Abstract
Primary epithelial tumors of the gallbladder are rarely reported in animals. In this study, 9 aged pigs (6–12 years old) were histopathologically examined for gallbladder proliferative lesions. At necropsy, a large gallstone occupied the lumen of the gallbladder of 3 pigs. Histopathological examination revealed chronic cholecystitis in all 9 pigs, mucosal hyperplasia in 2 pigs, adenoma in 1 pig, and adenocarcinoma in 2 pigs. Bacilli were detected in the gallbladder lumen of 6 pigs by Warthin-Starry stain. Mucosal hyperplasia, adenoma, and adenocarcinoma were characterized by papillary projections of the mucosa with occasional acinar structures. Tumor invasion of the surrounding tissue was observed in the cases of adenocarcinoma. On Alcian blue and periodic acid–Schiff double-stained sections, the acinar structure of gallbladder mucosa in chronic cholecystitis and mucosal hyperplasia was stained in a mosaic pattern, indicating pyloric gland metaplasia. The results of immunohistochemistry revealed a CD10-positive epithelial brush border and mucin (MUC) 2–positive goblet cells in chronic cholecystitis, adenoma, and adenocarcinomas, indicating intestinal metaplasia. Immunoreactivity of MUC5 AC and cytokeratin 19 was weaker in adenoma and adenocarcinomas compared with the normal and hyperplastic gallbladder mucosa. The number of p53-positive nuclei and the Ki-67 index were higher in adenocarcinomas compared with benign lesions. These results suggest that chronic cholecystitis associated with gallstones and/or bacterial infections may contribute to metaplastic changes and development of gallbladder tumors in aged pigs. Alteration of mucin, cytokeratin, and p53 profiles in gallbladder proliferative lesions in pigs were similar to that in humans, suggesting a common pathogenesis in tumor development.
Keywords
Primary epithelial tumors of the gallbladder are rarely reported in animals. According to a survey of neoplastic diseases in cattle, gallbladder adenomas are slightly more common in cows compared with other domestic animals. 3,4 Gallbladder adenocarcinomas have been reported in the dog, 6,8,19,28 cat, 27 cow 3,4 and pig. 2,30 In gallbladder adenocarcinomas of the cow and pig, mucin-secreting columnar epithelial cells were arranged in a papillary pattern. 2 –4,30 In gallbladder adenocarcinomas of the dog and cat, by contrast, neoplastic cells were composed of acinar or tubular structures. 8,27,28
Gallbladder tumors are also uncommon in humans. 24 Human gallbladder adenocarcinoma is an aggressive disease with a poor prognosis, and metastases to the lymph nodes and liver often occur. Prolonged exposure to gallstones can cause chronic inflammation of the mucosa, which is considered a major risk factor for developing gallbladder adenocarcinoma. 7,29 Repetitive stimulation to the mucosa and subsequent tissue repair result in metaplasia and/or dysplasia of the gallbladder mucosa, which are considered precancerous lesions progressing to adenocarcinoma. 14,33,36,37 Sequential histological and molecular changes over a period of several years or decades have been studied in human gallbladder diseases. 5 Mutation of TP53, a tumor suppressor gene, is commonly found in human gallbladder adenocarcinoma, and accumulation of p53 protein is observed in the tumor cells in more than 50% of patients. 5,15,17,18,21,38 In addition, the production of specific mucins lining the mucosal surface have been associated with tumor development and progression. 1,7,11
The present study examined hyperplastic and neoplastic lesions in the gallbladder of aged pigs and evaluated their association with the presence of gallstones and mucosal inflammation.
Materials and Methods
Cases and Samples
Nine pigs examined in the present report had been kept in the University Farm of the Graduate School of Agricultural and Life Sciences, the University of Tokyo. The pigs were raised on a farm at Fukushima prefecture during the great East Japan earthquake in March 2011 and had been subsequently kept in the university farm for observational studies. They were housed in groups of 3 or 4 in a room with free access to tap water and were fed a commercial diet (MARUCHIRAKKU; Chubu Shiryo, Aichi, Japan) at 1.0 to 1.2 kg per head twice a day. The pigs in this study were emaciated with anorexia and depression and were euthanized because of poor prognosis (Table 1). Complete necropsy was performed on all 9 pigs, which were 6 to 12 years old. At necropsy, visceral organs including the gallbladder were collected and fixed in 10% neutral-buffered formalin.
Signalment and Major Pathologic Findings in 9 Pigs With Lesions of the Gallbladder.
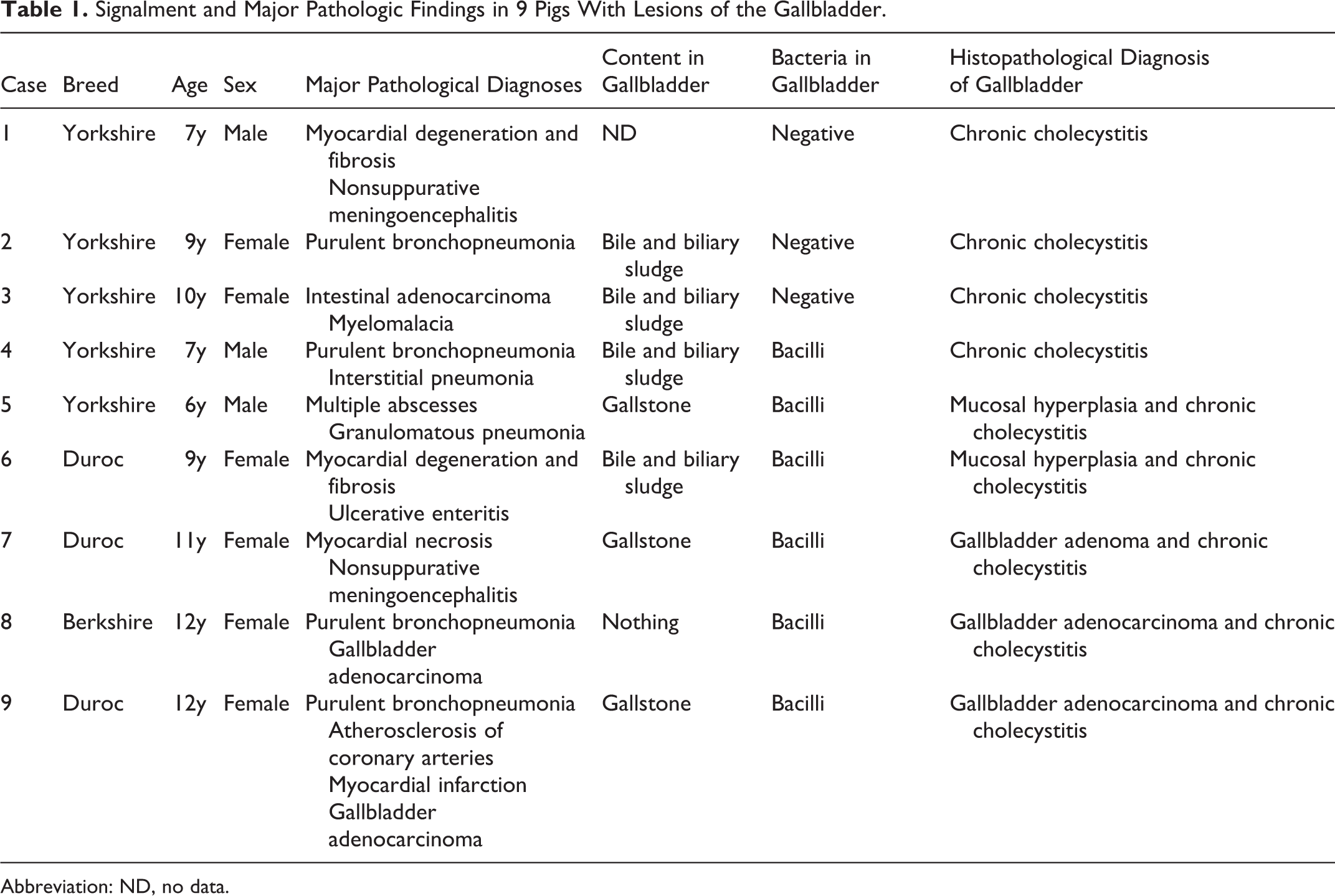
Abbreviation: ND, no data.
Histopathology
Formalin-fixed tissues were embedded in paraffin. Four-micrometer-thick sections were stained with hematoxylin and eosin (HE) for general pathological examinations. Further examinations for the detection of bacterial agents and mucins were performed on gallbladder tissue sections with Warthin-Starry staining and double staining of Alcian blue (AB) and periodic acid–Schiff (PAS), respectively. For AB-PAS double staining, deparaffinized sections were first stained with AB (pH 2.5) and subsequently stained with PAS. Tissue sections of the normal gallbladder, gastric pylorus, and duodenum from a 1-year-old 3-crossbred pig were also stained with AB-PAS.
Immunohistochemistry
Immunohistochemistry (IHC) was performed on gallbladder tissue sections as described in Table 2, according to the guidelines set forth by the American Association of Veterinary Diagnosticians Subcommittee on Standardization of Immunohistochemistry. 31 The detection system used in the present study was the peroxidase-conjugated EnVision Polymer Detection System (Agilent Technologies, Santa Clara, CA). Immunoreactivity of the tumor cells was evaluated for each marker as follows: –, negative; +, <25% of tumor cells are positive; ++, 25% to 50% of tumor cells are positive; +++, >50% of tumor cells are positive. The number of Ki-67–positive cells was counted in 1000 epithelial cells on each slide, and the percentage of Ki-67 positivity (Ki-67 index) was calculated in each case. The number of p53-nuclear positivity (p53-positive nucleus number) was counted in 1000 epithelial cells on each slide, and the percentage of p53-nuclear-positive cells was calculated in each case.
Primary Antibodies Used for Immunohistochemistry.
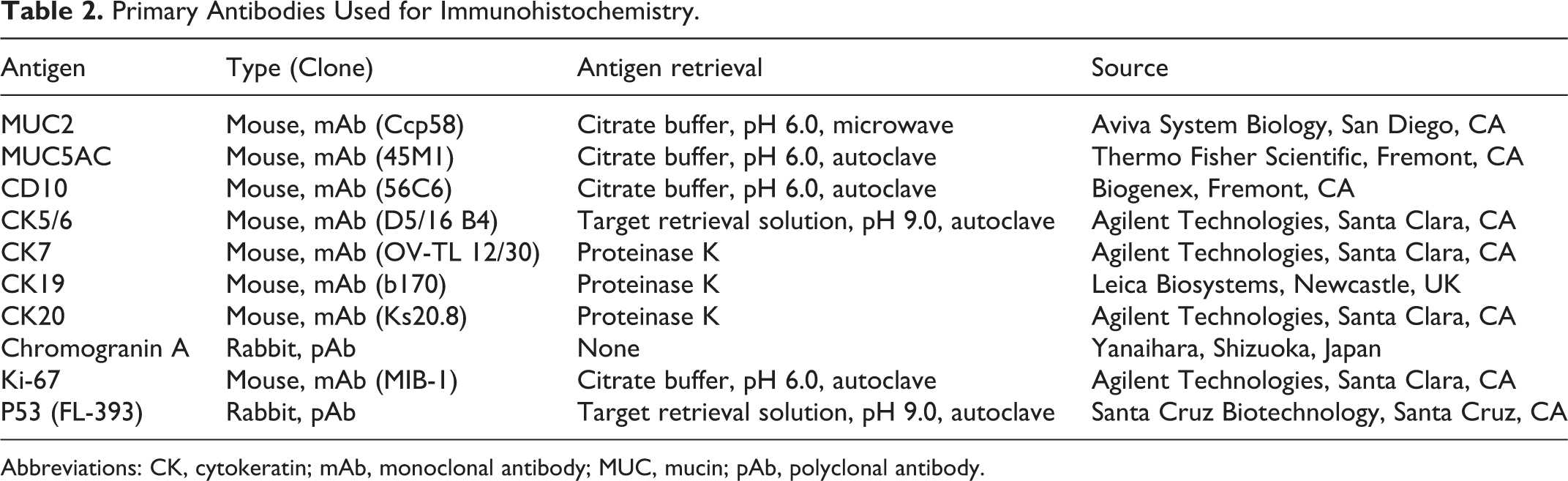
Abbreviations: CK, cytokeratin; mAb, monoclonal antibody; MUC, mucin; pAb, polyclonal antibody.
Results
Gross Findings
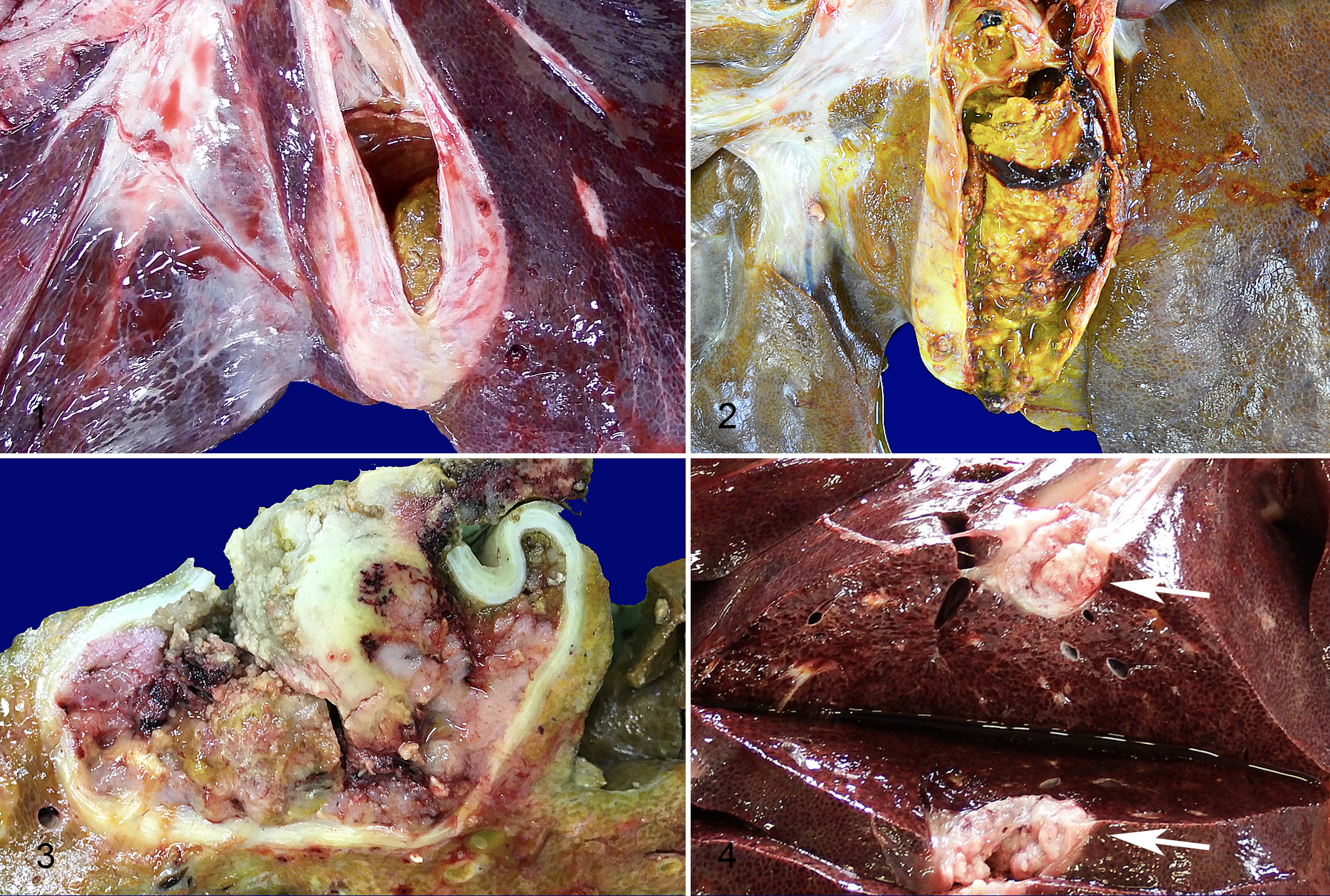
At necropsy, dark green friable biliary sludge mixed with bile filled the gallbladders of 4 pigs (cases 2, 3, 4, and 6). A large gallstone was observed together with bile in the gallbladder of 3 pigs (cases 5, 7, and 9; Figs. 1 and 2). The gallstones were yellow to ocher in color and irregular in shape (cases 5, 7, and 9; Figs. 1 and 2). There was no significant lesion in the gallbladder mucosa of cases 1 to 4. Gallbladder mucosa was slightly thickened in cases 5 and 6. In case 7, a focal nodule was located in the fundus of the gallbladder. In case 9, the gallbladder wall was diffusely and irregularly thickened, and multifocal hemorrhage was observed on the surface of the thickened mucosa (Fig. 3). The liver was yellowish, and the intrahepatic bile ducts were severely dilated with bile, indicating the biliary tract obstruction. In case 8, the lumen contained no bile fluid or gallstones but was replaced by the proliferation of soft white neoplastic tissues (Fig. 4).
Histopathology
Diffuse infiltration of lymphocytes and plasma cells as well as hyperplastic lymphoid follicles were observed in the gallbladder mucosa of all 9 pigs (Fig. 5). Warthin-Starry staining revealed bacilli in the gallbladder lumen of 6 pigs (cases 4–9) and in the gallstone of 1 pig (case 8). Diffuse or focal hyperplasia of the gallbladder mucosa with acinar structures and elongated folds was observed in 2 pigs (cases 5 and 6; Fig. 6). The acinar cells of the mucosa in chronic cholecystitis and mucosal hyperplasia had clear mucinous cytoplasm with an eccentric nucleus (Fig. 6, arrow). In contrast, surface epithelial cells composing the folds had the eosinophilic cytoplasm, and goblet cell-like cells were occasionally observed.
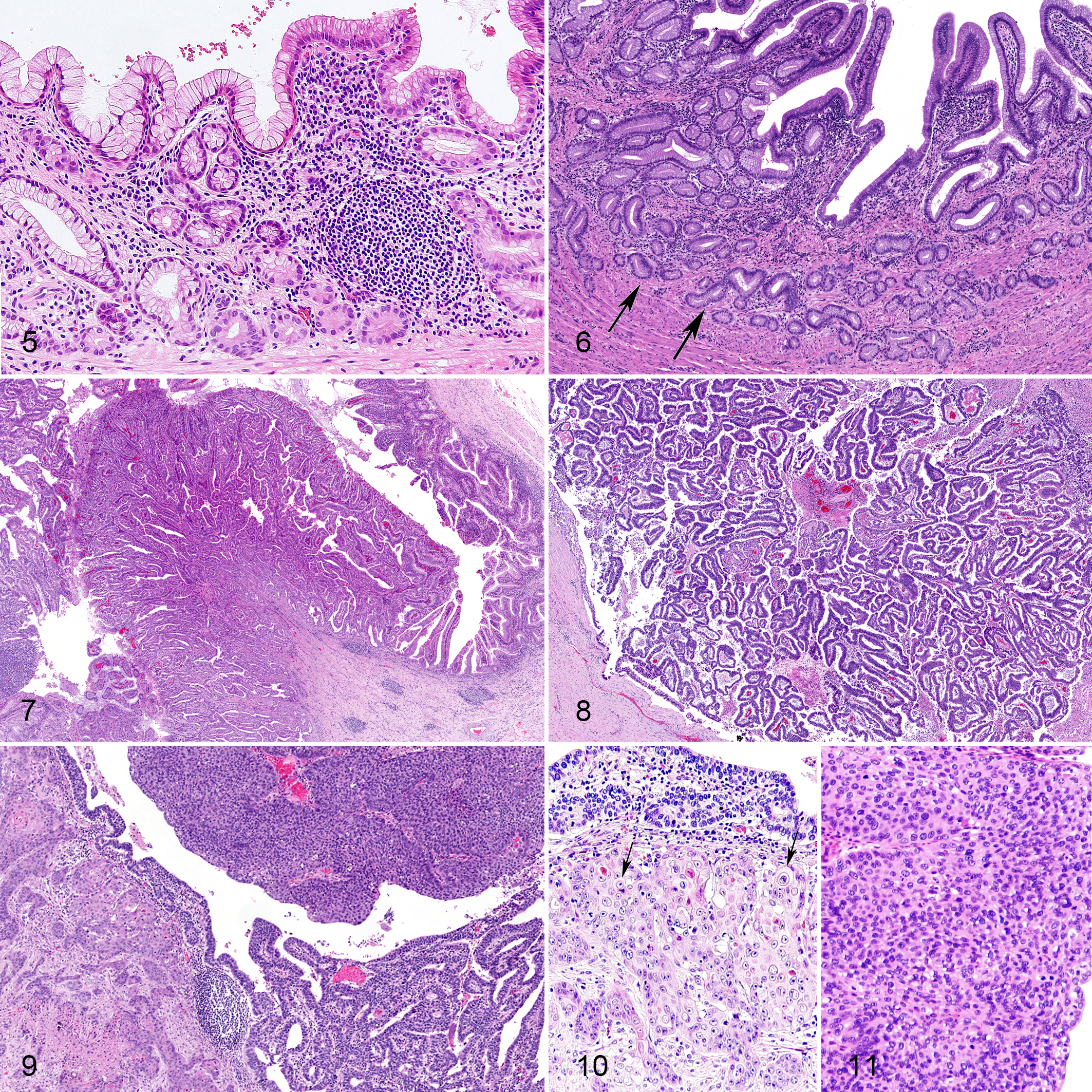
Neoplastic lesions of the gallbladder in 3 pigs included adenoma (case 7) and adenocarcinoma (cases 8 and 9). Adenoma was composed of tubular and papillary structures with thin stroma lined by a single layer of neoplastic epithelial cells (Fig. 7). Most of the neoplastic epithelial cells had eosinophilic cytoplasm, occasionally with a brush border on the cell membrane. Goblet cell-like cells were also occasionally observed. Anisokaryosis was mild, and nuclei were located in the basal side of the cell. In the pig with gallbladder adenoma (case 7), the extrahepatic duct was obstructed by a gallstone, and the mucosa was necrotic.
Adenocarcinomas were composed of papillary structures lined by single to multiple layers of epithelial cells that invaded the muscularis of the gallbladder (Figs. 8 and 9). In case 9, the tumor cells further invaded the liver and the gastrohepatic lymph node. Severe anisokaryosis and loss of cellular and nuclear polarity of the neoplastic cells were evident in adenocarcinoma. Squamous metaplasia with keratinization was occasionally observed in the invasive lesion of adenocarcinoma (Fig. 10, arrows), and basal cell-like tumor cells were arranged in solid sheet structures (Fig. 11).
Mucins and Immunohistochemical Profiles
Results of AB-PAS stain and IHC for normal tissues and inflammatory and proliferative gallbladder lesions are summarized in Table 3.
Mucin and Immunohistochemical Profiles of Epithelial Cells in Control Tissues and the Gallbladder Lesions.
Abbreviations: AB-PAS, double staining of Alcian blue and periodic acid–Schiff; MUC, mucin; CK, cytokeratin; Cg A, chromogranin A; NT, not tested.
aImmunohistochemical scoring: –, negative; +, 1%–25% positive cells; ++, 26%–50% positive cells; +++, >50% positive cells.
bPercentage of Ki-67 positivity (Ki-67 index) was calculated based on the number of Ki-67-positive cells counted in 1000 epithelial cells.
cPercentage of p53 positivity (p53-positive nucleus number) was calculated based on the number of p53-nuclear positive cells counted in 1000 epithelial cells.
On AB-PAS double-stained sections, acid mucin is stained blue with AB, neutral mucin stains magenta with PAS, and a mixture of both stains purple. Surface epithelial cells and glandular acinar cells of the normal gallbladder mucosa were stained purple in the luminal surface and in the cytoplasm (Suppl. Fig. S1). In the normal pyloric mucosa, surface epithelial cells were stained magenta (Suppl. Fig. S2), while cells of the pyloric gland were stained in a mosaic pattern of magenta and blue (Suppl. Fig. S3). In the normal duodenal mucosa, the brush border of the surface epithelial cells was stained magenta, and the cytoplasm of the goblet cells was stained purple (Suppl. Figs. S4 and S5).
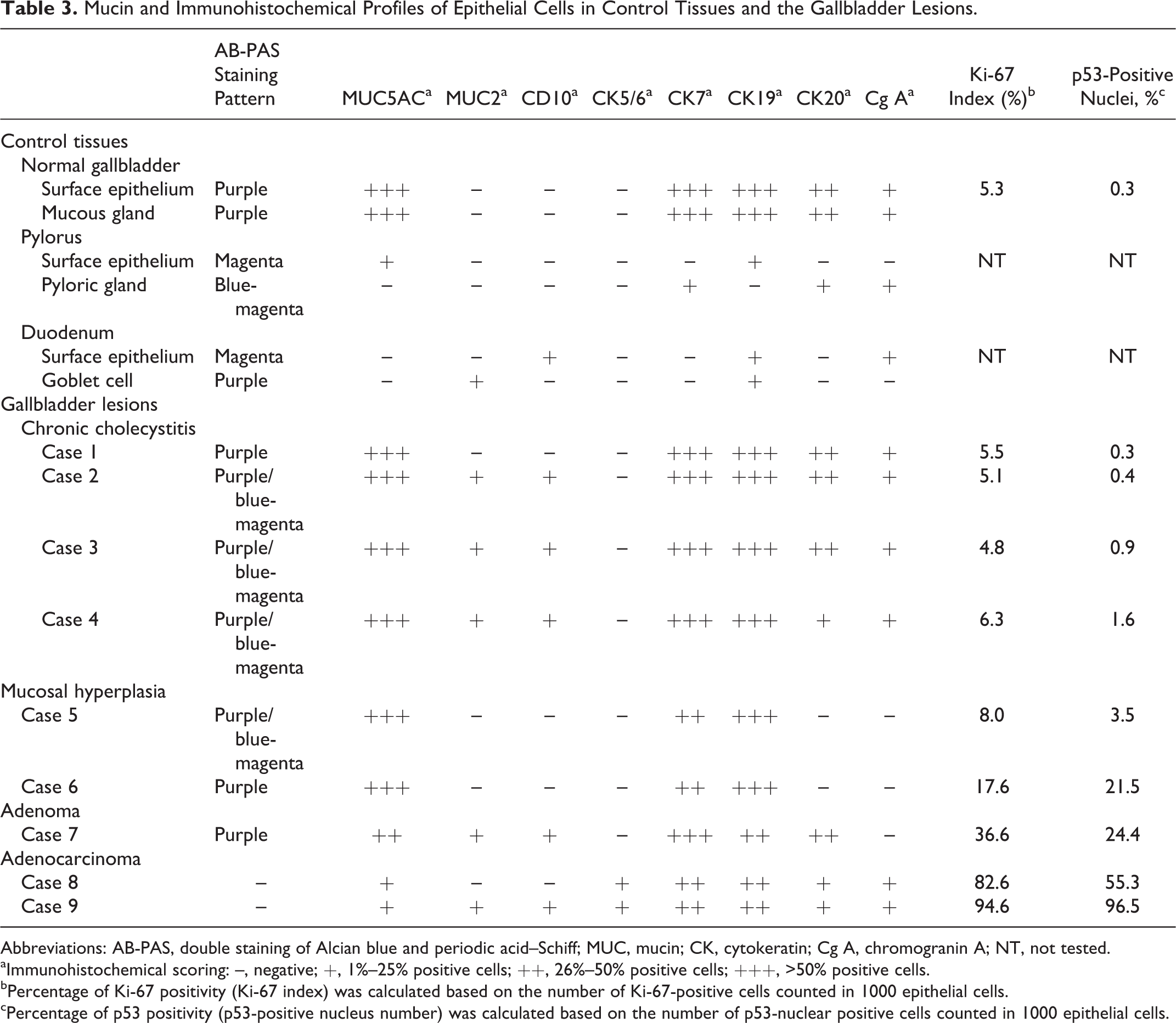
Most surface epithelial cells in chronic cholecystitis, mucosal hyperplasia, and adenoma were stained purple on the luminal surface and diffusely purple in the cytoplasm (Fig. 12), similar to the staining pattern of the normal gallbladder mucosa. In some areas of chronic cholecystitis and adenoma, goblet cell-like cells with purple cytoplasm and magenta brush border were observed (Fig. 13), which resembled the appearance of the duodenal mucosa. Most of the acinar cells of chronic cholecystitis, mucosal hyperplasia, and adenoma were stained purple on the luminal surface and in the cytoplasm. In some areas of chronic cholecystitis and mucosal hyperplasia, a mosaic pattern of magenta- and blue-stained acinar cells was observed (Fig. 14), which resembled the appearance of the pyloric gland. The cytoplasm of adenocarcinoma cells was negative for both AB and PAS.
The results of IHC revealed that surface epithelial cells and glandular acinar cells of the normal gallbladder mucosa were positive for mucin (MUC) 5 AC, cytokeratin (CK) 7, CK19, and CK20 (Suppl. Figs. S6, S7, and S8) and negative for MUC2, CD10, and CK5/6. Few chromogranin A–positive neuroendocrine cells were randomly observed in the gallbladder mucosa (Suppl. Fig. S9). Surface epithelial cells of the normal pyloric mucosa were positive for MUC5 AC (Suppl. Fig S10) and negative for MUC2 and CD10, while the epithelial cells of pyloric glands were negative for MUC5 AC, MUC2, and CD10. In the normal duodenal mucosa, the goblet cells were positive for MUC2 (Suppl. Fig. S11) and negative for MUC5 AC and CD10, and the brush border was positive for CD10 (Suppl. Fig. S12).
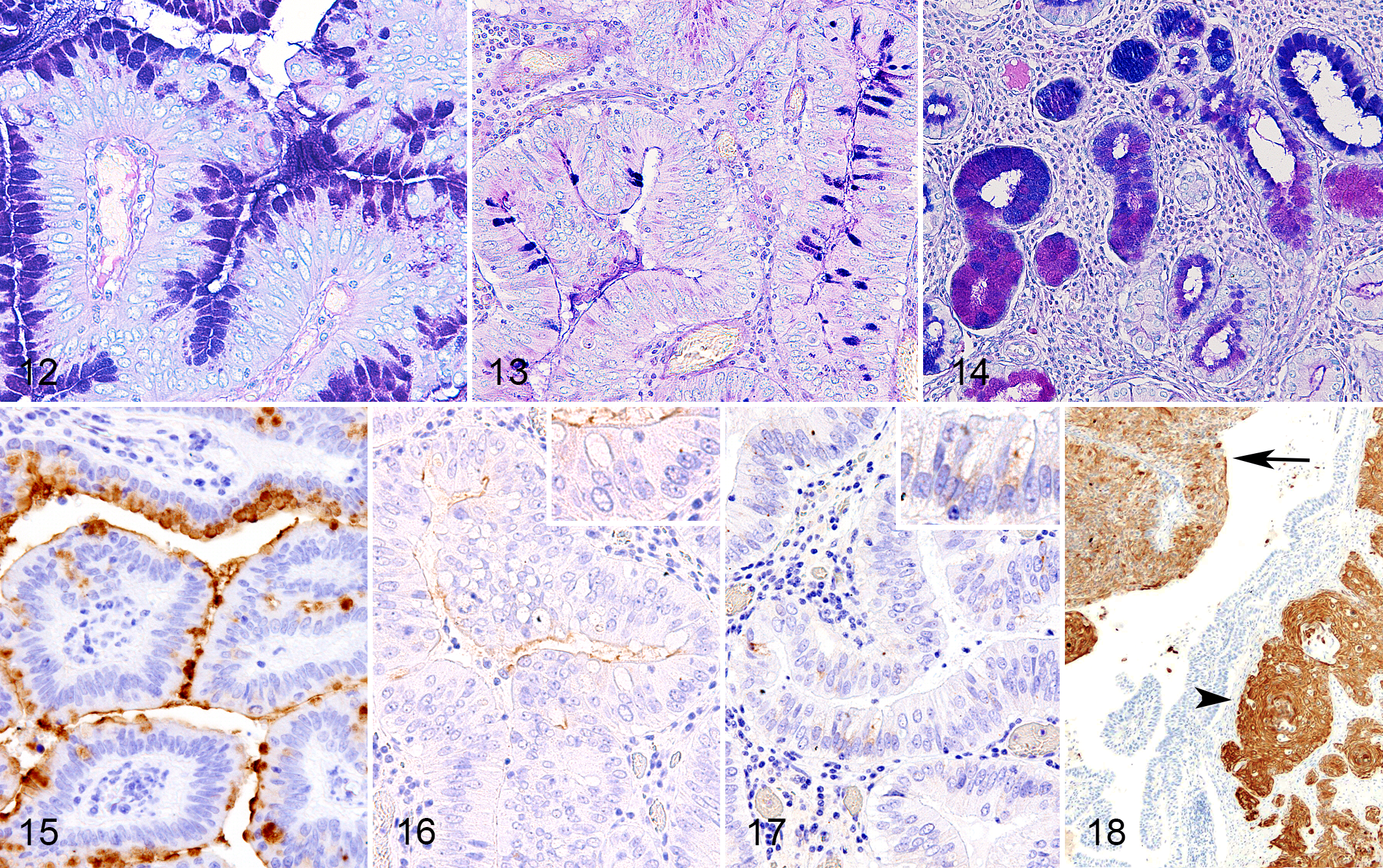
Surface epithelial cells and glandular acinar cells in chronic cholecystitis, mucosal hyperplasia, and adenoma were positive for MUC5 AC (Fig. 15), CK7, and CK19 but negative for CK5/6. Surface epithelial cells and glandular cells were also positive for CK20 in chronic cholecystitis and adenoma but negative in mucosal hyperplasia. The acinar cells that appeared in a mosaic pattern of blue and magenta by AB-PAS staining were negative for MUC5 AC in chronic cholecystitis and mucosal hyperplasia. In chronic cholecystitis and adenoma, the brush border of mucosal epithelial cells was positive for CD10 (Fig. 16), and the cytoplasm of goblet cell-like cells were positive for MUC2 (Fig. 17). In the lesions of adenocarcinoma, the mucosal epithelial cells were positive for CK7, CK19, CK20, and chromogranin A (Suppl. Figs. S13–S15). Basal cell-like neoplastic cells and the invasive cells in adenocarcinoma were positive for CK5/6 (Fig. 18). In some cells, cytoplasmic mucin was positive for MUC5 AC and MUC2. The brush border in the adenocarcinoma lesions was positive for CD10.
Ki-67 indices were 5.3% in the normal gallbladder mucosa; 5.5% (case 1), 5.1% (case 2), 4.8% (case 3), and 6.3% (case 4) in chronic cholecystitis (mean 5.4%); 8.0% (case 5) and 17.6% (case 6) in mucosal hyperplasia (mean 12.8%); 36.6% (case 7) in adenoma; and 82.6% (case 8) and 94.6% (case 9) in adenocarcinomas (mean 88.7%; Figs. 19–22).
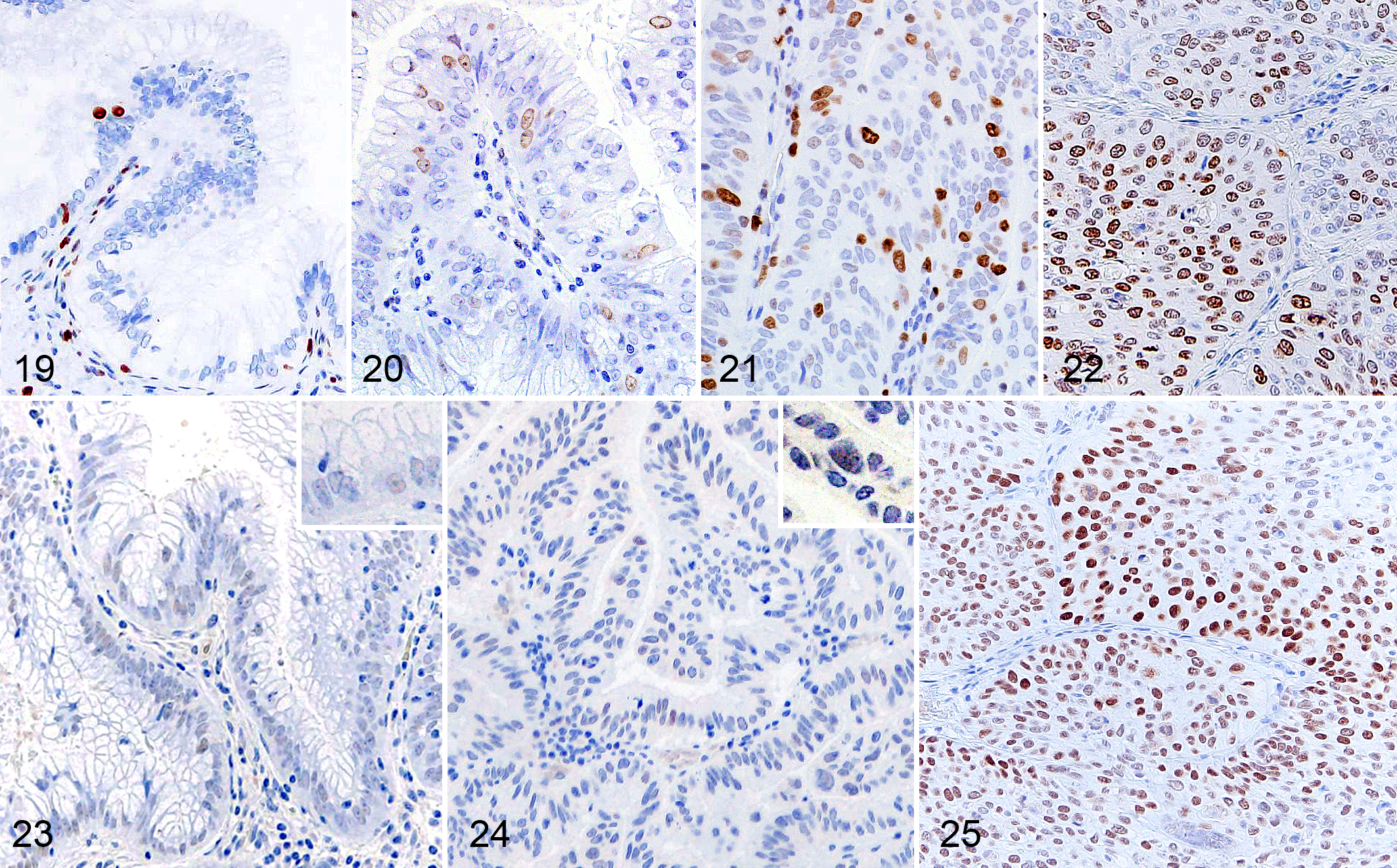
The normal gallbladder mucosa was negative for p53. Few p53-positive cells were observed in a hyperplastic gallbladder (case 6) and adenoma (case 7; Figs. 23 and 24). A large number of p53-positive cells were observed in adenocarcinomas, especially in basal cell-like cells and invasive cells (cases 8 and 9; Fig. 25).
Discussion
The present study describes histopathological findings of gallbladder epithelial tumors in 3 aged pigs: 1 adenoma and 2 adenocarcinomas. Also, gallbladder mucosal hyperplasia was observed in another 2 pigs. Chronic inflammation and metaplastic changes were also observed within these proliferative lesions and in the other pigs investigated. Although the pigs in the present study once lived in the evacuation zone in Fukushima Prefecture and might have been exposed to radioactive pollutants, we could not conclude the impact of radiation exposure on gallbladder carcinomas. Recently, pathological research of Japanese black cattle living in the same evacuation zone revealed that there was no significant relationship between the radiation effect and incidence of tumors. 32 Thus, we expect that radiation exposure is not likely related to the tumorigenesis of the gallbladder carcinoma in pigs.
Gallstones are classified into 3 types according to the amount of cholesterol and calcium bilirubinate: cholesterol gallstones, pigment gallstones, and mixed gallstones. 35 The gross color of cholesterol gallstones is yellow and that of pigment gallstones is brownish or black. 9 Cholesterol gallstones are formed and enlarged by biliary cholesterol supersaturation due to metabolic alterations and hypersecretion of mucins, 12 which can be induced by the intake of a cholesterol-enriched diet also in pigs. 10,23 In a study on spontaneous lesions in Göttingen minipigs, cholecystitis was observed especially in pigs older than 6 months. 20,22 The gallstones in the present pigs, based on gross inspection, are considered to be mainly composed of cholesterol, although the pigs were not fed cholesterol-enriched foods. The formation of cholesterol gallstones in aged pigs might be promoted by 2 factors: hepatic cholesterol synthesis and chronic cholecystitis-induced mucin hypersecretion.
The pathogenesis of gallbladder adenocarcinoma has been studied mainly in tissues from human patients, and 2 pathways are proposed. One is the adenoma-carcinoma cascade, which is considered a minor pathway because gallbladder adenomas are comparatively rare. 1 The other pathway is sequential steps of chronic inflammation, metaplasia, dysplasia, and finally adenocarcinoma. 37 This multistep pathogenesis is considered more plausible because chronic inflammation, hyperplasia, and metaplastic changes are often seen in the vicinity of adenocarcinoma in humans. Chronic inflammation commonly results from gallstones, changes in the bile nature, and bacterial infection. 14 In the present study, bacilli were detected in the gallbladder of 6 pigs, and 5 of those 6 pigs developed gallbladder mucosal hyperplasia (cases 5 and 6), adenoma (case 7), and adenocarcinoma (cases 8 and 9). Gallstone was detected in 3 pigs affected with gallbladder mucosal hyperplasia (case 5), adenoma (case 7), and adenocarcinoma (case 9). In 5 pigs, mucosal epithelial cells with CD10- and PAS-positive brush border and MUC2-positive goblet cells were observed, indicating intestinal metaplasia of the gallbladder mucosa. Two of the 5 pigs developed adenoma (case 7) and adenocarcinoma (case 9). In 4 pigs, a mosaic pattern of AB-PAS–stained acinar cells was observed in the gallbladder mucosa, indicating pyloric gland metaplasia. One of the 4 pigs developed mucosal hyperplasia (case 5).
Phenotypic alterations of mucins and cytokeratins have been studied in human gallbladder tumors. 11,33 The expression level of MUC5 AC is reduced in adenoma and adenocarcinoma, compared with the normal gallbladder mucosa that diffusely expresses MUC5 AC. In the present study, MUC5 AC was diffusely expressed in the normal, inflammatory, and hyperplastic gallbladder of the pig. Similar to the human gallbladder tumor, the expression of MUC5 AC was reduced in adenoma and adenocarcinoma of the pig. With regard to the cytokeratin profiles of tumor cells, loss of CK7 expression was associated with a poor survival of gallbladder adenocarcinoma patients. 11 Also, CK20 expression was associated with carcinomatous changes of gallbladder adenoma in human patients. In the present study on the pig gallbladder, CK7 and CK19 were diffusely and consistently expressed in the normal mucosa, chronic cholecystitis, mucosal hyperplasia, adenoma, and adenocarcinoma. CK20 was expressed also in the normal gallbladder, chronic cholecystitis, adenoma, and adenocarcinoma but absent in the hyperplastic gallbladder mucosa.
Chromogranin A expression was observed in adenocarcinoma lesions of 2 pigs (cases 8 and 9), suggesting the multipotent nature of the tumor cells. In humans, chromogranin A–positive adenoneuroendocrine carcinomas of the gallbladder have been reported, and multipotent stem cells in the gallbladder mucosa or neuroendocrine cells in the intestinal or gastric metaplasia lesions have been considered as the origin of the tumor. 13,26 In the present study, intestinal metaplasia was observed in 1 of the pigs with gallbladder adenocarcinoma (case 9).
Aberration of a tumor-suppressor gene, TP53, is frequently detected in human gallbladder adenocarcinomas (more than 90%). Moreover, TP53 abnormality has been detected in 57% of the gallbladder tissue from human gallstone patients with chronic cholecystitis and without adenocarcinoma; thus, it is considered to be one of the earliest changes during gallbladder tumor development. 25 Missense mutation of TP53 correlates with the higher level of p53 nuclear expressions in humans. 38 Moreover, the nuclear expression of p53 correlates with poor differentiation of tumor cells, tumor size, lymph node metastasis, and high invasiveness in human gallbladder adenocarcinoma. 16,34 In the present study, p53 expression was absent in the normal gallbladder of the pig. Focal expression of p53 was detected in pigs with hyperplastic lesions of the gallbladder (case 6) and also with gallbladder adenoma (case 7). Intense p53 expression was observed in gallbladder adenocarcinomas (cases 8 and 9). These results indicate that changes in the p53 expression may be associated with gallbladder carcinogenesis in the pig, as well as in humans. Also, p53 nuclear expression was highly detected in the invasive and solid lesions compared with the papillary lesions of adenocarcinomas, indicating that the p53 nuclear expression may be correlated with differentiation and invasiveness of gallbladder adenocarcinoma in the pig, as well as in humans.
In conclusion, persistent chronic cholecystitis associated with gallstones and bacterial infections may trigger the onset of hyperplasia and adenoma with metaplasia and ultimately develop into adenocarcinoma in the pig gallbladder, as well as in humans. It is likely that aged pigs develop gallbladder adenocarcinoma because this multistep carcinogenesis progresses over decades.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819875749 - Chronic Inflammatory and Proliferative Lesions of the Gallbladder in Aged Pigs
Supplemental Material, DS1_VET_10.1177_0300985819875749 for Chronic Inflammatory and Proliferative Lesions of the Gallbladder in Aged Pigs by Nanako Ushio, James K. Chambers, Ken-ichi Watanabe, Takuya E. Kishimoto, Takanori Shiga, Jun-You Li, Hiroyuki Nakayama and Kazuyuki Uchida in Veterinary Pathology
Footnotes
Acknowledgements
We thank Ms Shizuka Kato for her technical assistance.
Declaration of Conflicting Interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
