Abstract
We describe gross pathology, histopathology, ultrastructure, and molecular characterization of skunk adenovirus 1 (SkAdV-1) in 3 ferrets (Mustela putorius) submitted to the Infectious Diseases Laboratory, Zoo and Exotic Animal Pathology Service at the University of Georgia. Case 1 came from a cohort with a 6-week history of multiple ferrets with respiratory disease and mortality. Cases 2 and 3 were submitted 19 days later; they died 3 days after arrival at the vendor’s facility. Histopathological evaluation found evidence of suppurative bronchopneumonia, with intralesional gram-positive cocci in case 1. The others had evidence of subacute multifocal histiocytic and lymphoplasmacytic interstitial pneumonia. Viral isolation, polymerase chain reaction (PCR), and DNA sequencing identified SkAdV-1 in lung tissue. In situ hybridization confirmed adenovirus within the lung lesion, and adenovirus particles were visible under an electron microscope. This is the seventh species in which SkAdV-1 has been identified. Cross-species transmission events have important implications for wildlife management and multispecies facilities.
Adenoviruses (AdVs) are icosahedral, 70 to 90 nm diameter, nonenveloped, double-stranded DNA viruses that infect almost all known classes of vertebrates.12,15 Adenoviridae is organized into 6 genera: Mastadenovirus, Aviadenovirus, Ichtadenovirus, Atadenovirus, Siadenovirus, and Testadenovirus. Adenoviruses from Mastadenoviruses are commonly reported in mammals. Aviadenoviruses are usually found in birds. Atadenoviruses are found in birds, squamates, and ruminants. 12 Siadenoviruses are found in birds, tortoises, and a single frog. Testadenoviruses7,15 are found in tortoises (Testudinidae). Ichtadenoviruses contain the only known AdV with a fish host, sturgeon ichtadenovirus A. Adenoviruses, especially in humans, are typically associated with respiratory disease. They may also be associated with hepatitis, enteritis, pancreatitis, encephalitis, vasculitis, and conjunctivitis. 15
There is evidence of cross-species and zoonotic transmission of AdVs. 2 A recent study screened 327 published manuscripts for relevance to zoonotic and anthroponotic transmission potential and identified 74 for review; of 24 deemed relevant, 16 demonstrated evidence of zoonotic transmission of AdVs. 3 Six studies (25%) had evidence of AdVs transmitted by humans to nonhuman primates; 5 (21%) had evidence of AdVs transmitted by nonhuman primates to humans, including an outbreak of a novel AdV in a colony of titi monkeys (Callicebinae) which was transmitted to a researcher and 2 relatives. 5 Adenoviruses may cross species barriers, even among distantly related species. For instance, canine adenovirus 1 (CAdV-1) has caused disease in 3 mammalian families including foxes, wolves, and coyotes (Canidae); skunks (Mephitidae); and bears (Ursidae). 12
Skunk adenovirus 1 (SkAdV-1), of the Mastadenovirus genus, was first isolated in North America from a single skunk (Mephitis mephitis) in 2015. Initial sequencing of the virus revealed a nucleotide similarity of 64% with bat adenovirus 2 (BtAdV-2) and 64.1% with CAdV-1, but the virus was determined to be phylogenetically distinct and named as a new species, skunk mastadenovirus A (SkAdV-A). 14 Skunk adenovirus 1 has been documented in 6 mammalian families: Callitrichidae, Mephitidae, Erinaceidae, Erethizontidae, Canidae, and Procyonidae.4,10,14,17
In early 2019, the Zoo and Exotic Animal Pathology Service, at the University of Georgia Infectious Diseases Laboratory, received whole bodies of 3 male ferrets from a regional pet vendor for complete necropsy and histological examination. Case 1 was from a cohort with a 6-week history of multiple ferrets with respiratory disease and mortality; it was euthanized. Cases 2 and 3, received 19 days later, were from a group of 4 ferrets that died 3 days after arriving at the facility. Samples were collected from all individuals. Lung tissue was subdivided for influenza A polymerase chain reaction (PCR), aerobic bacterial culture, viral isolation, Streptococcus PCR, and AdV PCR. Additional samples collected included oropharyngeal and bronchial swabs and tissue from spleen, liver, kidney, lymph node, brain, large and small intestines, heart, trachea, thyroid, esophagus, stomach, thymus, adrenal gland, skeletal muscle, urinary bladder, adipose tissue, bone marrow, pancreas, salivary gland, tongue, and eye. Samples were frozen at −20°C or immersed in 10% neutral-buffered formalin, processed routinely for histopathology, and stained with hematoxylin and eosin for examination by light microscopy.
For viral isolation, a lung tissue homogenate was filtered through a 0.45 µm syringe filter onto a monolayer of Madin-Darby canine kidney (MDCK) cells grown in media supplemented with antibiotics. Cytopathic effect was observed on the MDCK cells 5 days after inoculation. The cell culture preparation was used for electron microscopy (EM) (UGA Georgia Electron Microscopy Core Facility). On EM, viral particles were observed in a purified cell culture from case 1, using the direct negative stain protocol. 13 A boiled supernatant of the cell culture from each of the 3 ferrets was amplified by conventional PCR using previously published protocols and AdV primers designed to amplify the adenoviral DNA-dependent DNA polymerase gene. 22 Concomitant with viral isolation and PCR, metagenomic, indexed, Illumina sequencing libraries 11 were prepared from lung tissue DNA of the 3 individuals and sequenced as a small portion of a paired-end 250 base run on an Illumina MiSeq Desktop Sequencer. For whole genome viral sequencing, lung tissue homogenate infected MDCK cells that were exhibiting cytopathic effect were frozen, thawed, and transferred to a 15-ml centrifuge tube and centrifuged at 500 × g to remove cell debris. A 1-ml portion of the supernatant was transferred to a 1.5-ml centrifuge tube and centrifuged at 21,000 × g for 5 minutes. The supernatant was removed, and the viral pellet was input into the QIAGEN DNeasy® tissue standard protocol (QIAGEN, LLC). About 200 ng of DNA was used to prepare Illumina sequencing libraries in the same manner as the metagenomic libraries. Sequencing reads were quality filtered and trimmed using fastp and randomly down-sampled from 11,000× to 235× using bbmap and assembled using SPADES v. 3.13.0. 20 and Geneious. For multiple sequence alignment, translated sequences were extracted from GenBank records aligned using Muscle v3.8.425 8 and edited/exported in Jalview. 21
Infectious Diseases Laboratory (IDL) conducted PCR testing 22 on lung tissue from case 1 for Streptococcus 16 and canine distemper virus (reference for primers 9 ) using PCR in combination with Förster resonance energy transfer, also known as fluorescence resonance energy transfer, or FRET, in which 2 fluorophores are brought in close proximity after hybridization (FRET probes are proprietary to IDL). The Athens Veterinary Diagnostic Laboratory (AVDL) (UGA College of Veterinary Medicine) used lung tissue from case 1 to conduct PCR for influenza A, and for aerobic bacterial culture. The AVDL conducted fluorescent antibody tests on cases 2 and 3 to rule out canine distemper virus (brain tissue), and coronavirus and parvovirus (small intestinal tissue).
Upon presentation, body condition scores ranged from 2/5 (n = 1) to 1/5 (n = 2). 1 Lung lobes of all 3 ferrets had multiple dark red foci affecting approximately 80% of the organ (Fig. 1a). Other observations included scant thick white exudate upon disarticulation of the atlanto-occipital joint and thickened leptomeninges, in case 1; bilaterally sunken eyes and left unilateral retropharyngeal lymphadenomegaly, in case 2; and splenic pallor and a shrunken thymus, in case 3.
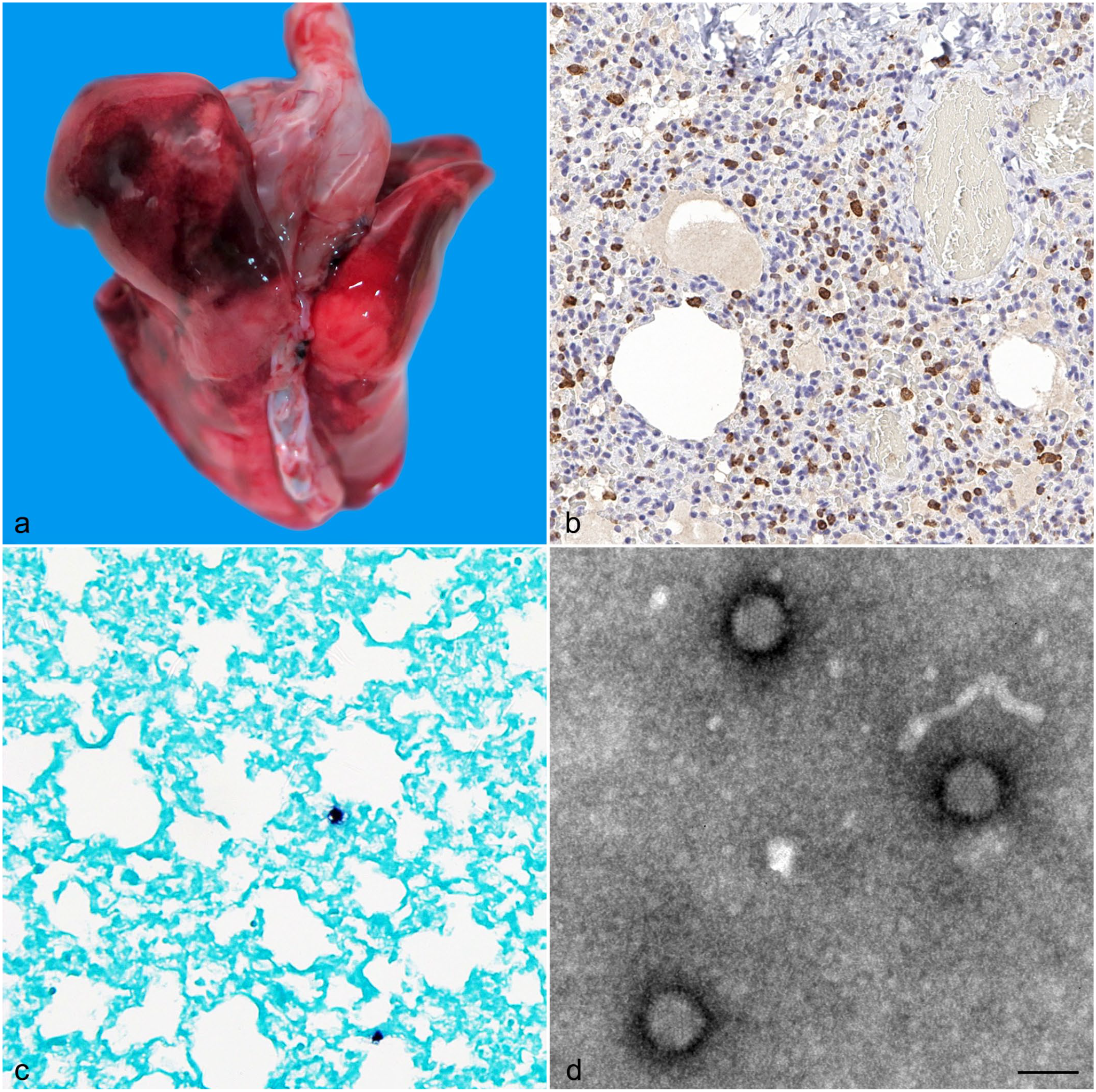
Skunk adenovirus 1 infection in domestic ferrets. (a) Multifocal to coalescing, red, irregular foci in the lungs of ferret 1. (b) Numerous cells within the interstitium had cytoplasmic immunoreactivity to IBA1 consistent with macrophage origin in the lung of ferret 2. (c) In situ hybridization detects adenovirus (dark brown reaction) in the lung lesions of ferret 1. (d) Electron microscopy of adenoviral particles showing the individual capsomers on the surface of these particles. Negative contrast using phosphotungstate. Bar = 100 nm.
We used IBA1, CD79a, and CD3 immunohistochemistry to characterize inflammation in the lung from macrophages, B-cells, and T-cells, respectively. Scattered cells were immunoreactive to CD79a (Supplemental Figure S1). Multiple cells were immunoreactive to CD3 and numerous cells had cytoplasmic immunoreactivity to IBA1 (Fig. 1b). Histologic examination of case 1 found evidence of suppurative bronchopneumonia, lymphoplasmacytic myocarditis, and suppurative meningitis. One-third of the lung sections contained viable and degenerate neutrophils with foamy macrophages, lymphocytes, fibrin, hemorrhage, and occasional eosinophils. Half of the bronchioles and few bronchi contained intraluminal infiltration with the previously described inflammatory population with the addition of rare gram-positive cocci (Supplemental Figure S2). Rare alveolar emphysema and overdistension were observed while the vasculature was diffusely congested. The cerebrum and cerebellum, concentrated mostly in the ventral meninges, had infiltration of viable and degenerate neutrophils, with fewer foamy macrophages, lymphocytes, fibrin, extravasated erythrocytes, and rare eosinophils. Meningeal vessels were congested. We observed gram-positive cocci similar to those in lung tissue. Brain tissue was not cultured, but it seems probable the organism could have crossed the blood-brain barrier. The heart was infiltrated with lymphocytes, and there was evidence of cardiomyocyte degeneration. Aerobic bacterial culture of lung tissue from case 1 resulted in heavy growth of Streptococcus equi subsp. zooepidemicus, which was validated by PCR. Virus isolation from case 1 was positive for AdV, which was confirmed by PCR/sequencing as SkAdV-1. Case 1 was negative for influenza A by PCR.
Histological examination of cases 2 and 3 revealed histiocytic and lymphoplasmacytic interstitial pneumonia, diffuse hepatocellular atrophy, and diffusely decreased zymogen granules within the acinar cells of the pancreas. The changes in the liver and pancreas are supportive of a negative energy balance. The lungs contained interstitial infiltrates composed of macrophages, lymphocytes, plasma cells, rare neutrophils, and necrotic cell debris (Supplemental Figure S3); alveoli had poorly defined septa with collapse (atelectasis). Lymph nodes contained tingible body macrophages and occasional erythrophagocytosis. The thymus exhibited lymphoid depletion and prominent tingible body macrophages. The stomach contained rare gland abscesses with mucosal glands being distended and containing eosinophilic cellular debris. Fluorescent antibody testing for canine distemper virus (brain tissue), canine coronavirus (small intestinal tissue), and canine parvovirus (small intestinal tissue) were negative for both animals. Cases 2 and 3 were positive for SkAdV-1 by PCR and sequencing. Cases 2 and 3 were not tested for influenza and lung tissue was not cultured. In situ hybridization was performed on lungs from cases 1, 2, and 3, confirming the presence of viral DNA of AdV within the pulmonary lesions (Fig. 1c) (see Supplemental Materials and Methods for full description and references).
Cytopathic effects were observed 5 days after inoculation of lung tissue of case 1 on MDCK cells. Electron microscopy (negative contrast) revealed viral particles which were morphologically consistent with AdV (Fig. 1d). The virions were nonenveloped, 76 to 82 nm in diameter. The icosahedral capsid consists of well-defined capsomers; the characteristic triangle profile was seen in some of the virions by negative contrast.
The amplified 406 bp DNA-dependent DNA polymerase sequence had a 99.75% identity to the base pairs from 6104 to 5699 of the complete genome of skunk AdV PB1 (KP238322.1) with a single nonsynonymous transversion substitution G→T at position 5749, changing a glutamine to a lysine. Metagenomic sequencing from lung samples from the 3 individuals each yielded SkAdV-1 sequence data and produced an additional ~1500 bp of paired sequence alignments dispersed throughout the skunk AdV genome that matched the PB1 regions with 100% identity. The genomic assembly from the infected cell culture DNA resulted in a viral genome of 31,754 bp (GenBank accession number: MZ182243) which had a 99.60%, 99.65%, and 99.74% nucleotide identity to the complete genomes of skunk AdV PB1, African pygmy hedgehog AdV 1 isolate HO-2018 (MK937781.1), and skunk AdV HUN/2009 (MN482116.1), respectively. As with the HUN/2009 skunk AdV recovered from a pygmy marmoset, 6 the genomic region with the most diversity was in the E3 ORFA gene and resulted primarily from a 50 bp deletion in the 3′ portion of the gene resulting in a frame change relative to the 3 other E3 ORFA genes and a divergent caboxyl-terminal part of the protein (Supplemental Figure S4). 19
The most significant lesions associated with the SkAdV-1 infections involved the respiratory tract, consistent with commonly reported lesions in other Mastadenovirus infections, including SkAdV-1.4,10,12,14,15,17,18 Viral inclusion bodies were not observed histologically in these ferrets. Case 1 had lymphoplasmacytic myocarditis and suppurative meningitis, both of which may have been caused by the Streptococcus equi subsp. zooepidemicus infection. 16 Case 1 also had a bacterial pneumonia, presumed secondary to immunosuppression or viral infection. A recent study documented 24 cases of SkAdV-1 (2015–2020) in multiple free-ranging species from North America. Necrotizing bronchopneumonia was the most common lesion, involving 22 individuals; 10 had no interstitial involvement. Secondary bacterial infections were diagnosed in 4 individuals, including 1 case of S. zooepidemicus. 4
An underlying, predisposing immunosuppressive condition cannot be ruled out for Case 1 (Supplemental Figure S2). Potentially, multiple contributing stressors could affect immunosuppression in these cases. All ferrets were emaciated. Cases 2 and 3 were recently acquired and stress associated with shipping may have led to transient immunosuppression. We do not know their ages, medical histories, nor whether they were exposed to other species. Per the vendor, the new arrivals were housed separately from the earlier cohort.
The pathogenesis and disease progression of SkAdV-1 is currently unknown, as to its role in clinical disease and whether it affects healthy individuals or only immunocompromised individuals. Skunk adenovirus 1’s prevalence, impact, geographic distribution, host range, and evolutionary origin in wildlife populations are also unknown. Over a decade, SkAdV-1 has demonstrated a wide geographic distribution and diverse, distantly related host range.4,10,14,17 Our study documents SkAdV-1 in a new species in North America.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231189722 – Supplemental material for Detection of skunk adenovirus-1 in domestic ferrets (Mustela putorius)
Supplemental material, sj-pdf-1-vet-10.1177_03009858231189722 for Detection of skunk adenovirus-1 in domestic ferrets (Mustela putorius) by Maria E. Orbay-Cerrato, Roger Alan Nilsen, Nicole Gottdenker, Rita McManamon, Jessica A. Elbert, Justin M. Stilwell, Mary B. Ard, Branson W. Ritchie and Brittany McHale in Veterinary Pathology
Footnotes
Acknowledgements
These findings were presented by Dr. Brittany McHale at the 2019 American Association of Zoo Veterinarians 51st Annual Conference in St. Louis, Missouri, United States; and by Maria E. Orbay-Cerrato in October 2020 at the Science of Veterinary Medicine Symposium, held at the UGA College of Veterinary Medicine in Athens, Georgia, United States. Ms. Orbay-Cerrato is a member of the UGA CVM’s DVM Class of 2022. Editing of this manuscript was conducted and coordinated by Kat Yancey Gilmore, senior science writer and editor for the UGA Infectious Diseases Laboratory, the UGA Emerging Diseases Research Group and the UGA New Materials Institute.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Diagnostic testing was paid for by the client of the UGA Infectious Diseases Laboratory. No other funding sources were utilized for this study.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
