Abstract
Tumors of the adrenal glands are among the most frequent tumors in cattle; however, few studies have been conducted to describe their characteristics. The aim of this study was to classify 41 bovine adrenal neoplasms from 40 animals based on macroscopic and histologic examination, including electron microscopy and immunohistochemistry for melan A, synaptophysin, chromogranin A, vimentin, pan-cytokeratin, 2′,3′-cyclic nucleotide-3′-phosphohydrolase (CNPase), and Ki-67. The tumors were classified as 23 adrenocortical adenomas, 12 adrenocortical carcinomas, 2 schwannomas, 2 pheochromocytomas (1 malignant), and 1 ganglioneuroma. Five histologic features were characteristic of metastasizing adrenocortical tumors: invasion of the capsule, vascular invasion, diffuse growth pattern, spindle-cell morphology, and nuclear pleomorphism. Adrenocortical tumors with at least 3 of these features were classified as malignant. Immunohistochemically, adrenocortical tumors expressed melan A (16/19), vimentin (14/26), cytokeratin (11/26), and chromogranin A (9/27), whereas pheochromocytomas expressed chromogranin A (2/2), synaptophysin (2/2), and vimentin (1/2). Both schwannomas expressed CNPase. An immunohistochemistry panel consisting of antibodies against melan A, synaptophysin, and CNPase was considered most useful to classify bovine adrenal tumors. However, the distinction between benign and malignant adrenocortical tumors was based on histologic features as in human medicine.
Keywords
Adrenal tumors are relatively common in cattle 7,11,22,23,33 but, in contrast to those in other animal species, 4,8,12,30 –32,40 are generally considered to be nonfunctional. 23,33 In addition, bovine adrenal tumors rarely metastasize or cause clinical manifestations 23,33,39 and are, therefore, mainly found during routine meat inspection. 7,11,23,26,33,39 Cortical adenomas, pheochromocytomas, and cortical carcinomas are the most frequently diagnosed. 7,11,22,23,26,33,39 Adrenal neuroblastomas, neurofibromas, and ganglioneuromas have also been described, 23,35,39 and it is not uncommon to observe secondary tumors (ie, metastases to the adrenal gland). 16
The fixative potassium dichromate and chromaffin stains have been used to diagnose bovine pheochromocytomas, 10,23,33,38,39 whereas immunohistochemistry has been applied to characterize and differentiate adrenal gland tumors in nonbovine species. 2,6,9,15,19,20,27,28,32 Major immunohistochemical tests in the diagnosis of adrenal gland tumors include chromogranin A, synaptophysin, inhibin-α, and melan A (clone A103). Chromogranin A is present in neuroendocrine cells and has been used to differentiate pheochromocytomas from adrenocortical tumors in dogs, ferrets, pigs, rats, and humans. 2,5,19 –21,27,28,32 Synaptophysin is also a marker of neuroendocrine tumors, and immunolabeling for synaptophysin has been demonstrated in pheochromocytomas in dogs, rats, and humans. 2,5,20,21,27,32 Variable immunoreactivity for synaptophysin has, however, been reported in adrenocortical tumors in ferrets and humans. 5,28 Finally, inhibin-α and melan A have been used for identification of adrenocortical tumors in ferrets 28,34 and dogs, 29 respectively.
Distinction between benign and malignant adrenal tumors is important and can be challenging in the absence of metastasis. 17,18,23 In human medicine, a combination of clinical and pathologic parameters has been used to differentiate adrenocortical adenomas from carcinomas, including size, 13,18,37 and histological features such as invasiveness, degenerative changes, mitotic rate, and growth pattern. 18 Finally, increased immunohistochemical expression of proteins associated with cell proliferation, such as Ki-67, has been associated with malignant behavior. 24,36 Similar criteria have been applied in dogs for differentiation of benign and malignant adrenocortical tumors. 17
In this study, we evaluated the usefulness of an immunohistochemical panel consisting of antibodies for melan A, synaptophysin, chromogranin A, vimentin, pan-cytokeratin, and 2′,3′-cyclic nucleotide-3′-phosphohydrolase (CNPase) to classify bovine adrenal gland tumors. Furthermore, we investigated various methods to differentiate between benign and malignant bovine adrenocortical tumors.
Materials and Methods
Forty-one adrenal gland tumors from 40 cattle slaughtered at Danish abattoirs from 1999 to 2010 were examined. The tumors were incidental findings during meat inspection, during which the adrenal glands are not routinely incised. Signalment and information regarding clinical disease, reported by the owner or discovered during visual inspection of the animals prior to slaughter, were recorded in the anamnesis. The material submitted to the Department of Veterinary Disease Biology consisted in each case of the tumor, part of the liver, the kidneys, and the regional lymph nodes. When lesions suspected to be metastases were present, all affected organs were also submitted and examined grossly and histologically.
At gross examination, the liver, kidney, adjacent vessels, and regional lymph nodes were inspected for invasion or metastasis. The size of the tumor was measured at its largest diameter, and the entire tumor was sliced to obtain representative regions for histology. One to 6 samples (in total, 100 samples) were taken from each tumor for histologic examination. Five normal bovine adrenal glands were used for comparison.
All samples were fixed in 10% neutral buffered formalin, processed conventionally, and embedded in paraffin. Samples from 2 tumors were fixed immediately after slaughter, whereas the rest of the samples were refrigerated, submitted to the Department of Veterinary Disease Biology for gross examination, and fixed 1 to 2 days postmortem. The freshly fixed tumors were used to evaluate the effects of delayed fixation. Samples with extensive autolytic changes were not included in the study. Furthermore, the effect of the duration of fixation on melan A immunohistochemistry was assessed in samples from 2 tumors and 2 normal adrenal glands fixed in formalin for 30 hours, 5 days, or 11 days. From each sample, 2- to 3-μm sections were cut and stained with hematoxylin and eosin (HE) for histologic assessment.
To differentiate adrenal adenomas from carcinomas, the adrenocortical tumors with histologically confirmed metastases were compared with those without metastasis, and the following histologic features were evaluated for each tumor: growth pattern, necrosis, hemorrhage, mineralization, cystic degeneration, capsular invasion, vascular invasion, cell morphology, nuclear pleomorphism, and mitotic index. Tumors with at least 3 of the following 5 features were classified as malignant: invasive growth into the capsule, vascular invasion, diffuse growth pattern, spindle-cell morphology, and nuclear pleomorphism.
The Ki-67 proliferation index was determined in benign and malignant pheochromocytomas, as well as in adrenocortical adenomas and carcinomas. In each sample, the Ki-67 proliferation index was calculated as the proportion of Ki-67–positive nuclei among 1000 cells counted using a grid in the area with the highest number of immunopositive cells (ie, “hot spots”).
Selected tumors were evaluated immunohistochemically with antibodies to the following antigens: melan A, synaptophysin, chromogranin A, CNPase, vimentin, and cytokeratin (Table 1). For immunohistochemistry, 1- to 2-μm-thick sections were mounted on adhesive-coated slides (Superfrost Plus; Menzel-Glaser, Braunschweig, Germany), processed through xylene, and rehydrated in ethanol. Antigen retrieval was done by boiling in a microwave oven (700 W) twice for 5 minutes in Tris-EDTA buffer (1.21 g TRIS base [A 1379; Applichem, Darmstadt, Germany] and 0.372 g EDTA [8418; Merck, Darmstadt, Germany] to 1 liter of distilled water), pH 9; Tris-EGTA buffer (1.21 g TRIS base [A 1379; Applichem] and 0.190 g EGTA [E3889; Sigma-Aldrich, St Louis, MO] to 1 liter of distilled water), pH 8; or 0.01M citrate buffer, pH 6. Endogenous peroxidase was blocked with 0.6% (v/v) H2O2 in Tris-buffered saline (TBS; pH 7.6) for 15 minutes at room temperature before the sections were incubated with the primary antibody for 1 hour or 24 hours at 4°C (Table 1). The detection systems UltraVision ONE, HRP polymer and UltraVision LP, HRP polymer were applied in accordance with the manufacturer’s instructions (Lab Vision, Thermo Fisher Scientific, Fremont, CA) (Table 1). The chromogen was 3-amino-9-ethylcarbazoloe (AEC-red) (Lab Vision, Thermo Fisher Scientific); sections were counterstained with Mayer’s hematoxylin. Slides were given two 5-minutes washes in TBS (pH 7.6) before addition of each reagent.
Immunohistochemical Methods.

aSynaptophysin and melan A (AbD Serotec, Düsseldorf, Germany); chromogranin A, vimentin, cytokeratin, and Ki-67 (DakoCytomation, Glostrup, Denmark); CNPase (Lab Vision, Thermo Fisher Science, Freemont, CA).
bLab Vision, Thermo Fisher Science.
Negative controls with the primary antibody substituted by 1% bovine serum albumin in TBS or with an irrelevant monoclonal (matching isotype) or polyclonal antibody (X0903; DAKO, Glostrup, Denmark) of the same concentration as the primary antibody were run in parallel. Normal bovine adrenal glands, fixed immediately after slaughter, were used as positive controls.
A subjective grading system was used to determine the intensity of immunoreactivity in neoplastic cells (0 = no reaction, 1 = weak reaction, 2 = moderate reaction, and 3 = intense reaction). In addition, a semi-quantitative assessment of the percentage of positive neoplastic cells, independent of intensity, was used (negative = <2%, 1 = 2%–9%, 2 = 10%–50%, and 3 = >50%).
For transmission electron microscopy, small samples (1 mm3) from 2 adrenocortical tumors were, immediately after slaughter, immersed in Karnovsky fixative (2.5% glutaraldehyde and 2.0% paraformaldehyde in 0.1 mM cacodylate buffer, pH 7.2) for 2.5 hours. After rinsing in cacodylate buffer, the samples were postfixed in 1.0% osmium tetroxide and stained with 1% uranyl acetate for 1 hour. Dehydration in a graded series of acetone was followed by infiltration by Epon, polymerization at 60°C, and ultrathin sectioning (60 nm). The sections were mounted on grids and contrasted with lead citrate for 2 minutes.
The χ 2 test was used to compare histomorphologic characteristics between metastasizing and nonmetastasizing adrenocortical tumors and to compare size, immunoreactivity, and proliferation index between tumors classified as adrenocortical adenomas and carcinomas. The limit for statistical significance was set at P < .05.
Results
The 41 adrenal gland tumors included 23 adrenocortical adenomas, 12 adrenocortical carcinomas, 2 pheochromocytomas (1 of which was malignant), 2 schwannomas, and 1 ganglioneuroma. In 1 case, a schwannoma and an adrenocortical adenoma component were diagnosed in the same adrenal tumor, and 1 cow had an adrenocortical adenoma in the left adrenal gland and a schwannoma in the right gland.
Clinical History and Gross Pathology
All but 1 animal with adrenal gland tumors were female, with no apparent breed predilection. The average age of animals with adrenocortical adenomas and carcinomas was 7.2 years (range, 2.5–15 years) and 7.5 years (range, 3.5–12 years), respectively. No clinical signs of hormonal functionality were reported, although a 2.5-year-old bull with an adrenocortical adenoma was slaughtered because of infertility.
On gross examination, the mean (SD) diameter of adrenocortical adenomas was 12.73 (3.65) cm (range, 5–17 cm); that of adrenocortical carcinomas was 12.88 (8.29) cm (range, 3–25 cm) (P = .52). Both adenomas and carcinomas were yellowish, lobulated, and soft or friable, and they comprised areas with hemorrhage (21/35), calcification (15/35), necrosis (10/35), and/or large cystic spaces (7/35). At the edge of the tumor, nonneoplastic adrenal gland tissue was usually identified (Fig. 1). The adenomas were well demarcated; some were surrounded by a thin fibrous capsule, and in 7 of 23, compression and thinning of the aortic or vena caval wall were noted. Invasion and thrombosis of the caudal vena cava were detected in 4 of 12 carcinomas, and 7 carcinomas had metastasized to the liver (3/7), lungs (3/7), or regional lymph nodes (2/7).
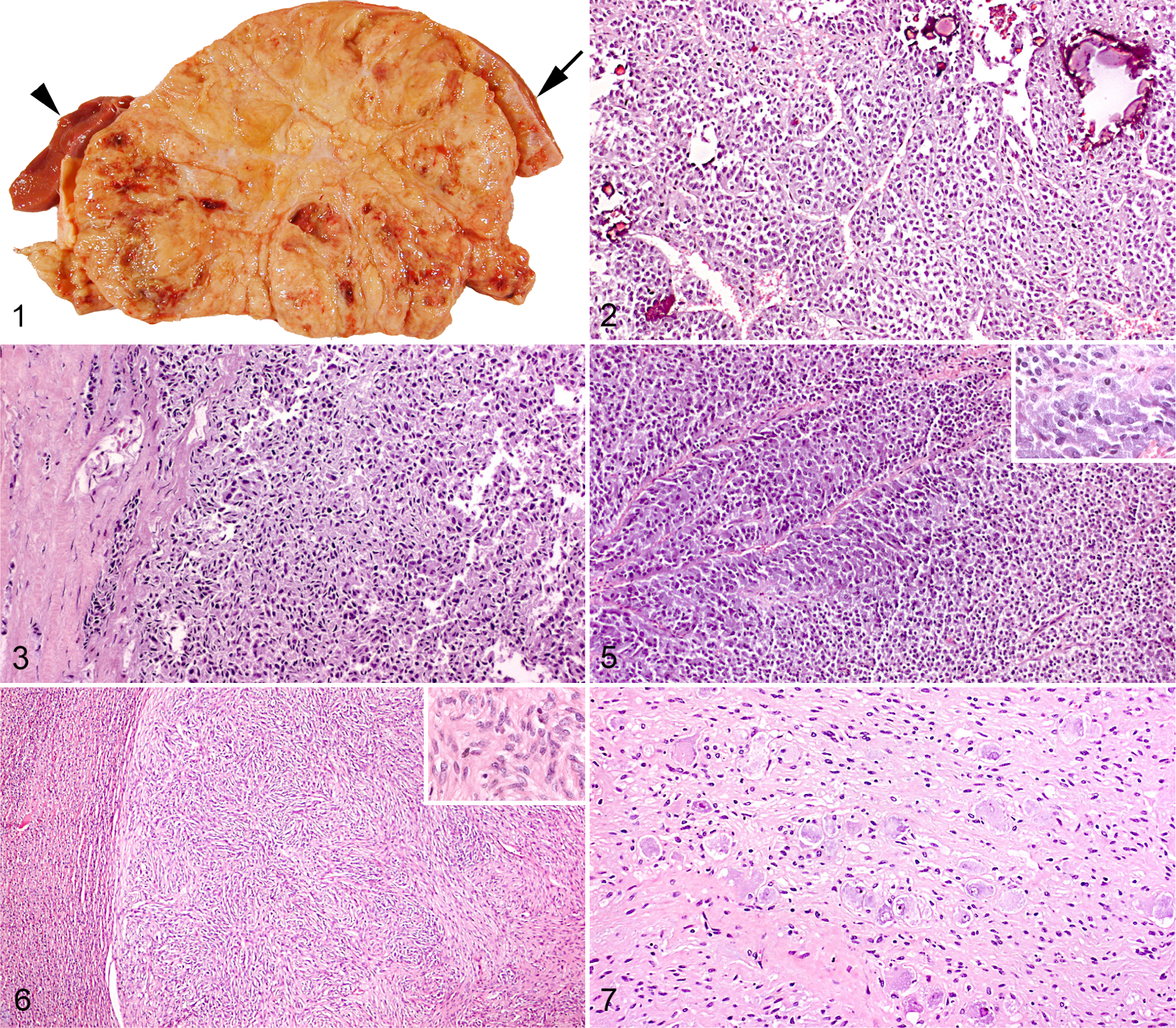
The 2 pheochromocytomas were 8 and 9 cm in diameter, respectively, reddish-brown, and firmer than cortical tumors. The schwannomas were circumscribed, white to yellowish nodules within an enlarged adrenal gland. The ganglioneuroma, found in a 13-month-old heifer, was well demarcated, 15 cm in diameter, firm, and marbled white to yellow.
Histopathology and Electron Microscopy
The adenomas were generally lobulated and composed of cells arranged in trabeculae, cords, or nests separated by a fine fibrovascular stroma (Fig. 2). The cells had uniform round to oval vesicular nuclei and an indistinct, pale, eosinophilic, and flocculent cytoplasm. A few cells contained large eosinophilic cytoplasmic granules, similar to those in the cells of the zona glomerulosa. 14
Most adenomas and carcinomas had moderate to marked necrosis, mineralization, and hemorrhage with cavernous spaces filled by plasma or blood. Three adenomas and 2 carcinomas had areas in which the neoplastic cells formed lumen-like spaces containing variable amounts of mucin-like material (myxoid differentiation; Table 2).
Prevalence of Selected Histologic Features in Bovine Adrenocortical Tumors.
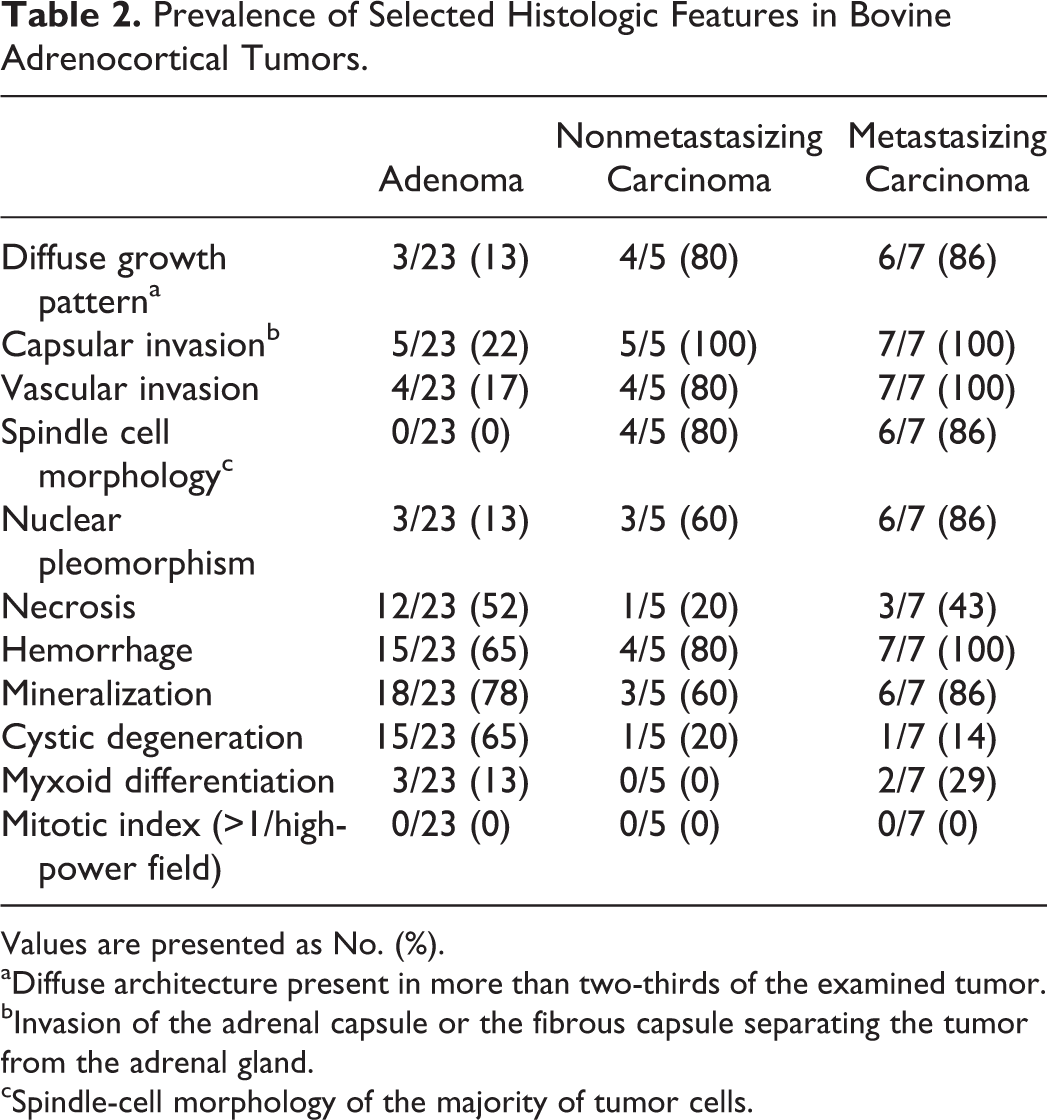
Values are presented as No. (%).
aDiffuse architecture present in more than two-thirds of the examined tumor.
bInvasion of the adrenal capsule or the fibrous capsule separating the tumor from the adrenal gland.
cSpindle-cell morphology of the majority of tumor cells.
In the periphery of the adrenocortical tumors, nonneoplastic medullary and/or cortical tissue was detected. In addition, marked fibrosis was present in the part of the medulla adjacent to the tumor, forming a discontinuous capsule between the tumor and the rest of the adrenal gland. In comparison with adrenocortical tumors that had not metastasized, the following histologic characteristics were more common in adrenocortical carcinomas with distant metastasis (P = .00–.02): (1) invasion of the adrenal capsule or the fibrous capsule separating the tumor from the rest of the gland (Fig. 3), (2) invasion of venous structures or sinusoids (the presence of few individual neoplastic cells in a vascular lumen was regarded as artifactual implantation), (3) diffuse architecture in more than two-thirds of the examined tumor (ie, absence of an organized arrangement of tumor cells in nests, cords, or trabeculae; Fig. 3), (4) spindle-cell morphology in the majority of tumor cells, and (5) moderate to marked nuclear pleomorphism (Table 2). Four of the 5 nonmetastasizing adrenocortical tumors classified as carcinomas had spindle-cell morphology, diffuse growth pattern, and/or vascular invasion, whereas 5 of 5 and 3 of 5 had invaded the capsule or had nuclear pleomorphism, respectively (Table 2). In 1 nonmetastasizing carcinoma as well as in 1 carcinoma with metastasis, only 3 of the 5 criteria were concurrently present. No difference was observed in the prevalence of the histologic features of hemorrhage, necrosis, mineralization, cystic degeneration, or mitotic index between metastasizing and nonmetastasizing adrenocortical tumors (P = .09–1.00; Table 2).
Ultrastructurally, the adrenocortical tumors were composed of cells with densely packed mitochondria with tubular cristae and abundant smooth endoplasmic reticulum. Concentric whorls of endoplasmic reticulum or neurosecretory granules were not detected (Fig. 4).
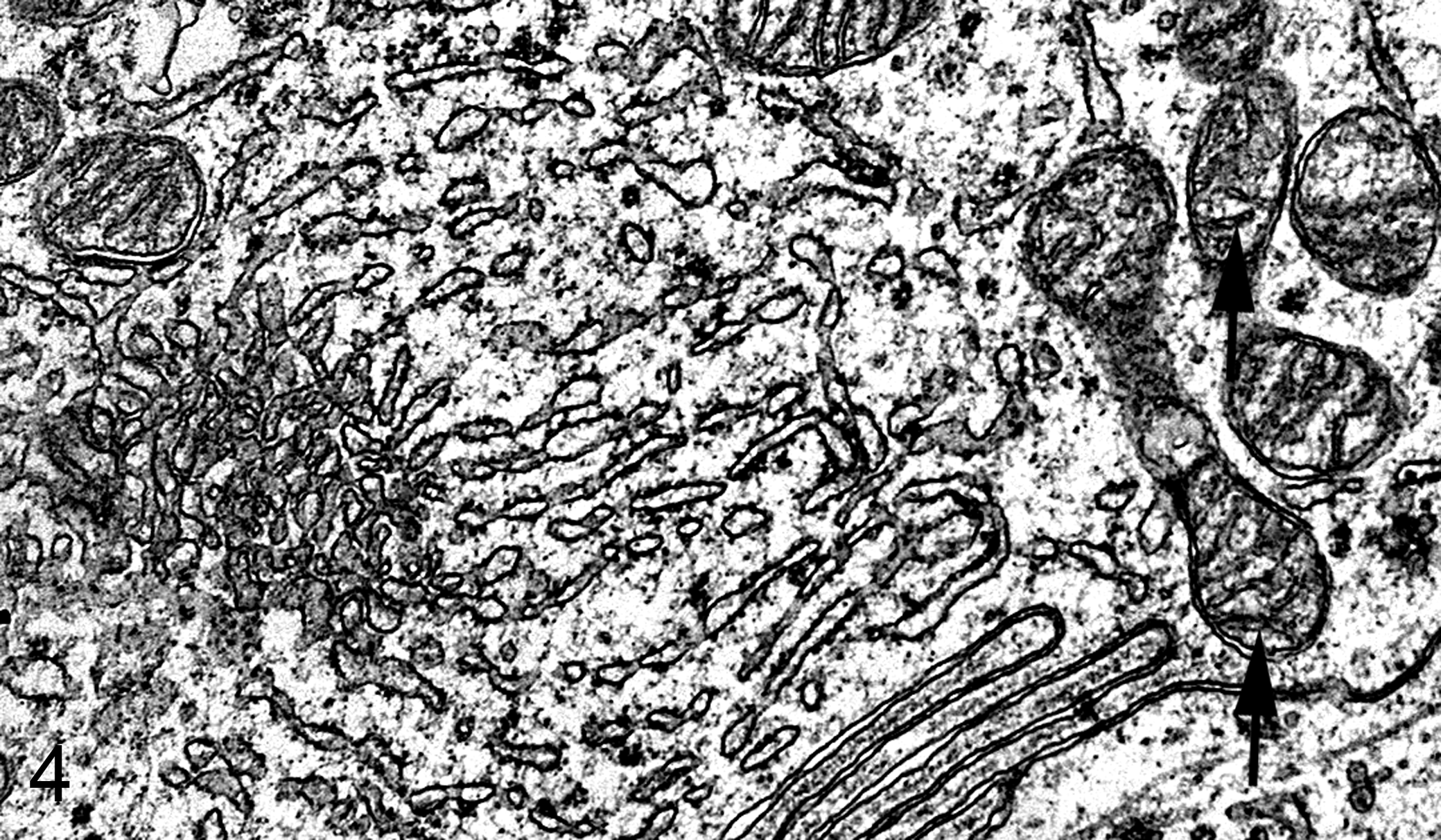
Bovine adrenocortical carcinoma cell. Note smooth and rough endoplasmic reticulum, Golgi apparatus, and mitochondria with primitive (tubular and shelf-like) cristae (arrows). Electron microscopy.
The pheochromocytomas were nonencapsulated, highly cellular, and arranged in small lobules by delicate fibrovascular septa. The neoplastic cells were small, had a round hyperchromatic nucleus, and resembled norepinephrine-producing cells of the normal adrenal medulla, whereas large polyhedral cells similar to the epinephrine cells formed sheets and palisades along sinusoids (Fig. 5). The benign pheochromocytoma expanded into the adrenal cortex, causing compression and atrophy. In the malignant pheochromocytoma, hemorrhage and vascular and capsular invasion were conspicuous features. The mitotic index was 0 and 1 in ten 400× high-power fields (hpf) in the benign and malignant pheochromocytoma, respectively.
The schwannomas were well demarcated and expansive, causing compression of the adjacent adrenal medullary or cortical tissue. They were composed of spindle-shaped cells with indistinct cell borders that were arranged in short interwoven fascicles or broad bundles (Fig. 6). Mitotic figures were not seen.
In the ganglioneuroma, numerous mature ganglion cells were interspersed between broad bundles of spindle-shaped cells and nerve fascicles (Fig. 7). Mitotic figures were rare (0 in 10 hpf).
Immunohistochemistry
The Ki-67 immunoreactivity was intense and confined to the nucleus. In the normal adrenal gland, positive cortical cells were mainly in the zona glomerulosa, and the mean proliferation index was 1.2%. In the normal adrenal medulla, the mean proliferation index was 2.08%. In the adrenal tumors, Ki-67–positive cells were diffusely distributed in the neoplastic tissue, and labeled cells were approximately the same size as the negative cells. There was no statistically significant difference (P = .39) between the mean proliferation index of adenomas (mean [SD], 5.5% [8.0%]; range, 0%–12%) and carcinomas (mean [SD], 4.6% [3.6%]; range, 2.1%–7.2%). The Ki-67 proliferation index was 13.6% and 11.3% in the benign and malignant pheochromocytoma, respectively.
In the normal bovine adrenal gland, melan A immunolabeling was in the cytoplasm of cortical cells, and the zona fasciculata and reticularis had more intense reactivity than the zona glomerulosa. Medullary cells as well as the adrenal capsule were negative (Fig. 8). Melan A immunoreactivity was detected in 6 of 8 adrenocortical adenomas and in 10 of 11 carcinomas (sensitivity, 84.2%). In contrast, no pheochromocytomas (0/2) or schwannomas (0/3) labeled positive. Melan A immunolabeling could not be used to distinguish between benign and malignant adrenocortical tumors (P = .55).
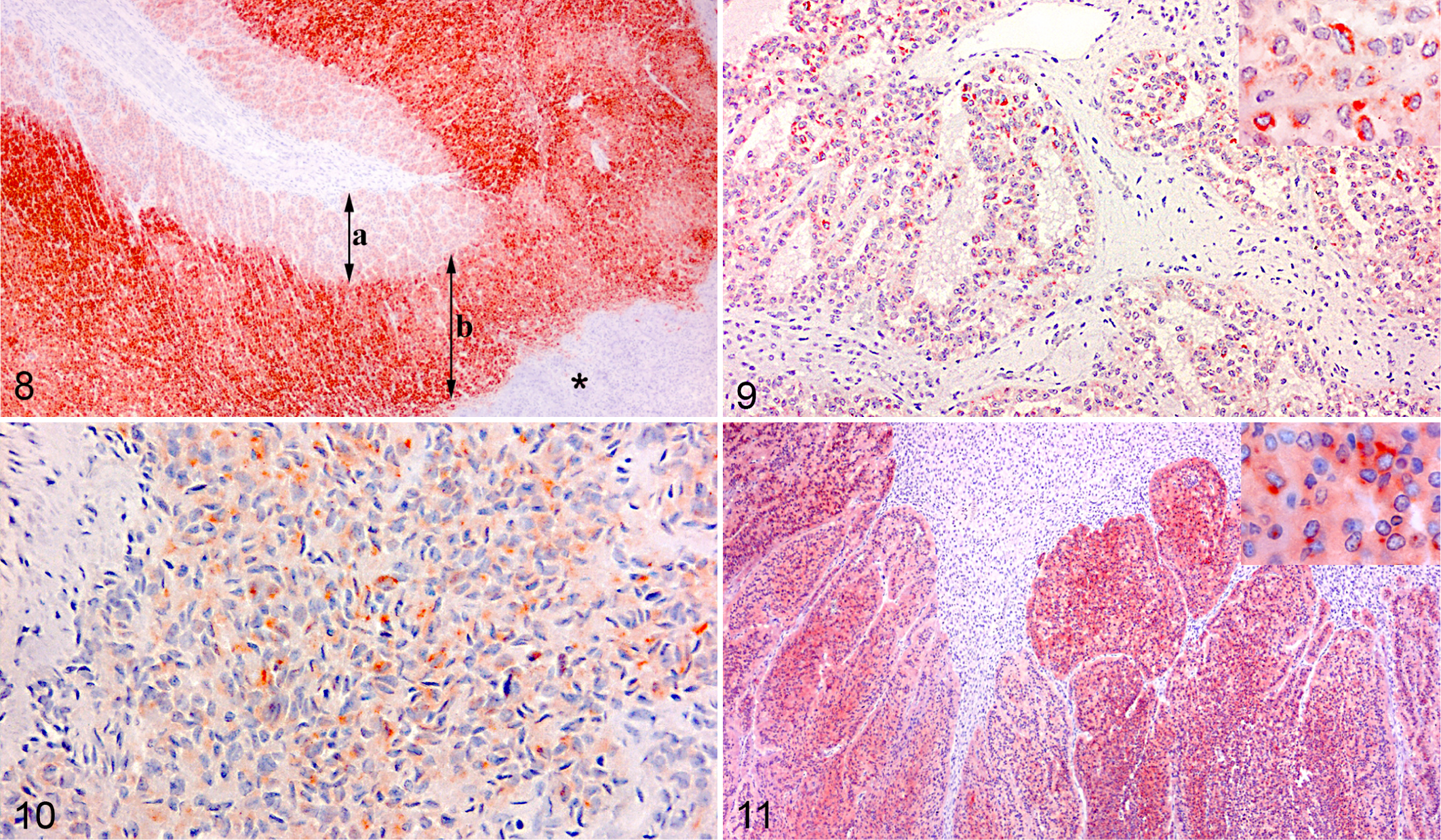
In both adenomas and carcinomas, the intensity of melan A immunoreactivity in the neoplastic cells varied from strong to weak or negative (Fig. 9). In general, the predominant labeling intensity of the neoplastic cells was weak (score 1) compared with that in cortical cells in the rest of the adrenal gland. Only 4 of 8 adenomas and 3 of 11 carcinomas had moderate to intense immunoreactivity (score 2 or 3) in more than 50% of the neoplastic cells (Table 3).
Immunohistochemistry Results for Selected Bovine Adrenal Tumors.

aThe labeling for all adrenocortical tumors was heterogeneous, with some areas strongly positive and others not stained at all. In most cortical tumors, the predominant labeling intensity was weak compared with cortical cells in the rest of the adrenal gland.
bLess than 10% of the cells were strongly positive.
NT, not tested.
There was no difference in the melan A immunolabeling among areas with spindle-cell morphology (Fig. 10), myxoid differentiation (Fig. 9), or cells arranged in diffuse or trabecular patterns. The melan A labeling intensities of adrenal gland tissue fixed in formalin for 30 hours, 5 days, and 11 days, respectively, were comparable. In contrast, the time between slaughter and initiation of fixation influenced the immunohistochemical results, as tumors fixed at the abattoirs had more intense labeling for melan A than those that were fixed 1 or 2 days later.
In the normal bovine adrenal gland, immunolabeling for synaptophysin was confined to the cytoplasm of the medullary cells. Moreover, the immunoreactivity of the outer zone with epinephrine-producing cells was consistently more intense compared with the inner zone of the adrenal medulla. Both pheochromocytomas had positive labeling for synaptophysin (intensity score 1–2 in more than 50% of the cells; Fig. 11), and the larger cells lining the sinusoids tended to react more intensely than the rest of the neoplastic cells. Adrenocortical tumors and schwannomas were negative for synaptophysin.
The cytoplasmic immunolabeling for chromogranin A was in the medullary cells of the normal adrenal gland, and as for synaptophysin, the epinephrine-producing cells had the most intense immunolabeling. Weak diffuse immunolabeling for chromogranin A was, however, also seen in the cortex, especially the zona glomerulosa. The immunolabeling for chromogranin A in the pheochromocytomas was similar to that for synaptophysin. Of 27 adrenocortical tumors, 2% to 10% of the neoplastic cells in 9 tumors labeled intensely positive for chromogranin A (Table 3).
Zona fasciculata and zona reticularis of the normal adrenal gland were strongly positive for vimentin (intensity score 3), whereas the labeling intensity of zona glomerulosa was weak (intensity score 1) or absent. The immunolabeling of the nodules of cortical cells, which often protruded into the medulla of the normal adrenal gland, was similar to the immunolabeling of the zona fasciculata. Nine of 15 adenomas and 5 of 11 carcinomas had multiple areas of intensely positive tumor cells (grade 3), which constituted 5% to 50% of the tumor sections. Positive immunolabeling for vimentin was seen in 3 of 3 schwannomas (intensity score 3 in >50% of the neoplastic cells) and in the benign pheochromocytoma (intensity score 3 in 15% of the neoplastic cells).
Small nests of elongated cells with large oval vesicular nuclei, which were interpreted as perineural cells, in the capsule and in relation to nerves in the medulla of the normal adrenal gland labeled positive for cytokeratin, and 8 of 15 adenomas and 3 of 11 carcinomas also labeled positive (Table 3). The cytokeratin immunolabeling was multifocal, was strongly positive (score 3), and seen in 5% to 50% of the neoplastic cells. Schwannomas and pheochromocytomas were negative for cytokeratin.
Only schwannomas were labeled with antibody to CNPase. Positive labeling was seen in all 3 schwannomas with an intensity score of 2 in >50% of the neoplastic cells.
Discussion
Twelve of 41 adrenal tumors (29%) in this study were classified as carcinomas, which is a much higher percentage than in other studies. 23,33,39 Because invasive growth and cellular pleomorphism were also criteria for classifying adrenocortical tumors as carcinomas in the other studies, the reason for the discrepancy might be that the adrenal tumors in the current study were all incidental findings at the abattoirs. The adrenal gland is not routinely incised during meat inspection, so small adrenocortical adenomas and peripheral nerve sheath tumors (PNSTs) might have been overlooked. In fact, none of the tumors submitted from abattoirs and examined in this study had a diameter less than 3 cm, which contrasts with the study by Wright and Conner, 39 who reported 64 of 146 adrenocortical adenomas and all PNSTs with a diameter less than 3 cm.
In contrast to other species, 17,28 carcinomas with distant metastasis have not been described in cattle. 23,33,39 However, in this study, 7 of 12 adrenal carcinomas metastasized. In other surveys, metastases could have been overlooked because organs such as the liver and lungs were not routinely incised at meat inspection. 39
It is difficult to distinguish adrenocortical carcinomas that have not metastasized from adenomas. 17,18,23 Tumor size has been proposed as indicative of malignancy in dogs and humans, 17,18 but no such correlation was apparent in the present study. Likewise, the Ki-67 proliferation index, used as an indicator of malignancy in dogs and humans, 17,18 was not higher in carcinomas than in adenomas. In humans, a set of histological guidelines known as the Weiss system has been used to differentiate benign adrenocortical tumors from tumors with malignant potential. 18 Likewise, several histomorphologic criteria were more common in canine adrenocortical carcinomas than in adrenocortical adenomas. 17 In this bovine study, 5 histologic features were significantly more common in adrenocortical tumors with distant metastasis. Therefore, in accordance with the Weiss system, 5 nonmetastasizing adrenocortical tumors that expressed at least 3 of these histologic features were classified as adrenocortical carcinomas. Vascular invasion was seen in 4 adrenocortical adenomas, and vascular invasion by cortical or medullary cells has also been described as common in the normal bovine adrenal gland. 14 This criterion alone was, therefore, not considered sufficient for classification as malignant. Likewise, the presence of fewer than 3 of the criteria was considered inadequate for the diagnosis of carcinoma because none of the metastasizing tumors had fewer than 3 of these histologic features. Other morphologic criteria evaluated in this study, such as necrosis, hemorrhage, or a high mitotic index, which in other tumor types are indicative of malignancy, were not useful to distinguish bovine adrenocortical carcinomas from adenomas because they were observed in both adenomas and metastasizing carcinomas.
The ability to identify those adrenal tumors with malignant potential is important to understand the pathogenesis of adrenal neoplasia and to compare adrenal tumors in cattle with those in humans and other animal species. One of the histologic features seen more often in metastasizing adrenocortical carcinomas than in adenomas was spindle-cell morphology. The presence of a spindle-cell component in adrenocortical carcinomas has also been described in goats 1 and ferrets, and in ferrets, the spindle cells are thought either to arise from smooth muscle cells in the adrenal capsule or to be adrenocortical cells with a different morphology. 3,9,28 The spindle cells of adrenocortical carcinomas in this study were labeled with immunohistochemistry for melan A and are, therefore, likely to be derived from adrenocortical cells. When the spindle-cell component is prominent, the tumors resemble PNSTs, which have been reported to occur frequently in bovine adrenal glands. 39 Antibodies for CNPase were used to differentiate spindle-cell adrenocortical carcinomas from schwannomas, as this antigen is a sensitive marker for neoplastic Schwann cells in bovine PNSTs. 25
Adrenocortical carcinomas with myxoid differentiation have been described in ferrets. 28 In the bovine adrenocortical tumors, a similar pattern was seen, but unlike in ferrets, myxoid differentiation was detected in both adenomas and carcinomas.
Because 84% of the adrenocortical tumors and none of the medullary tumors expressed melan A, this marker, as in humans and dogs, 5,29 can be useful to distinguish between bovine cortical and medullary adrenal tumors. In most cases, labeling intensity varied among different areas of the tumor, and it was often weaker than that of the normal cortical cells. In the normal adrenal gland, melan A labeling intensity was weaker in the zona glomerulosa than in either the zona fasciculata or reticularis. A similar heterogeneous labeling pattern has been reported in dogs. 29 The weak labeling could be caused in part by the delayed fixation of the tumors. On the other hand, the cortical cells, especially those in the zona fasciculata, had moderate to strong immunoreactivity, independent of the time of fixation. Therefore, weak labeling of adrenal cortical tumors could indicate origin from cells of the zona glomerulosa. This was corroborated by the presence of chromogranin A–positive cells in many tumors. In dogs and humans, chromogranin A is specific for adrenal medullary cells, and cortical cells are consistently negative. 5,6,21,29 In the normal bovine adrenal gland, however, cortical cells are reported to label weakly with antibodies for chromogranin A, 14 which was confirmed in the present study.
The weak melan A immunoreactivity and the presence of chromogranin A–positive cells in the tumors could also indicate that the cell of origin was medullary and not cortical cells. Ultrastructural examination of neoplastic cells in 2 of the tumors that expressed both melan A and chromogranin A, however, showed that these cells were adrenocortical cells.
It has also been suggested that bovine adrenocortical tumors derive from nodules of cortical cells in the medulla. 39 But in the medulla of adrenal glands examined in this study, these cells had a morphology and immunohistochemical staining properties that resembled the cells of the zona fasciculata. The cells of the zona fasciculata, in contrast to those of the zona glomerulosa, typically have strong immunoreactivity for vimentin and melan A, which was a feature of only a few areas in adrenocortical tumors in this study.
Both synaptophysin and chromogranin A labeling were weaker in pheochromocytomas than in normal bovine adrenal medullary cells, but immunolabeling for synaptophysin was consistently negative in adrenocortical tumors. Therefore, synaptophysin should be evaluated as a potentially more specific marker of bovine adrenal medullary tumors.
In conclusion, the most common bovine adrenal tumors were adrenocortical adenomas and carcinomas, in which the features of invasion of vessels and capsule, nuclear pleomorphism, diffuse growth pattern, and spindle-cell morphology were indicators of malignancy. An immunohistochemical panel of antibodies against melan A, synaptophysin, and CNPase was found to be useful to classify the more common adrenal tumors in cattle.
Footnotes
Acknowledgement
We are grateful to the staff at Danish Crown, Tønder, Denmark, and E. Munch at Aarhus Slagtehus A/S, Aarhus, Denmark, for help in collecting samples. We thank Lisbet Kiørboe, Dennis Brok, Department of Veterinary Disease Biology, Section of Pathology, Faculty of Health and Medical Sciences, University of Copenhagen, Denmark, and Nina Broholm, Department of Pathology, Rigshospitalet, Copenhagen University Hospital, Denmark, for technical assistance. A special thanks to Prof. Björn Rozell, Faculty of Health and Medical Sciences, University of Copenhagen, for critically reviewing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
