Abstract
Plasmacytoid and rhabdoid variants of urothelial carcinomas (UCs) of the urinary bladder have been described in humans with plasma cell–like or rhabdoid cellular appearance and aggressive clinical outcome. Canine UC of the bladder is generally classified as papillary/nonpapillary and infiltrating/noninfiltrating with limited information regarding other histological patterns. We report 3 cases of UC of the urinary bladder showing a unique discohesive cellular morphology with malignant behavior resembling the human plasmacytoid and rhabdoid variants of UC, which may raise some difficulties in diagnosis. Epithelial-mesenchymal transition and reduced E-cadherin expression were revealed by immunohistochemistry in 2 cases, possibly explaining the discohesive and invasive behavior of the tumor cells. The findings broaden the morphological spectrum as well as the distinct clinical features of canine UC of the urinary bladder.
Keywords
Human urothelial carcinomas (UCs) of the bladder have a variety of histologic variants aside from the papillary/nonpapillary forms. 2,5 The plasmacytoid and rhabdoid variants are documented as neoplasms comprising discohesive cells with plasma cell–like or rhabdoid morphology and aggressive presentation. 4,10,12,14,15,17 Canine UC has a strong resemblance to human invasive bladder cancer with respect to histopathology, molecular features, and behavior. 8 However, subtypes or variants of UC, other than papillary/nonpapillary and infiltrating/noninfiltrating, are rarely mentioned in animals. We have encountered 3 noteworthy cases with unique discohesive and round contour morphology and aggressive behavior in both primary tumor site and the metastatic sites. Two of the 3 cases resembled the plasmacytoid subtype, and the remaining one was similar to the rhabdoid variant of human UC.
Case No. 1 was an 11-year-old male castrated Beagle with 1 year of intermittent hematuria and 1 month of urinary incontinence; cystotomy revealed generalized thickening of the bladder wall. Case No. 2 was a 10-year-old female spayed Miniature Schnauzer, showing 1 month of dysuria and bilateral hydronephrosis; laprarotomy revealed nodular masses at the midline fascia and the neck and regions of ureter insertion of the bladder with bilateral ureteral obstruction. Case No. 3 was a 12-year-old female spayed Shetland Sheepdog, displaying 3 days of hematuria and dysuria; radiograph and ultrasound revealed dilated left renal pelvis and diffusely thickened bladder wall with 2 large masses present at the trigone. Surgical biopsies of the bladder were obtained by cystotomy in case Nos. 1 and 3. Samples in case No. 2 were taken from nodular masses at the midline fascia, rectum, and junction between the ureter and bladder. Following cystotomy and 3 subsequent chemotherapies with carboplatin, mitoxantrone, and carboplatin, respectively, case No. 1 was euthanized due to disseminated intravascular coagulopathy and full necropsy was performed. At necropsy, extensive petechial and ecchymotic hemorrhages were observed in the skin of the lower abdomen with several 1.5- to 2.5-cm poorly demarcated hemorrhagic nodules in the subcutis. The inguinal and sublumbar lymph nodes were swollen. The bladder was diffusely thickened with scattered 0.1- to 0.5-cm hemorrhagic nodules in the mucosa. The lungs were heavy and wet. The liver and spleen had several 1.0- to 2.5-cm hematomas. Case No. 2 was euthanized due to extensive metastasis and case No. 3 died shortly after cystotomy, and no necropsy was performed in either case.
All tissue samples were fixed in 10% neutral buffered formalin, processed routinely, sectioned at 5 μm, and stained with hematoxylin and eosin. Microscopically, the biopsies of the bladder from case Nos. 1 and 3 were characterized by numerous neoplastic cells diffusely distributing in the lamina propria of mucosa (Fig. 1a), while the urothelium was relatively well maintained and intact, showing no evidence of downward infiltration. In case No. 2, discohesive tumor cells infiltrated into the tissues of midline fascia, ureter insertion to the bladder, and rectum with vigorous desmoplasia (Fig. 2a). The tumor cells of case Nos. 1 and 2 were similar, characterized by sheets of individualized, medium to large, round to oval cells with distinct cell borders, moderate to abundant eosinophilic cytoplasm, and an eccentrically located large round nucleus with clumped coarse chromatin, morphologically resembling plasma cells. The tumor cells of case No. 3 were also individualized but polygonal in shape with a moderate to large amount of eosinophilic cytoplasm and a variably sized and shaped hyperchromatic nucleus (Fig. 3a). Many of the cells contained abundant bright eosinophilic cytoplasm and an eccentric crescent nucleus with variably conspicuous magenta nucleolus, morphologically compatible with the rhabdoid cells (Fig. 3a). Overall, the levels of anisocytosis and anisokaryosis were high in all 3 cases. The mitotic counts were about 20, 36, and 28 per 10 high-power fields for case Nos. 1, 2, and 3, respectively. Binucleated to multinucleated tumor cells were also frequent. The tissues from necropsied case No. 1 showed that the discohesive neoplastic cells had diffusely infiltrated the bladder wall with vascular invasion and metastases to the lungs, liver, kidney, spleen, stomach, skin, lymph nodes, and adrenal gland. The tumor cells that had invaded within the blood vessels and lymphatics were also individualized.
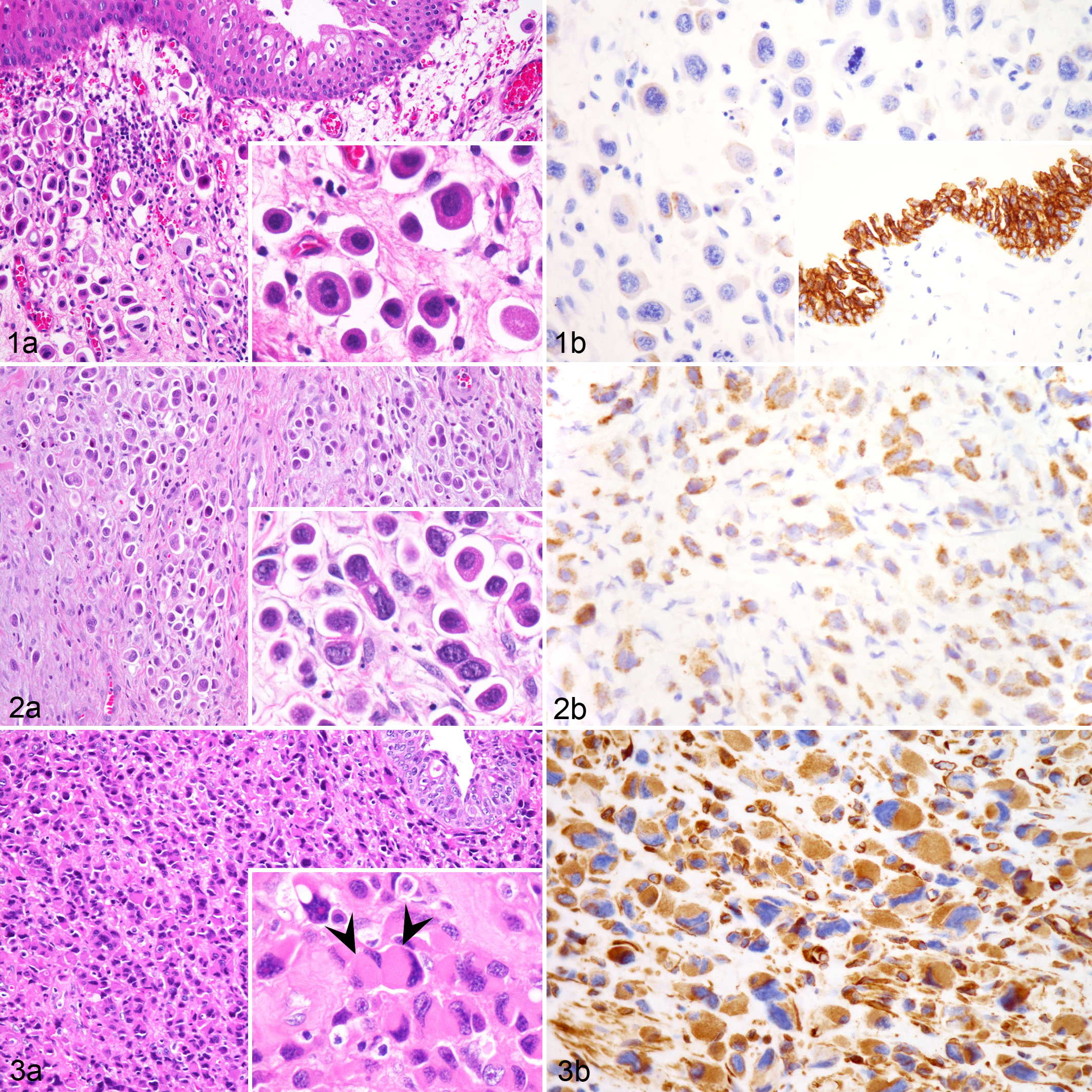
Immunohistochemistry (IHC) was performed on formalin-fixed, paraffin-embedded sections by using various monoclonal antibodies, including cytokeratin (clone AE1/AE3; Leica, United Kingdom), vimentin (clone V9; Leica), uroplakin III (clone AU1; Zytomed, Germany), CD138 (clone B-A38 [Zeta Coorporation, USA]; clone B-A38 [Ventana]; clone MI15 [Dako, Denmark clone MI15 [DakoCytomation, Glostrup, Denmark]]), E-cadherin (clone GM016; Genemed Biotechnologies, Inc., USA), MUM1 (clone MUM1p; Dako), CD79a (clone HM57; Abgent, Inc., USA), CD3 (clone F7.2.38; Dako), and Melan-A (clone A103; Dako). Hematoxylin was used for counterstain. The results are shown in Table 1. The neoplastic cells were strongly positive for CK AE1/AE3 in all 3 cases. For uroplakin III, case No. 2 showed diffuse moderate positivity (Fig. 2b) but case Nos. 1 and 3 were sporadically weakly positive. The results for MUM1, CD79a, CD3, and Melan-A were all negative. Both anti-CD138 clones failed to cross-react with canine plasma cells. The neoplastic cells in case Nos. 1 and 2 showed variable membranous and cytoplasmic positivity for E-cadherin in the biopsied tissues, while the E-cadherin intensity in the necropsied specimens of case No. 1 was reduced (Fig. 1b); both cases were vimentin negative. In case No. 3, the tumor cells exhibited lower E-cadherin intensity, mostly cytoplasmic, in comparison to the strongly positive bladder epithelial cells; however, they also displayed strong vimentin positivity (Fig. 3b).
Pathologic Findings in 3 Cases of Plasmacytoid or Rhabdoid Urothelial Carcinoma of the Urinary Bladder in Dogs.

Abbreviations: C, cytoplasmic labeling; CK, cytokeratin; M, membranous labeling; –, negative; +, weakly positive; ++, moderately positive; +++, strongly positive.
aE-cadherin labeling was diffuse/+ in tissues obtained at necropsy.
The diagnosis of canine UC is usually quite straightforward morphologically and clinically. 11 The initial differential diagnoses for the surgical biopsies of the 3 cases included UC and round cell tumors, especially poorly differentiated plasma cell tumor and multiple myeloma, due to cell individualization and morphology. A firm diagnosis was challenging because of the limited biopsy specimens with diffusely distributed individualized round tumor cells in the lamina propria of the urinary bladder with no detectable downward invasion by the urothelium (case Nos. 1 and 3) and the prominent desmoplasia at the metastatic sites (case No. 2). Even after examining serial sections of the thickened bladder wall of case No. 1 following necropsy, we failed to identify any downward epithelial invasion with transformation of coherent cells to individualized ones. However, it is possible that an abrupt transition from a noninvasive to an invasive component did occur in a rather small region prior to developing strong invasiveness and systemic spread. Not until performing IHC for pan-cytokeratin and uroplakin III was the urothelial origin of the tumor cells clarified.
The neoplastic cells in case Nos. 1 and 2 displayed a plasmacytoid appearance. Plasmacytoid UC in humans, named based on the unusual plasma cell–like morphology of the tumor cells, was initially described in 1991, 16,18 followed by other published case reports and large case series. 4,10,11,13,17 The tumor cells are round with abundant amphophilic to eosinophilic cytoplasm and eccentric nucleus that may raise difficulties in diagnosis, particularly with limited biopsied specimens. 2,12 The tumor cells are usually positive for CK7 and CK20 but might 10,12 or might not 4 be positive for CD138, and CD138 positivity should not be considered as the requirement for diagnosing plasmacytoid UC due to its broad expression profile. 13 In the present study, all 3 commercial anti-CD138 monoclonal antibodies from 2 different clones failed to cross-react with canine plasma cells.
Human rhabdoid UC is characterized by discohesive pleomorphic tumor cells that have large eccentric nuclei with vesicular chromatin and abundant cytoplasm containing an eosinophilic inclusion, which ultrastructurally consists of whorls of intermediate filaments. 9,14 Rhabdoid UC in humans should be differentiated from the true malignant extrarenal rhabdoid tumors, which usually have the characteristic mutation or deletion of the INI1 gene at chromosome 22q11 and the loss of nuclear INI1 expression immunohistochemically. 2,14,17 The rhabdoid feature is just a morphologic variant rather than a true myogenic differentiation in rhabdoid UC. The prognosis of rhabdoid UC in humans is generally poor, with an aggressive clinical course and short survival time. 2,6,14 The tumor cells of case No. 3 displayed similar rhabdoid features, including cell discohesion and individualization, abundant eosinophilic cytoplasm, vimentin positivity, and large eccentric crescent-shaped to bizarre nuclei. In humans, although the rhabdoid and plasmacytoid variants share somewhat similar characteristics of round cellular contour and discohesive feature, it has been pointed out that the abundant eosinophilic cytoplasmic inclusion with vimentin positivity is the major characteristic of the rhabdoid variant. 6,15
The discohesive character of rhabdoid and plasmacytoid UC has been reported to be associated with reduced E-cadherin expression. 1,3,4,17 E-cadherin is a calcium-dependent intercellular adhesion transmembrane glycoprotein that is important in epithelial differentiation and functions as a tumor metastasis suppressor. The loss of E-cadherin expression is related to increased invasiveness and epithelial-mesenchymal transition (EMT). EMT is involved in multiple steps of metastasis, characterized by switching from the expression of keratins as the major intermediate filament to the mesenchymal intermediate filament vimentin. 7 In humans, 84% of plasmacytoid UCs harbor truncated somatic alterations in CDH1, the gene encoding E-cadherin, leading to the enhanced migratory and invasive characteristics. 1 Such a pathognomonic gene alteration results in the lack of E-cadherin expression based on IHC. This may further explain the unique peritoneal tumor spread, wide dissemination, and poor clinical outcome of patients with plasmacytoid UC. 1 Our 3 cases all presented a discohesive appearance with aggressive tumor behavior, and case Nos. 1 and 2 exhibited peritoneal tumor spread and wide metastasis. However, the 3 cases displayed variable intensities in E-cadherin expression. Similar to human rhabdoid UC, 15,17 case No. 3 had reduced E-cadherin expression and concurrent vimentin and cytokeratin positivity. The biopsied specimens of case Nos. 1 and 2 showed variable intensities in membranous and cytoplasmic E-cadherin expression. On the contrary, the neoplastic cells retained in the bladder or spreading to other tissues/organs in case No. 1 when euthanized displayed a significantly reduced intensity compared to the biopsied specimens taken 2 months previously and the remaining normal urothelium. This difference suggests that E-cadherin expression may change as the disease progresses, although similar molecular and genetic alterations as human cases require further clarification. In human cases, although a complete negativity of E-cadherin was demonstrated immunohistochemically in 70% to 100% of cases, 3,4 the timeframe of loss of E-cadherin has not been reported. The strikingly similar plasmacytoid and discohesive features of the tumor cells in case Nos. 1 and 2 highlight the possibility of a distinct entity of plasmacytoid UC in dogs. Because the reduction in E-cadherin expression is not as prominent as in human cases, other mechanism(s) involving tumor cell discohesion may occur in canine bladder tumors.
In conclusion, here we report 3 cases of canine UC of the urinary bladder showing a unique discohesive cellular appearance with an infiltrative and invasive growth pattern. They are similar to human plasmacytoid and rhabdoid UC and may raise challenges in diagnosis. The discohesive cell arrangement and aggressive behavior of such plasmacytoid and rhabdoid features are notable, as they broaden the morphological spectrum of canine UC and aid in diagnosis of this clinically and pathologically unique entity.
Footnotes
Acknowledgements
We thank Dr. Wei-Hsiang Huang for his assisting in E-cadherin staining.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
