Abstract
Considering the high incidence of dogs with acute bacterial cystitis (BC) and the relationship among inflammation, genotoxicity, and carcinogenesis, we conducted a case-control study comparing the frequency of deoxyribonucleic acid (DNA) lesions assessed by the comet assay between disease-free animals (13 males and 13 females) and cytology-confirmed cases of acute BC (12 males and 12 females), which was mainly caused by Staphylococcus sp. (40%) and Escherichia coli (35%). The results show no increase in DNA damage in cells obtained by bladder washings and no influence of age, sex, and breed due to acute BC. In conclusion, DNA damage was seemingly not associated with the infection by specific bacteria.
Bacterial cystitis (BC) is the most frequent inflammatory disease affecting the urinary bladder of dogs and humans. 2 It is known that inflammatory processes cause genotoxicity, mainly by causing oxidative deoxyribonucleic acid (DNA) damage. 7 Although DNA mutations are strongly associated with cancer, the inflammatory process is only associated with malignancy when inflammation is chronic. 7 To investigate whether acute BC can induce genetic damage in canine urinary bladder epithelium, we measured primary DNA damage (i.e., strand breaks, alkali-labile sites, incomplete-repair sites, and cross-links) in exfoliated epithelial cells obtained by bladder washing. Damage was assessed by the comet assay, a simple and sensitive methodology for DNA damage estimation at the single-cell level. 11
Dogs were from the ambulatory service of the Veterinary Medicine Hospital UNESP, Botucatu, Brazil. The cytologic criteria used to define acute BC were the presence of numerous neutrophils (i.e., > 70% of cells obtained), presence of bacteria, and abnormalities in epithelial cells, such as hyperchromatic nuclei and perinuclear halos. Hyperplastic or dysplastic changes (cluster formation, nuclear variability, or increases in nuclear-cytoplasm ratio) were not observed in these specimens, excluding, therefore, the likelihood of chronic disease. 1 Twenty-four cytology-confirmed acute BC cases (12 males and 12 females) and 26 (13 males and 13 females) controls with negative urinary bladder cytology and uroculture were evaluated in this study. Dogs were of various breeds, and their ages varied between 8 months and 14 years. For each animal, we performed bladder washing with sterile 0.9% NaCl (injected and withdrawn five times; 22-G needle, and 8-ch Foley catheter) and cystocentesis. Studies were conducted with the approval of the Faculty of Veterinary Medicine Ethical Committee for Animal Research. No cell separation or enrichment for urothelial cells was conducted. The liquid obtained from the bladder washing (total of 15 ml) was divided into two aliquots: one (5 ml) was used for routine urinary cytology and other for the comet assay. 3,12 Cell viability was evaluated by trypan blue exclusion. After centrifugation of the second aliquot (for comet assay), the cell pellet (approximately 5 × 105 cells) was embedded in low–melting point agarose (0.5%), layered onto a microscope slide precoated with agarose, allowed to solidify at 4 C, and lysed for 24 hour at 4 C in a solution containing 2.5 M NaCl, 100 mM Na2 ethylenediaminetetraacetic acid, 10 mM Tris, pH 10, 2% sodium sarcosinate with 1% Triton X-100, and 10% dimethyl sulfoxide added just before use. Slides were incubated in alkali solution (pH > 13) for 30 minutes and electrophoresed at 0.66 V/cm for 20 minutes. Slides were then neutralized, fixed in absolute ethanol, stained with ethidium bromide, and analyzed under green light. The mean percentage of DNA in the tail of 50 comets per animal was evaluated by image analysis (Comet Assay II, Perceptive Instruments, Haverhill Suffolk, UK) and was used as the indicator of DNA damage.
Urine obtained by cystocentesis was cultured to confirm BC. 4,10 Cytology specimens were stained by Giemsa or Shorr methods. The Student's t-test or analysis of variance, followed by the least significance difference test, was performed when comparisons were made between two or more groups, respectively. When applicable, Pearson's correlation coefficients were calculated and verified for a trend by the t-test. Values were considered significant if the P value was smaller than 0.05.
Acute BC was confirmed cytologically in 100% of test group animals. One animal had a concomitant urinary bladder neoplasm and was excluded from the analyses. Four animals that were treated with antibiotics had negative urine cultures. The most frequent causal agents were Staphylococcus sp. (40%) and Escherichia coli (35%) (Table 1). This prevalence was different from that reported in most other studies, in which the presence of E. coli was found to be the leading cause of infection. 2,8,9 Cell viability was around 60% and did not differ between groups (57.1 ± 14.5 and 60.9 ± 16.7 for controls and dogs with acute BC, respectively; P > 0.10). Age, sex, and breed did not affect the results significantly (P > 0.10; data not shown). This study had a power (1 − β) of 97% to detect an increase from 10% to 20% in the mean percentage of DNA in the tail of comets. However, no significant difference was observed for DNA migration in the comet assay in the test group when compared with controls (P > 0.10, Table 1). Moreover, when the analysis was stratified by etiologic agent, there also was no significant difference with regard to DNA damage. However, stratification yielded very small sample size, hampering conclusions on this issue (P > 0.10; Table 1). Taking advantage of the single-cell data generated by the comet assay, we made histograms of DNA damage to verify the presence of a subpopulation of comets that might be related to the number of inflammatory cells in bladder washings of animals with acute BC, but no obvious subpopulation of comets was identified (data not shown). A problem with this study is that a high proportion of inflammatory cells might have been analyzed along with the urothelium from dogs with acute inflammation. It has been shown that there is a poor correlation between inflammatory cell DNA damage and other parenchymal cell damage. 5,10 Further studies would be required to verify the confounding potential of these inflammatory cells in dogs. Nevertheless, in humans, acute inflammation is unlikely to cause any shifts in DNA damage detected by the alkaline comet assay in true urothelial cells, as we have recently shown in a small group of patients. 5
DNA damage status (mean percentage of DNA in the tail of 50 comets/animal) of urothelial cells of dogs with acute BC according to different etiologies.
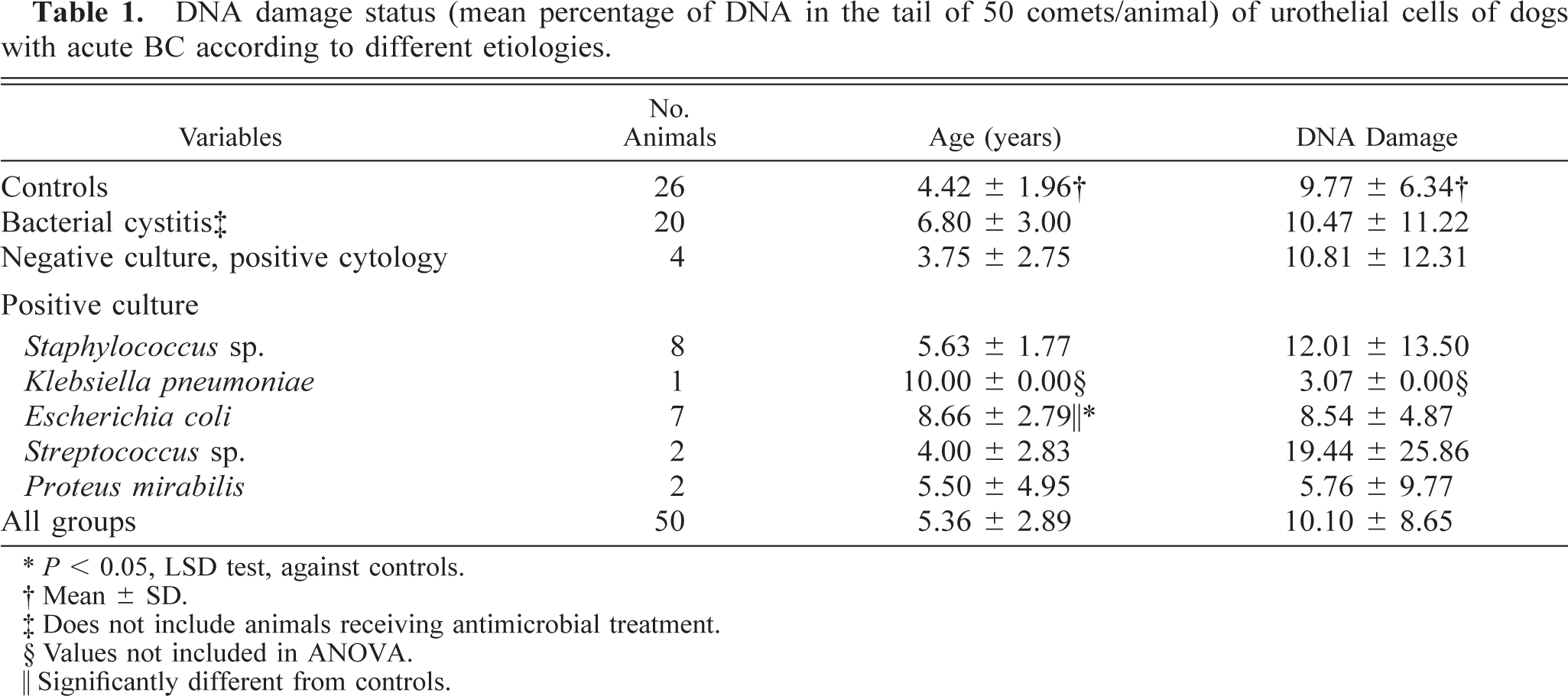
∗ P < 0.05, LSD test, against controls.
† Mean ± SD.
‡ Does not include animals receiving antimicrobial treatment.
§ Values not included in ANOVA.
‖ Significantly different from controls.
Increased DNA damage, as detected by the comet assay, has been shown in the urothelial cells of human patients at increased risk for urothelial cell carcinoma, such as smokers 5 and patients with a history of urothelial cell carcinoma but with normal-looking cytology. 3 We are unaware of prospective studies in the literature evaluating primary DNA damage as a potential risk factor for cancer. The biologic significance of primary DNA damage in viable cells is the risk for mutation accumulation and its relationship with genomic instability in vitro 6 and in vivo. 3 Our findings show no association between positive cytology for acute BC and increased DNA damage, suggesting that acute cystitis does not increase the risk for mutation in urothelial cells.
