Abstract
A 21-month-old domestic Hanford pig (Sus scrofa domestica) in a 1-year study for experimental myocardial infarction was euthanized at the end of the study. One week earlier, the animal had symptoms and elevated clinical chemistry results suggestive of hepatobiliary disease, which resolved after medical therapy. At necropsy, the gallbladder was markedly enlarged, discolored, and had a thickened wall. Within the gallbladder, there was abundant friable green-brown material. A culture of the gallbladder luminal material yielded Clostridium perfringens type A. Histopathology of the gallbladder demonstrated multifocal areas of necrosis of varying depths, admixed with an inflammatory infiltrate that was also observed on the serosa and within the associated adipose tissue. Luminal material was composed of cellular debris and bile sludge admixed with numerous bacterial rods. Smooth-muscle hypertrophy of numerous small arterioles with narrowed lumina was observed in the gallbladder. A diagnosis of acalculous cholecystitis presumably because of ischemia of the gallbladder with secondary clostridial infection was made. To the authors' knowledge, this is the first reported case of acalculous cholecystitis with evidence of vascular compromise in a pig, as well as cholecystitis secondarily attributed to C. perfringens type A.
Acalculous cholecystitis (AC), inflammation of the gallbladder without evidence of cholelithiasis, is an uncommon disease in humans. 3 When diagnosed, the disease is usually seen in critically ill patients, especially after cardiac and abdominal vascular surgery. 13 It was also documented in cases of severe trauma, burns, and septicemia. 3,8 Experimental induction of this disease in swine was documented. 15 A spontaneous case of AC with evidence of vascular compromise, and cholecystitis secondarily attributed to Clostridium perfringens type A (CPA) in a pig, is reported in the present study.
A 9-month-old male Hanford pig (Sus scrofa domestica) underwent coronary arterial coil embolization for induction of myocardial infarction. The procedure was approved by the animal care and use committee of the National Heart, Lung and Blood Institute of the National Institutes of Health. The animal recovered uneventfully from surgery and was placed on enteric-coated aspirin (325 mg), lisinopril a (5 mg), and atenolol b (25 mg) daily. Approximately 1 year later, the animal was anesthetized for an magnetic resonance imaging (MRI) with a cocktail of ketamine c (6 mg/kg), xylazine d (2 mg/kg), atropine e (0.02 mg/kg), and butorphanol f IV (0.2 mg/kg), and was maintained on isoflurane with mechanical-assisted ventilation. The animal recovered uneventfully from the procedure. The next day, the animal attempted to crawl through a primahedron enrichment device g that had been placed in its kennel but became lodged in the device. After several unsuccessful attempts to remove the device, the animal was sedated with a ketamine, xylazine, atropine, and butorphanol combination to facilitate removal. Three days later, the animal was depressed and anorexic, with a temperature of 39.8°C (reference [ref.] range: 38–39°C). The animal was icteric and had severe abdominal pain. The animal was sedated with Telazol h (5 mg/kg), blood was collected for a routine complete blood count (CBC) and serum chemistry, and an abdominocentesis and electrocardiogram were performed. The serum was icteric, but, otherwise, the CBC count was within laboratory reference interval. Serum biochemical abnormalities included elevated albumin 3.6 g/dl (ref. interval: 1.9–3.4 g/dl), total bilirubin 3.1 mg/dl (ref. interval: 0–1.0 mg/dl), and potassium 7.7 mEq/liter (ref. interval: 4.4–6.7 mEq/liter) concentrations, as well as elevated activities of alanine aminotransferase (ALT) 85 U/liter (ref. interval: 31–58 U/liter), aspartate aminotransferase (AST) 339 U/liter (ref. interval: 32–84 U/liter), lactate dehydrogenase (LDH) 1572 U/liter (ref. interval: 380–634 U/liter), and creatinine phosphokinase (CPK) 2833 (no ref. interval). The diagnostic laboratory reported that serum sample was mildly (1+) hemolyzed, which can contribute to elevations in total bilirubin (2–3 times normal) and potassium concentrations. In addition, AST and CPK activities may be increased. Dehydration is the most likely cause of the hyperalbuminemia. The most common causes of hyperbilirubinemia are hemolytic disease and hepatobiliary disease. Because hematologic parameters were within reference intervals, hepatobiliary disease was suspected. Although the total bilirubin concentration was only 3 times above the upper discrimination value of the reference interval, a test result of 3.1 mg/dl was significantly elevated over values typical for this colony of pigs, even when hemolysis was present, and it was interpreted to represent a real elevation. Elevations of ALT and AST activity are associated with hepatocellular damage from any cause. 18 Causes of hyperkalemia include increased potassium intake, translocation of potassium from intracellular to extracellular fluid (e.g., hemolysis), and decreased urinary potassium excretion. Because this pig was anorexic on presentation and had no other indications of renal insufficiency, the hyperkalemia could be because of trauma from being trapped in the enrichment device but was most likely secondary to the hemolysis of the blood sample. Lactate dehydrogenase activity is found in many body tissues, and nonspecific elevations of this enzyme are of little diagnostic value. Creatinine phosphokinase activity is a highly sensitive and specific indicator of muscle damage, although modest elevations in enzymatic activity, such as seen in this pig, can be a result of intramuscular injections. Because the results of the CBC, abdominocentesis, and electrocardiogram were within the reference intervals, a diagnosis of hepatobiliary disease was most probable.
The animal was started on treatment with fentanyl i (75 μg/hr transdermal) and cefazolin j (25 mg/kg). Two days later, the animal's attitude improved, and its appetite was normal. The research protocol ended 2 weeks later, and the animal was euthanized. The heart was harvested by the investigator, and the remaining carcass was submitted for pathologic evaluation. At necropsy, the gallbladder was turgid and distended to approximately 3 cm in diameter by 8–9 cm in length. The wall of the gallbladder was diffusely thickened 2–3 times normal size and was mottled, opaque, whitish-gray, and purple. Upon opening the gallbladder, abundant brown friable material admixed with fibrin was observed partially adhered to the mucosa. A culture of the friable material within the gallbladder yielded a pure culture of C. perfringens. Genotyping by an independent laboratory k revealed CPA. Other lesions included multifocal areas of fat necrosis within the mesentery adjacent to the stomach, multiple gastric ulcers of the pars esophagea, erosions of the esophageal mucosa, and mucosal reddening of the cecum and spiral colon. Bacterial cultures were not obtained from any other organ system.
Representative samples of numerous tissues were collected in 10% formalin, processed, and embedded in paraffin, and 5-μm hematoxylin and eosin–stained sections were evaluated. Microscopic evaluation of the gallbladder revealed coalescing areas of mucosal ulceration, with large foci of necrosis that extended down into the lamina propria and tunica muscularis. The necrotic foci were composed of cellular debris, numerous debris-laden macrophages, and degenerate neutrophils (Fig. 1). The remaining areas of the gallbladder were composed of moderate-to-marked lymphoplasmacytic infiltrates admixed with fewer neutrophils, eosinophils, and macrophages. Lymphoplasmacytic infiltrates of varying intensity were observed in the serosa and attached adipose tissue, with occasional granulomatous steatitis. The gallbladder lumen contained large amounts of necrotic cellular debris admixed with degenerate neutrophils and myriad colonies of large Gram-positive rod bacteria (Fig. 2). Numerous small and moderately sized arterioles of the submucosa had mild-to-moderate smooth-muscle hypertrophy, which caused narrowing of the vascular lumina (Fig. 3). The venous and lymphatic vessels were dilated and congested throughout the gallbladder. Other lesions observed included a mild typhlocolitis. The gross and histologic findings of the gallbladder were consistent with a diagnosis of AC.
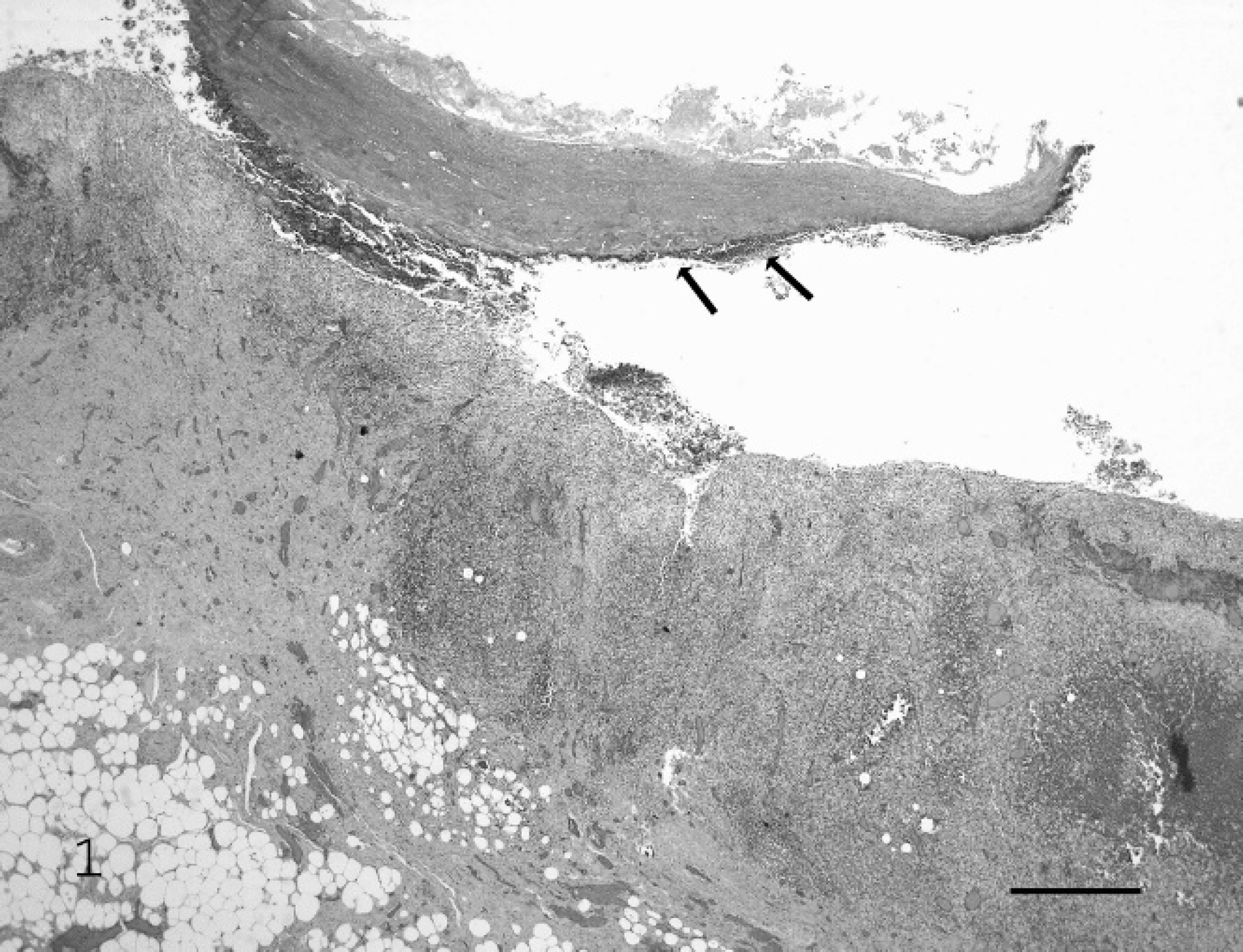
Gallbladder; pig. The wall of the gallbladder is markedly thickened with a cellular infiltrate that extends into the serosa adipose tissue. Note the abundant necrotic debris (arrows) in the lumen. Hematoxylin and eosin stain. Bar = 670 μm.
Acalculous cholecystitis has broad morphologic characteristics, which range from erosions and ulcerations with a neutrophilic-to-lymphoplasmacytic infiltrate, to gangrene and emphysema of the gallbladder. 6 It has been shown to be associated with trauma, 1 burns, hypovolemic and septic shock, 8 and coronary and abdominal vascular surgeries. 13 The prominent feature of AC is the absence of cholelithiasis, which, in general, is known to contribute to cholecystitis. The etiology of AC is believed to be multifactorial. Factors or mechanisms involved in the development of AC can be categorized as luminal-obstructive, ischemic, or infective.
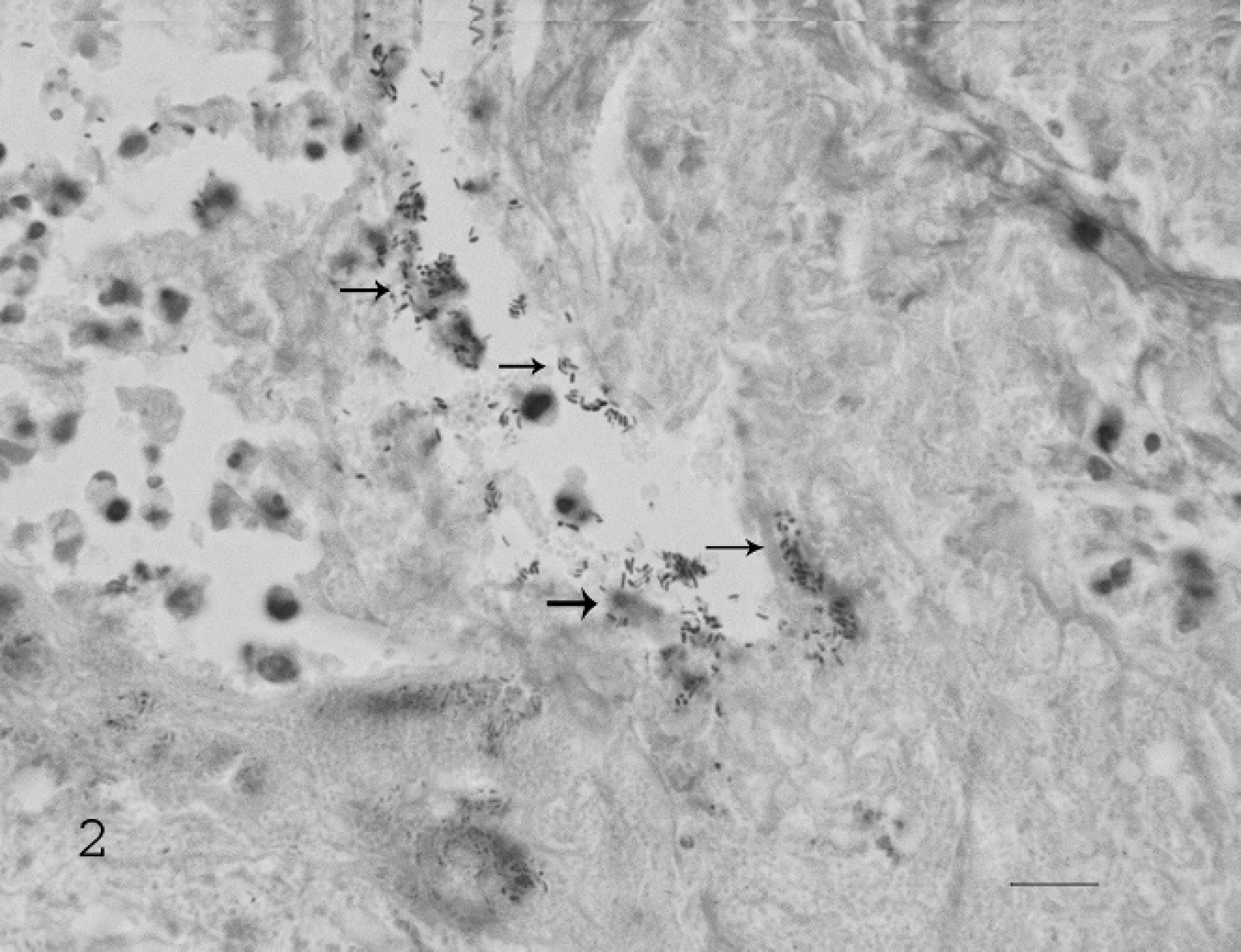
Gallbladder; pig. Large numbers of Gram-positive rods (arrows) present in the luminal necrotic debris. Gram stain. Bar = 50 μm.
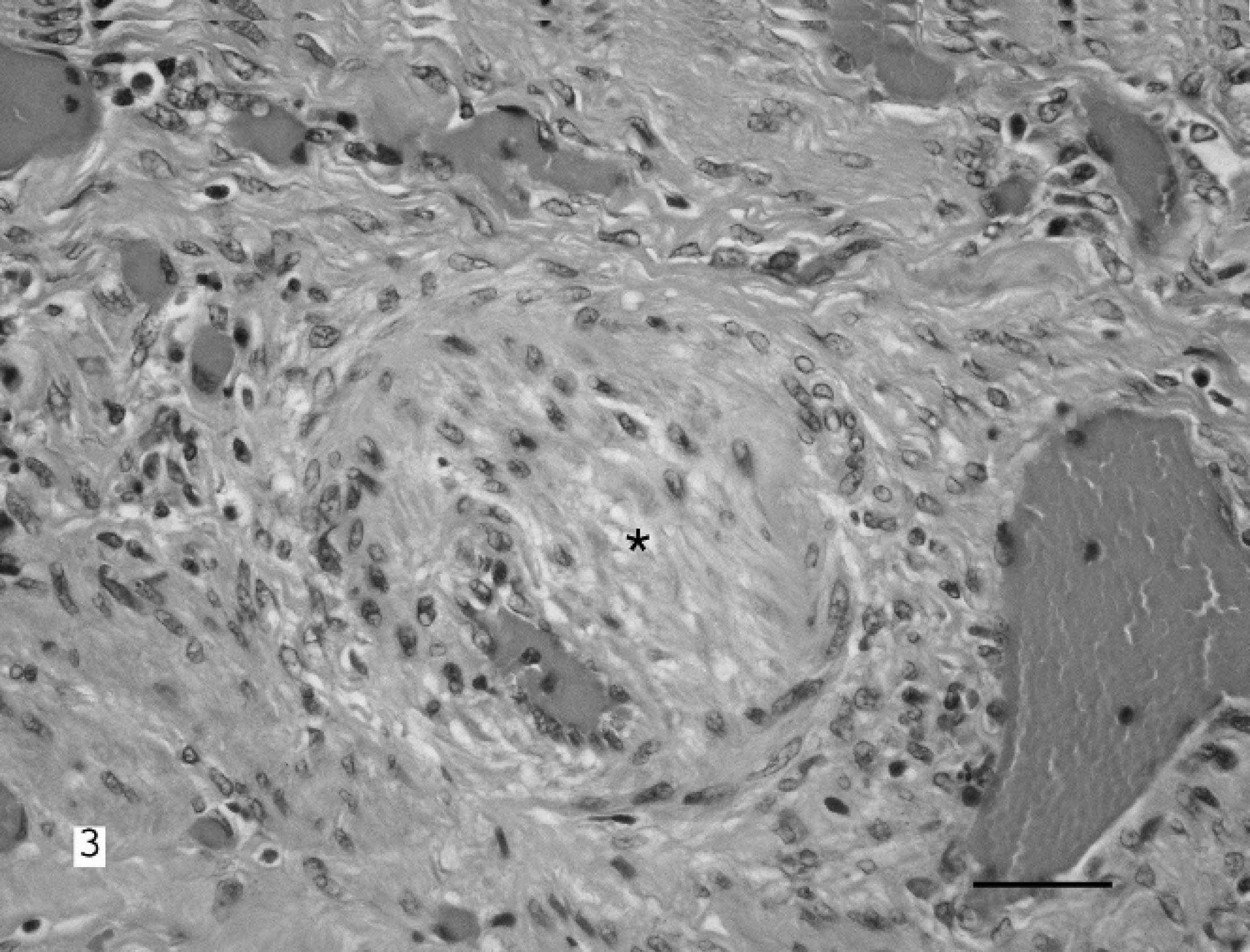
Gallbladder; pig. Submucosal arteriole with marked smooth-muscle hypertrophy (asterisk) and corresponding lumen narrowing. Diffuse ectasia and congestion of surrounding venules is also observed. Hematoxylin and eosin stain. Bar = 150 μm.
Results of studies suggest that vasospasm in the gallbladder wall may cause local ischemia during cardiogenic shock (arterial wall spasm, passive vasoconstriction that leads to edema). 2,17 In this pig, numerous small and moderately sized arterioles had mild-to-moderate smooth-muscle hypertrophy, which caused narrowing of the vascular lumina, which potentially could have contributed to the ischemic changes observed in the gallbladder. The exact cause for the gallbladder arteriole smooth-muscle hypertrophy was not determined. However, it may have been secondary to hemodynamic alterations caused by the experimentally induced myocardial infarction.
A few reports documented AC that occurred 1–3 weeks after cardiovascular surgery in humans. 10,13 Although the pig in the current report had coronary-vessel ligation, with subsequent myocardial infarction, this procedure had been performed 1 year previous to the diagnosis of AC and thus was most likely not a factor involved in the development. However, it is possible that the animal had an acute coronary event during an MRI or during the stressed situation it experienced when lodged in the enrichment device. Albeit unlikely, the trauma and stress induced from the animal having its body lodged in an enrichment device may have contributed to AC, because AC can occur after injury, such as when there has been abdominal trauma. 3
Porcine bile is sterile. The growth of bacterial organisms is normally inhibited by the bile-salt concentration. 8 However, cholestasis reduces bile-salt concentration, thus allowing bacteria to proliferate in the gallbladder. 7 Bacterial entrance into the biliary system occurs primarily through duodenal-biliary reflux. Normally, the sphincter of Oddi acts to prevent the entry of bacteria into the common bile duct from the duodenum. Continuous bile flow, with secretion of mucus and immunoglobulin, also acts to ensure bile-duct sterility. 5 Intestinal motility also helps prevent bacterial ascension. In addition to obstruction of the sphincter of Oddi, events that cause intestinal hypomotility, such as enteritis, metabolic, or respiratory acidosis 16 (which might have occurred from positive-pressure ventilation during an MRI procedure), or pharmacologic agents may allow duodenal-biliary reflux. Ketamine and opioid derivatives (i.e., butorphanol, buprenorphine, fentanyl) 11,12 have been shown to decrease gastrointestinal motility and may have contributed, in part, to bacterial entry into the gallbladder. Pathogenic bacteria cultured from the bile of diseased gallbladders in pigs have included Escherichia coli, Streptococcus spp., Staphylococcus spp., Klebsiella spp., Enterococcus spp., Serratia marcescens, Citrobacter spp., Proteus spp., Enterobacter spp., Salmonella spp., and Clostridium perfringens. 15 Clostridium perfringens type A is considered part of the normal flora of the porcine intestinal tract. 14 However, CPA has been implicated in enteritis in piglets and young animals up to 5–7 weeks of age. 9 Clostridium perfringens type A causes a transient diarrhea with moderate morbidity and low mortality. Although the intestinal tract was not cultured from the pig in the present study, gastrointestinal abnormalities were not clinically observed (i.e., diarrhea), and microscopic evaluation of the cecum and colon demonstrated a mild nonspecific typhlocolitis. Also, the age of the animal would not correlate with typical CPA enteritis. Clostridium perfringens type A has not been reported to cause hepatobiliary disease in pigs.
The use of narcotic agents, such as butorphanol, buprenorphine, and fentanyl, which were administered to the pig within 1 week of euthanasia, may be responsible for or may have compounded the AC by causing constriction or spasm of the gallbladder neck and cystic duct, with resultant elevation in intrabiliary pressure. 4 The increased pressure can lead to cholestasis and subsequent formation of bile sludge, which alone can contribute to mucosal damage. The increased gallbladder-wall tension can also cause impairment of mucosal perfusion. Mucosal perfusion impairment occurs by compromising arterial, lymphatic, and venous flow, which can leave it susceptible to infection, gangrene, or necrosis. The venous and lymphatic vessels were dilated and congested throughout the gallbladder in this pig, which indicated a possible outflow obstruction. Fasting may also cause cholestasis. Normally, food that enters the duodenum stimulates the duodenal mucosa to secrete cholecystokinin, which causes the gallbladder to contract. 5 The animal had been anorexic for 5 days and then began eating normally 2 weeks before euthanasia.
Acalculous cholecystitis has been proposed to be a multifactorial disease process. The pig in the current study had been exposed to a number of predisposing factors, including opioid derivatives, mechanical ventilation, myocardial infarction, trauma, vascular changes, bacterial infection, and fasting. Acalculous cholecystis as well as CPA cholecystitis should be considered in the differential diagnoses for a novel cause for porcine gallbladder disease.
Acknowledgements. The authors would like to thank Jorge Chavez, Annie Merriweather, and Frances Benedetti for technical assistance. This work was supported by the Intramural Research Program of the National Institutes of Health, Office of Research Services.
Footnotes
a.
West-Ward Pharmaceutical Corp., Eatontown, NJ.
b.
Sandoz Inc., Princeton, NJ.
c.
Fort Dodge Animal Health, Fort Dodge, CO.
d.
Lloyd Inc., Shenandoah, IA.
e.
APP Pharmaceuticals Inc., Schaumburg, IL.
f.
Vedco Inc., St. Joseph, MO.
g.
Primate Products Inc., Woodside, CA
h.
Fort Dodge Animal Health, Fort Dodge, CO.
i.
Janssen Pharmaceutical Products LP, Titusville, NJ.
j.
Baxter International Inc., Deerfield, IL.
k.
University of Missouri Veterinary Medical Diagnostic Laboratory, Columbia, MO.
