Abstract
Evidence of nitric oxide synthase (NOS) 2 activity was determined by formation of nitrotyrosine (a reaction product of peroxynitrite) and by activation of poly(ADP-ribose) synthetase (PARS) in NOS2-expressed pleuropneumonic lungs from 20 pigs naturally infected with Actinobacillus pleuropneumoniae using immunohistochemistry. Intense immunostaining for nitrotyrosine residue was seen within the lung lesions from A. pleuropneumoniae-infected pigs, but it was minimal in the unaffected parts of the lung from A. pleuropneumoniae-infected pigs and in the normal lung from control pigs. Staining was especially strong in neutrophils and macrophages in the periphery of the lesions and within the alveolar spaces. There was close cell-to-cell correlation when serial sections were examined by immunohistochemistry for NOS2 and nitrotyrosine in each of the 20 lung samples. Expression of PARS was always present within inflammatory lesions but was minimal in the unaffected lung of A. pleuropneumoniae-infected pigs. Macrophages in alveolar spaces frequently exhibited strong staining for PARS. Colocalization of nitrotyrosine and PARS antigen was especially prominent in macrophages in the periphery of lesions. NOS2 expression in pleuropneumonic areas associated with protein nitrosation and PARS suggests that NOS2 is functionally active during infections caused by A. pleuropneumoniae.
Keywords
Actinobacillus pleuropneumoniae is a facultative anaerobic, nonspore-forming, encapsulated, nonmotile, and pleomorphic gram-negative coccobacillus. Infection with this bacterium is often associated with severe fibrinohemorrhagic to necrotizing pleuropneumonia in growing pigs between 2 and 6 months of age. 34 Histologically, necrotic areas in the lung are surrounded by darkly stained bands of streaming alveolar leukocytes, and similar cells often line subpleural lymphatics. 10 , 34 A. pleuropneumoniae is detected primarily within neutrophils and alveolar macrophages at the periphery of foci of acute coagulative necrosis and at the edge of granulation tissue in chronic lesions. 7
Reactive oxygen species and reactive nitrogen intermediates, such as superoxide (O2
−), nitric oxide (NO), and peroxynitrite (ONOO−), are released from activated neutrophils and macrophages during the inflammatory process.
1
,
15
Among these reactive nitrogen intermediates, NO exhibits diverse biological functions in the regulation of vascular tone, neuronal signaling, and host defense mechanisms.
24
NO is synthesized from
NO reacts rapidly with superoxide to produce peroxynitrite, which, in turn, reacts with proteins to produce nitrotyrosine. 2 , 13 , 18 Peroxynitrite is a highly oxidizing and cytotoxic molecular species when compared with NO. 13 , 18 Peroxynitrite induces cellular injury and necrosis through several mechanisms including peroxidation of membrane lipids, protein denaturation, and glutathione depletion. 8 , 26 , 28 Moreover, peroxynitrite can also cause DNA damage pleuropneumonia, 14 , 29 and this results in the activation of the nuclear enzyme poly(ADP-ribose) synthetase (PARS). Activation of PARS results in the depletion of nicotinamide adenine dinucleotide (NAD+) and adenine triphosphate (ATP) and in the death of multiple cell types, including macrophages. 35 There is evidence that the activation of PARS may also play an important role in inflammation. 32
Recently, expression of NOS2 messenger RNA (mRNA) and antigen has been identified in lungs from pigs naturally infected with A. pleuropneumoniae, 5 , 6 indicating that NO may play a role in the pathophysiology of pleuropneumonia. However, no one has reported evidence of NOS2 activity in pleuropneumonia. Therefore, the objective of this study was to detect and localize nitrotyrosine and PARS antigen by immunohistochemistry to propose a functional role of NOS2 in pigs naturally infected with A. pleuropneumoniae.
Materials and Methods
Animals
Samples were obtained at necropsy from pigs submitted to the Department of Veterinary Pathology of Seoul National University from January 1998 to December 2001. Twenty pigs (Nos. 1–20), approximately 80–160 days of age, from 20 different herds were selected on the basis of clinical signs, characteristic lesions, bacteria isolation, and serotype. All 20 pigs were negative for Mycoplasma hyopneumoniae, Pasteurella multocida, and porcine reproductive and respiratory syndrome virus by culture and in situ hybridization. 4 , 20
Positive control sections were from pigs that had been naturally infected with A. pleuropneumoniae. 5–7 , 23 Two additional positive control samples at 24 and 48 hours after inoculation were from pigs that had been experimentally infected with A. pleuropneumoniae. Negative control sections were prepared from a 1-day-old colostrum-deprived pig that had not been exposed to any viral and bacteria pathogens and from conventional 60-day-old and 80-day-old pigs that were negative for bacteria isolation.
Reverse transcription–polymerase chain reaction
The reverse transcription–polymerase chain reaction (RT-PCR) was carried out as previously described. 6 RNA was extracted from lungs with Trizol LS reagent (GIBCO-BRL, Grand Island, NY) according to the manufacturer's instructions. The forward and reverse primers were 5′-CGTTATGCCACCAACAAT-3′ and 5′-ACTTCCTCCAGGATGTTGTA-3′, respectively. The primers amplified a 491–base pair (bp) complementary DNA (cDNA) fragment. Positive control of NOS2 was prepared as previously described. 6
Tissue processing
Samples of lung, liver, kidney, palatine tonsil, mediastinal lymph node, and large and small intestines were collected from infected and noninfected animals, fixed in 10% (w/v) neutral buffered formalin for 24–48 hours, and embedded in paraffin according to standard laboratory procedures. Sections were cut 4 μm thick, floated on a water bath containing diethylpyrocarbonate-treated water, and mounted on positively charged slides (Superfrost/Plus slide, Erie Scientific Co., Portsmouth, NH). The sections were deparaffinized in xylene, rehydrated through graded alcohols, and air-dried.
Immunohistochemistry of NOS2
Immunohistochemical examination was carried out as previously described. 5 Polyclonal rabbit anti-NOS2 antibody (Biotechnology Inc., Santa Cruz, CA) was used as the primary antibody.
Immunohistochemistry of nitrotyrosine
Endogenous alkaline phosphatase was quenched with 20% glacial acetic acid for 2 minutes at 4 C. All slides were then treated with proteinase K (GIBCO-BRL) 100 μg/ml in phosphate-buffered saline (PBS; 0.1 M, pH 7.4) for 20 minutes at 36 C. All slides were incubated with 10% normal goat serum (Sigma Chemical Company, St. Louis, MO) in PBS for 30 minutes at room temperature to saturate nonspecific protein-binding sites. Monoclonal mouse anti-nitrotyrosine antibody (Zymed Laboratories, Inc., CA), which was stated by the manufacturer to react with porcine nitrotyrosine, was diluted 1 in 100 in PBS containing 0.1% Tween 20. The slides were incubated with antibody overnight at 4 C in a humid chamber.
After three washes with 0.1% Tween 20 in PBS, sections were flooded and incubated for 1 hour at 36 C with alkaline phosphatase–conjugated goat anti-mouse IgG (Dako, Glostrup, Denmark) diluted 1 in 200 in PBS containing 0.1% Tween 20. The slides were washed in 0.1% Tween 20 in PBS. Then, sections were equilibrated with Tris buffer (pH 9.5) for 5 minutes at room temperature. The final reaction was produced by immersing the sections in a solution of red substrate (Boehringer Mannheim, Indianapolis, IN) at room temperature. The sections were lightly counterstained with Mayer's hematoxylin.
Immunohistochemistry of PARP
Endogenous alkaline phosphatase was quenched with 20% glacial acetic acid for 2 minutes at 4 C. All slides were incubated with 10% normal goat serum (Sigma Chemical Company) in PBS for 30 minutes at room temperature to saturate nonspecific protein-binding sites. Polyclonal rabbit anti-PARP antibody (Roche Molecular Biochemicals, Mannheim, Germany), which was stated by the manufacturer to react with porcine PARP, was diluted 1 in 100 in PBS containing 0.1% Tween 20. The slides were incubated with antibody overnight at 4 C in a humid chamber.
After three washes with 0.1% Tween 20 in PBS, sections were flooded and incubated for 1 hour at 36 C with biotinylated goat anti-rabbit IgG (Dako) diluted 1 in 200 in PBS containing 0.1% Tween 20. The slides were washed in 0.1% Tween 20. Sections were flooded and incubated for 1 hour at 36 C with streptavidin–alkaline phosphatase conjugate (Roche Molecular Biochemicals) diluted 1 in 200 in PBS containing 0.1% Tween 20. They were then equilibrated with Tris buffer (pH 9.5) for 5 minutes at room temperature. The final reaction was produced by immersing the sections in a solution of red substrate (Boehringer Mannheim) at room temperature. The sections were lightly counterstained with Mayer's hematoxylin.
Results
Amplification of template cDNA with primers of NOS2 resulted in amplified products corresponding to those of the predicted size, namely, 491 bp. PCR products were sequenced and their identity confirmed as NOS2. To determine expression of NOS2, RT-PCR analyses were performed using RNA extracted from lung tissues of pigs that had been infected with A. pleuropneumoniae. NOS2 was consistently detected in lung tissues of the 20 pigs with naturally occurring pleuropneumonia (Fig. 1). In contrast, NOS2 was not detected in lungs from negative control pigs.
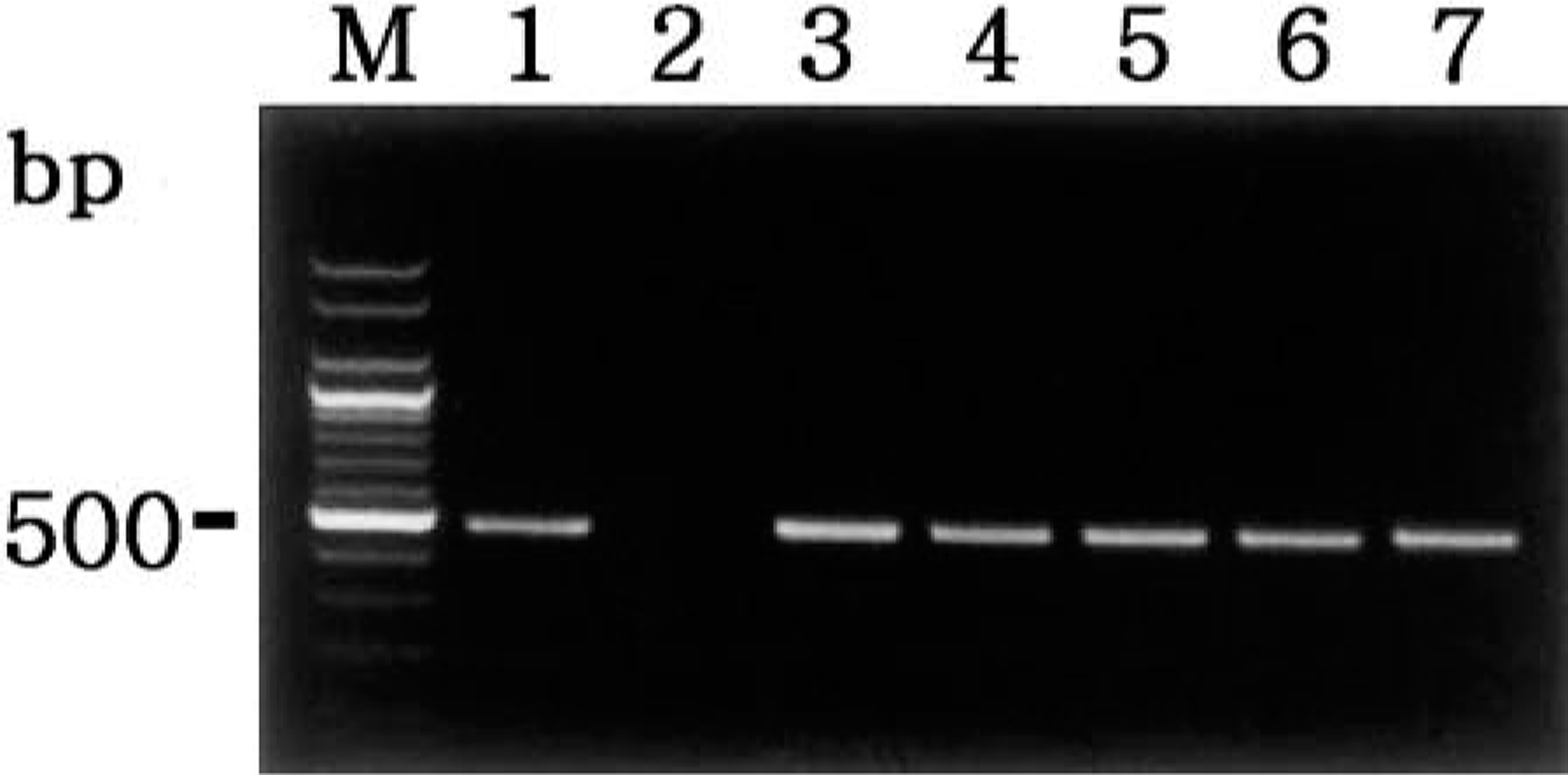
Agarose gel electrophoresis of PCR-amplified NOS2 cDNA products. From left to right: M = 100-bp DNA ladder; lane 1 = positive control from lipopolysaccharide-treated macrophages; lane 2 = negative control from normal macrophages; lane 3 = positive NOS2 cDNA from pig No. 2; lane 4 = positive NOS2 cDNA from pig No. 6; lane 5 = positive NOS2 cDNA from pig No. 9; lane 6 = positive NOS2 cDNA from pig No. 13; lane 7 = positive NOS2 cDNA from pig No. 15.
The 20 pigs infected with A. pleuropneumoniae had distinct and positive immunohistochemical staining for NOS2, nitrotyrosine, and PARS. Signal intensity varied within and between anatomical structures in any one section and between pigs. Positive cells were identified as a red reaction product without background staining.
Immunohistochemical staining of NOS2 protein was particularly intense in “oat cells,” the clustered leukocytes with streaming nuclear chromatin that are a characteristic histologic feature of porcine pleuropneumonia. Less intense immunoreactivity was seen in the dense zone of degenerate cells in granulation tissue surrounding the necrotic areas. The positive cells generally possessed large oval nuclei and abundant cytoplasm that are characteristic of macrophages. Macrophages that had infiltrated the alveolar spaces frequently exhibited a strong signal. Intense signal was also seen in the neutrophils in the alveolar spaces (Fig. 2) NOS2 protein was always associated with macrophages and neutrophils in the pleuropneumonic lung lesions, but it was minimal in the unaffected parts of the lung of A. pleuropneumoniae–infected pigs and in the normal lung from control pigs. Immunolabeling for NOS2 protein was consistently detected in lungs from positive control pigs, but it was not detected in lungs from negative control pigs.
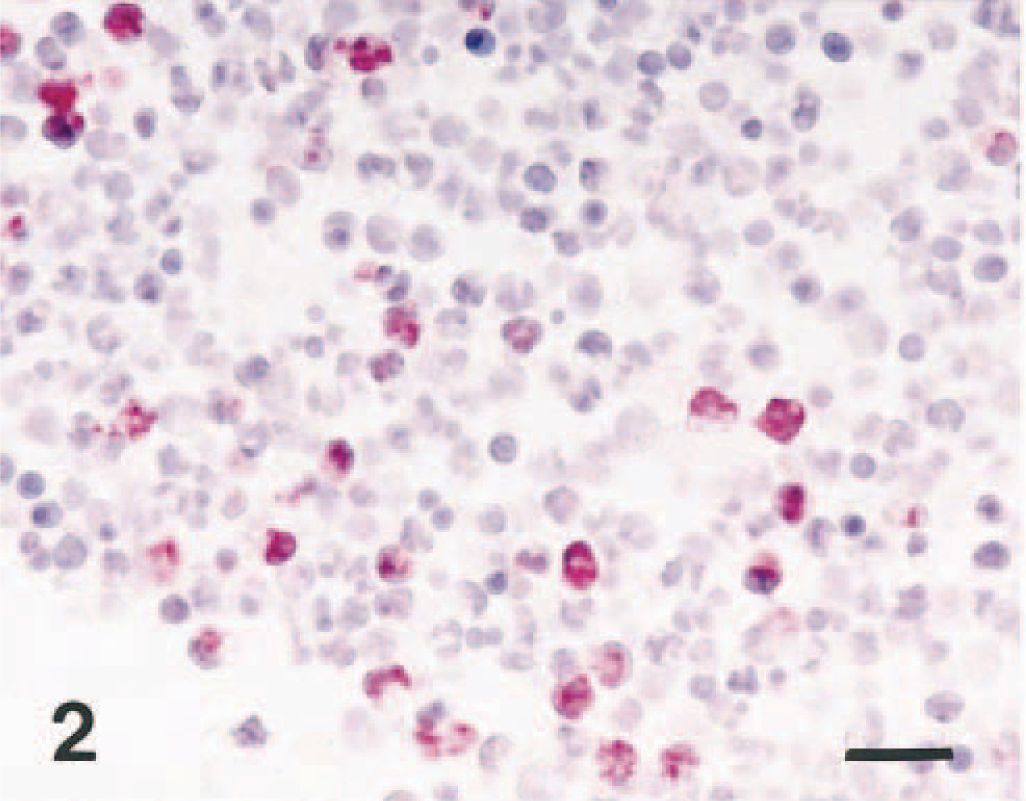
Lung; pig No. 2, naturally infected with A. pleuropneumoniae serotype 2. NOS2 antigen (red reaction) was detected in neutrophils of alveolar spaces. Immunohistochemistry; alkaline phosphatase, red substrate, hematoxylin counterstain. Bar = 30 μm.
Intense immunostaining for nitrotyrosine residue was seen within the lung lesions from A. pleuropneumoniae–infected pigs. Staining was especially strong in neutrophils and macrophages in the periphery of the lesions and within the alveolar spaces (Fig. 3). Minimal staining was seen in oat cells, the clustered leukocytes with streaming nuclear chromatin. There was close cell-to-cell correlation when serial sections were examined by immunohistochemistry for NOS2 and nitrotyrosine in each of the 15 lung samples (Fig. 4a, b). Colocalization of NOS2 and nitrotyrosine antigen was especially prominent in neutrophils and macrophages in the periphery of lesions. Nitrotyrosine antigen was always associated with macrophages and neutrophils in the pleuropneumonic lung lesions, but it was minimal in the unaffected parts of the lung of A. pleuropneumoniae–infected pigs. Sections from negative control pigs did not exhibit an immunohistochemical staining for nitrotyrosine.
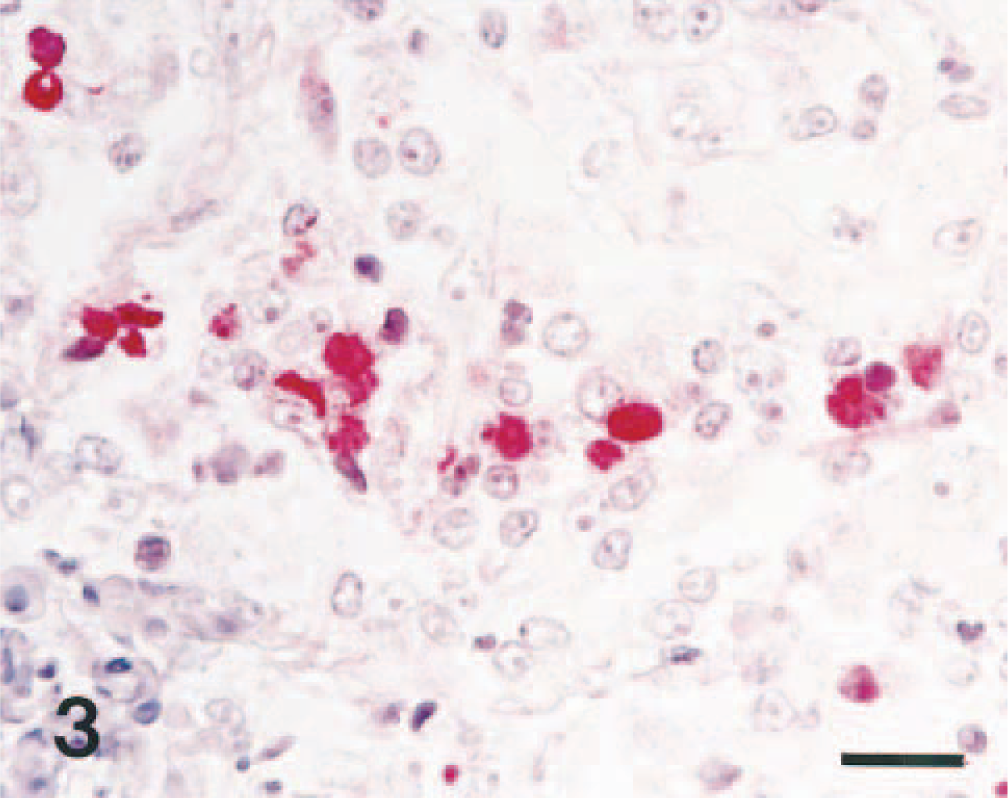
Lung; pig No. 4, naturally infected with A. pleuropneumoniae serotype 2. Nitrotyrosine antigen (red reaction) was detected in macrophages of alveolar spaces. Immunohistochemistry; alkaline phosphatase, red substrate, hematoxylin counterstain. Bar = 30 μm.
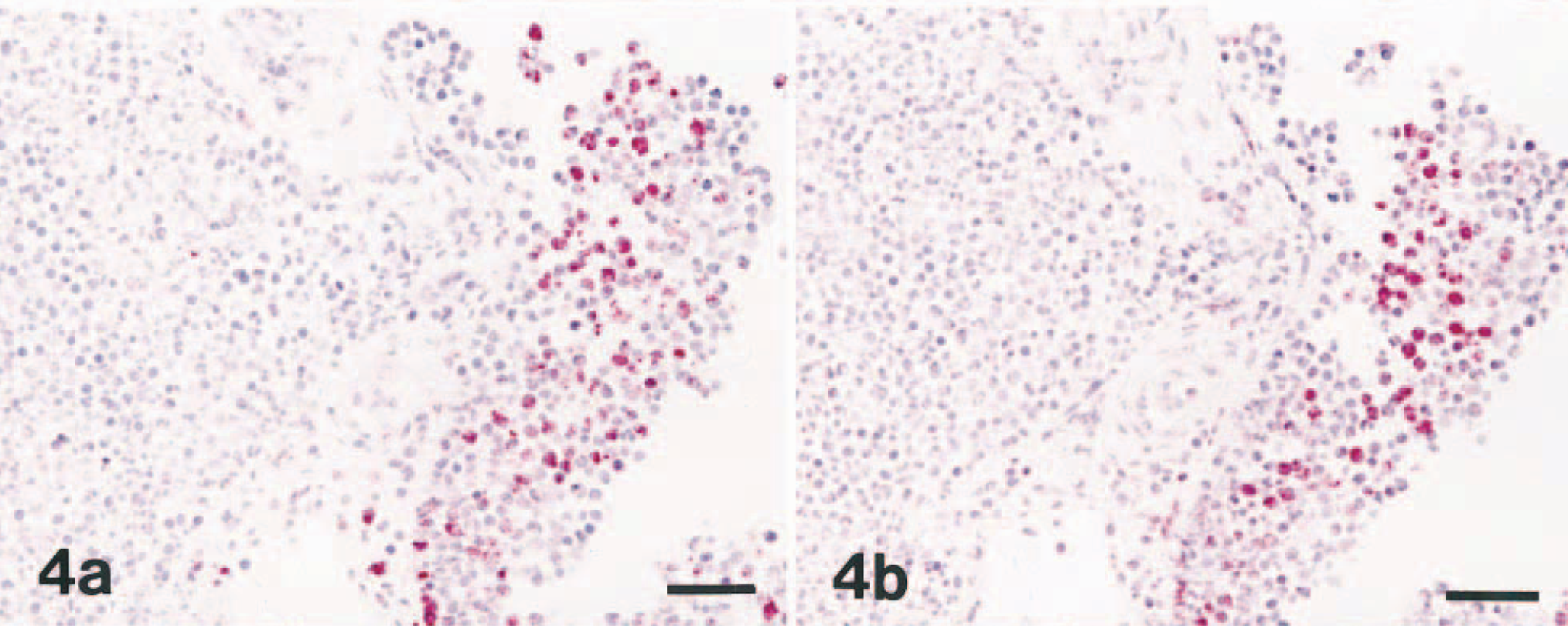
Lung; pig No. 6, naturally infected with A. pleuropneumoniae serotype 5. Fig. 4a. NOS2 antigen (red reaction) was detected in inflammatory cells. Immunohistochemistry; alkaline phosphatase, red substrate, hematoxylin counterstain. Bar = 55 μm. Fig. 4b. Serial section of the cells in Fig. 4a showing the same cells positive for nitrotyrosine antigen (red reaction). Immunohistochemistry; alkaline phosphatase, red substrate, hematoxylin counterstain. Bar = 55 μm.
Expression of PARS was always present within the inflammatory lesions but was minimal in the unaffected lung of A. pleuropneumoniae–infected pigs. Macrophages in alveolar spaces frequently exhibited a strong staining for PARS. The majority of areas containing NOS2-positive cells also have numerous PARS-positive cells in serial sections of lungs. Immunohistochemistry of nitrotyrosine and PARS with serial sections of lung tissues indicated a close cell-to-cell correlation for nitrotyrosine and PARS antigen (Fig. 5a, b). Colocalization of nitrotyrosine and PARS antigen was especially prominent in macrophages in the periphery of lesions. Sections of lung tissue from negative control pigs did not exhibit an immunohistochemical staining for PARS.
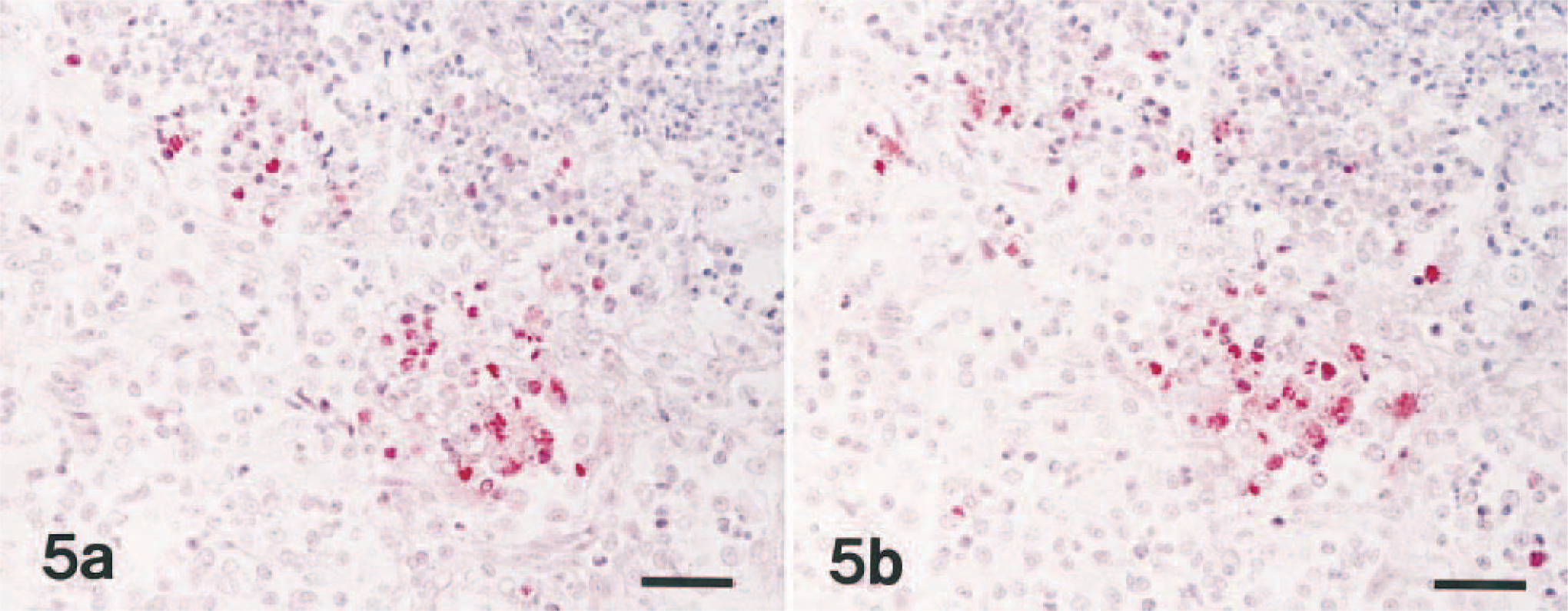
Lung; pig No. 9, naturally infected with A. pleuropneumoniae serotype 5. Fig. 5a. Nitrotyrosine antigen (red reaction) was detected in inflammatory cells. Immunohistochemistry; alkaline phosphatase, red substrate, hematoxylin counterstain. Bar = 55 μm. Fig. 5b. Serial section of the cells in Fig. 5a showing the same cells positive for poly(ADP-ribose) synthetase (red reaction). Immunohistochemistry; alkaline phosphatase, red substrate, hematoxylin counterstain. Bar = 55 μm.
Discussion
Expression of NOS2 was markedly upregulated at the mRNA and protein levels in lung tissue from pigs with lesions caused by A. pleuropneumoniae. Simultaneous detection of NOS2 mRNA and protein also provides a scientific evidence that these cells produce NO. Because expression of NOS2 was greater in the inflammatory lesions than in the unaffected parts of lung, it would appear that induction of NOS2 is a direct response to A. pleuropneumoniae. Furthermore, immunohistochemistry and in situ hybridization of serial sections of lung indicated that the majority of areas containing numerous NOS2-positive cells also have numerous A. pleuropneumoniae DNA-positive cells (data not shown).
The present immunohistochemical investigation using a specific anti-nitrotyrosine antibody clearly demonstrated the presence of nitrotyrosine in neutrophils and macrophages in the lesions of A. pleuropneumoniae–induced pleuropneumonia, especially in the periphery of inflammation. The localization of nitrotyrosine-positive cells in the periphery of the inflammation suggests that peroxynitrite is generated mainly in expanding areas of inflammation, and it is due to the accumulation of newly recruited inflammatory cells in areas where molecular oxygen can be effectively used for the biosynthesis of NO and superoxide anion.
Immunohistochemistry of serial sections of the lung indicates that the majority of areas containing numerous NOS2-positive cells also have numerous nitrotyrosine-positive cells and PARS-positive cells. Inflammatory cells such as neutrophils and macrophages express NOS2, produce NO, 22 and they also produce superoxide. 16 , 22 Peroxynitrite is a product of the interaction of superoxide and NO; peroxynitrite is formed in inflammatory lesions in porcine pleuropneumonia by the formation of nitrotyrosine, a product of the reaction of peroxynitrite with tyrosine residue. Inflammatory cells infiltrating the alveolar spaces express NOS2 and colocalize in cells with nitrotyrosine immunoreactivity. Because NOS2 and nitrotyrosine colocalize in inflammatory cells in alveolar spaces, it is possible that NO and superoxide produced by inflammatory cells react to form peroxynitrite, which, in turn, reacts with proteins to produce nitrotyrosine. Nitrotyrosine formation was initially proposed as a relatively specific marker for the detection of the endogenous peroxynitrite formation because peroxynitrite is impossible to measure in vivo because of its high reactivity. 2 Detection of nitrotyrosine in tissues is most often achieved by immunostaining, although HPLC- or GC-based spectrophotometery can be used. 19 However, other reactions can induce tyrosine nitration irrespective of the presence of peroxynitrite. The reaction of nitrite with hypochlorous acid and the reaction of myeloperoxidase with hydrogen peroxide can lead to the formation of nitrotyrosine. 11 , 30 Increased staining for nitrotyrosine is therefore considered to be an indication of increased nitrative stress rather than a specific marker of the generation of peroxynitrite.
Although NO, per se, may not be detrimental, it can react with superoxide in a diffusion-limited fashion to produce peroxynitrite, which has been shown to cause cellular injury. Speculation that NOS2 is a detrimental molecule has often been based on the assumption that NOS2 induction means the production of peroxynitrite. Peroxynitrite breaks strands of DNA, and this triggers energy-consuming repair mechanisms and activates the nuclear enzyme PARS. Activation of PARS results in the depletion of its NAD+ substrate and a reduction in the rate of glycolysis. This depletion of NAD+ leads to a rapid decrease in intracellular ATP, 35 cellular injury, and dysfunction. 3
Besides causing cytotoxicity, PARS may also play a role in the inflammatory process. Inhibition of PARS reduces tissue injury caused by the infiltration of neutrophils, 9 , 33 and the reduction in tissue injury by PARS inhibitors may be associated with a reduction in both oxygen- and nitrogen-centered free radical production. One mechanism by which PARS modulates neutrophil tissue infiltration may relate to the effect of PARS activation on the expression of intercellular adhesion molecules. 33 , 36
Upregulation of NOS2 mRNA and protein by macrophages is strictly associated with their capacity to produce NO and its microbicidal activity in vitro and in vivo. 22 Evidence that NOS2 is strongly expressed in lesions caused by A. pleuropneumoniae indicates that this enzyme is involved in the cellular defense mechanisms elicited against A. pleuropneumoniae. Moreover, the observation that NOS2 expression in pleuropneumonic area is associated with protein nitrosation and PARS suggests that NOS2 is functionally active during infections caused by A. pleuropneumoniae.
Footnotes
Acknowledgements
This research was supported by a grant of the Korea Health 21 R&D Project (02-PJ1-PG3-20799-0001), the Ministry of Health and Welfare, Republic of Korea.
