Abstract
Introduction:
Sepsis is a severe disease with a high morbidity and mortality. Baicalin, an active compound of Chinese medicine,
Methods:
The production of NOx, PGE2, and pro-inflammatory cytokines, the mRNA and protein expression of inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2), and the nuclear translocation of NF-κB in LPS-stimulated macrophages or endotoxic mice were determined. The model of severe endotoxic mice was established by injection of LPS (60 mg/kg, i.p.).
Results:
Baicalin significantly inhibited the production of NO, PGE2, and pro-inflammatory cytokines, including TNF-α, IL-1β, and IL-6 in LPS-stimulated macrophages. Baicalin treatment also markedly suppressed LPS-induced iNOS and COX-2 expression at the transcriptional and translational levels, and the nuclear translocation of NF-κB in macrophages. Similarly, the serum concentrations of NOx, PGE2, and pro-inflammatory cytokines, and the lung myeloperoxidase activity were greatly reduced in baicalin-treated endotoxic mice. Notably, after LPS injection, the 3-day survival rate of mice treated with pre- or post-administration of baicalin (50 mg/kg, i.p.) remarkably increased to 100% and 90%, respectively compared with LPS-injected alone mice with a survival rate of 0%.
Conclusion:
Baicalin has a potent anti-inflammatory activity in LPS-stimulated macrophages and endotoxic mice. Moreover, treatment with baicalin dramatically increased the survival in the severe septic mice, suggesting that baicalin may be a potential agent for sepsis therapy.
Keywords
Introduction
Sepsis is characterized by hypotension, systemic inflammatory responses, organ ischaemia, which in turn often causes multiple organ failure. Sepsis is a leading cause of death in intensive units with an overall mortality of sepsis exceeding 30% despite the much advances in intensive medical care.1,2 So far, current drugs are not considered to have a promising effect to reduce the high mortality of sepsis, effectively. Therefore, development of potential therapeutic agents to alleviate the severe systemic inflammation, thereby increasing the survival of sepsis is urgently needed.
Nitric oxide (NO), synthesized by nitric oxide synthase (NOS), plays an important role in the regulation of several physiological and pathological responses. 3 There are two distinct NOS isoforms, including constitutive Ca2+/calmodulin-dependent NOS (cNOS) and inducible Ca2+/calmodulin-independent NOS (iNOS). The release of NO by cNOS is thought to modulate many physiological functions, including regulation of blood pressure, organ blood flow distribution, and platelet aggregation. 3 However, iNOS-derived high amount of NO activated by pro-inflammatory cytokines and lipopolysaccharide (LPS) is a crucial mediator promoting the development of bacterial endotoxemia. 4
Cyclooxygenase (COX) catalyses the conversion of arachidonic acid to prostaglandin H2 that is the precursor of a variety of biological active mediators, including prostaglandin E2 (PGE2), prostacyclin, and thromboxane A2. 5 Two distinct COX isoforms are found. COX-1 is constitutively present in most tissues and is responsible for regulating normal physiological functions. 6 In contrast, COX-2, an inducible isoform, is activated by LPS and pro-inflammatory cytokines and it is highly expressed in inflammatory tissues and LPS-stimulated macrophages. 7 The COX-2-derived excessive formation of pro-inflammatory PGE2 is implicated in the pathogenesis of inflammation-associated diseases such as sepsis. 8
The LPS, a characteristic component of the cell wall of Gram negative bacteria, is capable of triggering systemic inflammation, which is similar to the clinical features of sepsis. Accordingly, LPS is often used to induce sepsis in vitro and in vivo. In response to LPS, the formation of pro-inflammatory cytokines, including tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), and interleukin-6 (IL-6), increased markedly, which ultimately upregulates iNOS and COX-2.9,10 It is well known that nuclear factor-κB (NF-κB) is a key transcription factor accounting for induction of various inflammatory genes, including iNOS, COX-2, pro-inflammatory cytokines. 11 Taken together, drugs with an ability to inhibit NF-κB-activated pro-inflammatory gene expression and mediator formation may have a potential to prevent the detrimental consequences of sepsis.
The
Materials and methods
Reagents
Baicalin (purity: 98%), LPS and other chemicals and reagents were purchased from Sigma-Aldrich (St. Louis, MO, USA).
Cell culture
Murine macrophage cell line RAW 264.7 (American Type Culture Collection ATCC, TIB71, Rockville, MD, USA) was cultured in Dulbecco’s modified Eagle’s medium (DMEM) without phenol red, supplemented with 10% fetal calf serum, 4 mM
Cell viability assay
Cell respiration, an indicator of cell viability, was assessed by the mitochondrial dependent reduction of 3-(4,5-dimethylthiazol-2-yl)-diphenyltetrazolium bromide (MTT) to formazan. The extent of reduction of MTT to formazan with cells was quantified spectrophotometrically by measurement at OD550.
Nitrite and serum nitrite + nitrate (NOx) and PGE2 measurements
Various concentration of baicalin was dissolved in physiological saline was incubated with RAW 264.7 cells for 24 h in the presence of LPS (0.1 µg/ml), the culture medium was collected for the determination of nitrite, an index of NO production, and PGE2 concentrations. Nitrite was measured by the Griess reaction as previously described. 15 In in vivo study, 4 h after LPS (60 mg/kg, i.p.) injection the concentrations of serum NOx in mice were determined using a Sievers Nitric Oxide Analyzer (Sievers 280 NOA, Boulder, CO, USA). PGE2 was measured using an enzyme immunoassay (EIA) kit (Biosource International, Camarillo, CA, USA).
Reverse transcription–polymerase chain reaction (RT–PCR)
RNA was isolated from the RAW 264.7 cells using TRIzol reagent and the concentration of RNA was quantified by measurement of optical density at 260 nm. The RNA was then reverse transcribed in a solution containing 3 µg total RNA, 1 µl 10 mM deoxy-NTPs, and 1 µl oligo-dT (500 µg/ml ) in diethylpyrocarbonate (DEPC) H2O (12 µl). The mixture was incubated for 10 min at 70°C and then placed on ice for another 5 min. An aliquot of 8 µl reverse transcriptase mix (4 µl first strand buffer, 2 µl 0.1M dithiothreitol, 1 µl RNaseOUT, 40 units/ml, and 1 µl Superscript reverse transcriptase) was added to each tube, and this was incubated at 42°C for 60 min, followed by a 15 min incubation at 70°C. PCR was conducted in a reaction mixture containing 1U
The sequences of the primers were as follows:
iNOS forward: 5’–ATGGACCAGTATAAGGCAAGCACC–3’;
iNOS reverse: 5’–GCTTCTGGTCGGATGTCATGAGCAA–3’;
COX-2 forward: 5’–TTTGTTGAGTCATTCACCAGACAGAT–3’;
COX-2 reverse: 5’–CAGTATTGAGGAGAACAGATGGGATT–3’;
β-2m forward: 5’–TGACCGGCTTGTATGCTATC–3’;
β–2m reverse: 5’–CAGTGTGAGCCAGGATATAG–3’.
Western blot
Total cellular protein was prepared using lysis buffer (10 mM Tris-HCl, pH 7.4, 0.15 M NaCl, 1 mM EDTA, 10 mM 3-[(3-Cholamidopropyl)-dimethylammonio]-1-Propanesulphonate, 10 µg/ml Aprotinin and 0.02 mM phenylmethylsulphonyl fluoride). The protein (12 µg) was separated on sodium dodecylsulphate-polyacrylamide minigels (7.5% for iNOS and 12.5% for COX-2, respectively) and then transferred to polyvinylidene difluoride membranes (Millipore, Bedford, MA, USA). The membranes were then incubated with anti-iNOS (1:5000 dilution, Transduction Lab., Lexington, KY) or anti-COX-2 (1:250 dilution, Transduction) antibodies, respectively. The bands of iNOS and COX-2 were visualized by exposure of the membrane to a horseradish peroxidase-conjugated secondary antibody (Transduction) followed by addition of ECL reagent (Amersham International Plc., Buckinghamshire, UK). The relative optical density of bands was quantified by densitometric scanning of the western blots using Image-Pro Plus software. The nuclear extracts of different groups were prepared as described previously.11 Then, the nuclear extract was incubated with NF-κB antibody (Cell Signaling Technology, Beverly, MA, USA) followed by the processes of Western blotting assay.
Animal model of sepsis
ICR mice (weighing 26–30 g) were purchased from the National Animal Center (Taipei, Taiwan). The study was approved by the local Institutional Review Board of National Defense Medical Center (Taipei, Taiwan) according to the recommendations from Helsinki and the internationally accepted principles in the care and use of experimental animals. The mice were randomized into three groups: mice administrated with vehicle (saline, i.p., the LPS group) or baicalin (50 mg/kg, i.p., the LPS + Baicalin group) 30 min before LPS injection (60 mg/kg, i.p.). The saline-alone injected mice acted as the control group. Each group contained six mice. The calculation of sample size of animals selected for the study was performed and a statistical test (Fisher exact test, two tail) was used according to the method described previously. 16 Six mice were required for each group to achieve a power of 80% with a significance level of 0.05.
Cytokine measurements
In LPS-treated mice, our preliminary data showed that the injection of LPS resulted in a bell-shaped curve in serum TNF-α concentrations, which peaked at 1.5 h and subsequently decreased slowly. Therefore, unless otherwise indicated, TNF-α was measured at 1.5 h. Other cytokines, including IL-6 and IL-1β, were measured 4 h after the injection of LPS. The cytokines in medium or serum were measured using rat EIA kits (Genzyme Corporation, Cambridge, USA).
Myeloperoxidase (MPO) activity assay
Four hours after the injection of LPS, the lungs of mice were removed and washed with sterile normal saline, blotted dry of surface blood, weighed, and homogenized in ice-cold 0.5% hexadecyltrimethylammonium bromide (HTAB) in 50 mM phosphate buffer (pH 6.0, 5 ml HTAB/g tissue) on ice using a homogeniser (Pro model 200, Monroe, CT, USA) at a setting of 4 for 10 s. The resulting homogenate was sonicated and then centrifuged at 15,000 g for 15 min at 4°C. The amount of protein in the supernatant was determined using a dye-binding assay (Bio-Rad) with bovine serum albumin as a standard. The supernatant was mixed at 1:30 (supernatant: assay buffer) and read spectrophotometrically at 460 nm wavelength. The assay buffer consisted of 100 mM potassium phosphate, pH 6.0, 0.083 ml H2O2 (Sigma; 30% stock diluted 1:1000) and 0.834 ml
Survival rate measurement
To evaluate whether baicalin improves the survival rate in LPS-induced endotoxic mice, lethal dose of LPS (60 mg/kg, i.p.) was used under the administration of baicalin (50 mg/kg, i.p. at 30 min before and 6, 12 h after LPS injection or 50 mg/kg, i.p. at 4 and 10 h after injection of LPS) or vehicle. Then, the survival of mice was measured for 3 days. Each group contained 20 mice.
Statistical analyses
The data was expressed as mean ± standard error of mean (SEM). One-way ANOVA with post hoc Bonferroni test was used for statistical analysis. The
Results
Effects of baicalin on cell viability, and nitrite and PGE2 production in LPS-stimulated macrophages
Treatment with baicalin (10–50 µM) for 24 h dose-dependently inhibited the accumulation of nitrite (IC50 = 21 µM) and PGE2 (IC50 = 36 µM) in LPS-stimulated RAW 264.7 macrophages (Figure 1(a)). In the presence of LPS, baicalin (25, 50 µM) did not significantly affect the cell viability evaluated by MTT, suggesting that the effects of baicalin were not due to its cytotoxicity (Figure 1(b)).
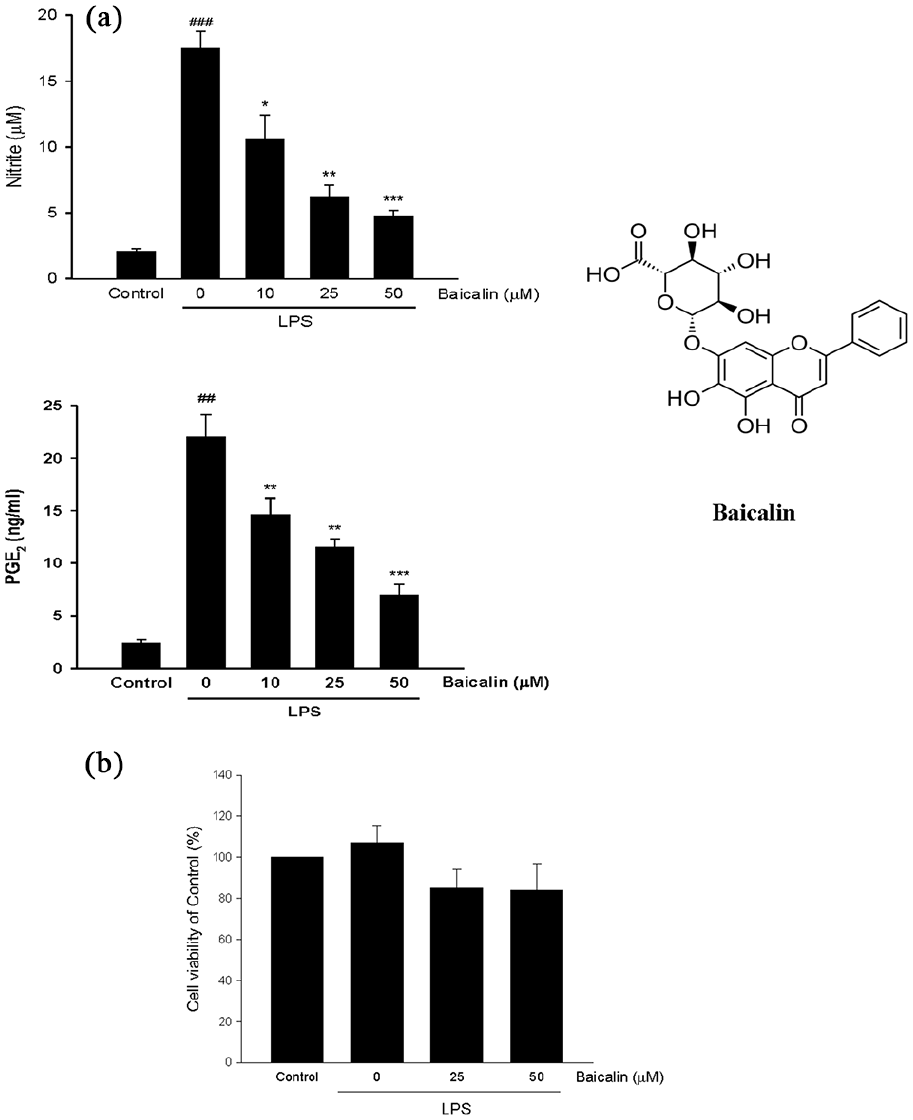
Effects of baicalin on the nitrite and PGE2 production (a), and cell viability (b) in LPS-stimulated RAW 264.7 macrophages. Baicalin (10–50 µM) or vehicle (Control) was added simultaneously with LPS (0.1 µg/ml), then incubated for 24 h. The cell viability, and the levels of nitrite and PGE2 in the medium were measured. The resting cells acted as the control group. Values are expressed as means ± SEMs (
Effects of baicalin on the mRNA and protein expression of iNOS and COX-2 and NF-κB activation in LPS-stimulated macrophages
Next, we determined whether the attenuation of nitrite and PGE2 production by baicalin was due to downregulation of iNOS and COX-2. The RT-PCR analyses and Western blot were used to examine the changes of mRNA and protein levels of iNOS and COX-2. In unstimulated macrophages, the amounts of iNOS and COX-2 mRNA were very low or undetectable. Our preliminary data showed that addition of LPS resulted in a remarkable increase in iNOS and COX-2 mRNA expression with a peak at 6 h and then declining. Thus, the iNOS and COX-2 mRNA expression was examined at the 6 h time point. We found that treatment with baicalin dose-dependently inhibited the elevation of iNOS and COX-2 mRNA in LPS-stimulated macrophages (Figure 2). Similarly, LPS-activated iNOS and COX-2 protein expression was also suppressed by baicalin in a dose-dependent manner. However, baiclain did not significantly affect the protein expression of cNOS and COX-1 (Figure 3(a)). Thus, the inhibitory effects of baicalin on LPS-induced NO and PGE2 production may attribute to inhibition of iNOS and COX-2 expression. The nuclear translocation and subsequent activation of NF-κB is required for promoting the transcription of iNOS and COX-2. Compared with unstimulated cells, stimulation with LPS for 1 h markedly elevated the nuclear level of NF-κB, which was reduced by baicalin in a concentration-dependent manner (Figure 3(b)). These findings suggest that inhibition of NF- κB activation may contribute to downregulation of iNOS and COX-2 by baicalin in LPS-stimulated macrophages.

Effects of baicalin on iNOS and COX-2 mRNA expressions in LPS-stimulated macrophages. Cells were incubated with LPS (0.1 µg/ml) for 6 h in the absence or presence of various doses of baicalin. The mRNA levels of iNOS, COX-2 and β-2 m were determined by RT-PCR. Lane 1, resting cells as Control; lane 2, LPS alone cells; lane 3, LPS + baicalin (10 µM); lane 4, LPS + baicalin (25 µM); lane 5, LPS + baicalin (50 µM). The base pair standard is shown in lane M. Band intensities were measured using densitometry. ###

Effects of baicalin on target gene expressions and NF-κB nuclear translocation in LPS-stimulated macrophages. Cells were incubated with LPS (0.1 µg/ml) for 24 h at 37°C in the presence or absence of baicalin (25 or 50 µM). The protein expressions of cNOS, iNOS and COX-1, and COX-2 (a), as well as the nuclear NF-κB (b) were determined by western blotting. ##
Effects of baicalin on pro-inflammatory cytokine formation in LPS-stimulated macrophages
Excessive production of pro-inflammatory cytokines plays a vital role in the pathogenesis of sepsis. 17 Upon LPS stimulation for 24 h, the levels of pro-inflammatory cytokines, including TNF-α, IL-6, and IL-1β were markedly increased in activated macrophages, which were significantly inhibited by baicalin in a dose-depend manner (Figure 4(a)).

Effects of baicalin on pro-inflammatory cytokine formation in LPS-stimulated macrophages, and the levels of serum NOx, PGE2, pro-inflammatory cytokines, as well as lung MPO activity in the septic mice. (a) After different concentrations of baicalin or vehicle (Control) was added simultaneously with LPS (0.1 µg/ml) and incubated for 24 h, the amounts of TNF-α, IL-1β, and IL-6 in medium were determined. ##
Effects of baicalin on serum levels of NOx, PGE2, and pro-inflammatory cytokines and lung MPO activity in endotoxic mice
Similarly, compared with the LPS-injected alone mice, the mice treated with baicalin (50 mg/kg, i.p.) 30 min before injection of LPS (60 mg/kg, i.p.) and for 4 h had a significantly lower serum concentrations of NOx and PGE2, and TNF-α, IL-6, and IL-1β, as well as the MPO activity in lungs (Figure 4(b)).
Baicalin increased the survival rate of endotoxic mice
On day 3 after LPS injection, the survival rate of mice treated with pre- or post-administration of baicalin markedly increased to 100% and 90%, respectively compared with the 0% in LPS-injected alone mice (Figure 5). Thus, baiclain is an effective agent to improve the survival in the severe septic mice.
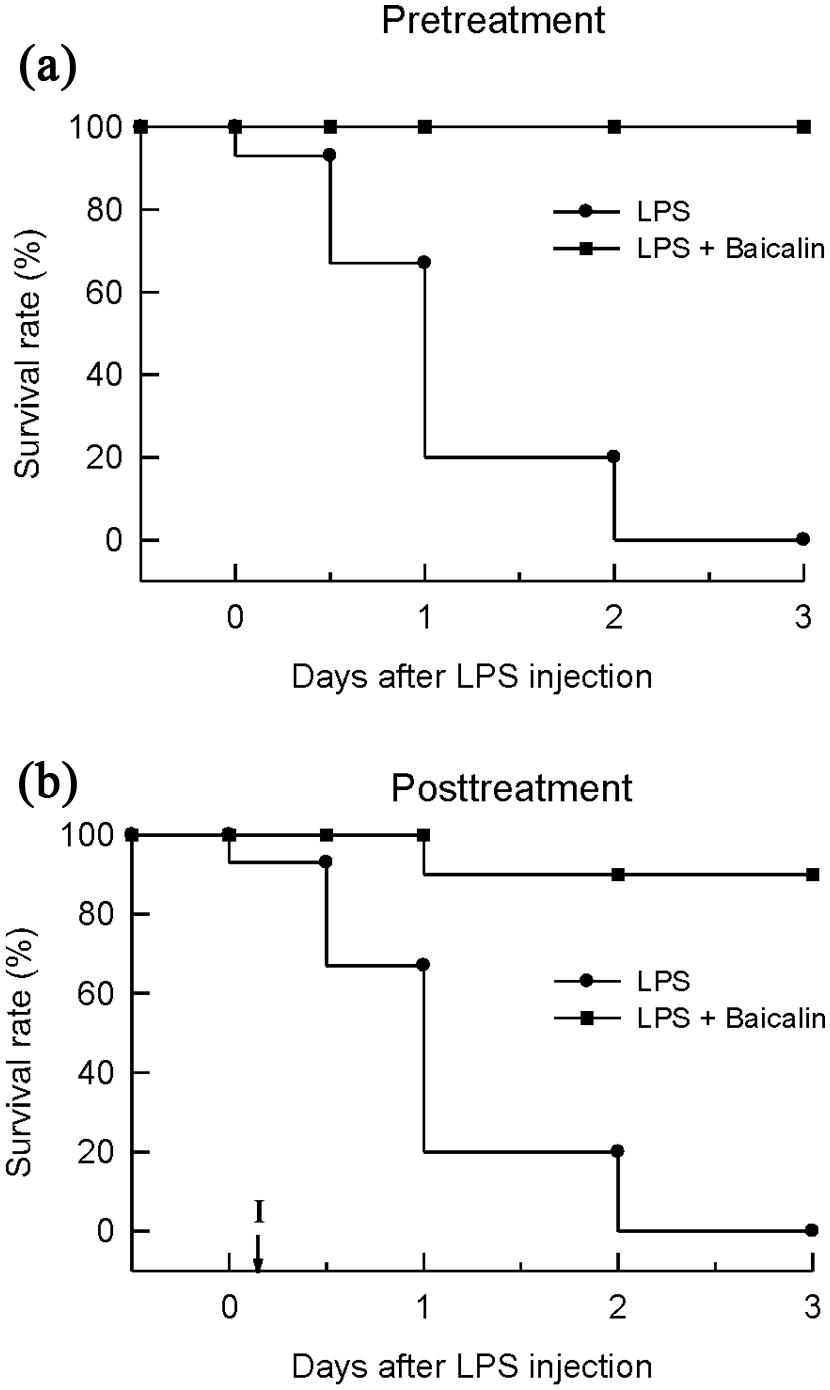
Effects of baicalin on the survival rate of LPS-induced septic mice. (a) LPS (60 mg/kg, i.p.) was injected with vehicle (saline, i.p.) or baicalin (50 mg/kg, i.p. at 30 min before and 6, 12 h after LPS injection). (b) Mice were administrated with vehicle or baicalin 50 mg/kg, i.p. at 4 and 10 h after LPS injection. Survival was monitored for 3 days after LPS injection. Data are expressed as percentage of mice surviving at the observed time point. Each group consisted of 20 mice.
Discussion
Exposure to bacterial endotoxins such as LPS, macrophages are activated and subsequently produce various inflammatory mediators, including NO, PGE2, and pro-inflammatory cytokines. Thus, macrophages have been regarded as an important source for releasing inflammatory substances during sepsis.18,19 In the present study, we demonstrated that baicalin is able to inhibit iNOS/NO and COX-2/PGE2 cascades, pro-inflammatory cytokine formation, and NF-κB activation in LPS-stimulated RAW 264.7 macrophages. Therefore, the anti-inflammatory effects of baicalin might be at least partly resulted from suppressing macrophages-dependent inflammatory processes. Notably, baicalin could dramatically improve the survival in the severe endotoxic mice. These findings suggest that baicalin seems to be a potential agent in treating sepsis in mice.
It is believed that uncontrolled inflammation is a critical pathological factor causing organ injury/dysfunction and high mortality during sepsis. 20 In response to endotoxin, iNOS and COX-2-derived excessive formation of NO and PGE2 from macrophages is thought to play a vital role in the development of characteristic features of sepsis. 21 The concept was supported by the fact that selective inhibition of iNOS induction or activity, or treatment with selective COX-2 inhibitor (NS-398) or PGE2 receptor antagonists greatly reduced the progres-sion and mortality in a murine model of sepsis or trauma.22,23 Here we confirmed that baicalin significantly inhibits LPS-induced NO and PGE2 production in macrophages and endotoxic mice. Moreover, the mRNA and protein levels of iNOS and COX-2 were significantly inhibited by baicalin. These results indicated that reduction of NO and PGE2 formation by baiclalin might be mediated by downregulating iNOS and COX-2 both at the transcriptional and translational levels. Furthermore, baiclain did not affect constitutive cNOS and COX-1 expression, which may be beneficial for maintaining their normal physiological functions. The inhibition of iNOS/NO cascade and PGE2 production by baicalin observed in this study is consistent with other similar study, but the COX-2 protein expression was not change after baicalin treatment. 24 In contrast, a recent research found a similar inhibition on COX-2 protein expression by baicalin. 25 The reasons resulting in the difference are unknown and the study design and experimental technique may be involved.
It is well known that the pathogenesis of sepsis is closely related to exacerbated release of pro-inflammatory cytokines such as TNF-α, IL-1β and IL-6. 17 Since, the release of TNF-α predominantly produced by activated macrophages starts within 30 min after LPS stimulation, TNF-α may act an early regulator of immune responses and subsequently promotes the secretion of other pro-inflammatory cytokines such as IL-1 and IL-6 in an autocrine an paracrine manner.26,27 As expected, injection of TNF-α could trigger many sepsis-related symptoms in animals. 28 Blocking TNF-α greatly abolishes LPS-induced IL-6 production in vitro and in vivo, which further supports that IL-6 may be secondary to TNF-α synthesis. 29 Several studies have reported that treatment with anti-TNF-α, anti-IL-1β or IL-1 receptor antagonists not only inhibits iNOS and COX-2 expression in LPS-stimulated macrophages but also improves the survival in a rat model of endotoxin shock.30,31 Additionally, IL-6 is able to activate B and T lymphocytes and trigger LPS-evoked fever. 32 Therefore, inhibiting the formation of these pro-inflammatory cytokines is a promising strategy to prevent sepsis progression. The results of the present study indicated that baicalin significantly inhibited the formation of TNF-α, IL-1β, and IL-6 both in LPS-stimulated macrophages and septic mice, which was the same as the reports by others.33,34
NF-κB located in the cytoplasm is an inactive complex bound to IκB-α. In the presence of inflammatory stimuli such as LPS, IκB-α is phosphorylated and degraded, leading to dissociation with NF-κB and release of activated NF-κB that then translocates into nucleus, where it activates the transcription of downstream target genes, including iNOS, COX-2, and pro-inflammatory cytokines. 35 Interestingly, TNF-α binding to TNF receptor can further induce NF-kB-p65 nuclear translocation, suggesting that there is a positive loop between TNF-α and NF-κB activation. 10 Treatment with baicalin was found to significantly inhibit NF-κB nuclear translocation in LPS-stimulated macrophages. Considering the importance of NF-κB on inflammation and in the development of sepsis, the inhibitory effects of baicalin on these genes associated with inflammation may be at least in part mediated by suppressing NF-κB activation. A marked elevation in activated neutrophil infiltration occurred in sepsis is a major source for oxygen-derived free radical formation, resulting in iNOS and COX-2 induction via activation of NF-κB. 36 Accordingly, inhibition of lung MPO activity (an indicator of neutrophil infiltration) by baicalin may provide a mechanism contributing its protective effect against sepsis. The additional effects of baicalin, including activation of nuclear factor erythroid 2-related factor 2 (NRF2) and its regulated antioxidant genes such as heme oxygenase-1 may be also involved in its anti-inflammatory effects.37,38
Although pro-inflammatory cytokines exert deleterious effects in sepsis, some clinical trials have indicated that treatment with TNF-α or IL-1β antagonists might worsen the survival of septic patients and the true reasons remain unclear. 39 It has been proposed that initially sepsis is characterized by increased inflammatory responses but as it persists, there is a shift toward an immunosuppressive state. 40 Therefore, cytokines may exert different effects at specific time points during sepsis progression. Similarly, endothelium-derived NO production represents a triphasic response: overproduction, normal and then subnormal after the onset of sepsis, suggesting that different stages of sepsis have their characterized amounts of cNOS, iNOS, and NO production. 41 Accordingly, if the NOS inhibitors or cytokine antagonists are treated at different stage of sepsis, the outcomes and the survival may be different. 42 Collectively, sepsis is a mutifactorial and dynamic process involving a complex interaction between various cells and systems. Since, no single treatment or drug is considered to have a satisfactory therapeutic effect to reduce the mortality of sepsis, how to improve the survival rate in septic patients remains a big problem. 43 An animal model of polymicrobial sepsis induced by cecal ligation and puncture (CLP) has been used to determine whether baicalin increases the survival. The results of the previous study indicated that treatment with baicalin (100 mg/kg, i.p.) at 1 h, 6 h, and 12 h after CLP could increase the 8-day survival rate (81.25%) compared with that of CLP mice (25%), and inhibition of inflammation, apoptosis and microbial clearance may be involved in its beneficial effect. 44 In this study, the results of survival rate indicated that all mice were dead after injection of high dose of LPS (60 mg/kg, i.p.) for 3 days, suggesting that the septic model is more severe than CLP model. Notably, pre- or post-administration of baicalin (50 mg/kg, i.p. at 30 min before and 6, 12 h after LPS injection), dramatically increased the 3-day survival rate to 100% and 90%, respectively. Our and other studies supported that baicalin is a potential agent for sepsis therapy.
Limitations of this study may include (1) Other molecular mechanisms underlying the anti-inflammatory activity of baicalin in sepsis were not investigated, despite its actions on sepsis-associated inflammatory mediators. (2) The protective effects of baicalin on other different septic animal models were not examined. Therefore, further studies should be done toward these objectives.
Conclusion
We demonstrated that baicalin exhibited anti-inflammatory effects by inhibiting iNOS and COX-2 induction and NF-κB activation, NO, PGE2, and pro-inflammatory cytokine production, and tissue neutrophil infiltration in LPS-stimulated macrophages and septic mice. Additionally, administration of baicalin could markedly improve the survival of endotoxic mice. Taken together, baicalin may be an effective agent or lead compound in the prevention and treatment of inflammation-associated diseases such as sepsis.
Footnotes
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
The study was approved by the Animal Ethics Committee of National Defense Medical Center (ENDMC-11-115).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partly supported by a research grant from the Taipei Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation (TCRD-TPE-108-45).
