Abstract
Urothelial carcinoma (UC) is the most common neoplasm of the canine urinary tract. Clinical presentation of UC is shared with several other, more common urinary tract disorders, and this often delays diagnosis of the UC. Definitive diagnosis of UC requires histopathologic examination of a biopsy specimen, but the cost and invasiveness for these diagnostic tests often result in most diagnoses being made on the basis of clinical findings, diagnostic imaging, and cytologic examination of urine sediment. Regardless of the diagnostic process used, most UCs currently are not diagnosed until they are at an advanced clinical stage and so are associated with poor prognosis. Improved methods for earlier and less invasive detection are needed. In a previous study, the authors demonstrated the presence of highly recurrent DNA copy number aberrations (CNAs) in canine UC and hypothesized that detection of these CNAs in tumor cells can be used as a molecular diagnostic for UC. In this study, a multiplexed droplet digital polymerase chain reaction (ddPCR) assay was detected to detect and quantify CNAs of specific regions of canine chromosomes 8, 13, 19, and 36. The assay was effective at differentiating 31 neoplastic and 25 nonneoplastic bladder tissues based on copy number, with 100% sensitivity and specificity in tissue samples. CNAs were also detected by ddPCR in 67% (12 of 18) of urine DNA specimens derived from UC patients. The findings show that ddPCR is a useful molecular technique to detect CNAs and may be used as a noninvasive molecular diagnostic test for canine UC.
Keywords
Urinary bladder cancer accounts for approximately 1% to 2% of all malignancies in dogs. Urothelial carcinoma (UC), also referred to as transitional cell carcinoma, is the most common form of urinary bladder malignancy. 16 Due to nonspecific clinical signs and unreliable diagnostics, the diagnosis of UC is often delayed, hindering therapeutic intervention. By the time of diagnosis, the majority of bladder tumors (>90%) have invaded the bladder wall, with >20% displaying regional or distant metastases. 17 Survival time of dogs diagnosed at advanced stage has been shown to be significantly shorter than that of dogs diagnosed at early stage, implying that early detection of UC may improve the prognosis of affected dogs. 17
Definitive diagnosis of UC requires histopathologic examination of a tumor biopsy, which may involve general anesthesia and expensive and invasive surgical procedures, such as cystotomy or cystoscopy. Although cystoscopy is considered less invasive than cystotomy, the necessary skills and equipment may not be available in all veterinary clinics. In addition, the size and sex of affected dogs may limit the application of cystoscopy, as it is often difficult to obtain sufficient amount of biopsy specimens for histopathologic evaluation from small or male dogs. 8 Although needle aspiration of the bladder is not recommended due to the risk of tumor dissemination, 21,23 cytopathology of urine sediments can be used for making a clinical diagnosis of UC in combination with other diagnostic modalities; however, 1 study reported that neoplastic cells were recognized in the urine of only 30% of dogs with UC. 20 A urine antigen test for canine UC (V-BTA) has been reported to be highly sensitive (80%–90%), but its low specificity limits its diagnostic value. 6,15 Therefore, the need for a reliable noninvasive diagnostic test for canine UC remains paramount.
Numeric and structural chromosomal abnormalities are hallmarks of cancer. Such changes to the genome have been utilized widely as diagnostics and prognostics in a range of human cancers, 10,14,19 including UC. 13 In a previous study, we demonstrated that 3 regions of the canine genome were subject to highly recurrent copy number changes in canine UC. In that study, gain of canine chromosomes (Canis familiaris) CFA 13 and 36 and loss of CFA 19 were present in 97%, 84%, and 77% of the UC cohort, respectively. 22 Since these numeric chromosomal changes are specific to neoplastic urothelium, detection of such aberrations offers a means to develop a molecular diagnostic test for canine UC. Although cytogenomic and fluorescence in situ hybridization (FISH) approaches are considered the standard methods for detecting chromosomal abnormalities, both are costly and labor intensive, potentially limiting their application as routine diagnostics in a clinical setting.
Digital polymerase chain reaction (PCR) is an emerging molecular technique, with applications including DNA/RNA quantification and rare allele detection. 4,5,9,24 Digital PCR is performed by partitioning the PCR mixtures into a large number of compartments (eg, droplets) so that each compartment contains either ≥1 (positive) or 0 (negative) target sequences. After thermal cycling amplification, each compartment is classified as either positive or negative by fluorescence signal intensity at the end point. The numbers of positive and negative compartments, together with Poisson distribution, are used to calculate absolute number of target sequence in a sample without the need for calibration standards. With these features, droplet digital PCR (ddPCR) offers a simple, objective, and precise method to determine copy number changes.
In this study, we developed a multiplexed ddPCR assay to assess copy number imbalance characteristic of canine UC, and we evaluated the assay as a potential molecular diagnostic for canine UC using tissue and urine samples obtained from UC and non-UC dogs.
Materials and Methods
DNA Specimens
A total of 31 cases of canine UC DNA samples from a previous study cohort 22 were accessed for this study; 15 samples were from fresh-frozen material (case Nos. 1–15), and 16 were from formalin-fixed paraffin-embedded (FFPE) tissues (case Nos. 16–31). As nonneoplastic controls, DNA was isolated from canine bladder epithelium of a total of 25 dogs, each obtained by necropsy with no evidence of neoplastic changes on histopathologic evaluations (21 normal, 3 cystitis [2 mild and 1 severe] and 1 mild urothelial hyperplasia and dysplasia with granulation). To assess the effect of DNA quality on the ddPCR assay, DNA was isolated from paired fresh-frozen and FFPE materials from 12 of 25 control specimens. For the other 13 control samples, DNA was isolated from either fresh-frozen materials (3 dogs) or FFPE tissues (10 dogs).
To evaluate copy number imbalance in cells recovered from free catch urine, genomic DNA was isolated from the urine sediment of 18 dogs with UC, 5 clinically healthy dogs, and 2 dogs with urinary tract infections. Free-catch urine samples were collected (with informed consent) from dogs evaluated through the Clinical Studies Core of the North Carolina State University College of Veterinary Medicine or from private veterinary hospitals with an informed consent. Of the 18 UC dogs, 4 were diagnosed by histopathology of the bladder mass, and 14 were diagnosed according to clinical findings of lower urinary clinical signs, presence of bladder mass on ultrasound, and cytologic evidence of abnormal epithelial cells in urine sediments.
Genomic DNA was isolated from cell pellets of urine specimens after being washed with phosphate-buffered saline (11 dogs) or following fixation with 3:1 methanol acetic acid used for chromosomal analysis (14 dogs), some of which were used in the previous study. 22 To compare copy number imbalance detected in primary tumor tissue with cells shed into the urine, DNA was also isolated from FFPE tumor blocks of 4 dogs with histologic diagnosis of UC. Signalment of dogs used for urine DNA isolation and urine DNA source are summarized in Supplemental Table 1.
DNA was extracted with the DNeasy Blood and Tissue Kit (Qiagen, Hilden, Germany) or the QIAmp DNA FFPE Tissue Kit (Qiagen). Spectrophotometry (NanoDrop; Thermo Scientific, Wilmington, DE), and agarose gel electrophoresis were used to determine DNA quantity and integrity of all tissue-derived DNA. Integrity of urine DNA was also assessed in the same way if the DNA yield was >1 µg.
Digital PCR Assay
A “test” ddPCR assay comprising 2 PCR primers and a TaqMan probe was designed within the genome sequence coordinates of highest penetrance of copy number change along each of CFA 13, 19, and 36. In addition, a fourth ddPCR assay was designed within the copy number–neutral 1-Mb region of CFA 8 described previously. 22 Primers and TaqMan probes were designed with PrimerQuest software (http://www.idtdna.com) after confirming the absence of known single-nucleotide polymorphisms in the target sequences (https://www.broadinstitute.org/mammals/dog/snp2). Each ddPCR assay was processed through the University of California–Santa Cruz In-Silico PCR tool (http://genome.ucsc.edu) to confirm the absence of nonspecific binding of primers and probes. All primers and TaqMan probes were custom synthesized by Integrated DNA Technologies (Coralville, IA, USA). For each assay, a double-quenched probe (5′-FAM or HEX/ZEN/3′-IBFQ) was used to reduce background fluorescence. Sequences and locations of primers and probes used in this study are shown in Table 1.
Nucleotide Sequences of Primers and Probes Used to Detect Copy Number Imbalance Characteristic of Canine Urothelial Carcinoma.

Abbreviation: CFA, Canis familiaris chromosome.
aIn parentheses, amplicon length in base pairs.
Three duplex ddPCR reactions were performed with a combination of 1 of the FAM-labeled CFA 13, CFA 19, or CFA 36 primer/probe set with HEX-labeled CFA 8 primer/probe set. Each reaction mixture comprised 1× Droplet Supermix (Bio-Rad, Richmond, CA, USA), 500 nM of each primer, 250 nM of each probe, and ∼55 ng of genomic DNA for tissue and 30 to 55 ng for urine specimens. The PCR reaction mixtures were partitioned into an emulsion of ∼13 000 droplets (mean ± SD: 13 012 ± 945 droplets/reaction) via a QX100 Droplet Generator (Bio-Rad). PCR was performed on T100 Thermal Cycler (Bio-Rad) with the following thermal cycle condition: denaturation at 95°C for 10 minutes, 40 cycles of 94°C for 30 seconds and 58°C for 60 seconds, 98°C for 10 minutes.
Post-PCR, droplets were analyzed on a QX100 Droplet Reader (Bio-Rad). The ratios of CFA 13:CFA 8 (ratio13/8), CFA 19:CFA 8 (ratio19/8), and CFA 36:CFA 8 (ratio36/8) were calculated on the Poisson distribution with Quantasoft software (v1.3.2.0; Bio-Rad) after counting positive and negative droplets (for the assignment of positive and negative droplets; see Fig. 1). To maximize the detection of copy number imbalance, ratios of CFA 13:CFA 19 (ratio13/19) and CFA 36:CFA 19 (ratio36/19) were calculated indirectly by dividing ratio13/8 by ratio19/8 and ratio36/8 by ratio19/8, respectively.
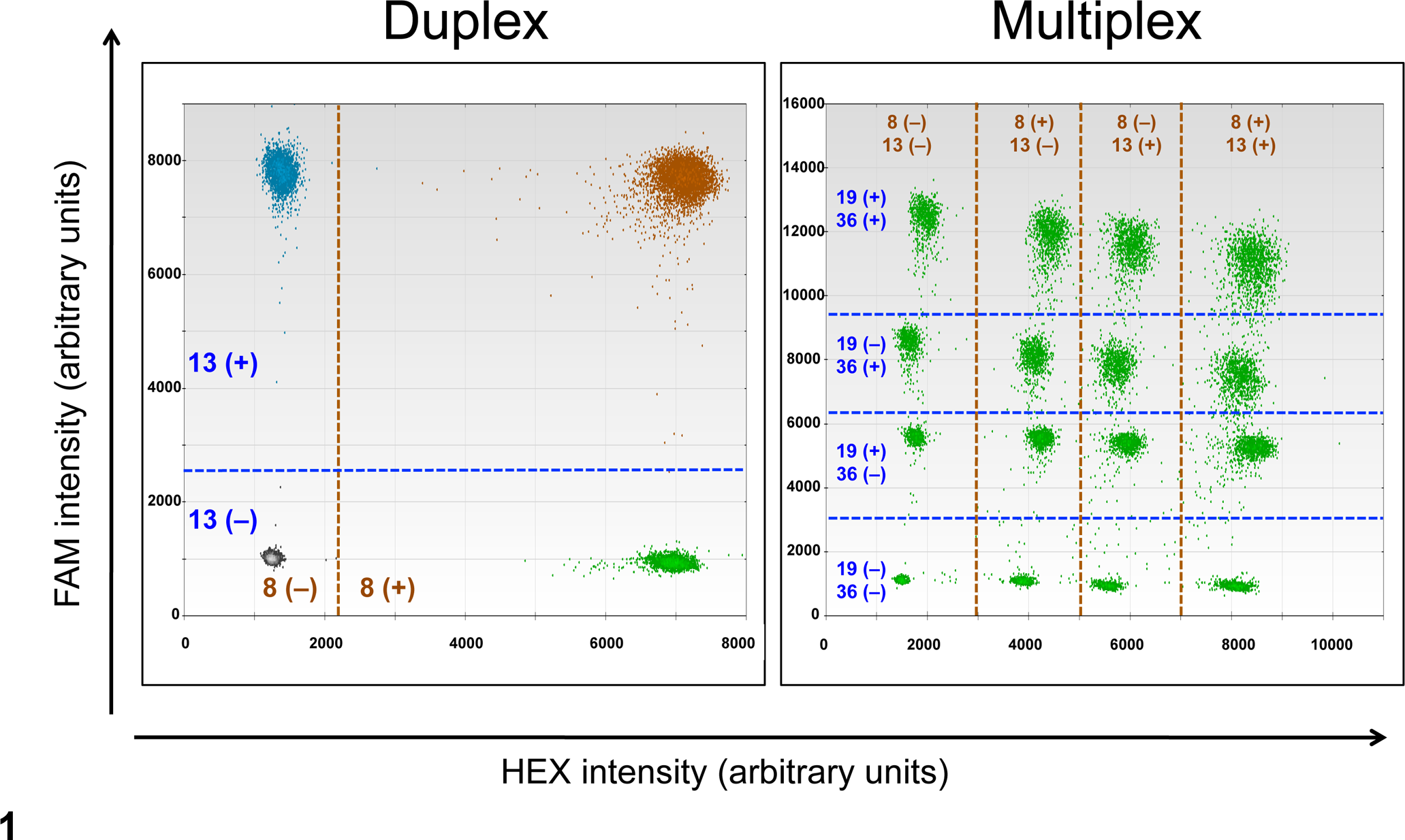
Two-dimensional scatter plots of a droplet digital polymerase chain reaction assay: duplex (left: CFA 8, 13) and multiplex (right: CFA 8, 13, 19, 36). Droplets were divided into 4 clusters in the duplex assay, based on presence or absence of the corresponding amplicon: double negative, CFA 8(–)CFA 13(–); mixed positive and negative, CFA 8(+)CFA 13(–) and CFA 8(–)CFA 13(+); and double positive, CFA 8(+)CFA 13(+). Similarly, droplets were divided into 16 clusters (as shown) in the multiplex analysis, based on the fluorescence intensity. CFA, Canis familiaris chromosome.
Comparison With Oligonucleotide Array Comparative Genomic Hybridization Data
Data obtained by genome-wide oligonucleotide array comparative genomic hybridization (oaCGH) of 31 UC cases 22 were used to assess correlation of copy number ratios determined by oaCGH and ddPCR. The mean log2 ratio of the 5 consecutive oaCGH probes surrounding each ddPCR primer/probe set was used as a representative log2 ratio for each region. After log2 ratio was converted into copy number, oaCGH-based copy number ratios were calculated according to the copy number of 2 regions.
Dilution Series of Tumor DNA in Normal Cell DNA
One UC DNA sample (case No. 1) was diluted into DNA obtained from a normal fresh-frozen bladder to make a dilution series with tumor DNA: normal DNA = 100:0, 75:25, 50:50, 30:70, 20:80, 10:90, 5:95, and 0:100. The expected copy number ratio was calculated for each dilution.
Multiplex ddPCR Assay
To consolidate the use of ddPCR into a single-tube assay for the detection of neoplastic UC cells, a multiplex ddPCR assay was developed by modification of primer/probe combinations and concentrations as follows: 500 nM of each primer and 100 nM of CFA 8–HEX and CFA 19–FAM and 200 nM of CFA 13–HEX and CFA 36–FAM probes. Each droplet was scored as positive or negative for each primer/probe set according to the corresponding location on a 2-dimensional plot (Fig. 1).
Statistical Analysis
The correlation of 2 values was evaluated with Pearson correlation coefficient analysis. The Wilcoxon rank-sum test was used for the comparison of 2 groups. The Wilcoxon signed-rank test was used to assess the effect of DNA fragmentation in 12 paired DNA samples from fresh-frozen and FFPE tissues. To determine the suitable threshold discriminating UC from nonneoplastic bladder tissues, a receiver operating characteristic curve analysis was performed for each copy number ratio. Results are shown as mean ± standard deviation. Differences were considered significant when P < .05. Statistical analysis was performed with JMP software (v11; SAS Institute, NC, USA).
Results
Duplex ddPCR for direct measurement of ratio13/8, ratio19/8, and ratio36/8 and indirect measurement of ratio13/19 and ratio36/19 were optimized with 4 DNA samples isolated from nonneoplastic fresh-frozen canine bladder tissues. The intra-assay (3 different reactions per plate) and interassay (1 of each reaction in 3 different plates) variability in 4 nonneoplastic bladder DNA samples was determined. While there were very small differences in the 5 ratios, all were close to 1.0 (0.98–1.03), and the coefficients of variation were very low: 1.07% to 2.00% for the intra-assay and 1.73% to 3.19% for the interassay variations (Suppl. Table 2).
The effect of DNA quality on the ddPCR assay was assessed with 12 paired fresh-frozen and FFPE DNA. Since the difference in copy number ratios between DNA samples of 2 different DNA qualities were comparable (difference: 0.05 ± 0.04), the data from fresh-frozen and FFPE DNA were combined. For each of the 12 normal fresh/FFPE pairs, the mean of the respective fresh-frozen and FFPE copy number ratios was utilized for further analysis.
Copy number ratios derived from oaCGH and ddPCR analyses were well correlated (R 2 = 0.86–0.96; Fig. 2), and there was clear linearity of the ratios obtained by ddPCR with the tumor/normal DNA dilution series (R 2 = 0.97–1.00; Fig. 2).
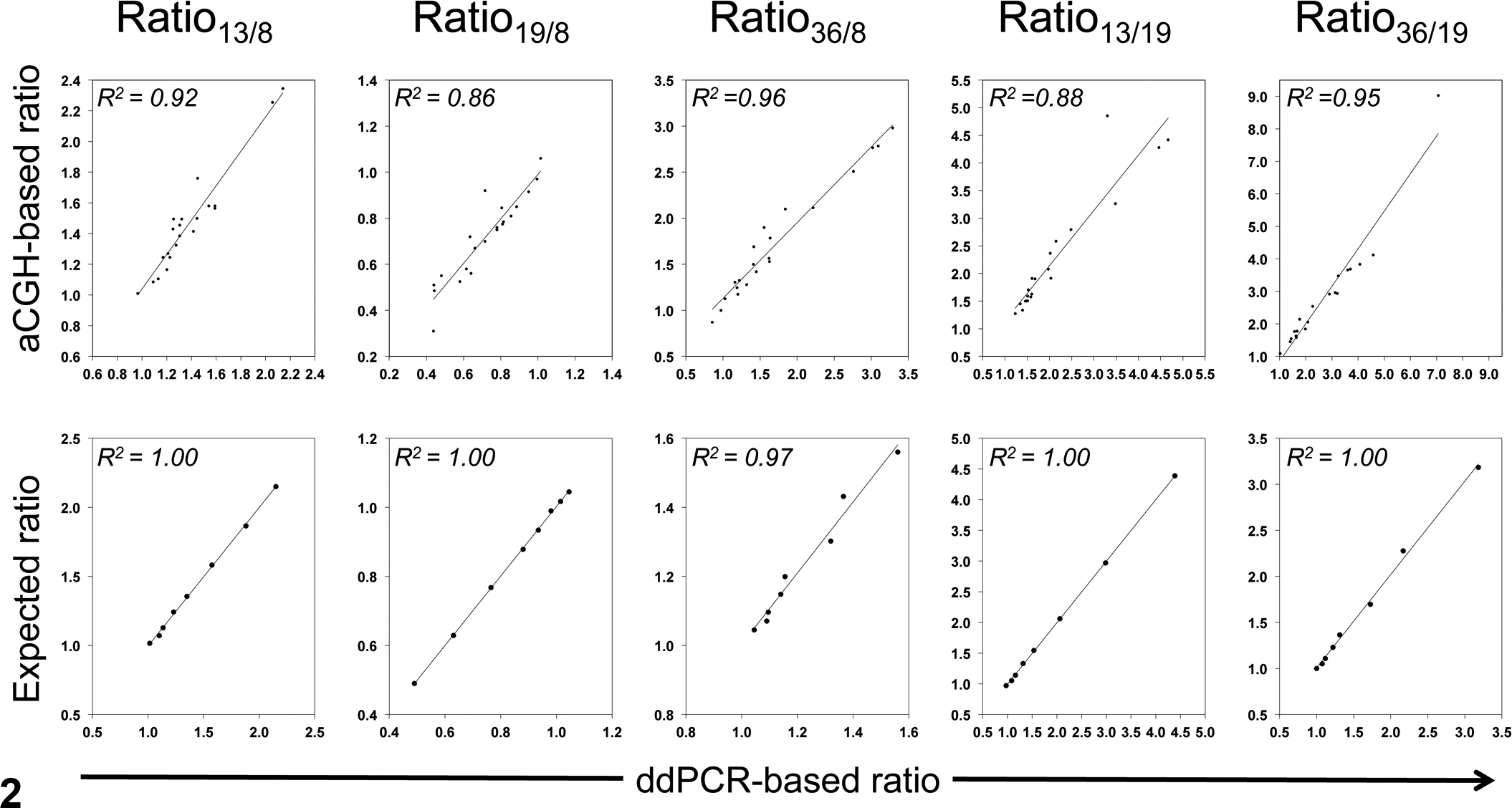
Performance of the droplet digital polymerase chain reaction (ddPCR) assays. Correlation with oligonucleotide array comparative genomic hybridization (oaCGH)–based ratio (upper row: x-axis, ddPCR-based ratio; y-axis, oaCGH-based ratio) and correlation with expected ratio in tumor/normal DNA dilution series (lower row: x-axis, ddPCR-based ratio; y-axis, expected ratio based on tumor DNA proportion) of ratio13/8, ratio19/8, ratio36/8, ratio13/19, and ratio36/19. Ratio indicates that between chromosomes (eg, CFA 13:CFA 8 = ratio13/8).
After the performance of ddPCR for this application was examined, the copy number ratios obtained from DNA derived from 25 nonneoplastic bladder tissues, and 31 UC tissues were assessed. UC samples showed significant increase in ratio13/8, ratio36/8, ratio13/19, and ratio36/19 (all P < .0001), and ratio19/8 showed significant decrease (P < .0001), as compared with nonneoplastic bladder tissue (Fig. 3). Receiver operating characteristic curve analysis was performed, and the thresholds that hold specificity of 100% (no false positives) were determined, as shown in Table 2. To simplify the analysis, deviation ≥0.25 (≥1.25 in ratio13/8, ratio13/19, ratio36/19, and ratio36/8 or ≤0.75 in ratio19/8) was used as a universal threshold of copy number imbalance. At this threshold, all UC cases had copy number imbalance in at least 1 of the 5 ratios. Copy number imbalance in each tumor and aberration frequencies in the cohort are summarized in Table 3. When a “positive” outcome was defined as the presence of at least 1 copy number imbalance, the sensitivity and specificity of UC detection were both 100% (31 of 31 and 25 of 25, respectively).
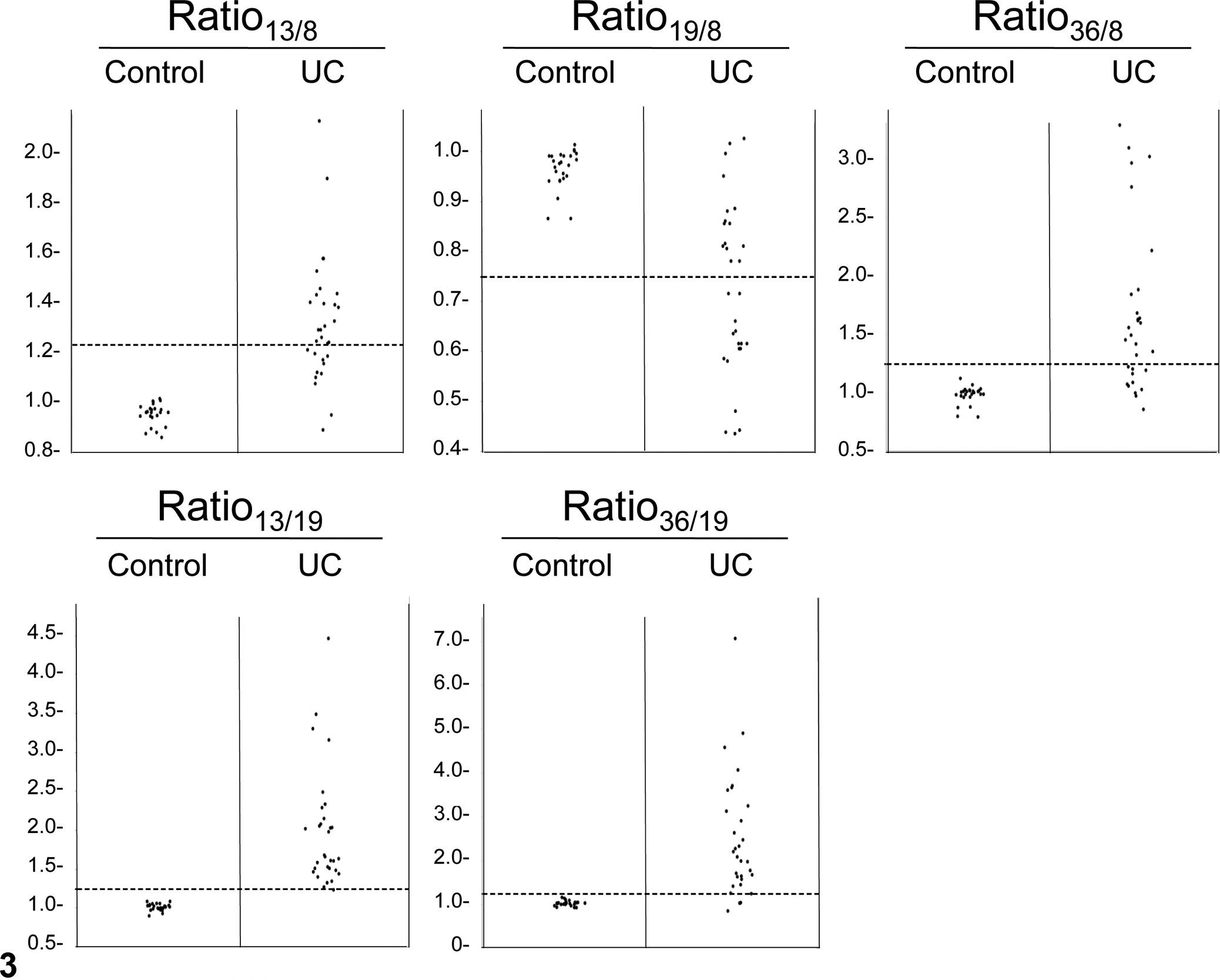
Copy number ratios in canine urothelial carcinoma (UC) and control bladder tissues. Broken lines indicate 0.25 difference from 1: the threshold value identified to separate normal and abnormal copy number ratios. Ratio indicates that between chromosomes (eg, CFA 13:CFA 8 = ratio13/8).
Receiver Operating Characteristic Curve Analysis and the Threshold for Copy Number Imbalance for Diagnosis of Canine Urothelial Carcinoma.
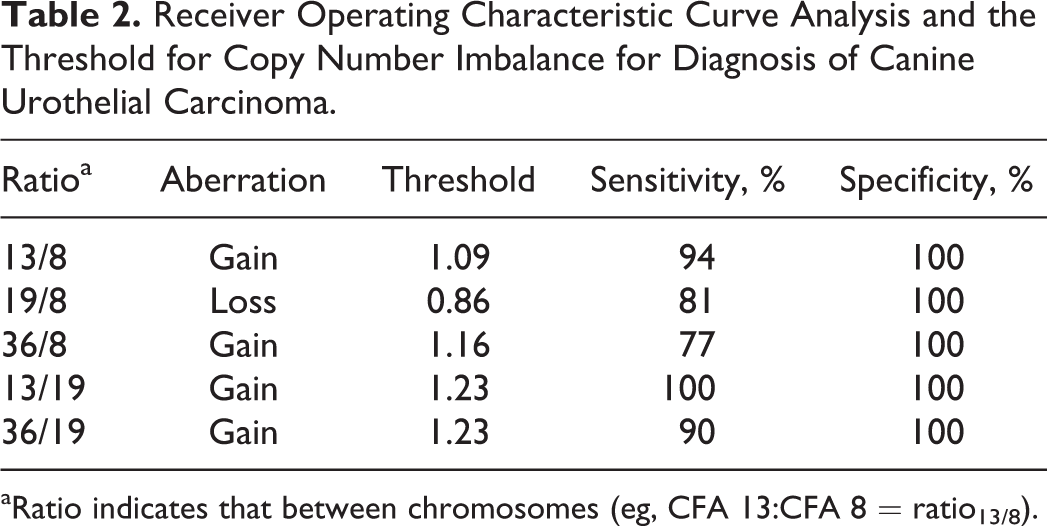
aRatio indicates that between chromosomes (eg, CFA 13:CFA 8 = ratio13/8).
DNA Copy Number Imbalance and Aberration Frequencies in Canine Urothelial Carcinoma Cases.a
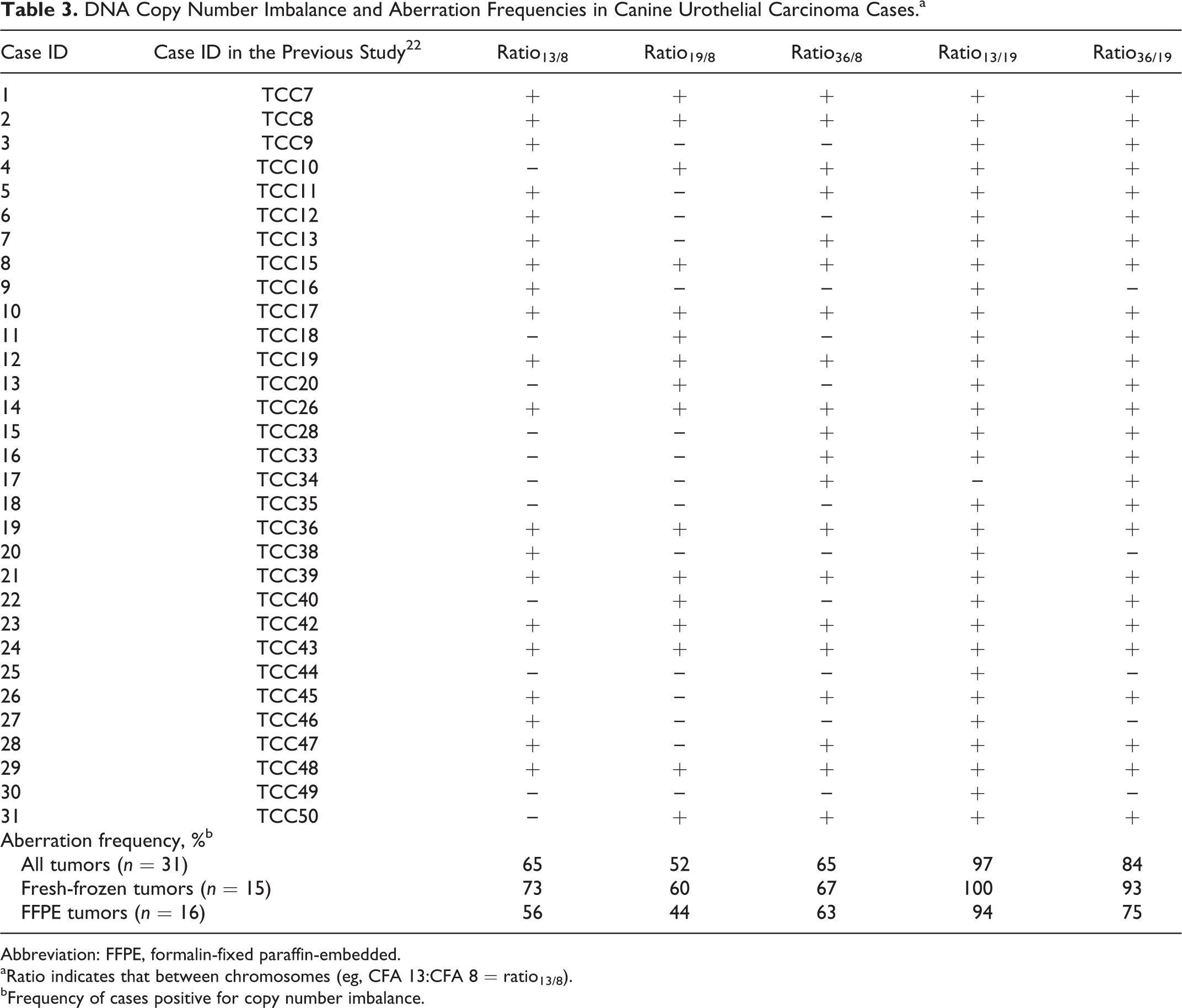
Abbreviation: FFPE, formalin-fixed paraffin-embedded.
aRatio indicates that between chromosomes (eg, CFA 13:CFA 8 = ratio13/8).
bFrequency of cases positive for copy number imbalance.
To consolidate these independent assays into a single tube, we developed a multiplex ddPCR assay and reevaluated all nonneoplastic and UC DNA samples. All the ratios were well correlated between duplex and multiplex ddPCR (R 2 = 0.98–1.00; Suppl. Fig. 1). With the same cutoff value, all 25 DNA samples derived from nonneoplastic bladder epithelium still showed normal copy number ratios. There were 5 minor discrepancies in the results of the UC cases when assessed with the multiplexed assay, with all 5 cases showing small ratio changes (0.22–0.32). All UC cases still showed copy number imbalance in at least 1 ratio in the multiplex analysis, maintaining a sensitivity and specificity of 100% in tissue samples.
Using the ddPCR assay developed in this study, we analyzed urine samples obtained from UC or non-UC (healthy control and urinary tract infection) dogs. Integrity of urine DNA was assessed by gel electrophoresis, and most of the urine-derived DNA specimens were of high molecular weight (mean fragment size >10 kilobases; Suppl. Fig. 2). First, we analyzed 4 tissue- and urine-matched samples of UC dogs to assess the copy number changes. Copy number imbalance was detected in both urine and tissue samples in 3 dogs, but in 1 case only the tumor sample had aberrant ratios (case No. 35; Table 4). These data demonstrate that our ddPCR assay is able to detect copy number imbalances in UC biopsy-derived DNA and, in most cases, the same imbalance is detectable in DNA obtained from free-catch urine of the same canine UC patient.
Comparison of DNA Copy Number Imbalance in Matched Tissue- and Urine-Derived DNA Samples in 4 Urothelial Carcinoma Cases.a
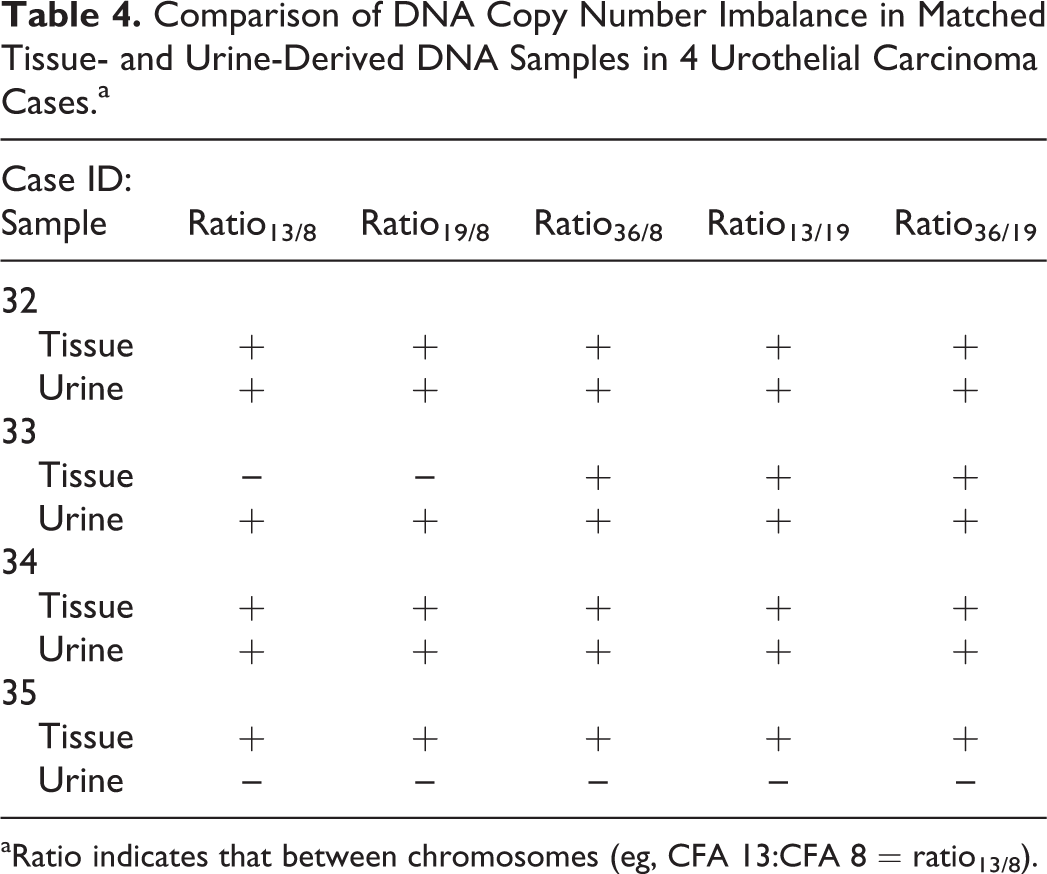
aRatio indicates that between chromosomes (eg, CFA 13:CFA 8 = ratio13/8).
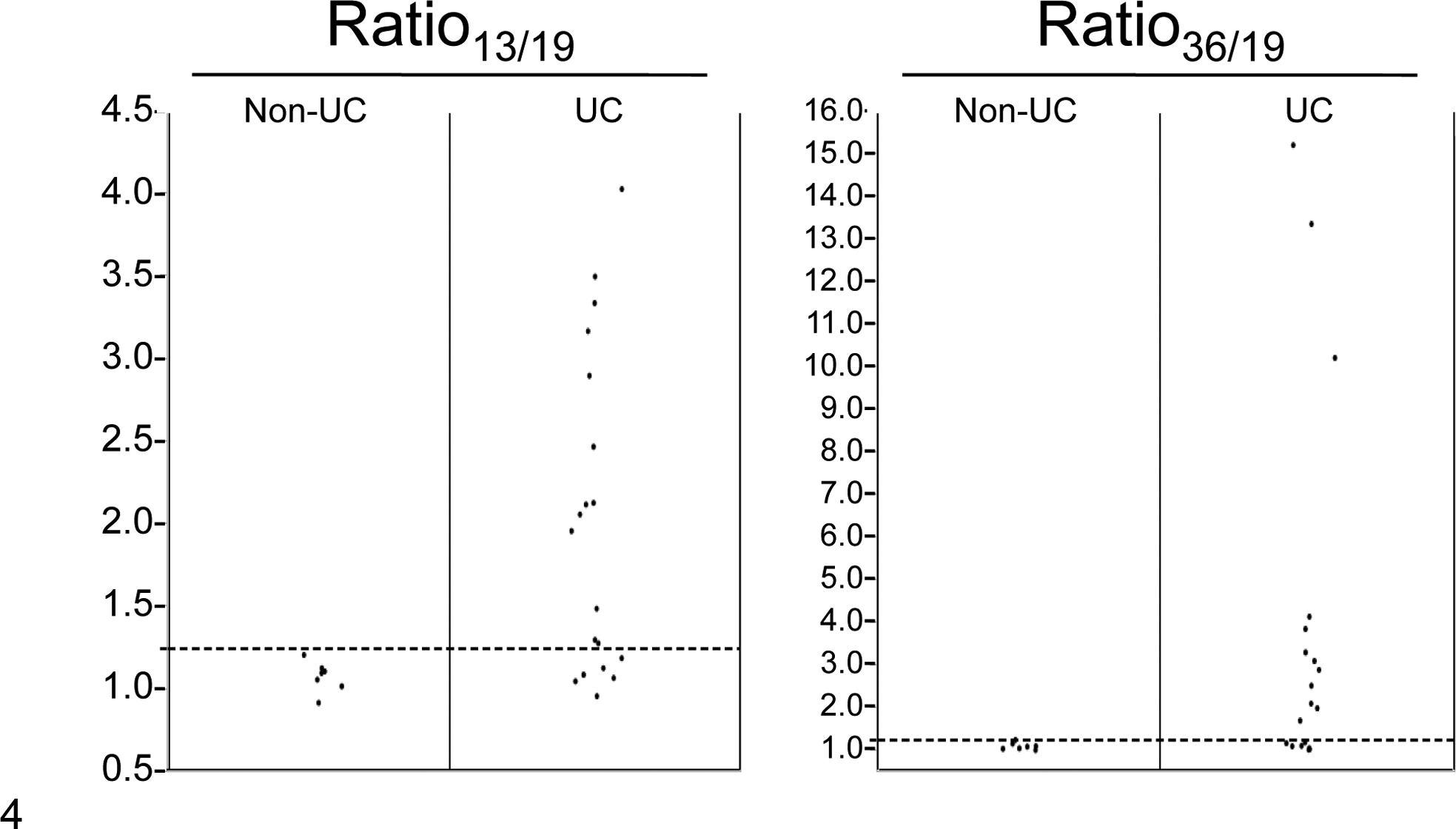
Next, we evaluated DNA obtained from an additional 14 UC and 7 non-UC urine samples. Unexpectedly, we identified 1 each of UC and non-UC samples that showed decreased ratios of 13:8, 19:8, and 36:8 at equal degree (ratios of 2:3 [∼0.67] and 2:4 [∼0.5], respectively), indicating the copy number gain of the CFA 8 region rather than simultaneous loss of the CFA 13, 19, and 36 regions. This copy number imbalance may indicate a disease-associated change or the presence of a natural copy number variation (gain) encompassing this CFA 8 “control” region in dog genome. Since all UC tissue samples evaluated had shown increase in their ratio13/19 and/or ratio36/19, these 2 ratios alone were considered sufficient to hold enough diagnostic value for differentiating UC and non-UC. Therefore, we evaluated only ratio13/19 and ratio36/19 for further analysis. Significant increases of ratio13/19 and ratio36/19 in UC urine specimens were observed as compared with non-UC urine specimens (Fig. 4; P = .0131 and .0168, respectively). In an additional 14 UC-derived urine specimens, 8 showed increased ratio13/19 and ratio36/19, and 1 showed increased ratio13/19 but normal ratio36/19, while none of 7 non-UC dog urine showed copy number imbalance. Combined with the result of the 4 tissue- and urine-matched UC samples, copy number imbalance was detected in 67% (12 of 18) of urine samples from dogs with UC.
Copy number ratios in urine samples obtained from canine urothelial carcinoma (UC) and non-UC patients. Broken lines indicate ratio of 1.25: the threshold value identified to separate normal and abnormal copy number ratios. Ratio indicates that between chromosomes (eg, CFA 13:CFA 8 = ratio13/8).
Discussion
Advances in molecular biotechnology have provided insight into the pathogenesis of various diseases at a molecular level. The knowledge of molecular mechanisms has also been translated and integrated into diagnostics, prognostics, and therapeutic selection in human medicine. Molecular pathology, in combination with conventional morphologic pathology, is becoming an integral part of clinical practice. In veterinary medicine, however, the knowledge of molecular mechanisms of many diseases is still limited, and molecular pathology has yet to become integrated into routine clinical diagnosis, with the exception of certain hematopoietic malignancies and infectious diseases. 2,18
Early diagnosis of bladder cancer (UC) and posttreatment monitoring pose a clinical challenge in human and veterinary medicine. As in veterinary medicine, histopathologic examination of a bladder mass obtained by cystoscopy is the gold standard for diagnosis of human bladder cancer. Due to the high recurrence rate of human bladder tumors, however, frequent cystoscopy is clinically and economically impractical. For this reason, molecular biomarkers to detect tumor cells in urine have been investigated as a noninvasive and cost-effective diagnostic means for bladder cancer in human medicine, including chromosomal alterations, somatic mutations of cancer-related genes, epigenetic alterations, and expression profiles of microRNA, mRNA, and protein. 7 Among various molecular markers, UroVysion—which utilizes a multicolor FISH assay for detection and enumeration of chromosomal changes of high frequency in human bladder cancer—has been approved by the Food and Drug Administration for the diagnosis of human bladder cancer and detection of tumor recurrence. 13 In a meta-analysis of 14 studies, the UroVysion assay demonstrated superior sensitivity (72%) and slightly decreased specificity (83%) when compared with routine cytologic analysis of urine samples (sensitivity 42%, specificity 96%) for detecting bladder cancer. 12
As in human bladder cancer, specific chromosomal alterations occur at high frequency in canine UC. 22 Conventional cytogenetic tools used to detect and quantify copy number changes, such as FISH and oaCGH, require highly skilled operators and are time-consuming and low throughput. Although FISH remains the gold standard of copy number enumeration in human cytogenetics, the limited throughput is associated with increased costs. Limited budgets for veterinary clinical practice mean that cost-effectiveness is often a driving factor for the clinical application of diagnostic assay. In this study, we demonstrated that ddPCR is a high-throughput, faster, and thus potentially less costly approach to determine DNA copy number imbalances without sacrificing accuracy. As digital PCR gains in popularity, several researchers are examining the potential of this technique for human oncology. For example, ddPCR has been used to detect HER2 gene amplification in human breast cancer, with data comparable to those derived from the conventional FISH-based assay. 3,11 We propose that ddPCR has the potential to be of clinical value as a rapid method to detect copy number imbalance in canine UC.
In this study, we developed a series of 3 custom duplex ddPCR assays to evaluate the presence and prevalence of 3 canine chromosome aberrations in canine UC: gain of CFA 13 and 36 and loss of CFA 19. The assays were used independently to detect and quantify DNA copy number in histopathologically confirmed cases of canine UC and nonneoplastic urothelium. The ddPCR-derived DNA copy number had high concordance with that indicated by oaCGH and showed high repeatability and linearity. We were able to multiplex the 3 duplex assays into a single quad-plex assay with minimal impact on the scoring of cases as positive or negative for the presence of ≥1 of the copy number aberrations evaluated. DNA isolated from all 25 nonneoplastic bladder tissues, including normal bladder and bladder with nonneoplastic lesions, showed minimal deviation from normal copy number, whereas UC cases showed variable degrees of copy number imbalance. With these data, we showed that these numeric copy number changes were restricted to the neoplastic urothelium, demonstrating a proof of concept that detection of such changes can be used as a specific molecular marker for UC. With a cutoff ratio difference of 0.25, sensitivity and specificity of the assay for the diagnosis of UC were both 100% in tissue specimens.
We next evaluated the ddPCR assay as a noninvasive diagnostic for use with free-catch urine samples derived from UC and non-UC canine patients. Unexpectedly, we encountered copy number change of the control region of CFA 8 against CFA 13, 19, and 36 in both UC and non-UC urine DNA. This is most likely a natural copy number variation in dog genome, 1 as the change was found in both neoplastic and nonneoplastic DNA, although the possibility of disease-associated change cannot be ruled out. Due to this unexpected finding, we evaluated only ratio13/19 and ratio36/19 to differentiate UC and non-UC urine. With these 2 ratios, copy number imbalance was detected in 67% of UC but absent in non-UC urine specimens. Although the sample size of this study is small and analysis of additional cohorts is necessary to assess the sensitivity and specificity of the assay, these data demonstrate that copy number imbalance consistent with canine UC was detectable in urine specimens of canine UC patients, suggesting that detection of such copy number imbalance may be a useful molecular marker for canine UC.
Despite the high sensitivity of the ddPCR assay in UC tissue samples, copy number imbalance was not detected in 33% of urine DNA obtained from dogs diagnosed with UC. This most likely resulted from an over abundance of copy number neutral inflammatory cells in these urine specimens, present due to coincidental secondary bacterial infections, which are common in dogs with UC. 16 Since the degree of copy number imbalance in UC specimens was relatively small (generally, copy number ratio <10), any imbalance would be diluted to an undetectable level in the presence of a large number of copy number–neutral nonneoplastic cells. To increase sensitivity of the ddPCR assay in such samples, enrichment of tumor cells via immunomagnetic beads or flow cytometry with antibodies for epithelial cell markers such as cytokeratins may be necessary. In a heterogeneous sample, however, gold standard FISH will remain preferable for accurate evaluation of copy number changes, as only epithelial cells need be analyzed by microscopy.
In conclusion, we developed a multiplexed ddPCR assay for the detection of copy number imbalances characteristic to canine UC. Additional research is warranted to evaluate the ddPCR assay as a molecular diagnostic test for canine UC in a clinical setting, using a cohort of dogs with UC and nonneoplastic urinary diseases.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Cancer Genomics Fund of the North Carolina State University College of Veterinary Medicine (M.B.).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
