Abstract
A cholecystectomy was performed on a 10-year-old spayed female mixed-breed dog with chronic weight loss, persistently increased liver enzyme activities, and cholecystomegaly identified by ultrasonographic examination. A subsequent diagnosis of a biliary carcinoid was made based on a neuroendocrine-type histologic pattern, cytoplasmic argyrophilia by Grimelius staining, immunopositivity for chromogranin A, and the ultrastructural finding of cytoplasmic secretory granules in neoplastic cells. Extrahepatic biliary carcinoid tumors are rare tumors of humans and have not been documented in domestic animals.
Keywords
In humans and animals, carcinoid tumors are uncommon neoplasms that arise from dispersed cells of the neuoroendocrine system in the gastrointestinal tract, biliary system, pancreas, and lung. These cells belong to one of two functional groups: 1) the amine precursor uptake and decarboxylation cells that produce serotonin and adrenocorticotrophic hormone, or 2) those cells capable of synthesizing low molecular weight polypeptide or protein hormones such as chromogranin, cholecystokinin and secretin. Definitive diagnosis of carcinoid tumors is based on histologic features, immunostaining for secretory products such as chromogranin, and ultrastructural identification of neuroendocrine secretory granules.2 In dogs, hepatic, gastrointestinal, and pulmonary carcinoids have been reported.5–7 Intestinal carcinoids have also been documented in a horse and a cow.2 In horses, three cases of maxillary sinus carcinoid tumors have been reported as well.8 In humans, carcinoid tumors are frequently found in the gastrointestinal system (74%) and the bronchopulmonary system (25%). Within the gastrointestinal tract, the most common sites include the small bowel (29%), appendix (19%), and rectum (13%).3
In this case, a 10-year-old spayed female mixed-breed dog initially presented for weight loss and persistent vomition. No abnormalities were noted on physical examination. Serum biochemistry abnormalities included increased liver enzyme activities, including aspartate aminotransferase (625 U/L; reference range 10–60 U/L), alanine aminotransferase (2,225 U/L; reference range 8–57 U/L), lactate dehydrogenase (531 U/L; reference range (0–250 U/L), and alkaline phosphatase (4,460 U/L; reference range 10–140 U/L) as well as hyperbilirubinemia (Total bilirubin 5.6 mg/dL; reference range 0–0.6 mg/dL) consistent with hepatocellular damage and cholestasis. During the next 8 weeks, progressive weight loss was accompanied by lethargy, intermittent fever, and increased liver enzyme activities. Abdominal radiographs demonstrated generalized hepatomegaly with abnormal calcification. Subsequent ultrasonographic examination identified gallbladder wall calcification accompanied by intraluminal contents suggestive of cholelithiasis. The liver and the remainder of the abdominal examination were otherwise within normal limits with no mass lesions detected.
On the basis of these findings, a cholecystectomy was performed 10 weeks after initial presentation. The gallbladder was enlarged (5 cm in diameter) with a thickened wall containing an exophytic, intraluminal mass 3 cm in diameter (Fig. 1). The solid mass had a sessile attachment at the neck of the gallbladder and largely occluded the lumen. Several smaller nodular masses (3–5 mm in diameter) of similar consistency were attached to the serosal surface of the gallbladder. In addition, the gallbladder mucosa was diffusely hyperplastic. An exploratory laparotomy performed at the time of cholecystectomy did not identify other abdominal lesions.
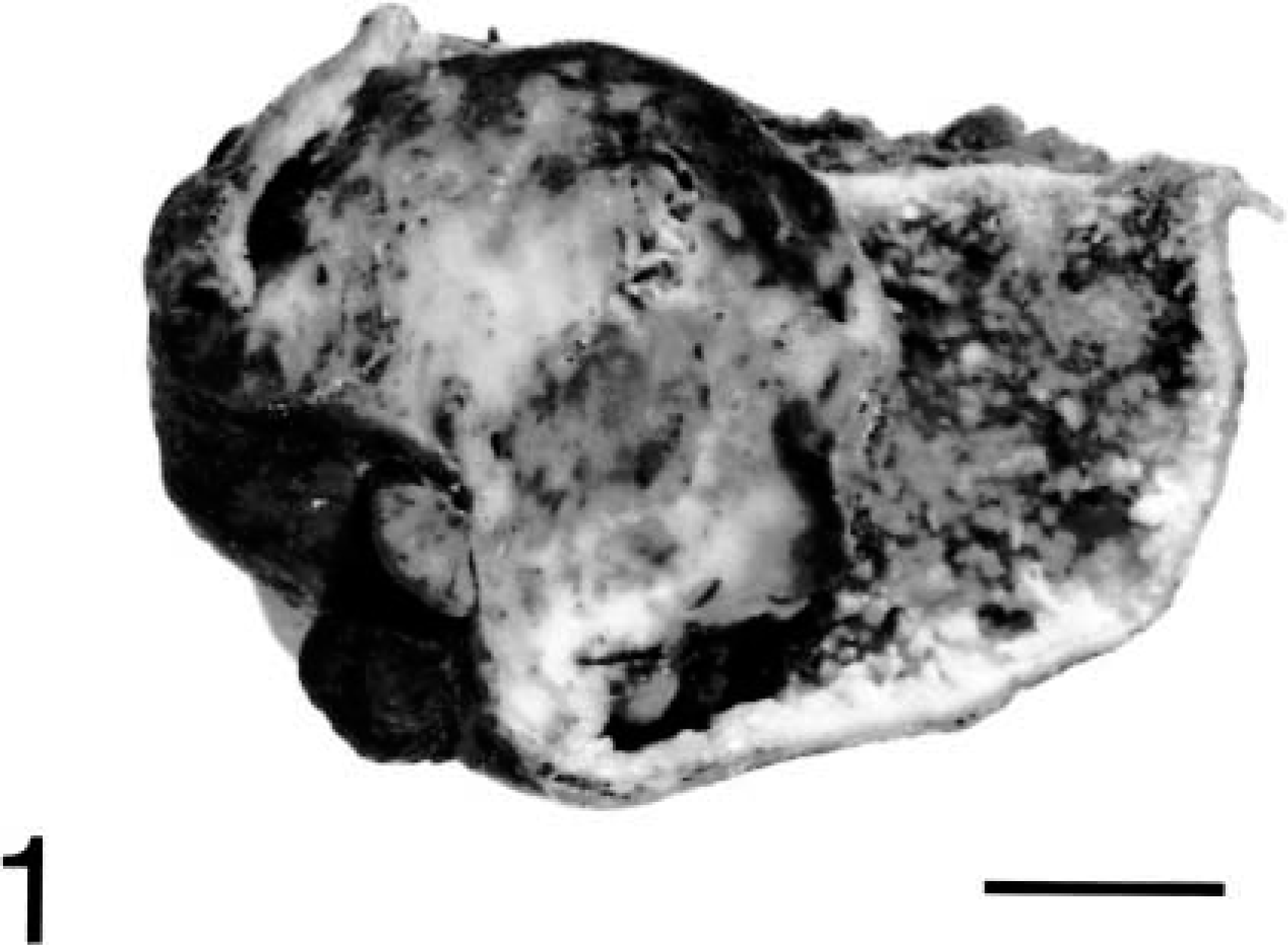
Gallbladder, cross-section; doe. A solid round mass attached to the neck of the gallbladder extends into the gallbladder lumen. Diffuse cystic mucosal hyperplasia is also apparent. Bar = 1 cm.
Microscopically, a transmural, nonencapsulated, circumscribed mass extended into and occluded the gall bladder lumen (Fig. 2). The mass was composed of closely packed, monomorphic round cells arranged in small nests and packets subdivided by a fine fibrovascular stroma (Fig. 3). The cytoplasm contained fine, punctate eosinophilic granules admixed with multifocal vacuolated regions. Nuclei were uniformly round, centrally located, and ranged from normochromatic to hyperchromatic. One to two nucleoli were present in most nuclei. Mitotic activity was rare (<1 per five 400× microscopic fields). Neoplastic cells also were present within small- and medium-sized vessels in the gall bladder wall (Fig. 2). The remaining gallbladder mucosa exhibited cystic mucosal hyperplasia with cystic structures often containing mineralized debris. Subjacent submucosa was edematous and diffusely infiltrated by lymphocytes. The luminal surface of the mass was covered by necrotic epithelium mixed with granulation tissue, multifocal hemorrhage, and a mixed inflammatory infiltrate including neutrophils and lesser numbers of lymphocytes.
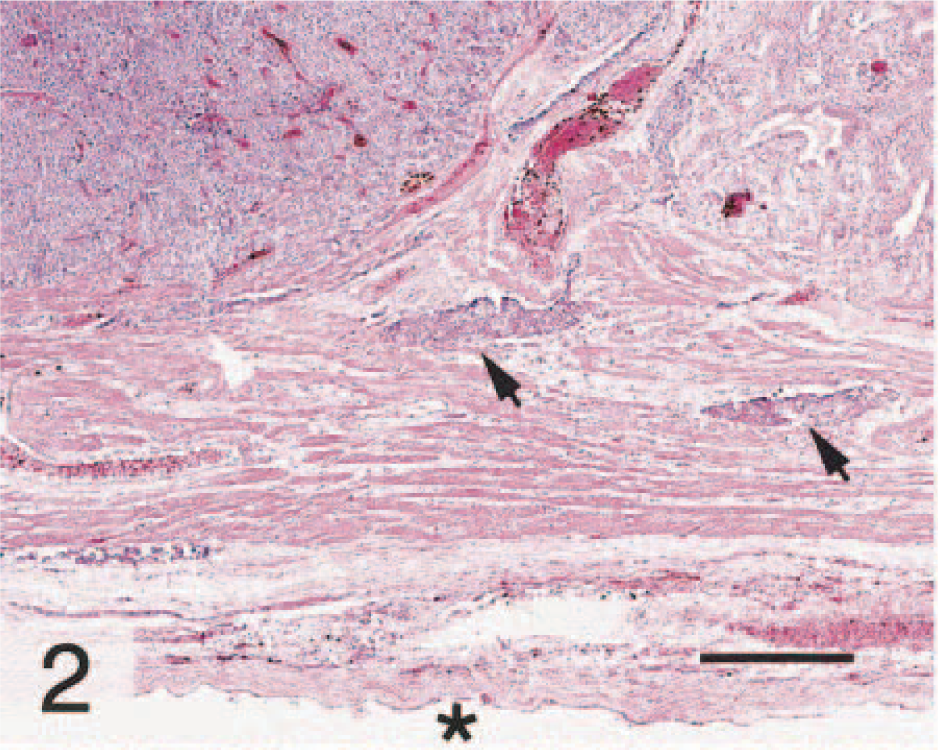
Gallbladder; doe. HE. The mass (white arrowhead) extends from the lumen deep into the gallbladder wall, with neoplastic cells also located within mural vessels (black arrows). An asterisk denotes the serosal surface. Bar = 320 μm.
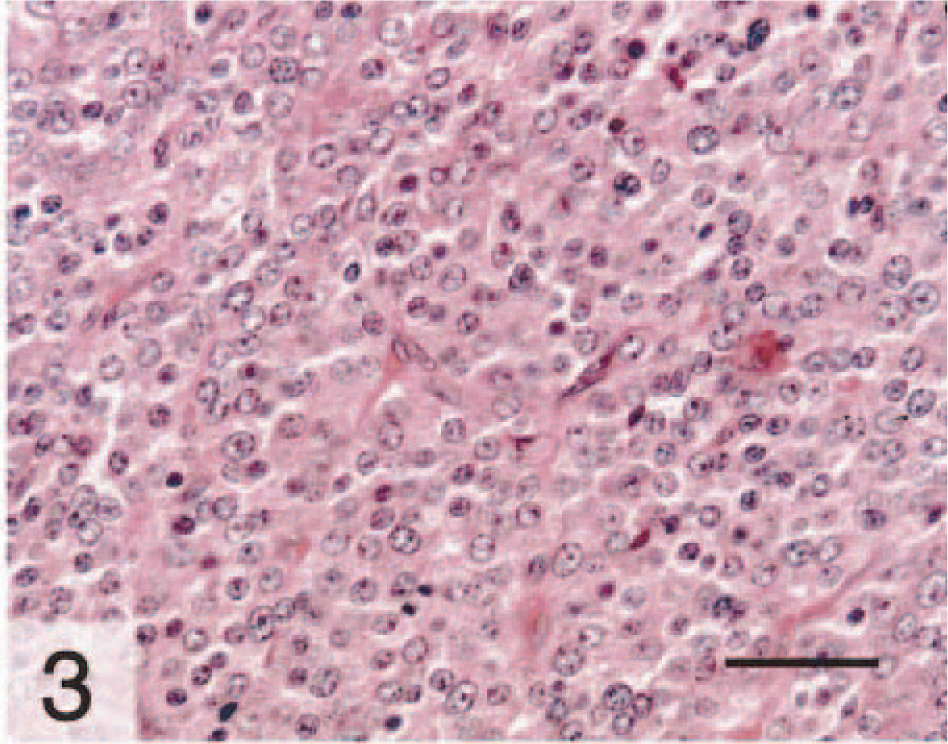
Gallbladder; doe. Closely packed, monomorphic round cells with round nuclei are arranged in small nests and packets subdivided by a fine stroma, a pattern consistent with a neuroendocrine origin. HE. Bar = 60 μm.
As these morphologic features were suggestive of a neuroendocrine origin, additional stains and electron microscopy were used to confirm this diagnosis. Argyrophilia was detected in the cytoplasm of neoplastic cells by the Grimelius reaction, a technique used to identify cytoplasmic argyrophilic granules in cells of neuroendocrine origin (Fig. 4). Additionally, neoplastic cells were immunopositive for chromogranin A following immunohistochemical staining with an antibody (Zymed, So. San Francisco, CA; diluted 1:50) that detects chromogranin A (Fig. 5). Immunohistochemistry was performed using the avidin–biotin–peroxidase conjugate technique. Transmission electron microscopy confirmed the presence of numerous intracytoplasmic, round, membrane-bound, electron-dense secretory granules with a dense central core approximately 200–250 nm in diameter scattered in the cytoplasm of the neoplastic cells (Fig. 6). All of these findings are consistent with a diagnosis of a carcinoid tumor.
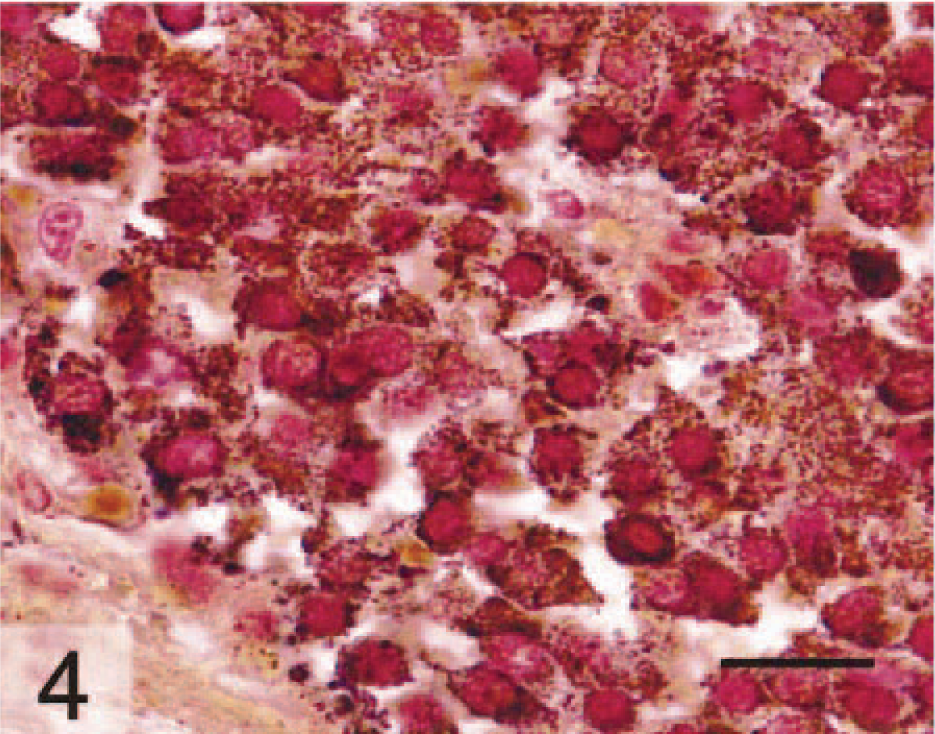
Gallbladder; doe. Cytoplasmic argyrophilia (dark brown granules) in neoplastic cells denotes presence of neurosecretory granules. Modified Grimelius stain. Bar = 30 μm.
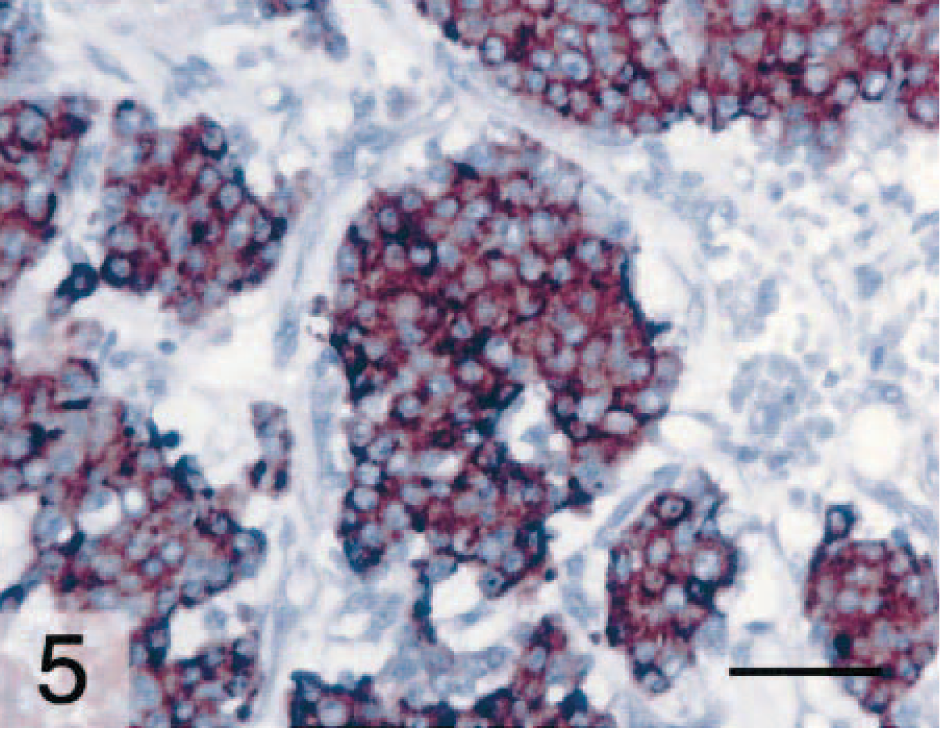
Gallbladder; doe. The cytoplasm of neoplastic cells is immunopositive (brown) for chromogranin A. ABC method, hematoxylin counter-stain. Bar = 50 μm.
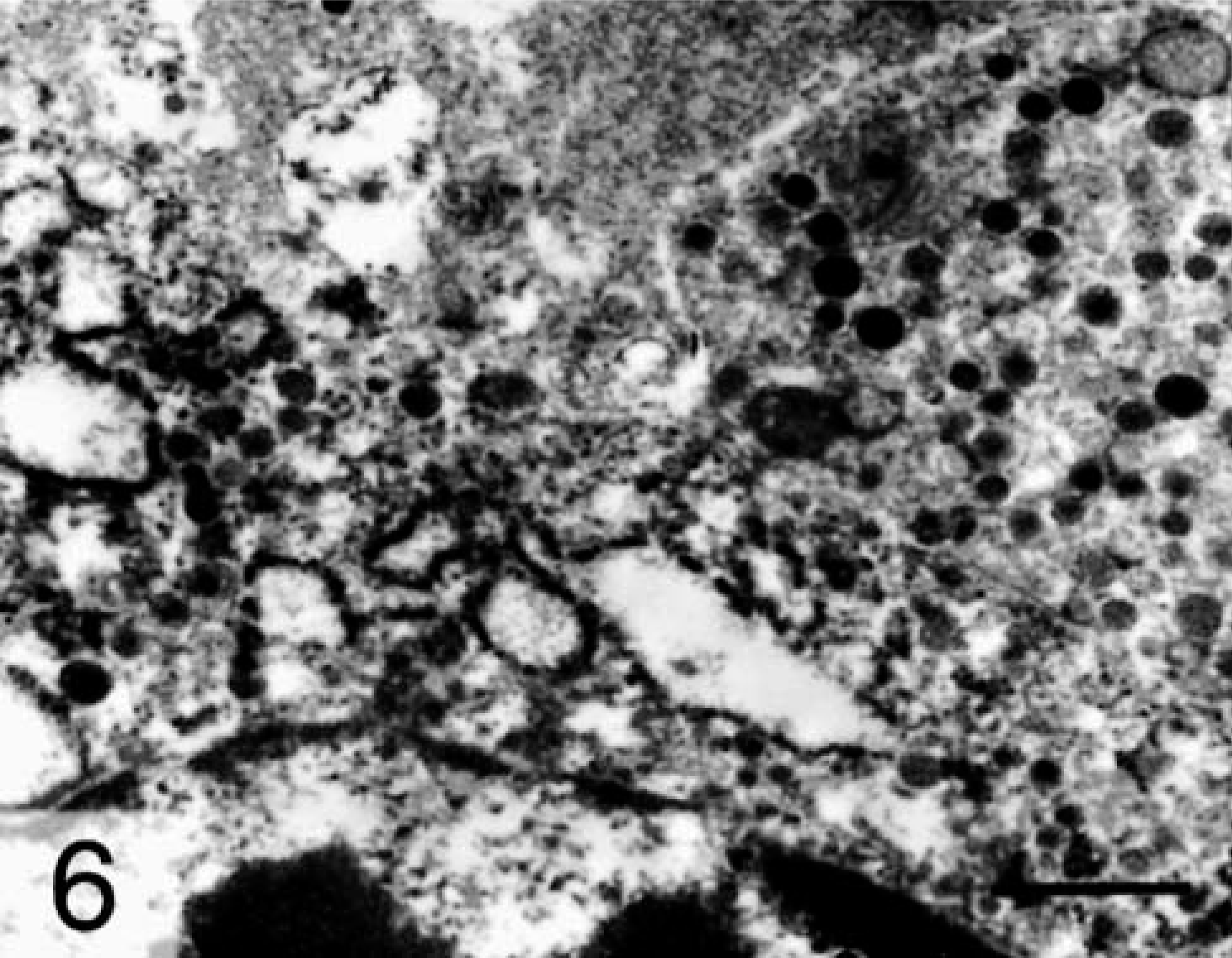
Gallbladder; doe. Numerous membrane-bound secretory granules with dense cores are scattered in the cytoplasm of a neoplastic cell. The nucleus is located at the bottom of the field. Transmission electron micrograph, Bar = 0.8 μm.
After cholecystectomy, the dog developed intermittent vomiting and diarrhea and was euthanatized 8 months later. On the basis of the clinical evaluation and the unlikelihood of metastasis to the gallbladder, this carcinoid tumor appeared to originate in the gallbladder, but this could not be demonstrated conclusively because the owners declined a postmortem examination. Endocrine cells have been identified in the mucosa of the gallbladder neck and may have been the origin of this neoplasm.4 Biliary carcinoid tumors are rare neoplasms of the extrahepatic biliary tree including the gallbladder with approximately 30 cases reported in humans and none in domestic animals.1 Although potentially malignant in humans, carcinoid tumors are usually slow growing with a 5-year survival rate of 94% if localized, 64% if regional metastasis is present, and 18% if distant metastasis has occurred at the time of diagnosis.3 In this case, the presence of neoplastic cells within vessels suggested the potential for metastatic dissemination. Although adenomas and adenocarcinomas arising in the gallbladder mucosa have been reported in dogs, carcinoid tumors also should be included in differential diagnoses when extrahepatic biliary masses are identified.2
Footnotes
Acknowledgements
We would like to thank Dr. John D. Strandberg for assistance with electron microscopy and Mr. Fred Dubs for photography. This work was supported by NIH RR07002.
