacular cells in 10 HPF; 4,24 to avoid overestimation of counts, sustentacular cells nearby or within vessels and necrotic and hemorrhagic areas were not considered. Canine mammary myoepithelial cells were used as positive control and canine mammary luminal cells as negative control.
Statistical Analysis
Information pertaining to signalment, clinical and laboratory findings, metastasis, tumor location, vascular invasion as identified by the radiologist or surgeon, histologic and immunohistochemical findings, and short- and long-term outcome of dogs with pheochromocytoma were noted on a spreadsheet. An exploratory investigation was carried out to compare PASS and each immunohistochemical marker between dogs that survived to discharge from hospitalization and those that died, between young and old dogs, between dogs with and without vascular invasion, and between dogs with and without invasion of the adrenal capsule. With regard to age, dogs were arbitrarily considered old if ≥10 years. 17 For vascular and capsular invasion, they were considered present if identified by the pathologist. Because information regarding vascular invasion was also provided by the radiologist based on ultrasound or computed tomography of the abdomen, the analysis was also repeated by combining both microscopic and macroscopic identification of vascular invasion and by considering only the latter. In addition, the PASS was compared within the results of each immunohistochemical marker, and associations between immunohistochemical findings were explored. The PASS was compared between groups using a t test with Levene’s test to assess equality of variances; because it includes assessment of vascular and capsular invasion, 22 each of the 2 criteria was excluded from the score to compare tumors with and without microscopic vascular or capsular invasion, respectively. Data were tested for normal distribution using the Shapiro-Wilk test, and nonnormal data were log-transformed and then analyzed using parametric tests. For each immunohistochemical marker, the frequency of positive staining or of each degree of staining intensity was calculated and compared between groups with Fisher’s exact test. Analyses were performed if at least 5 cases per group were available (eg, the analysis for age was performed if at least 5 dogs were young and 5 were old). Furthermore, Spearman’s correlation analysis was performed to investigate an association between PASS and immunohistochemical markers with quantitative data (ie, number of tumor cells positive for Ki-67 and the number of sustentacular cells). Last, survival to discharge from hospitalization was compared between pheochromocytoma-bearing dogs with and without vascular invasion (microscopic and macroscopic) as well as between those with and without capsular invasion using Fisher’s exact test. Significance was set at P < .05. Commercial software was used for statistical analysis (GraphPad Prism 4.0; GraphPad Software, San Diego, CA).
Results
Dogs
Twenty-four dogs that underwent adrenalectomy and had a pheochromocytoma diagnosed based on histology were included. The mean age was 11 ± 3 years; age was not available in 1 dog (14 dogs were ≥10 years old and 9 dogs were younger). Sex was known in 23 dogs; 12 (52%) were intact males, 10 (44%) were spayed females, and 1 (4%) was an intact female. Breed was available in 20 cases; 12 (60%) were purebred and 8 (40%) were crossbred; among the former, 3 were Labrador Retriever, 2 were West Highland White Terrier, and there was 1 each of American Cocker Spaniel, Boxer, Jack Russell Terrier, Lagotto Romagnolo, Miniature Schnauzer, Shih-Tzu, and Siberian Husky.
Information regarding distant metastasis prior to adrenalectomy based on diagnostic imaging was available in 20 dogs and was observed in none. Pheochromocytoma affected the left adrenal gland in 12 (60%) dogs and the right adrenal gland in 8 (40%) dogs; information was not obtained in 4 cases.
Ten (56%) dogs survived to discharge from the hospital and 8 (44%) dogs died during surgery or hospitalization; in 6 dogs, short-term outcome was unknown. Information pertaining to clinical and laboratory results, as well as long-term survival, was incomplete in 18 (75%) dogs and was not used for further investigation. For the 10 dogs with a favorable outcome, 5 were still alive at the time of writing with a mean survival of 406 ± 365 days and 5 died with a survival of 540 ± 390 days; cause of death was unknown in 4 cases and was unrelated to pheochromocytoma in the fifth dog (car accident).
Vascular invasion was documented by the radiologist or surgeon in 12 dogs (54%) and was not observed in 10 (46%) dogs; data were not obtained in 2 cases. Among the 12 dogs with vascular invasion, 6 involved the phrenicoabdominal vein, 5 the caudal vena cava, and 1 the renal vein.
Histology
Hematoxylin and eosin–stained sections from the 24 dogs with pheochromocytoma were reviewed for morphologic assessment and to generate the PASS (Table 1). 22 None of the tumors showed formation of a complete capsule; they all compressed or invaded the surrounding tissue. Tumor cells breaking through the capsule of the adrenal gland and invading the surrounding tissue was observed in 19 (79%) cases (Fig. 1). In addition, invasion of the periadrenal adipose tissue by neoplastic cells was noted in 5 (21%) cases. Vascular invasion was identified in 17 (71%) pheochromocytomas; affected vessels were observed either within the adrenal capsule or beyond the tumor mass.
Morphologic Criteria, Points Assigned, and Calculated PASS in Dogs With Pheochromocytoma.a
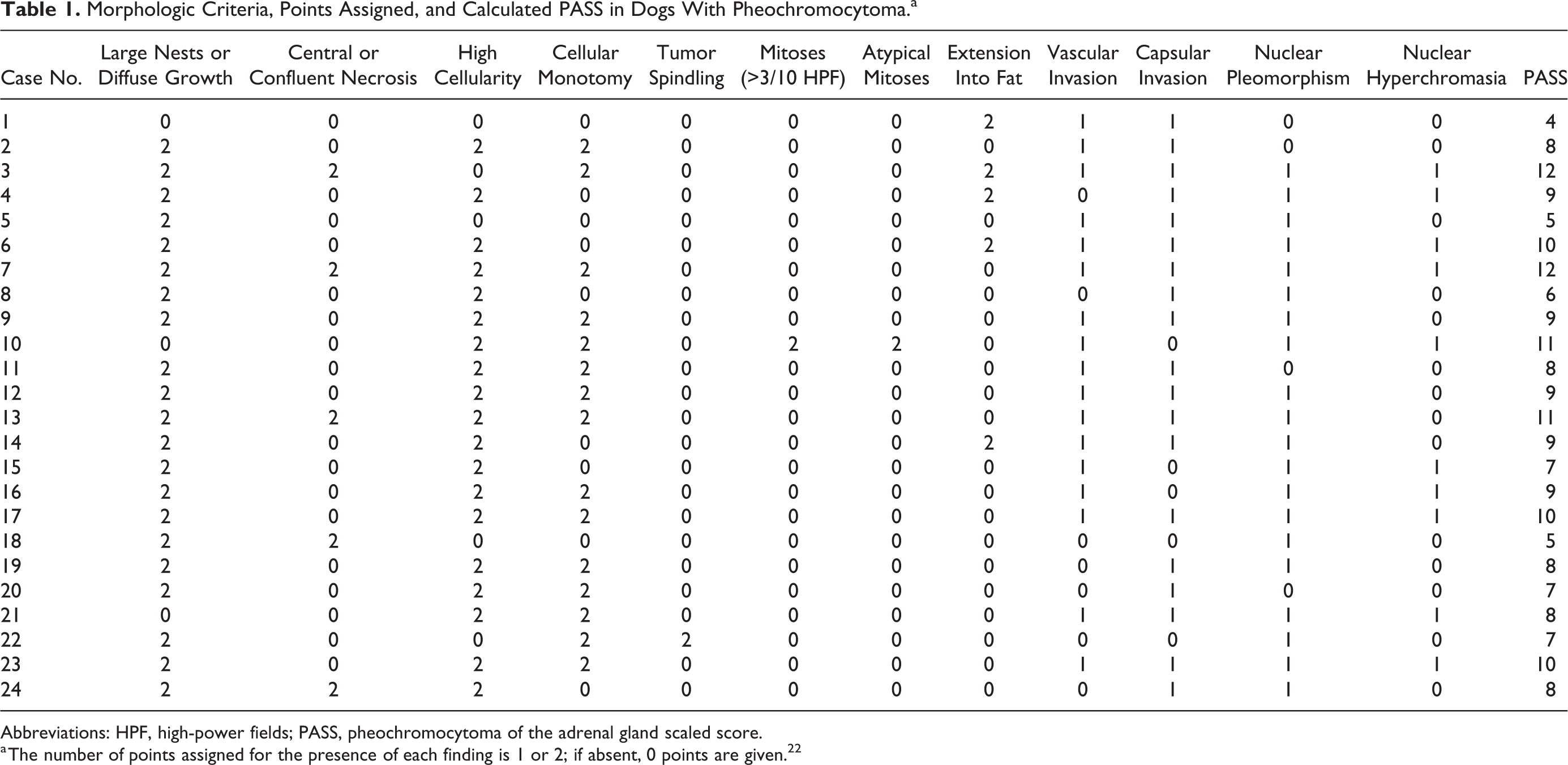
Abbreviations: HPF, high-power fields; PASS, pheochromocytoma of the adrenal gland scaled score.
a The number of points assigned for the presence of each finding is 1 or 2; if absent, 0 points are given. 22
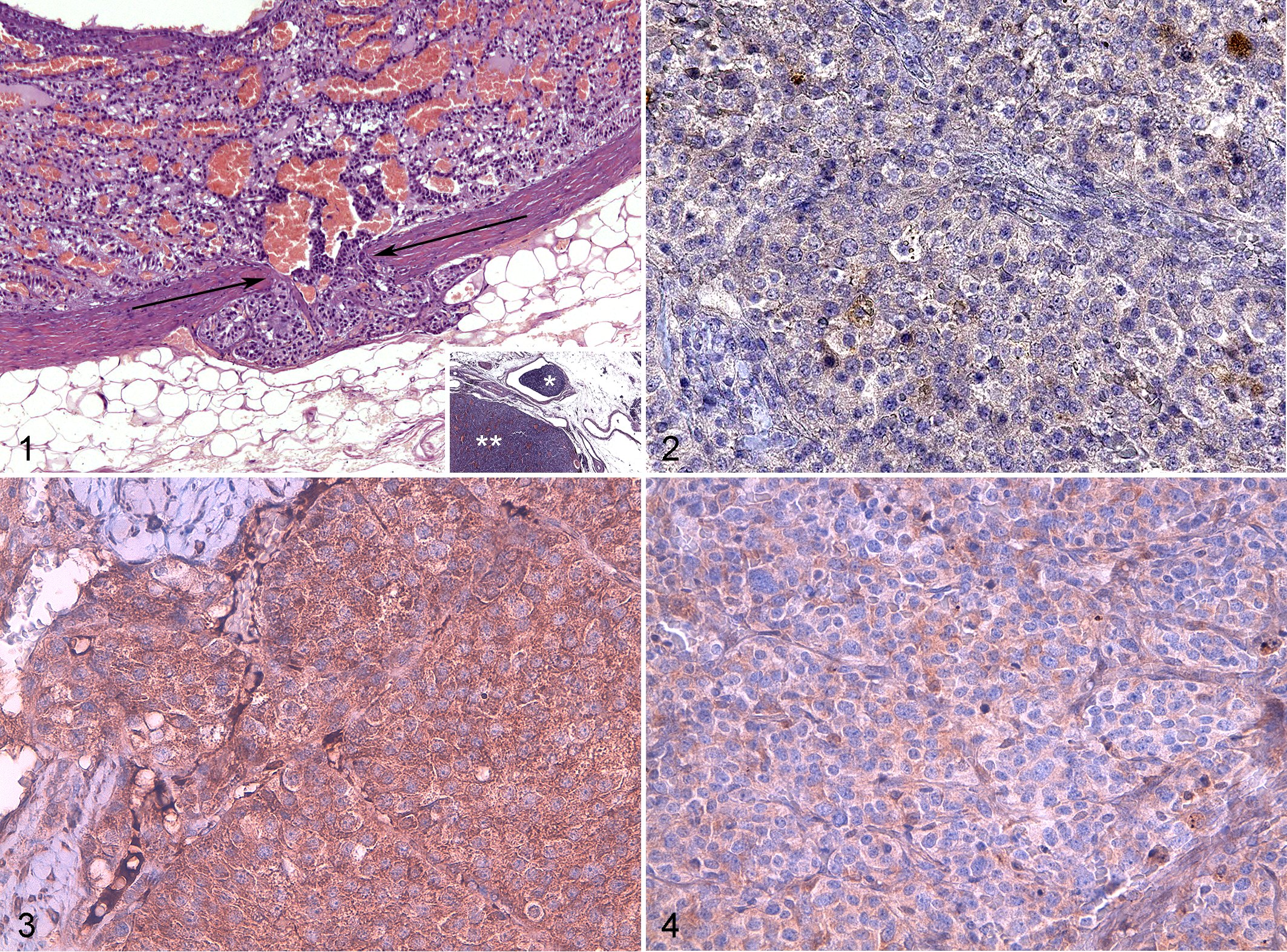
Pheochromocytoma, adrenal gland, dog.
With regard to histological patterns, tumor cells were predominantly distributed in large nests or diffuse growth surrounded by thick acellular bands of fibrosis in 21 (88%) dogs with pheochromocytoma, while in the remaining 3 (12%), they were distributed in small nests. Diffuse or confluent necrosis was observed in 5 (21%) cases. The cellularity was high in 19 (79%) tumors and moderate in the other 5 (21%) cases. Tumor cell spindling was observed in only 1 dog.
In all 24 dogs, the cytoplasm of tumor cells was uniformly granular. The granules were generally eosinophilic but were occasionally lightly eosinophilic or basophilic. Nuclear pleomorphism included enlarged nuclear size, irregular shape, and bizarre forms and was noted in 20 (83%) cases. Cellular monotony was observed in 15 (62%) tumors, while some cell variability was observed in the remaining 9 (38%). Nuclear hyperchromasia was observed in 10 (42%) tumors. Only 1 dog had more than 3 mitoses in 10 HPF; the same case showed atypical mitotic figures. The mean PASS of the 24 dogs was 8 ± 2; the lowest score was 4 (1 dog), and the highest was 12 (2 dogs).
Immunohistochemistry
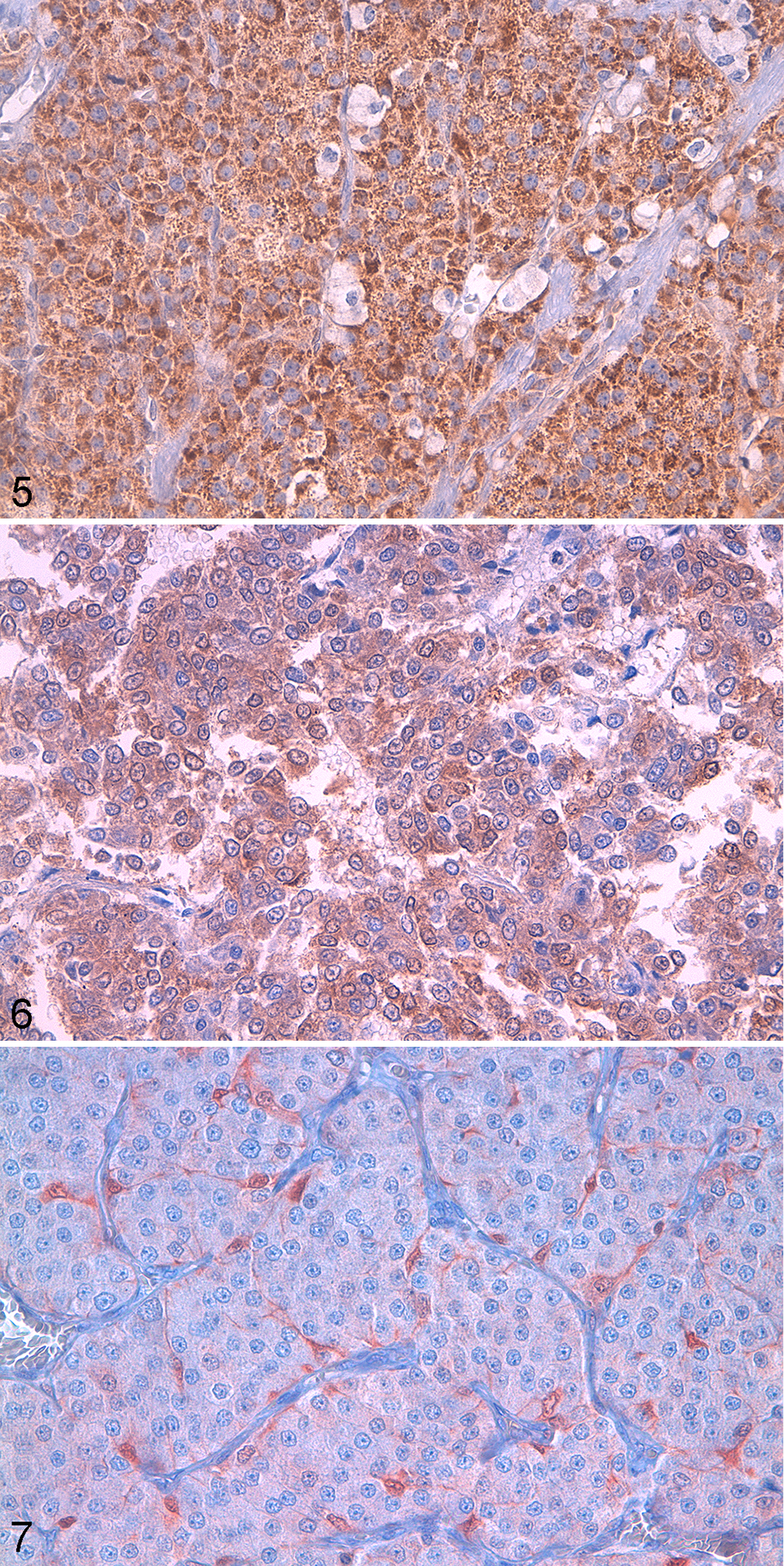
Expression of chromogranin A was observed in all 24 dogs with pheochromocytoma, being mildly positive in 12 (50%) and moderately to strongly positive in 12 (50%) cases. The proliferation marker Ki-67 was positive in 13 of 24 cases (54%); of these 13 tumors, the mean percentage number of Ki-67–positive cells was 2.2% ± 2.1% in 12 tumors but was much higher (ie, 22%) in 1 tumor (this case had capsular but not vascular invasion). COX-2 was positive in 4 of 24 (17%) cases (Fig. 2).
The tumor suppressor gene product p53 was evaluable in 21 of 24 (88%) dogs with pheochromocytoma; in 3 cases, positivity was not assessed due to nonspecific labeling; p53 was negative in 14 of 21 (67%) tumors, mildly positive in 3 (14%), and moderately to strongly positive in 4 (19%). The proto-oncogene product BCL-2 was expressed in all 24 tumors, being positive in <50% cells in 8 (33%) and positive in >50% cells in 16 (67%) cases (Fig. 3). The proto-oncogene product c-erbB-2 was positive in 3 of 24 (13%) tumors (Fig. 4). Expression of VEGF was negative in 13 (54%) tumors, mildly positive in 4 (17%), and moderately to strongly positive in 7 (29%) (Fig. 5).
Pheochromocytoma, adrenal gland, dog.
Expression of S100 was evaluated among tumor cells and was negative in 11 of 24 (46%) cases, mildly positive in 7 (29%), and moderately to strongly positive in 6 (25%) (Fig. 6). Furthermore, the presence of sustentacular cells was evaluated in 19 of 24 (79%) tumors (Fig. 7), while in 5 cases, assessment was not possible due to excessive nonspecific labeling or because the neoplastic tissue was strongly positive to S100, making sustentacular cells not distinguishable. Based on morphologic criteria and S100 immunolabeling, positive sustentacular cells were observed in 6 of the 19 tumors (32%); the mean percentage of sustentacular cells (of 1000 cells forming the tumor) was 3.6% ± 1.9%, and the absolute number was 15.5 ± 9.7 per 10 HPF.
Statistical Analysis
The PASS did not differ between pheochromocytoma-bearing dogs that survived to discharge from hospitalization and those that died (PASS score: 9 ± 2 vs 8 ± 2; P = .229), between young and old dogs (9 ± 2 vs 8 ± 2; P = .110), between tumors with and without microscopic vascular invasion (9 ± 2 vs 7 ± 1; P = .058), or between those with and without invasion of the adrenal capsule (9 ± 2 vs 8 ± 2; P = .478). In addition, the PASS was not different between pheochromocytomas with or without macroscopic vascular invasion (9 ± 3 vs 8 ± 2; P = .449); the analysis combining microscopic and macroscopic vascular invasion was not performed due to the limited number of tumors without invasion. Possible associations were also investigated for each morphologic criterion of the PASS and survival to discharge, age, and vascular and capsular invasion; none was significantly associated.
Similar to PASS, the immunohistochemical markers were not different between groups (Table 2); data pertaining to survival to discharge, age, vascular invasion, capsular invasion, and immunohistochemistry of all dogs are provided in Table 3.
Immunohistochemical Markers in Dogs With Pheochromocytoma and Comparison Between Groups.a
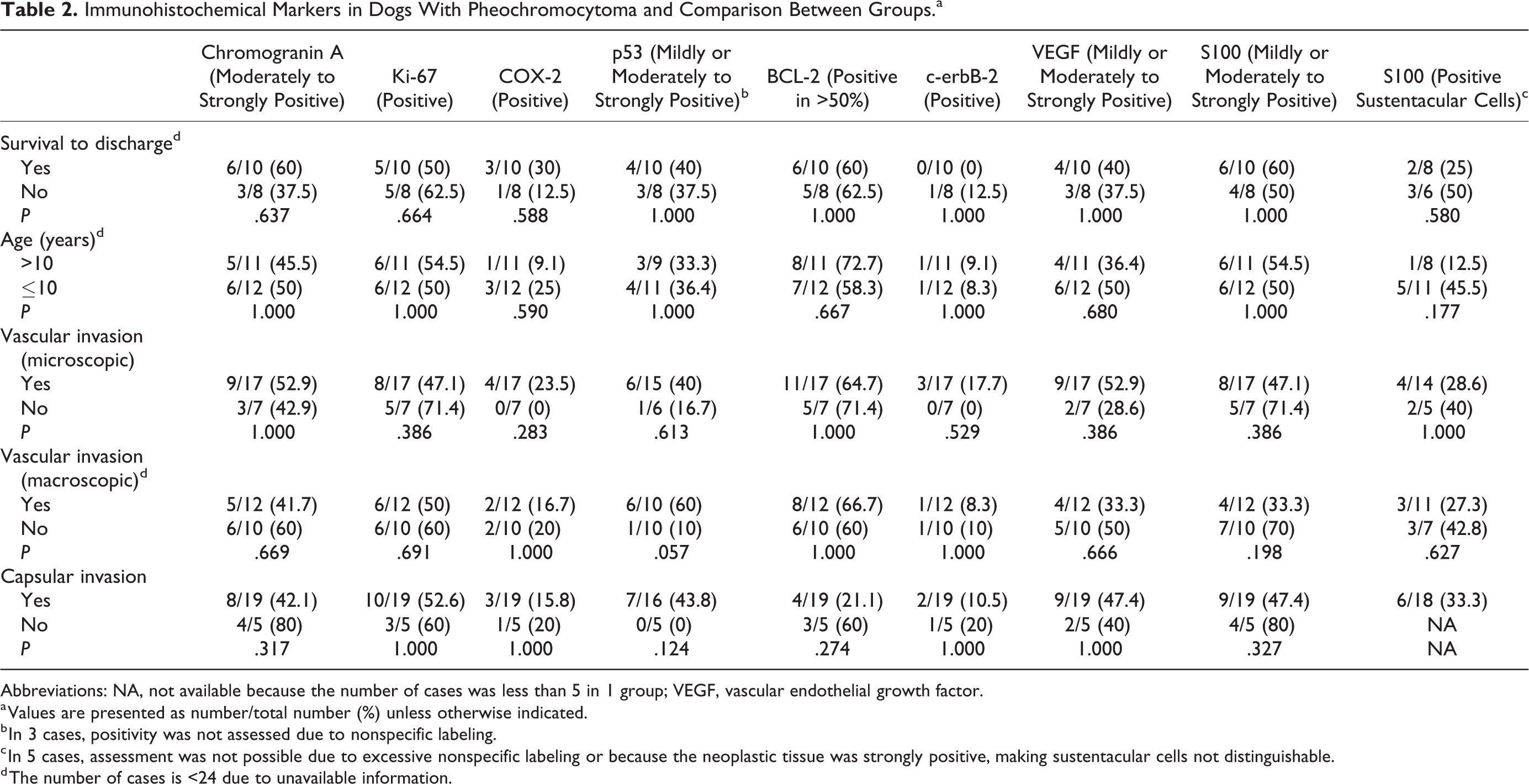
Abbreviations: NA, not available because the number of cases was less than 5 in 1 group; VEGF, vascular endothelial growth factor.
a Values are presented as number/total number (%) unless otherwise indicated.
b In 3 cases, positivity was not assessed due to nonspecific labeling.
c In 5 cases, assessment was not possible due to excessive nonspecific labeling or because the neoplastic tissue was strongly positive, making sustentacular cells not distinguishable.
d The number of cases is <24 due to unavailable information.
Survival to Discharge, Age, Vascular Invasion, Capsular Invasion, and Immunohistochemistry of 24 Dogs with Pheochromocytoma.a
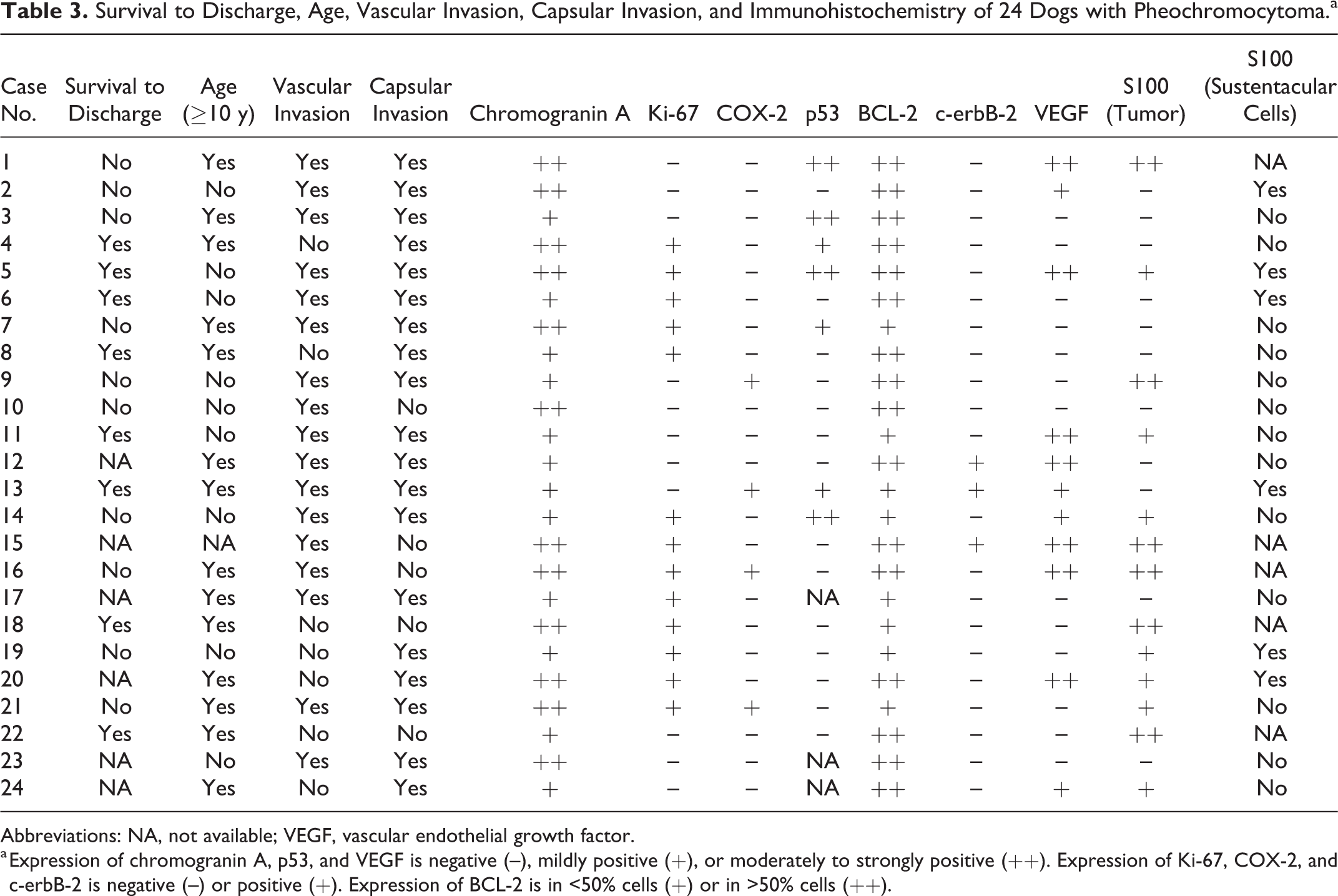
Abbreviations: NA, not available; VEGF, vascular endothelial growth factor.
a Expression of chromogranin A, p53, and VEGF is negative (–), mildly positive (+), or moderately to strongly positive (++). Expression of Ki-67, COX-2, and c-erbB-2 is negative (–) or positive (+). Expression of BCL-2 is in <50% cells (+) or in >50% cells (++).
The PASS was lower in dogs with tumor cells showing BCL-2 expression in >50% cells than those with BCL-2 expression in <50% cells (7 ± 2 vs 9 ± 2; P = .011; Fig. 8). As mentioned above, none of the dogs had tumors that were negative for BCL-2. Dogs with mild or moderate to strong staining intensity of tumor cells for S100 had a higher PASS than those with tumor cells negative for S100 (10 ± 2 vs 7 ± 2; P = .001; Fig. 9). The PASS did not differ among the results of the other immunohistochemical markers. Correlations were not documented between PASS and the number of tumor cells positive for Ki-67 or the number of sustentacular cells. Associations between immunohistochemical markers were not identified.
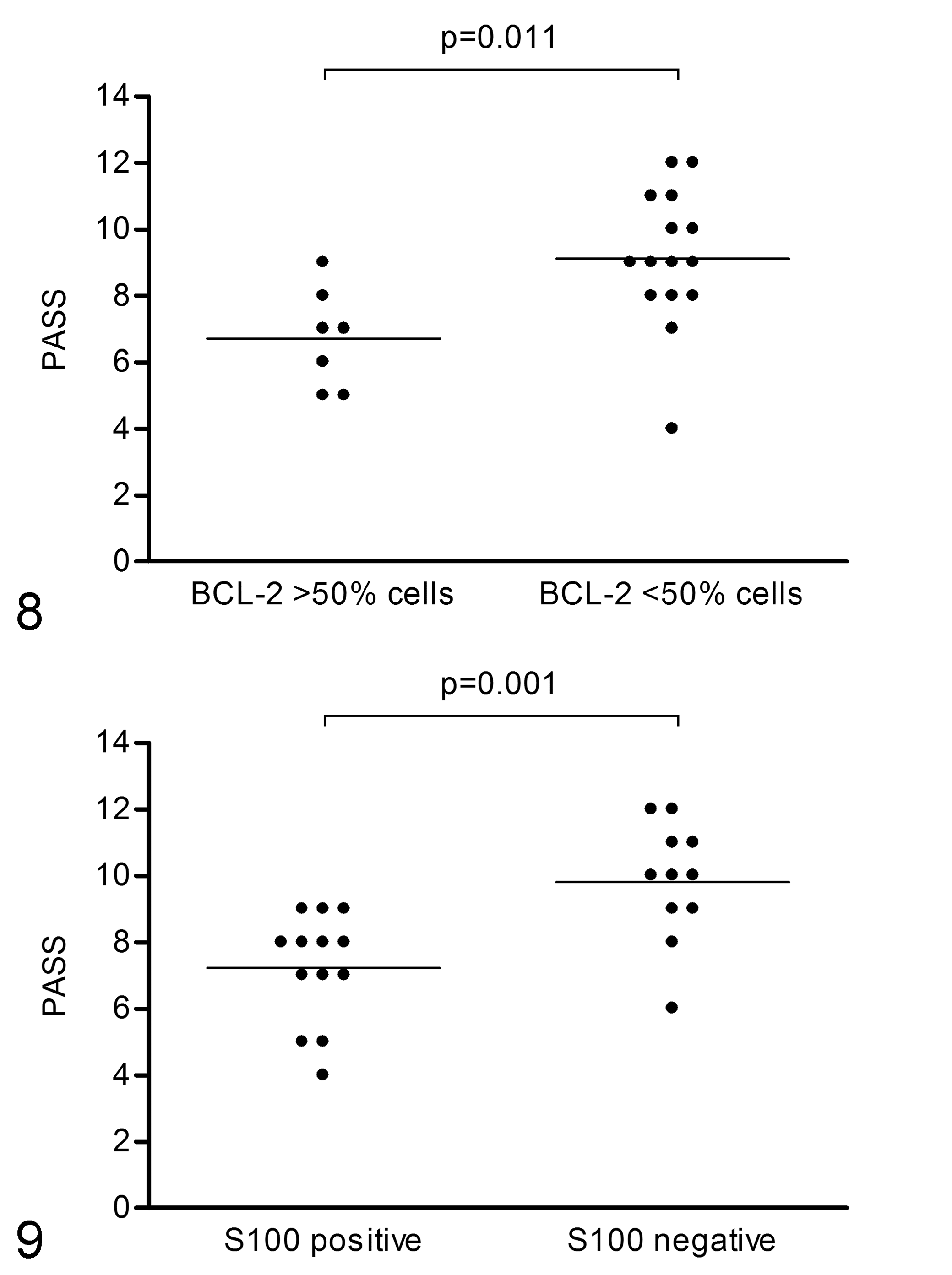
Pheochromocytoma of the adrenal gland scaled score (PASS) in pheochromocytomas with immunolabeling for BCL-2 in >50% cells and in <50% cells. The horizontal line is the mean value.
The rate of survival to discharge from hospitalization did not differ between pheochromocytoma-bearing dogs with and without microscopic (9 of 10, 90% vs 4 of 8, 50%; P = .118) or macroscopic (4 of 10, 40% vs 6 of 8, 75%; P = .188) vascular invasion or between those with and without capsular invasion (8 of 10, 80% vs 6 of 8, 75%; P = 1.000). Age did not differ between survivors and nonsurvivors (10 ± 3 years vs 11 ± 2 years; P = .725).
Discussion
Pheochromocytoma is a frequent tumor of the adrenal gland that carries a guarded prognosis in dogs. Up to now, few histological investigations have been performed in affected dogs, and morphologic criteria do not appear to reliably predict a malignant course. 14 The PASS, a score developed by Thompson 22 for humans with pheochromocytoma based on 12 different morphological features, was applied to canine pheochromocytoma along with immunohistochemical markers used in human pheochromocytomas, including chromogranin A, Ki-67, COX-2, p53, BCL-2, c-erbB-2, VEGF, and S100. 7,19,22,23 Unfortunately, the use of these parameters was not able to differentiate between pheochromocytoma-bearing dogs that survived to discharge from the hospital and those that died following adrenalectomy. Therefore, the scoring system and immunohistochemical markers used in humans to differentiate patients with pheochromocytoma associated with a malignant course from those with a benign course did not provide survival information in affected dogs.
One reason might be that in humans, the reliability of PASS and the listed immunohistochemical markers is not always clear-cut, which might be true in dogs also. Indeed, the PASS was not shown to be associated with outcome in some studies. 1,18 Furthermore, in humans with pheochromocytoma, the most frequently used cutoff of PASS is 4, with higher scores carrying a more guarded prognosis. 8,13 In the present series, only 1 dog had a PASS of 4 while the remaining 23 had higher scores, with a mean overall value of 8 ± 2. Hence, another explanation for the lack of usefulness of PASS in dogs is that the intrinsic behavior of pheochromocytoma in this species may differ from that in humans, with more morphological abnormalities already present by the time of surgery in dogs. This might suggest that pheochromocytomas tend to be more malignant in dogs. It is remarkable that approximately half of our dogs died before discharge from hospitalization, while over 90% of pheochromocytomas in humans are benign and removed without complications. 6
Similar to PASS, some of the listed immunohistochemical markers, such as Ki-67, p53, BCL-2, and c-erbB-2, have not been invariably associated with a malignant course in humans with pheochromocytoma; hence, the histological and the immunhistochemical evaluations are generally performed and evaluated in combination to better predict the prognosis. 7,18,21 Alternatively, the lack of association between PASS or immunohistochemical markers and survival to discharge may be explained by the relatively low number of dogs included, which may have decreased the chance to detect significant differences. It is worth noting that data pertaining to long-term survival were limited in these dogs; the high frequency of death prior to discharge may have masked the association between PASS or immunohistochemical markers and long-term survival.
Of note, survival to discharge might be influenced by a number of reasons other than the tumor. The expertise of the surgeon, anesthetist, and intensive care unit also plays a role. Hence, the short-term outcome of dogs undergoing adrenalectomy likely reflects a combination of factors and may not be solely due to the pheochromocytoma itself. Furthermore, in humans with pheochromocytoma, an important outcome predictor is the presence of distant metastasis or its development during follow-up; metastasis can occur at 5 years or longer after surgery. 6 In our canine series, cases were treated by adrenalectomy if distant metastasis was not documented at admission, and in most of those that survived surgery, it was unclear if metastasis developed during the subsequent months or years. Therefore, this group may be biased by the fact that none had known metastasis at the beginning and information was very limited during follow-up. The retrospective nature of the study did not allow us to obtain a complete history of the dogs, particularly after surgical excision of the tumor.
In addition to outcome, the use of PASS and the immunohistochemical markers did not yield any difference between pheochromocytomas with and without microscopic or macroscopic vascular invasion. The importance of vascular involvement is well known in dogs with pheochromocytoma, since renal veins and caudal vena cava are frequently affected. Macroscopic invasion was reported in half of cases, and of these, 50% infiltrated the caudal vena cava. Even if vascular invasion makes surgery more difficult, increasing the risk of acute and fatal bleeding, 17 in the present study, pheochromocytoma-bearing dogs with and without vascular invasion had a similar chance to survive to discharge from the hospital. Regardless of prognosis, pheochromocytomas with vascular invasion might have higher PASS or more frequent tumor cells positive for p53, BCL-2, c-erbB-2, VEGF, or Ki-67, although this hypothesis was not statistically confirmed in the present series. Differently from dogs, invasion of the inferior vena cava by pheochromocytoma is very rare in humans. 12 Thus, as considered above, it is possible that pheochromocytomas in dogs are more aggressive than in humans.
The use of PASS and immunohistochemical markers did not yield any difference between pheochromocytomas with and without invasion of the adrenal capsule. As for vascular invasion, differences would also be expected for capsular invasion. In humans with pheochromocytoma, capsular invasion is associated with a higher proliferation rate of tumor cells based on Ki-67. 18
Furthermore, there were no differences for PASS or immunohistochemical markers between young and old dogs. A better outcome has been previously documented in younger than older dogs with pheochromocytomas, 11 while in the present investigation, age was not different between survivors and nonsurvivors. In humans with pheochromocytoma, those arising at a younger age are more likely to be malignant, have a higher PASS, and be associated with worse prognosis. 13 Further studies are needed to clarify the role of age in dogs with pheochromocytoma, possibly including a larger number of cases. The limited number of dogs and the arbitrary cutoff of 10 years might have partly biased the results.
With regard to comparison of PASS with the frequency of labeling for each immunohistochemical marker, it was observed that dogs with more than half of tumor cells showing expression of the antiapoptotic product BCL-2 had a lower score than those with less expression. This result is unexpected because in humans with pheochromocytoma, both PASS and BCL-2 increase. 7,21,22 To clarify this finding, associations between BCL-2 expression and each morphologic criterion of PASS were investigated but none was documented (data not shown). In addition, dogs with tumor cells positive for S100 had higher PASS than those with tumor cells negative for S100. The interpretation of this result is difficult, based on the notion that in humans, pheochromocytoma cells with higher PASS are more likely to become S100 negative. 8 This finding may suggest that pheochromocytomas developing more morphologic changes also acquire S100 expression in dogs.
It was possible to identify sustentacular cells within approximately 30% of canine pheochromocytomas. Their presence was not associated with any other finding. As in humans, they were fusiform to stellate and individually localized between tumor cells or around nests. 4,24 They are believed to represent supporting elements but also participate in the modulation of the activity of the adrenal medulla, probably through paracrine effects. 9 In humans, a higher number of sustentacular cells in malignant pheochromocytomas have been described. 23
There are limitations to this study. Information on long-term survival was limited in this case series, and short-term survival may be more related to surgical factors than to the inherent nature or grade of the tumor. Thus, the high frequency of death prior to discharge from the hospital may have obscured a true association between PASS and long-term survival. The exclusion of dogs with known metastasis at the time of surgery did not allow us to obtain more information about potential associations between PASS or immunohistochemical markers and the disease course; however, in a clinical setting, adrenalectomy is normally not proposed to owners if metastases are evident. In addition, the absence of information about weight and size of the tumor prevented study of their possible associations with all the other features investigated. The lack of necropsy in dogs that died also represents a limitation of the study; this information might have provided additional insights into the prognostic role of pheochromocytoma. Furthermore, in most cases, the attending surgeon submitted only part of the tumor and fixation time was not standardized; hence, it cannot be excluded that some of the findings would have differed if the entire tumor was available for analysis or the fixation of samples was similar between cases. Finally, the dogs were not examined and treated by the same surgeon and radiologist, possibly biasing clinical details and survival.
In conclusion, in dogs with pheochromocytoma, outcome, age, and vascular and capsular invasion were not associated with PASS or with scoring of selected immunohistochemical markers. The higher PASS in S100-positive tumors may indicate that pheochromocytomas developing morphologic changes acquire S100 expression. The significance of lower PASS in tumors with elevated BCL-2 expression is uncertain. Overall, the use of PASS and the present immunohistochemical markers may not be useful in dogs with pheochromocytoma.
Footnotes
Acknowledgements
We are grateful to the veterinarians who provided information about the cases.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
