Abstract
Cyclooxygenase-2 (COX-2) and nitric oxide synthase 2 (NOS2) were detected and localized in 20 pigs with ulcerative colitis caused by natural infection with Salmonella typhimurium. Evidence of NOS2 activity was determined by the formation of nitrotyrosine, a reaction product of peroxynitrite, in NOS2-expressing ulcerative colons by immunohistochemistry. Transcript RNA of COX-2 and NOS2 was consistently detected in colonic tissues from the 20 pigs with ulcerative colitis by using reverse transcription-polymerase chain reaction. Immunohistochemical signals for COX-2 and NOS2 were detected in the ulcerated area of all 20 pigs. Expression of COX-2 and NOS2 was identified continuously within inflammatory intestinal lesions but was minimal in unaffected regions of the colon of S. typhimurium—infected pigs. The immunohistochemistry of serial sections of intestine indicated that the majority of colons containing numerous COX-2—positive cells also had numerous NOS2-positive cells. Localization of NOS2 and a nitrotyrosine antigen was prominent in neutrophils and macrophages in the periphery of the lesions. Simultaneous detection of COX-2 and NOS2 RNA and protein indicated functional activity of prostaglandin and NO production in vivo. This study suggested that COX-2 and NOS2 expression may play a role in the pathophysiologic processes in ulcerative colitis caused by S. typhimurium.
Keywords
Salmonella is important both as a pathogen of swine and as a zoonotic food-borne pathogen of humans. 11,39 The two major forms of clinical salmonellosis in swine include septicemia and enterocolitis. Septicemia is usually caused by S. choleraesuis. In contrast, enterocolitis is most commonly caused by S. typhimurium, although it has also been observed in pigs infected with S. typhisuis, S. agona, and S. choleraesuis. Enterocolitic salmonellosis most frequently occurs in pigs from weaning to about 4 months of age. 39 Histopathologic changes associated with enterocolitic samonellosis are characterized by diffuse catarrhal colitis with deep focal hemorrhage and shallow erosions covered with a fibrinonecrotic membrane in the colon and rectum. More severe lesions consist of deep mucosal ulcers, which may be focal or coalescing. 33,45
Cyclooxygenase (COX), a key enzyme required for the synthesis of prostaglandin (PG), has two distinct isoforms: the constitutive enzyme designated as COX-1 and the inducible isoform known as COX-2. 12 COX-1 appears to be responsible for the production of PGs that are physiologically important for homeostatic functions, such as the maintenance of mucosal integrity and mucosal blood flow. 44 In contrast, COX-2 is not constitutively expressed in most tissues but is dramatically upregulated during intestinal inflammation. 13
Nitric oxide synthase (NOS) is an enzyme that catalyzes the synthesis of NO from the terminal guanidinonitrogen of
COX-2 and NOS2 are present after the stimulation of host cells with bacteria or bacterial products such as lipopolysaccharide (LPS) or inflammatory cytokines such as tumor necrosis factor-α (TNF-α) and interleukin-1 (IL-1). 34,36,40 LPS is an essential component of the outer membrane of Salmonella and a major determinant of its virulence. 10 TNF-α is produced mainly by monocytes/macrophages, in response to S. typhimurium infection. 42,43 The objective of this study was to detect and localize COX-2, NOS2, and nitrotyrosine by immunohistochemistry in pigs with ulcerative colitis due to natural infection with S. typhimurium.
Samples were obtained at necropsy from pigs submitted to the Department of Veterinary Pathology of Seoul National University from January 2000 to December 2002. Twenty pigs (Nos. 1–20) approximately 80–160 days of age, from 20 different herds, were selected on the basis of clinical signs, characteristic lesions, bacteria isolation, and serotype. All 20 pigs were negative for Lawsonia intracellularis, Brachyspira hyodysenteriae, and B. pilosicoli by polymerase chain reaction (PCR). 9,20 Positive control sections were from pigs that had been naturally infected with Actinobacillus pleuropneumoniae. 4–8 Negative control sections were prepared from a 1-day-old colostrum-deprived pig that had not been exposed to any viral and bacterial pathogens and conventional 60 and 80-day-old pigs that were negative for bacteria isolation.
The reverse transcription–PCR of COX-2 and NOS2 was carried out as previously described. 4,8 RNA was extracted from colons with Trizol LS reagent (GIBCO BRL, Grand Island, NY) according to the manufacturer's instructions. For COX-2 the forward and reverse primers were 5′-GGAGAGACAGCATAAACTGC-3′ (nucleotides 413–432) and 5′-GTGTGTTAAACTCAGCAGCA-3′ (nucleotides 830–849), respectively. The primers amplified a 437–base pair (bp) complementary DNA (cDNA) fragment. For NOS2 the forward and reverse primers were 5′-CGTTATGCCACCAACAAT-3′ and 5′-ACTTCCTCCAGGATGTTGTA-3′, respectively. The primers amplified a 491-bp cDNA fragment. Positive control of COX-2 and NOS2 was prepared as previously described. 4,7
Immunohistochemical examination for COX-2, NOS2, and nitrotyrosine was carried out as previously described. 5,6,8 Three antibodies were used: 1) polyclonal rabbit anti-mouse COX-2 antibody (Cayman Chemical, Ann Arbor, MI), 2) polyclonal rabbit anti-NOS2 antibody (Biotechnology Inc., Santa Cruz, CA), and 3) monoclonal mouse anti-nitrotyrosine antibody (Zymed Laboratories Inc., San Francisco, CA). Lung tissues from pigs naturally infected with A. pleuropneumoniae was used as a positive control of COX-2, NOS2, and nitrotyrosine. 5,6,8
Immunohistochemical signal of COX-2 and NOS2 was given a ranked score of 0–3 by a simple subjective system: 0 = no immunohistochemical signal, 1 = minimal immunohistochemical signal, and 2 = intense immunohistochemical signal. Scores of immunohistochemical signals were subjected to Pearson correlation analysis to assess relationships. P < 0.05 was considered to be statistically significant.
In the colon, necrosis frequently extended to involve muscularis mucosa, submucosa, and lymphoid follicles. Necrotic tissue was separated from the healthy mucosa by a marginal zone of leukocytes, and the deeper tissues became heavily infiltrated by neutrophils. The lamina propria and submucosa contained numerous neutrophils and macrophages. Degeneration and necrosis of vascular endothelium in the intestinal serosa were observed and were often associated with vascular thrombosis.
Amplification of template cDNA with primers for COX-2 and NOS2 resulted in amplified products corresponding to those of the predicted size, namely, 437 and 491 bp, respectively. PCR products were sequenced and their identity confirmed as regions of the COX-2 and NOS2 genes. To monitor the expression of COX-2 and NOS2, PCR analyses were performed using RNA extracted from the colonic tissues of pigs that had been infected with S. typhimurium. COX-2 and NOS2 transcripts were consistently detected in the colonic tissues from all 20 pigs with S. typhimurium–induced ulcerative colitis (Figs. 1, 2). In contrast, COX-2 and NOS2 messenger RNAs (mRNAs) were not detected in colonic tissues from the negative control pigs.
Agarose gel electrophoresis of PCR-amplified COX-2 cDNA products. From left to right: M, 100-bp DNA ladder; lane 1, positive control from LPS-treated macrophages; lane 2, negative control from normal macrophages; lane 3, positive COX-2 cDNA from pig No. 1; lane 4, positive COX-2 cDNA from pig No. 5; lane 5, positive COX-2 cDNA from pig No. 12; lane 6, positive COX-2 cDNA from pig No. 15; lane 7, positive COX-2 cDNA from pig No. 20.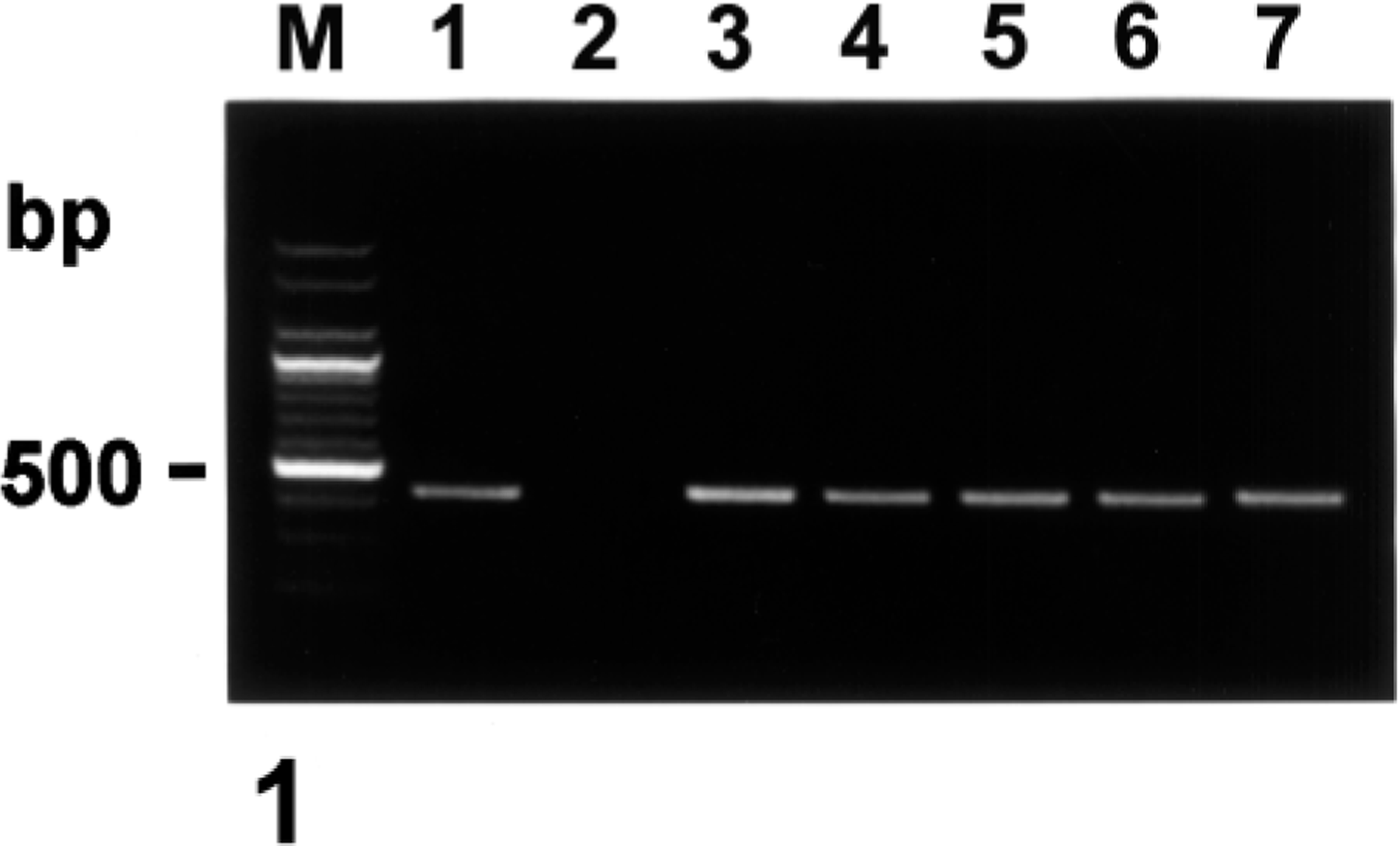
Agarose gel electrophoresis of PCR-amplified NOS2 cDNA products. From left to right: M, 100-bp DNA ladder; lane 1, positive control from LPS-treated macrophages; lane 2, negative control from normal macrophages; lane 3, positive NOS2 cDNA from pig No. 3; lane 4, positive NOS2 cDNA from pig No. 9; lane 5, positive NOS2 cDNA from pig No. 11; lane 6, positive NOS2 cDNA from pig No. 17; lane 7, positive NOS2 cDNA from pig No. 18.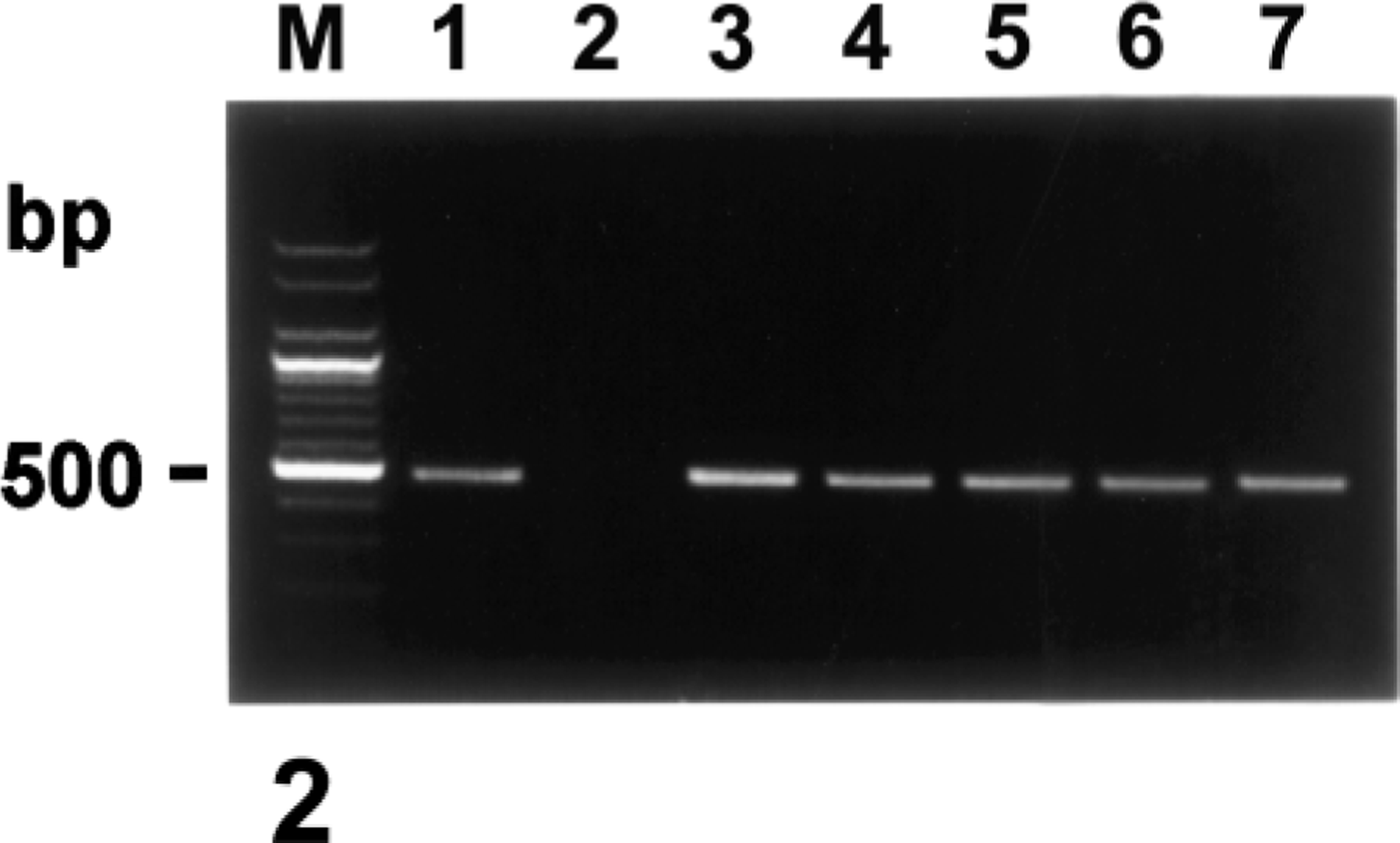
A distinct positive immunohistochemical signal for COX-2 and NOS2 was detected in all 20 pigs. The signal intensity varied within and among histologic structures in any one section and among pigs. Positive cells typically exhibited a red reaction product without background staining. The immunohistochemical signal for COX-2 and NOS2 was detected in ulcerated regions. Expression of COX-2 and NOS2 was always present within inflammatory intestinal lesions but was minimal in unaffected parts of the colon of S. typhimurium–infected pigs. Immunoreactivity of the COX-2 protein was particularly intense in the colonic epithelial cells in ulcerative tissues surrounding the dense zone of degenerative inflammatory cells (Fig. 3). Identification of cell types expressing COX-2 and NOS2 protein (Fig. 4) was occasionally difficult; however, positive cells generally had large oval nuclei and abundant cytoplasm. Comparison with hematoxylin and eosin–stained sections from the same block indicated that most of the positive cells were macrophages. The mean score of immunohistochemistry is 2.35 ± 0.75 for COX-2 and 2.2 ± 0.77 for NOS2, respectively. The score of immunohistochemical signal in COX-2 was not correlated with the score of immunohistochemical signal in NOS2 (rs = 0.1472, P > 0.05). Localization of both NOS2 and the nitrotyrosine antigen was especially prominent in neutrophils and macrophages from the periphery of the lesions (Fig. 5). The nitrotyrosine antigen was also consistently associated with macrophages and neutrophils in the ulcerative lesions but was minimal in the unaffected parts of the colon from S. typhimurium–infected pigs. Sections from positive control pigs showed strong immunohistochemical signals for COX-2, NOS2, and nitrotyrosine. Conversely, sections from negative control pigs showed no immunohistochemical signal for COX-2, NOS2, and nitrotyrosine.
Colon; pig No. 2, naturally infected with S. typhimurium. COX-2 antigen (red reaction) was detected in colonic epithelial cells in ulcerative tissues surrounding the dense zone of degenerative inflammatory cells. Immunohistochemistry; alkaline phosphatase, red substrate, hematoxylin counterstain. Bar = 30 µm.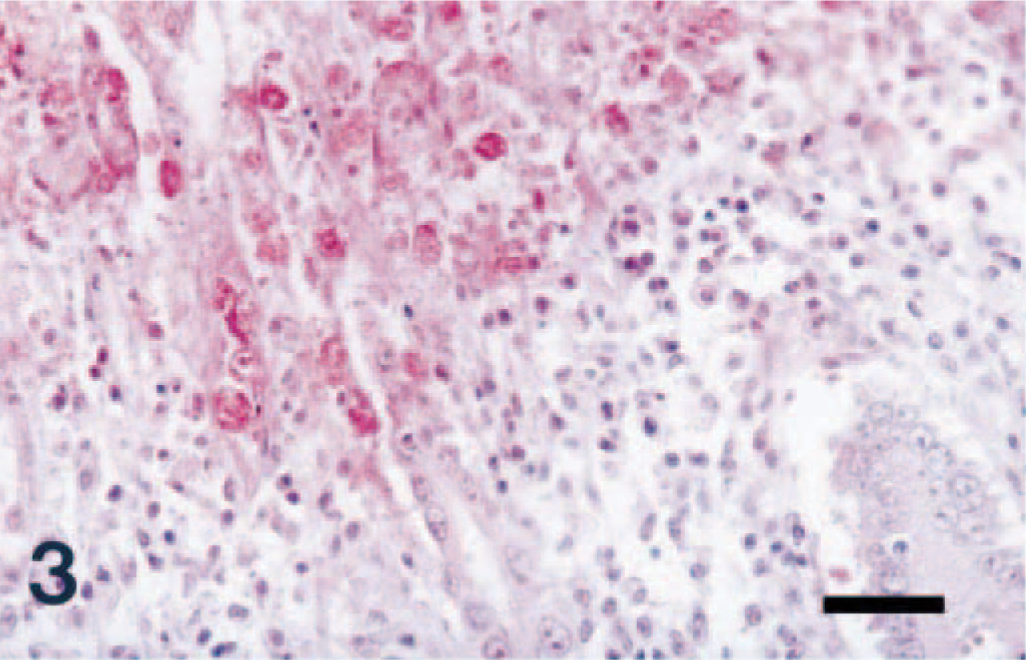
Colon; pig No. 5, naturally infected with S. typhimurium. NOS2 antigen (red reaction) was detected in inflammatory cells in the lamina propria. Immunohistochemistry; alkaline phosphatase, red substrate, hematoxylin counterstain. Bar = 30 µm.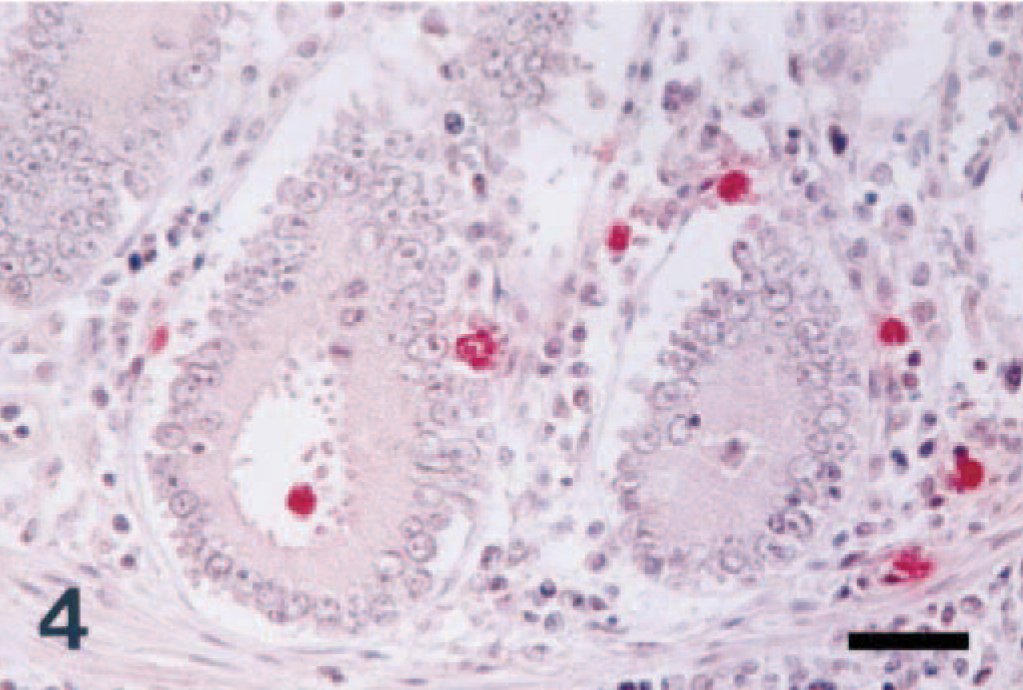
Colon; pig No. 19, naturally infected with S. typhimurium. Nitrotyrosine antigen (red reaction) was detected in inflammatory cells at the periphery of the inflammatory area. Immunohistochemistry; alkaline phosphatase, red substrate, hematoxylin counterstain. Bar = 55 µm.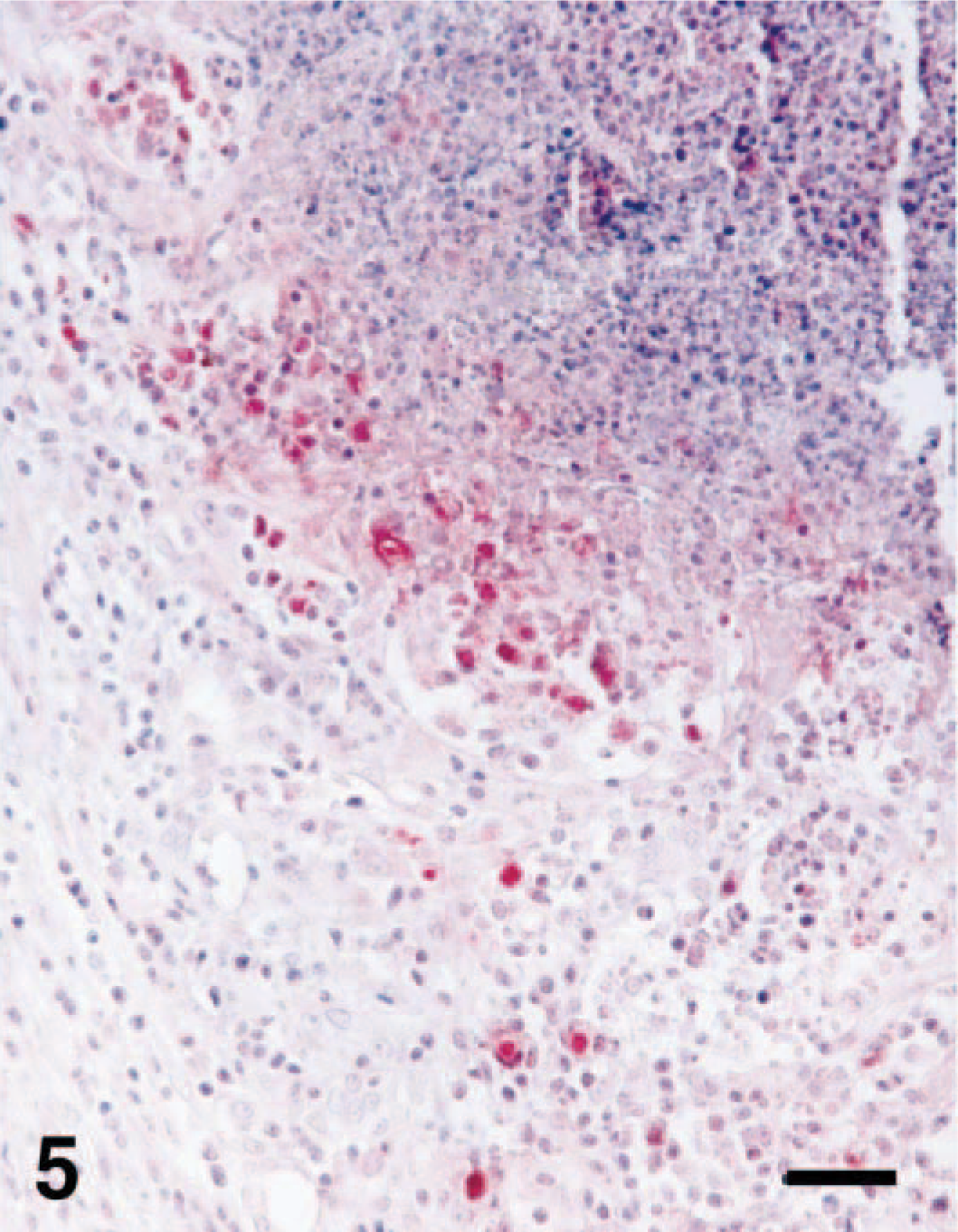
The results of this study suggest that COX-2 and NOS2 were markedly upregulated at the transcriptional and translational levels in ulcerative colitis compared with the normal colon. Intense and consistent expression of the COX-2 and NOS2 proteins was detected in colonic macrophages. Immunoreactivity of COX-2 was much more intense than that of NOS2. There have been several observations that suggest an interaction between COX-2 and NOS2. 23,37
Expression levels were significantly higher in ulcerative colitis compared with the normal colon, suggesting that induction of COX-2 and NOS2 may be a specific response to S. typhimurium. However, this association between COX-2 and NOS2 expression and histopathologic findings does not provide a cause and effect scenario. Alternatively, S. typhimurium–stimulated expression of COX-2 and NOS2 may be secondary to the effects of proinflammatory cytokines produced in response to S. typhimurium. COX-2 and NOS2 have been shown to be expressed after host cells were exposed to proinflammatory cytokines (e.g., TNF-α and IL-1). 34,36,40 Expression of TNF-α was also detected in monocytes/macrophages after exposure to S. typhimurium LPS. 42 Although the manner in which TNF-α mediates this response is not completely understood, it is likely that it acts synergistically with the S. typhimurium LPS to induce COX-2 and NOS2 expression by inflammatory cells in infected colons.
Although the detailed functions of COX-2 in the pathogenesis of ulcerative colitis due to S. typhimurium remain to be elucidated, there is evidence here supporting a protective role for COX-2 in mucosal inflammation. Immunohistochemistry showed significantly increased COX-2 expression in the ulcerative lesions, suggesting that endogenous PGs produced by COX-2 may be involved in spontaneous ulcer healing. This concept is in keeping with previous studies showing that larger amounts of PGE2 were detected at the site of ulceration than in nonulcerated musoca. 3,28 Futhermore, pretreatment with indomethacin, a nonselective inhibitor of COX, significantly suppressed PGE2 generation and markedly delayed the healing of the ulcers. 3,28
The functional role of NO produced by NOS2 owing to the pathogenesis of ulcerative colitis caused by S. typhimurium is unknown. Some evidence suggests that NO may diminish epithelial damage. NO can scavenge oxygen radicals, thus ameliorating their injurious effect.
16
Inhibition of NO synthesis promotes damage induced by HCl,
22
ethanol,
30
and LPS,
18
suggesting that NO acts to protect the gut when it is injured. In addition, perfusion of the inflamed intestine with N
G-nitro-
Inflammatory cells, such as neutrophils and macrophages, express NOS2, produce NO, and also generate superoxide. 21,29 NO reacts rapidly with superoxide to form peroxynitrite, which, in turn, reacts with proteins to produce nitrotyrosine. 1,17,24 Thus, peroxynitrite formation has been shown indirectly to be the result of the colocalization of NOS2 and nitrotyrosine immunoreactivity. 41 Peroxynitrite has bactericidal properties that may aid the killing mechanisms of macrophages and neutrophils. 2 Because the reaction of nitrite with hypochlorous acid and the reaction of myeloperoxidase with hydrogen peroxide can both lead to the formation of nitrotyrosine, 14,38 increased staining for nitrotyrosine is therefore considered to be an indication of increased nitrative stress rather than a specific marker of the generation of peroxynitrite.
Simultaneous detection of mRNA and COX-2 and NOS2 proteins indicated the functional activity of PG and NO production in vivo. Moreover, the observation that NOS2 expression in ulcerative area is associated with protein nitrosation suggests that NOS is also functionally active during infections caused by S. typhimurium. Therefore, this study concludes that COX-2 and NOS2 expression may play a role in the pathophysiologic processes in ulcerative colitis caused by S. typhimurium.
Footnotes
Acknowledgements
This research was supported by contract research funds from Research Institute for Veterinary Science (RIVS), College of Veterinary Medicine, Seoul National University, and Brain Korea 21 Project, Republic of Korea.
