Abstract
A cohort of 12 dogs with severe transmural gallbladder necrosis is described. All dogs had cholecystectomies performed. In six dogs, perforation of the gallbladder was noted at surgery. Eight dogs survived the immediate postoperative period, and four dogs died. Histologically, inflammation was absent or minimal in all cases, suggesting that cholecystitis was not the cause of necrosis. Thrombi (n = 2) and atheromatous vascular changes (n = 1) represent possible vascular causes of this condition. The findings of coagulative necrosis in these 12 dogs are compatible with “gallbladder infarction,” and the authors propose this term to describe the histopathologic appearance of the gallbladder.
Cholecystitis is an uncommon clinical entity in dogs. 4,7,10 Cholecystitis may or may not be associated with cholelithiasis 1–3 and is rarely associated with biliary cancer. 3 Cholecystitis can result in gallbladder necrosis, perforation, and subsequent bile peritonitis. 2,8,9 Histopathologic descriptions of canine cholecystitis are limited. The majority of case series and reports that detail histologic changes of cholecystitis describe a lymphocytic/plasmacytic inflammation with varying degrees of neutrophil infiltration, mucosal hyperplasia, and pericholecystic inflammation. 7,10 Other studies refer to “necrotic” or “necrotizing” cholecystitis without a detailed description of the histopathologic findings. 4,8 To the authors' knowledge, the histopathology associated with severe, diffuse, transmural necrosis of the gallbladder wall has not been previously reported in dogs. This article details the clinical and pathologic features of a cohort of dogs that had this unique, previously undescribed gallbladder histopathology. This condition appears to have a vascular pathogenesis, and the authors propose the term “gallbladder infarction” to describe this entity.
As part of a larger study, records of all dogs with a surgical diagnosis of canine extrahepatic biliary disease between 1990 and 2003 at the Matthew J. Ryan Veterinary Hospital, University of Pennsylvania, were reviewed. Gallbladder biopsy samples from 25 dogs with a biopsy diagnosis of cholecystitis were evaluated by a single pathologist (M. J. Hendrick). From this group of 25 dogs, a cohort of 12 dogs had gallbladder histopathology compatible with infarction.
Affected breeds included mixed-breed dogs (n = 6), Cocker Spaniel (n = 1), German Shepherd (n = 1), Toy Poodle (n = 1), Lhasa Apso (n = 1) Cairn Terrier (n = 1), and Dachshund (n = 1). Age of affected dogs ranged from 4 to 14 years (median, 7 years). There were seven female spayed and five male castrated dogs. Preexisting medical conditions in these dogs included hypothyroidism (n = 3) and hyperadrenocorticism (n = 1). The presenting complaints for affected dogs included vomiting (n = 12), anorexia (n = 10), and diarrhea (n = 7) including hematochezia (n = 1), icterus (n = 4), and seizures (n = 1). The duration of clinical signs before presentation ranged from 1 to 14 days (median = 4 days).
Four dogs were febrile at presentation, with temperatures ranging from 103 to 105° F. All 12 dogs had a complete blood count and serum biochemical profile performed at admission. Ten dogs had an initial neutrophilia on complete blood counts ranging from 16,000 to 40,000 × 103 cells/µl. A left shift was present in three of these cases. Two dogs were mildly anemic (packed cell volume on admission = 24 and 26). Biochemical abnormalities at presentation included elevated alkaline phosphatase (n = 12, range 178–8,237, normal 20–155 U/liter), elevated alanine amino transferase (n = 8, range 96–4,211, normal 16–91 U/liter), hyperbilirubinemia (n = 7, range 1.4–24.1, normal 0–1 mg/dl), hypoalbuminemia (n = 5, range 1.1–2.0, normal 2.3–3.9 g/dl), and hypercholesterolemia (n = 4, range 420–900, normal 150–250 mg/dl). Eleven dogs had coagulation screens performed; of these, three were abnormal with 100, 100, and 200% elevations of the partial thromboplastin time, respectively.
Nine dogs had abdominal ultrasonography performed; abnormalities were seen in the gallbladder in seven of these cases. At surgery, six dogs had obviously ruptured gallbladders; the gallbladder was described as “distended” in the remaining six cases. There was no evidence of gallbladder torsion at surgery. Cholecystectomy was performed in all dogs. Two dogs had splenectomies performed because of obvious thrombosis of the splenic vessels. Only one dog had a cholelith identified in the gallbladder at surgery. Bacteria were cultured from three of the 12 dogs, which included Escherichia coli and Clostridium sp. Eight dogs survived and four dogs died during the immediate postoperative period of 2 weeks.
Histologically, there was transmural coagulative necrosis of the gallbladder in all dogs (Fig. 1). Inflammation was absent or minimal in all 12 cases. When present, scant neutrophils were present at the periphery of the serosa, often merging into a zone of young granulation tissue (Fig. 1). In 11 of 12 cases, all histologic sections examined from the gallbladder had complete diffuse coagulative necrosis. In the remaining case, there was an abrupt area of transmural necrosis adjacent to viable wall (Fig. 2). In this and one other case, organized fibrin thrombi were visible in an artery supplying the gallbladder (Figs. 2, 3). In another dog, arterioles adjacent to the gallbladder had atheromatous change characterized by deposits of foamy macrophages and cholesterol clefts in the wall (Fig. 4). One vessel had a mild acute vasculitis associated with these atheromas. When pericystic liver was included in the biopsy sample, the immediately adjacent parenchyma had multifocal to coalescing zones of hepatocyte loss with granulation tissue, fibrosis, and bile duct proliferation. These changes suggest an attempt by the liver to “wall off” the necrotic gallbladder. In addition, two dogs had histopathologic evidence of splenic thrombosis and infarction, and one dog had a concurrent hepatocellular carcinoma.
Gallbladder; canine. There is complete transmural coagulative necrosis of the gallbladder with adjacent granulation tissue. Bar = 200 µm.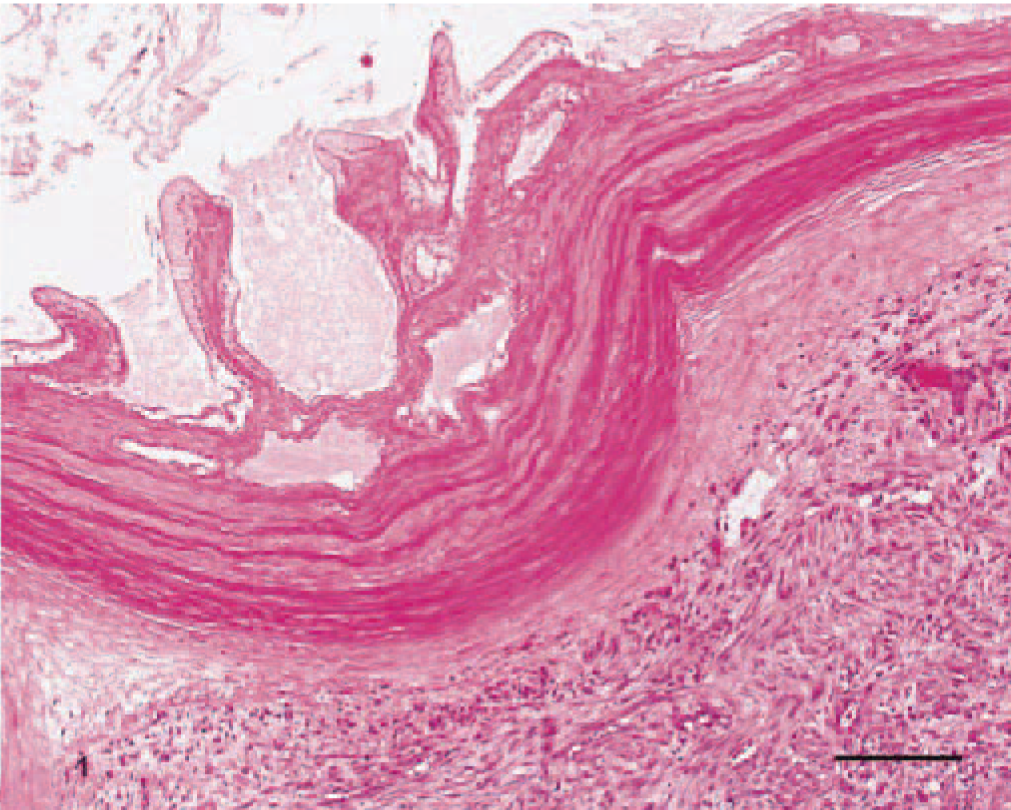
Gallbladder; canine. There is focally extensive transmural necrosis adjacent to viable wall. Note the thrombus at lower left (arrow). Bar = 400 µm.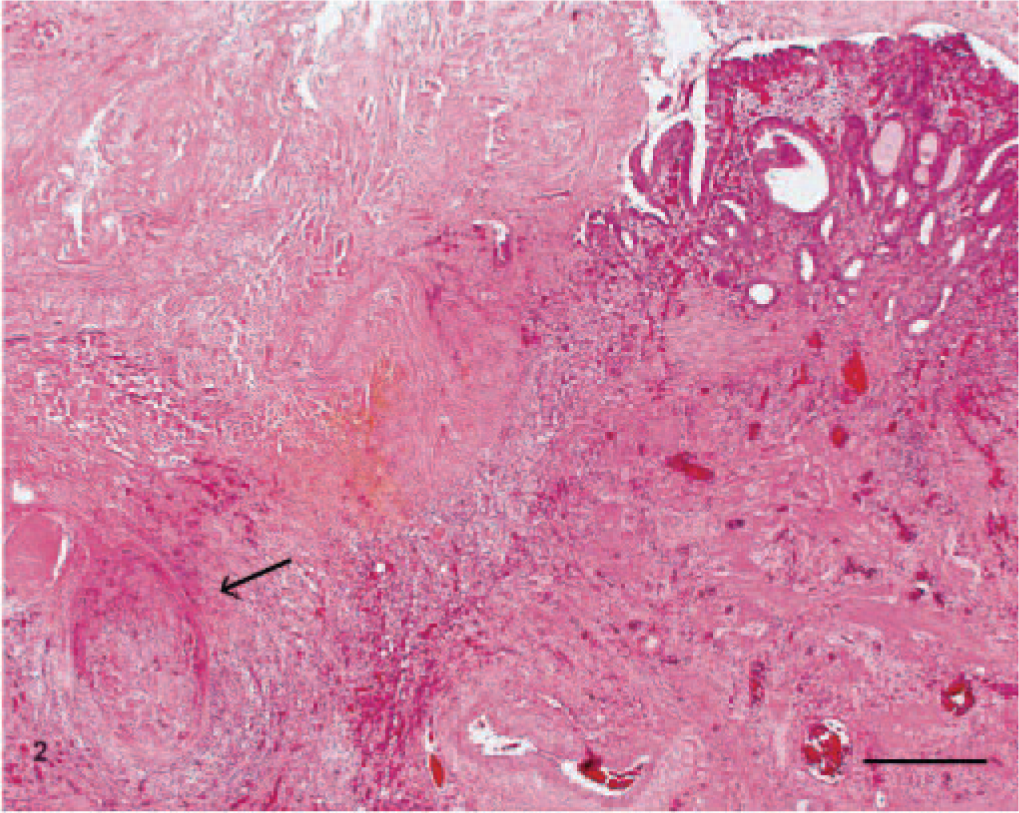
Gallbladder; canine. High-magnification view of organized fibrin thrombus adjacent to infarcted gallbladder wall. Bar = 200 µm.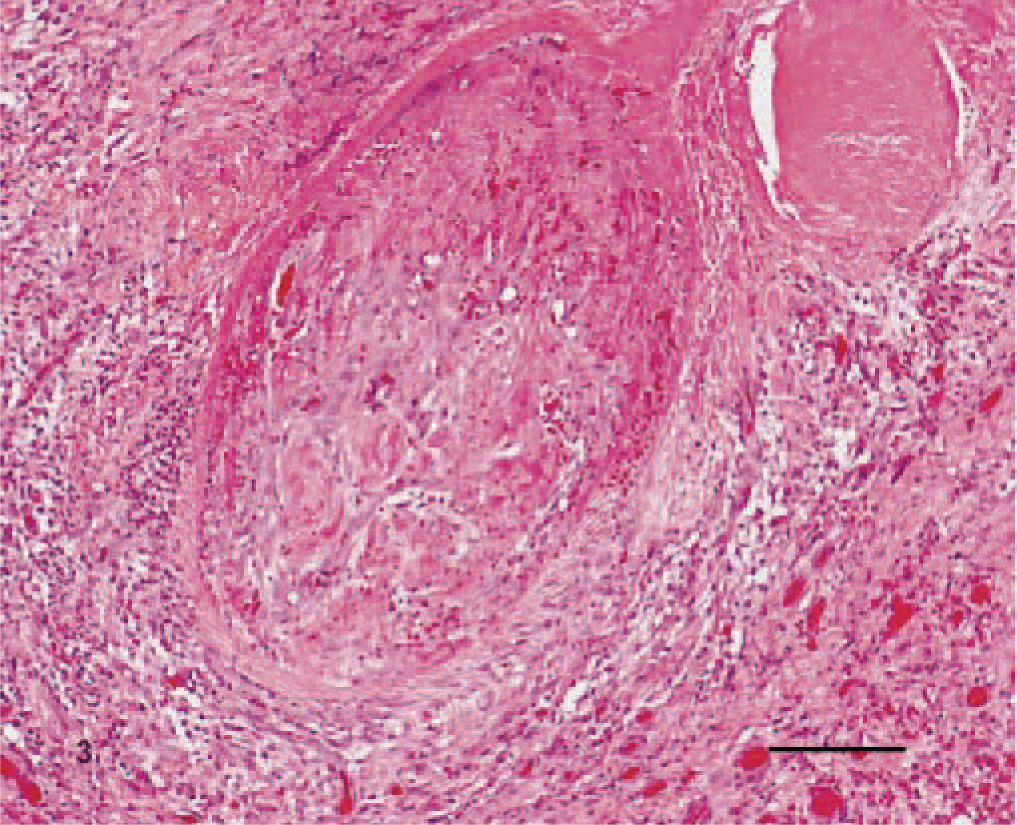
Gallbladder; canine. Atheromatous change of an artery adjacent to the gallbladder wall. Bar = 200 µm.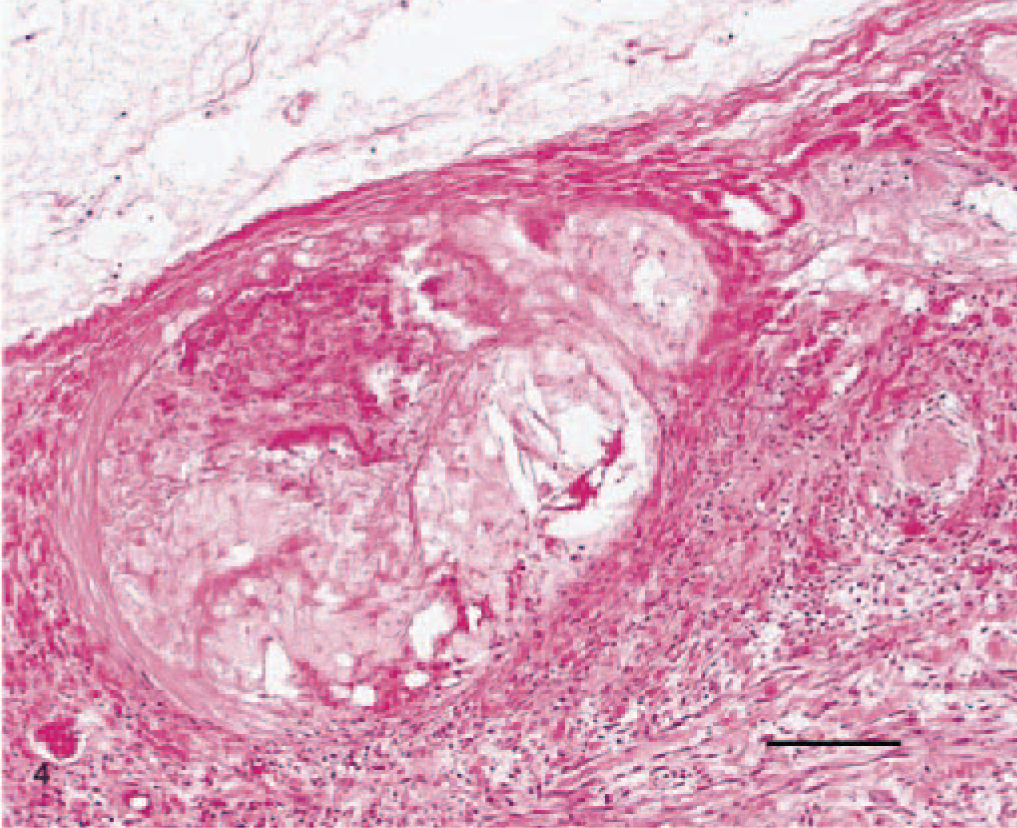
The histopathologic findings of coagulative necrosis in these 12 dogs are compatible with gallbladder infarction. Although coagulative necrosis can be caused by ischemia, anoxia, or direct toxin exposure, the finding of thrombi in two dogs and atherosclerosis in one dog supports ischemia and subsequent infarction. Thrombosis and infarction were also present in the spleen in two other dogs, suggesting a more generalized thrombotic state in those two cases. Hypercoagulability and thrombosis usually occur secondary to an underlying disease process. One dog in this report was being treated for hyperadrenocorticism, a disease associated with hypercoagulability.
The closest human corollary to the condition seen in these dogs is acalculous cholecystitis. The more severe form of this condition, in which the gallbladder becomes necrotic and perforates, is sometimes referred to as “gangrenous cholecystitis.” 5 Ischemia plays a role in this condition as evidenced by microangiographic studies showing poor filling of the gallbladder capillaries; this is in contrast to calculous cholecystitis, where regular filling of the gallbladder capillary network and dilated arterioles were visualized. 6
In humans, the ischemia in acalculous cholecystitis is multifactorial and most often secondary to hypovolemic or septic shock, major trauma, or cardiopulmonary bypass. 1,6 None of these conditions were present in the dogs in this study, although many of them had subjective and objective indicators of poor systemic perfusion. In contrast to human acalculous cholecystitis, these perfusion deficits appeared to be associated primarily with the biliary disease and subsequent peritonitis rather than major trauma or a primary insult to another organ system. In addition, the gallbladder necrosis seen in dogs in this report was more severe than that generally seen in human acalculous cholecystitis and had less associated inflammation than reported in the human disease. 1 Three dogs had obvious reasons for the gallbladder ischemia. In one dog, arterioles adjacent to the gallbladder had atheromatous change, presumably associated with the dog's hypothyroidism and hypercholesterolemia. Two other dogs had evidence of organized fibrin thrombi in an artery supplying the gallbladder. Neither of these dogs had abnormalities on preoperative coagulation profiles. It is impossible to determine whether the thrombi were primary causative lesions or secondary to regional injury associated with bile leakage and peritonitis.
Experimentally, severe transmural gallbladder necrosis cannot be easily produced in dogs by ischemia alone. Gallbladder necrosis has been experimentally created in dogs using a model of prolonged gallbladder ischemia and reperfusion. 11 To achieve the degree of gallbladder necrosis seen in our clinical cases, the hepatic artery and cranial mesenteric vein were occluded for either 45 or 90 minutes followed by 90 and 180 minutes of reperfusion, respectively. 11
This report describes a cohort of 12 dogs with severe transmural gallbladder necrosis that was associated with gross perforation at surgery in six cases. The cause of this condition was unclear in the majority of cases, although a vascular pathogenesis was possible in some dogs. The authors propose the term gallbladder infarction to describe the histopathologic appearance of the gallbladder. The prognosis for this condition is guarded with a 33% mortality rate in the dogs in this report.
Footnotes
Acknowledgements
The authors gratefully acknowledge the support of the Commonwealth of Pennsylvania.
