Abstract
Urothelial carcinoma (UC) is the most common tumor affecting the urinary bladder of dogs. Protein overexpression of ErbB2 (the canine homolog of HER2) has been observed in dogs with UC. However, no study regarding ErbB2 copy number aberration (CNA) is reported in dogs with UC. In this study, a digital PCR assay for detecting CNA of canine ErbB2 was developed. DNA samples were isolated from 83 formalin-fixed, paraffin-embedded urinary bladder tissues (36 UC, 8 polypoid cystitis, and 39 normal) and 94 urinary sediments (54 UC, 30 nonneoplastic, and 10 normal). The copy number of canine chromosome 8 (CFA8) was used as a control. In the urinary bladder tissues, ErbB2 CNA was detected in 12 of 36 (33%) UC, 2 of 8 (25%) polypoid cystitis, and 0 of 39 (0%) normal controls. In the urinary sediments, ErbB2 CNA was also detected in 19 of 54 (35%) UC; however, no ErbB2 CNA was detected in nonneoplastic diseases or normal controls. The sensitivity and specificity of ErbB2 CNA in urinary sediment for the detection of UC were 35% and 100%, respectively. There was a positive correlation between the copy number ratios of ErbB2 to CFA8 in the urinary bladder tissues and urinary sediments. Our findings indicate that the digital PCR assay of urinary sediments may be a useful, noninvasive method for detecting ErbB2 CNA in dogs with UC.
Urothelial carcinoma (UC) is a malignant tumor derived from transitional epithelial cells of the urinary tract. It accounts for approximately 1% to 2% of all canine malignant tumors and is the most common tumor affecting the urinary bladder of dogs, 5,9 accounting for 74% of all primary bladder tumors. 25 The vast majority of canine UC (>90% of cases) are intermediate- to high-grade invasive types, and low-grade superficial types are uncommon. 33,47 At the time of diagnosis, metastases to lymph nodes or distant tissues (eg, lungs, bone, liver) are reported in approximately 15% of cases. 18 Unfortunately, most dogs with UC die or are euthanized. 19,25 In a previous study, we analyzed urinary bladder tissues of healthy dogs and dogs with UC by RNA sequence analyses and showed that the pathway for ErbB2 (the canine homolog of HER2) oncogene was significantly activated during UC carcinogenesis. 22
ErbB2 is a cell surface receptor tyrosine kinase belonging to the ErbB family (ErbB1–4) and has the strongest catalytic kinase activity, forming the most active signalling complex in the family after dimerization with other members. 10,12 Under normal circumstances, ErbB2 is involved in cell proliferation, differentiation, angiogenesis, and migration. 13 ErbB2 copy number aberration (CNA) is caused by gene amplification and polysomy, and ErbB2 protein overexpression is observed in a variety of human malignancies such as bladder, breast, stomach, ovary, lung, and colon cancer. 43,50,51 Moreover, CNA and protein overexpression of ErbB2 are associated with poor prognosis. 30,32,35,45 Recent studies have shown that ErbB2 protein overexpression was observed in 56% to 61% of dogs with UC, 26,46 which was similar to that observed in humans; however, there have been no reports regarding ErbB2 CNA in dogs with UC.
The copy number of ErbB2 is generally detected using fluorescence in situ hybridization. However, this method is time-consuming, labor intensive, and expensive. On the other hand, recent studies have shown that digital PCR (dPCR) could help detect ErbB2 CNA. 14,17,49,52 dPCR is performed by partitioning PCR mixtures into a large number of compartments, with each compartment containing either ≥1 (positive) or 0 (negative) target sequence. After conventional amplification with thermocycling, each compartment is identified as being either positive or negative by assessing the fluorescence signal intensity at the endpoint. The number of positive and negative compartments for each fluorochrome and Poisson’s distribution are then used together to calculate the absolute number of target sequences in each sample, without using calibration standards. 48
In this study, we developed a dPCR assay for routine assessment of canine ErbB2 CNA. Using this assay with DNA samples isolated from formalin-fixed, paraffin-embedded (FFPE) urinary bladder tissues and urinary sediments, we evaluated the presence of ErbB2 CNA in dogs with UC.
Materials and Methods
DNA Samples
All procedures in this study were conducted in accordance with the guidelines of the Animal Care Committee of the Graduate School of Agricultural and Life Sciences, the University of Tokyo (approval number: P17-108, 2 October 2017). In total, FFPE urinary bladder tissues of 83 dogs (36 UC, 8 polypoid cystitis, and 39 normal) were used in this study (Supplemental Table S1). Diagnoses were based on histopathological evaluation. DNA was isolated with the QIAmp DNA FFPE Tissue Kit (QIAGEN, Hilden, Germany) according to the manufacturer’s instructions. A previous study, using a dPCR assay, has shown that detection of the v-raf murine sarcoma viral oncogene homologue B1 (BRAF) mutation is of high sensitivity (75%–83%) and specificity (100%) for diagnosis of UC. 28 To examine BRAF mutation, a portion of the tissue DNA samples was sent to the Molecular Diagnostic Laboratory, the University of Tokyo. 8 Although primers and probes sequence of the dPCR BRAF mutation assay are slightly different from that reported in the previous studies (Table 1), 8,28 the sensitivity (76%) and specificity (100%) for diagnosis of UC are similar. Of 36 UC, 22 (61%) UC were BRAF mutation-positive, whereas 14 (39%) UC were BRAF mutation-negative. No BRAF mutation was detected in polypoid cystitis and the normal controls.
Nucleotide Sequences of Primers and Probes Used in this Study.

Abbreviations: bp, base pairs; CFA8, canine chromosome 8.
A total of 94 canine urinary sediments (54 UC, 30 nonneoplastic, and 10 normal) were also used in this study (Supplemental Table S2). Urine samples were collected with urethral catheters from 54 dogs with UC presenting at the Veterinary Medical Center of the University of Tokyo from June 2015 to August 2017. Of the 54 UC, 22 were diagnosed by histopathological evaluation of excised mass lesions, whereas 32 UC were diagnosed based on clinical signs, presence of bladder mass on ultrasound, cytological evidence of abnormal epithelial cells in urinary sediments, and the evidence of tumor progression (invasion into bladder wall, lymph node involvement, or distant metastasis) during the follow-up period. Urine samples from 30 dogs with nonneoplastic diseases (including 16 cystitis, 10 polypoid cystitis, and 4 cystolithiasis) and 10 clinically healthy dogs were used as disease and normal controls, respectively. DNA was isolated with the DNeasy Blood and Tissue Kit (QIAGEN), according to the manufacturer’s instructions. A portion of the urinary DNA samples was sent to the Molecular Diagnostic Laboratory to examine BRAF mutation. Of 54 UC, 38 (70%) UC were BRAF mutation-positive, whereas 16 (30%) UC were BRAF mutation-negative. No BRAF mutation was detected in the disease and normal controls.
dPCR
Primers and TaqMan MGB probes of ErbB2 and a reference gene were designed with Primer Express Software (Thermo Fisher Scientific, MA, USA). 29 Since ErbB2 is located in canine chromosome 9, the reference gene was designed within the copy number-neutral 1-Mb region of canine chromosome 8 (CFA8). 42 Sequences of the primers and probes used in this study are shown in Table 1. The ErbB2 and CFA8 probes were labeled with 6-carboxyfluorescein (FAM) and 4,7,2′-trichloro-7′-phenyl-6-carboxyfluorescein (VIC), respectively. All primers and probes were custom-made by Thermo Fisher Scientific.
PCR was performed on a QuantStudio 3D Digital PCR system (Thermo Fisher Scientific), as described previously. 16 In brief, each 15-µL reaction mixture contained 2 × QuantStudio 3D Digital PCR Master Mix v2 (Thermo Fisher Scientific), 900 nM of each primer, 200 nM of each probe, and 20 ng of genomic DNA from FFPE urinary bladder tissues or 20 to 30 ng of genomic DNA from urinary sediments. The reaction mixtures were loaded onto a QuantStudio 3D Digital PCR 20 K Chip v2 (Thermo Fisher Scientific) using a QuantStudio 3D Digital PCR Chip Loader (Thermo Fisher Scientific). The 20 K chip contained 20 000 individual wells, within which DNA was randomly and uniformly distributed. The chip containing the reaction mixture underwent thermocycling in a ProFlex 2× Flat PCR System (Thermo Fisher Scientific) under the following conditions: Denaturation was performed at 96°C for 10 minutes; 39 cycles were carried out at 60°C for 2 minutes, 98°C for 30 seconds, and 60°C for 2 minutes, after which the temperature was maintained at 10°C. After the dPCR assay, the PCR chip was loaded onto a QuantStudio 3D Digital PCR Instrument (Thermo Fisher Scientific). The positive and negative plots were counted, and ErbB2:CFA8 ratios were calculated using Poisson’s distribution with QuantStudio 3D AnalysisSuite version 3.1.2 (Thermo Fisher Scientific, Fig. 1).
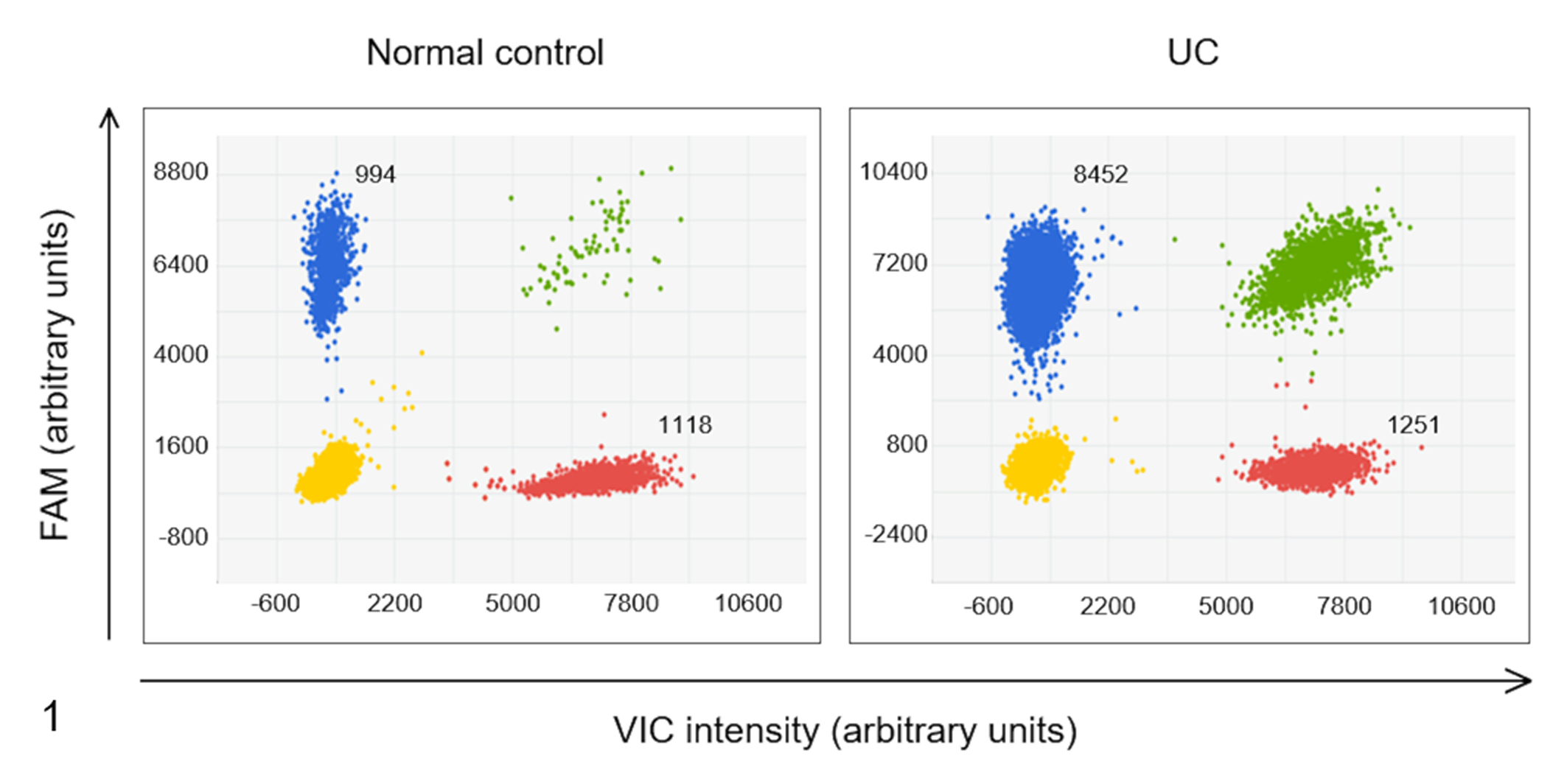
Two-dimensional scatter plots of the digital polymerase chain reaction assay for detecting ErbB2 copy number aberration in urine DNA samples obtained from a healthy control dog and a dog with urothelial carcinoma (UC). ErbB2 was labeled with 6-carboxyfluorescein (FAM), whereas the reference gene in CFA8 was labeled with 4,7,2′-trichloro-7′-phenyl-6-carboxyfluorescein (VIC). Four clusters were identified to be single-positive for FAM (blue) and VIC (red), double-positive (green), and double-negative (yellow). The ratio of blue plot points to red plot points in the dog with UC (8425 to 1251) was much higher than that in the healthy control (994 to 1118).
Immunohistochemistry for ErbB2
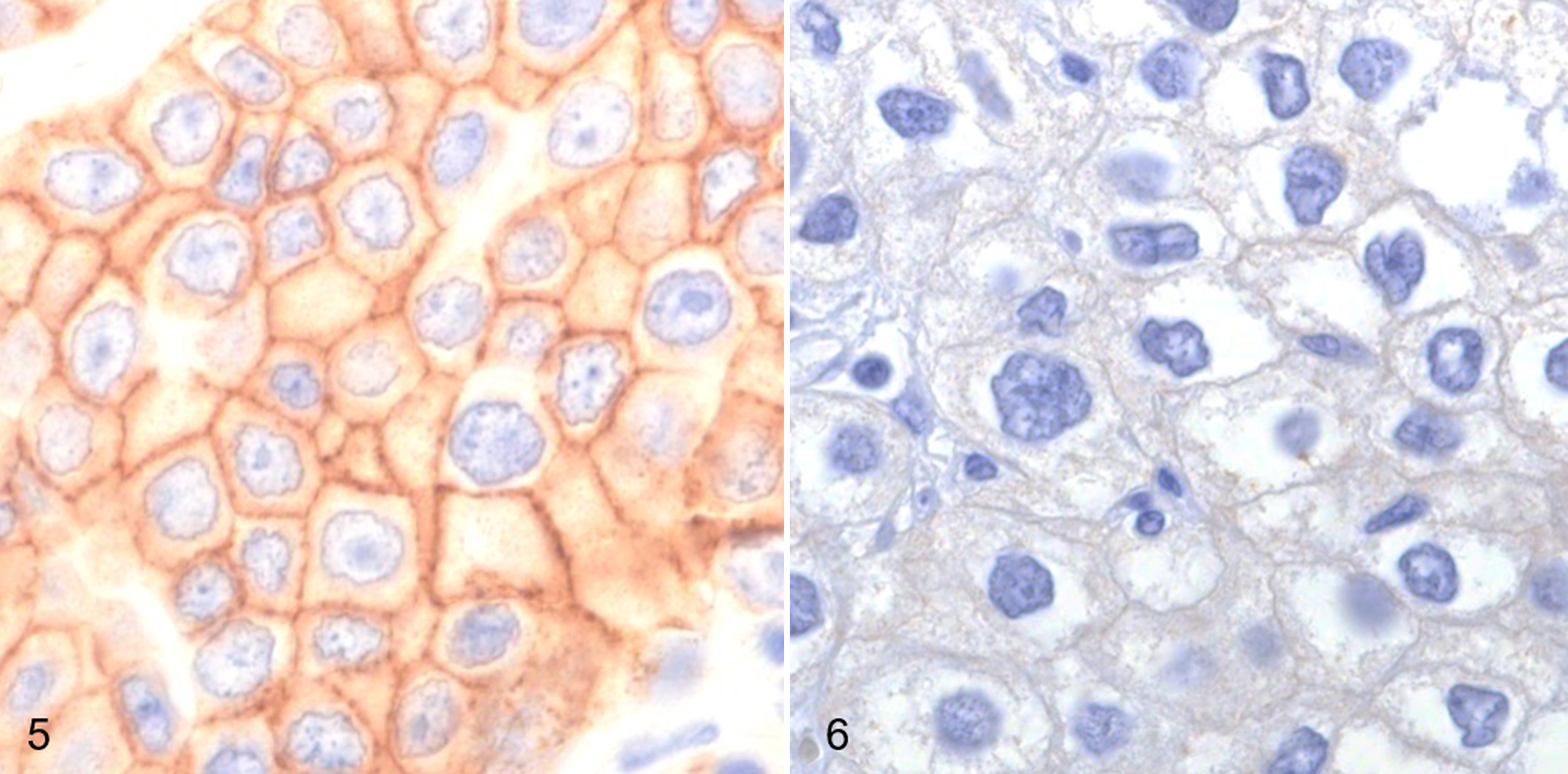
In this study, tissue DNA samples were extracted from 36 UC cases. In 22 of these cases, ErbB2 protein expression had been evaluated in our previous study. 46 Briefly, 4-µm sections were deparaffinized, rehydrated, and treated with 3% hydrogen peroxide-methanol at room temperature for 5 minutes and then heated in a water bath at 98°C for 40 minutes in target retrieval solution (pH 9.0, Dako, Glostrup, Denmark). After being cooled for an hour, the sections were washed in Tris-buffered saline (TBS) and then incubated in 8% skimmed milk-TBS at 37°C for 40 minutes. The sections were incubated with rabbit polyclonal anti-human c-erbB-2 oncoprotein (HER2/neu) antibody (1:100 in dilution, Dako) at 37°C for 40 minutes and then with Envision horseradish peroxidase-labeled anti-rabbit IgG polymer (Dako) at 37°C for an hour. The reacted products were visualized using 3.3’-diaminobenzidine (Dojindo, Kumamoto, Japan) and 0.03% H2O2 in TBS. Counterstaining was conducted with Mayer’s hematoxylin. ErbB2 protein overexpression was defined as intense immunoreactivity in ≥10% of tumor cells. Positive controls were prepared with canine mammary carcinoma tissue, which is known to show ErbB2 overexpression according to a previous report. 23 Negative controls were performed by omitting the primary antibody or adding isotype control antibody instead of primary antibody.
Clinical Outcomes in Dogs With UC
Survival analyses included the dogs with UC that underwent total cystectomy and subsequent urinary diversion to the prepuce or vagina 37 or partial cystectomy to remove tumor. The cases that underwent partial cystectomy only for biopsy were excluded from the analyses. Information regarding the current status (alive, dead, or lost) and disease recurrence at the end of the study (13 September 2017) was obtained for each UC case based on the medical records or a fax interview with the referring veterinarians. Diagnosis of recurrence was based on ultrasonography and/or urine cytology. Disease-free survival (DFS) was defined as the duration from surgery until recurrence or death at the end of the study. Overall survival (OS) was defined as the interval between surgery and the established cause of death of the animal at the end of the study (13 September 2017).
Statistical Analyses
To determine the differences in ErbB2:CFA8 ratios among the 3 groups, the Kruskal-Wallis test and post hoc Dunn’s multiple comparison test were performed. Fisher’s exact test was used to determine the association between ErbB2 CNA and BRAF mutation. Survival curves were generated with the Kaplan-Meier method, and survival rates were compared using the log-rank test with Bonferroni correction. The correlation of 2 values was evaluated with Spearman rank correlation analysis. These statistical analyses were performed using Prism software, version 5.0.1 (Graph Pad Software). Statistical significance was defined as P < .05.
Results
Intra- and Interassay Variations of the Canine ErbB2 Copy Number Assay
The dPCR assay used to measure the ErbB2:CFA8 ratio was optimized with DNA samples isolated from the urinary sediments of 4 normal controls. Tests to determine intra-assay (3 different reactions per plate) and interassay (1 of each reaction in 3 different plates) variations were performed. The intra-assay and interassay coefficients of variations were low: 2.69% to 4.52% and 1.75% to 4.65%, respectively.
ErbB2 Copy Number in Urinary Bladder Tissues
Tissue ErbB2:CFA8 ratios in UC were significantly higher than those in the normal controls (P < .0001) but were not significantly higher than those in polypoid cystitis (Fig. 2). There were no significant differences between the tissue ErbB2:CFA8 ratios in the normal controls and polypoid cystitis. To determine universal threshold of ErbB2 CNA in urinary bladder tissues, the mean +3 standard deviation of the tissue ErbB2:CFA8 ratios in the normal controls was calculated (1.00). At this threshold, ErbB2 CNA was detected in 12 of 36 (33%) UC and 2 of 8 (25%) samples of polypoid cystitis (Table 2), but no ErbB2 CNA was detected in the normal controls. The sensitivity and specificity of tissue ErbB2 CNA for differentiating UC from massive lesions of the urinary bladder were 33% and 75%, respectively. ErbB2 CNA was detected in 8 of 22 (36%) BRAF mutation-positive and 4 of 14 (29%) BRAF mutation-negative UC. There was no association between ErbB2 CNA and BRAF mutation in UC (P = .7272; Supplemental Table S3).
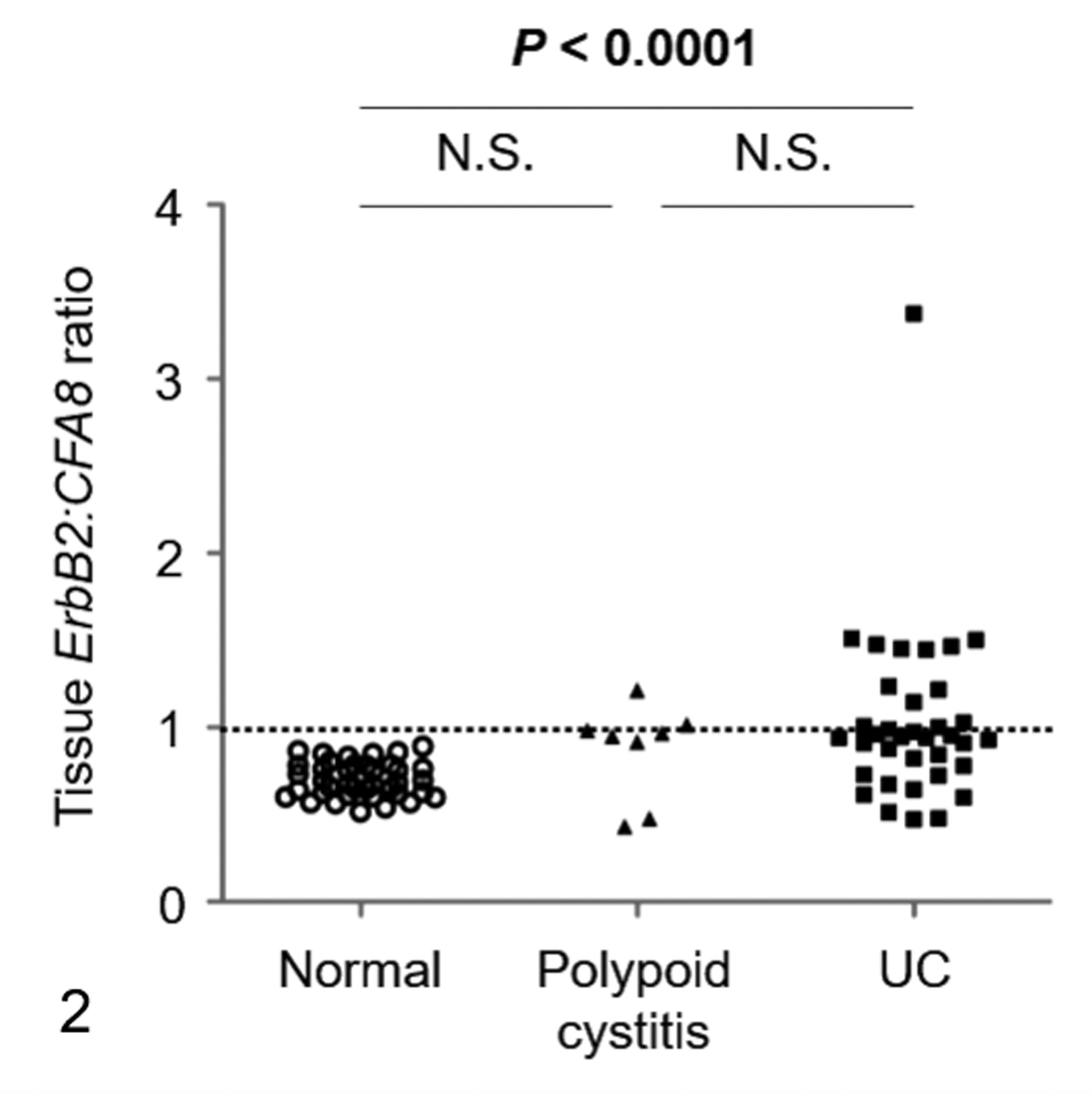
ErbB2:CFA8 ratios in formalin-fixed paraffin-embedded (FFPE) tissues of normal urinary bladder (n = 39), polypoid cystitis (n = 8), and urothelial carcinoma (UC, n = 36). The dotted line indicates a universal threshold (>1.0) for detecting ErbB2 copy number aberration. N.S., not significant.
ErbB2 Copy Number Aberration (CNA) in Urinary Bladder Tissues and Urinary Sediments of Dogs.
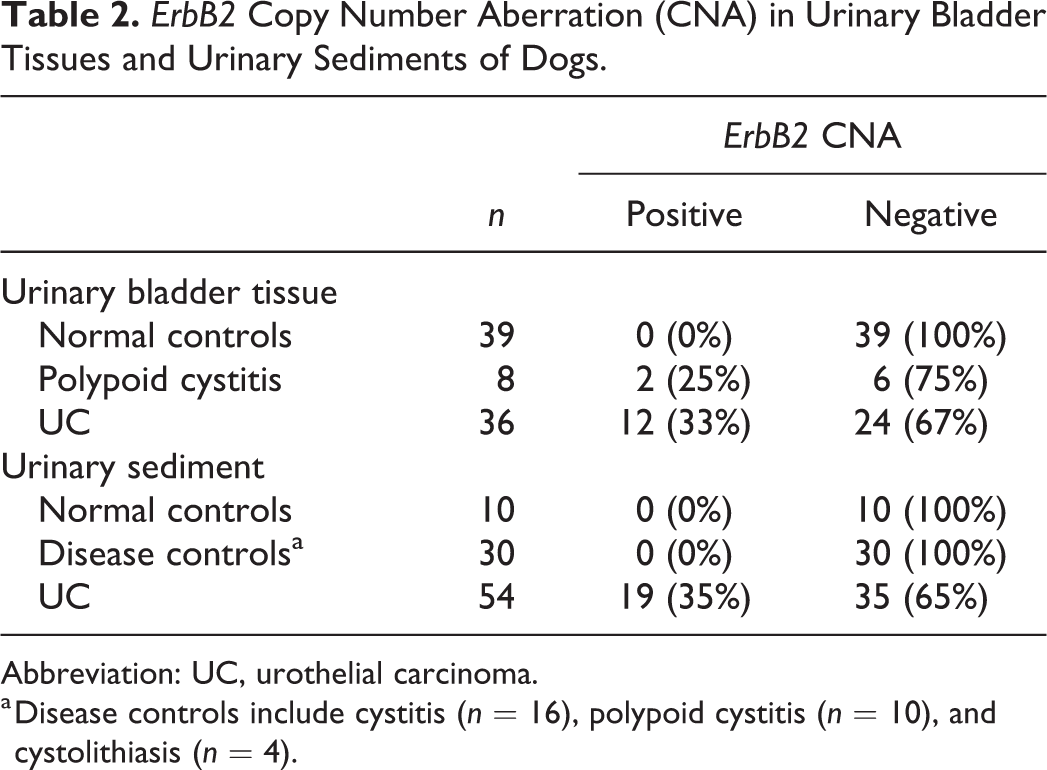
Abbreviation: UC, urothelial carcinoma.
a Disease controls include cystitis (n = 16), polypoid cystitis (n = 10), and cystolithiasis (n = 4).
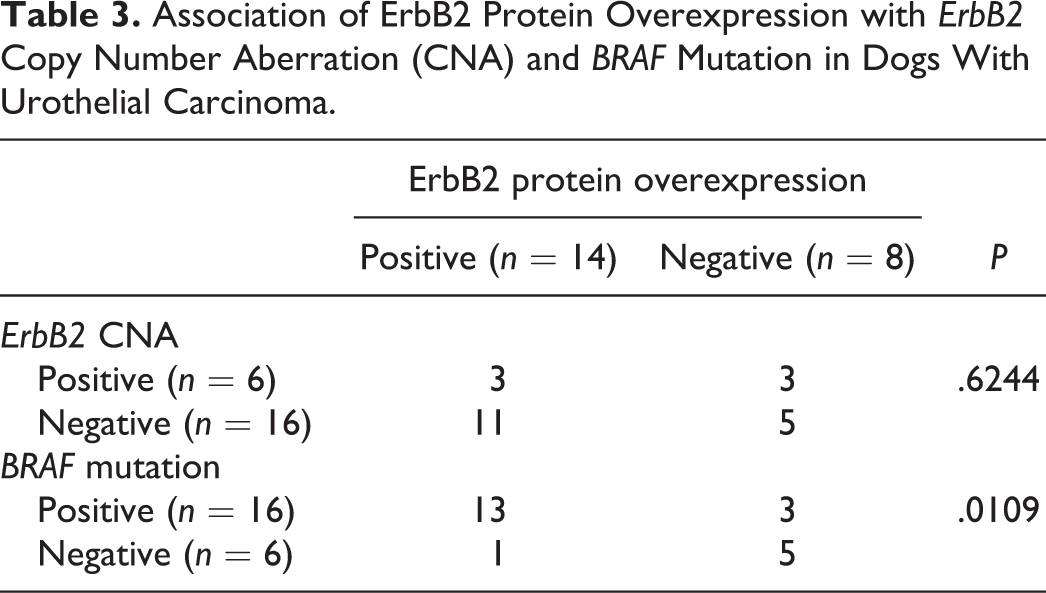
Of the UC, ErbB2 protein expression was examined in 22 cases. 46 ErbB2 protein overexpression was observed in 14 of 22 (64%) cases (Figs. 5, 6). ErbB2 CNA was detected in 3 of 14 (21%) cases with ErbB2 overexpression. On the other hand, ErbB2 overexpression was observed in 3 of 6 (50%) cases with ErbB2 CNA. There was no significant association between CNA and protein overexpression of ErbB2 (Table 3). On the other hand, BRAF mutation was detected in 13 of 14 (93%) cases with ErbB2 overexpression. There was significant association between BRAF mutation and ErbB2 overexpression (Table 3).
Association of ErbB2 Protein Overexpression with ErbB2 Copy Number Aberration (CNA) and BRAF Mutation in Dogs With Urothelial Carcinoma.
ErbB2 Copy Number in Canine Urinary Sediments
Urinary ErbB2:CFA8 ratios were significantly higher in UC than those in the normal and disease controls (P < .05; Fig. 3). There was no significant difference between the urinary ErbB2:CFA8 ratios in the normal and disease controls. To determine universal threshold of ErbB2 CNA in urinary sediments, the mean +3 standard deviation of the urinary ErbB2:CFA8 ratio in the normal controls was calculated (1.13). To simplify the analysis, urinary ErbB2:CFA8 ratio >1.1 was defined as the universal threshold. At this threshold, ErbB2 CNA was detected in 19 of 54 (35%) UC (Table 2), but no ErbB2 CNA was detected in the normal and disease controls. The sensitivity and specificity of urinary ErbB2 CNA for differentiating UC from the other urinary bladder diseases were 35% and 100%, respectively. ErbB2 CNA was detected in 14 of 38 (37%) BRAF mutation-positive and 5 of 16 (31%) BRAF mutation-negative UC. There was no association between ErbB2 CNA and BRAF mutation in UC (P = .7639; Supplemental Table S3).
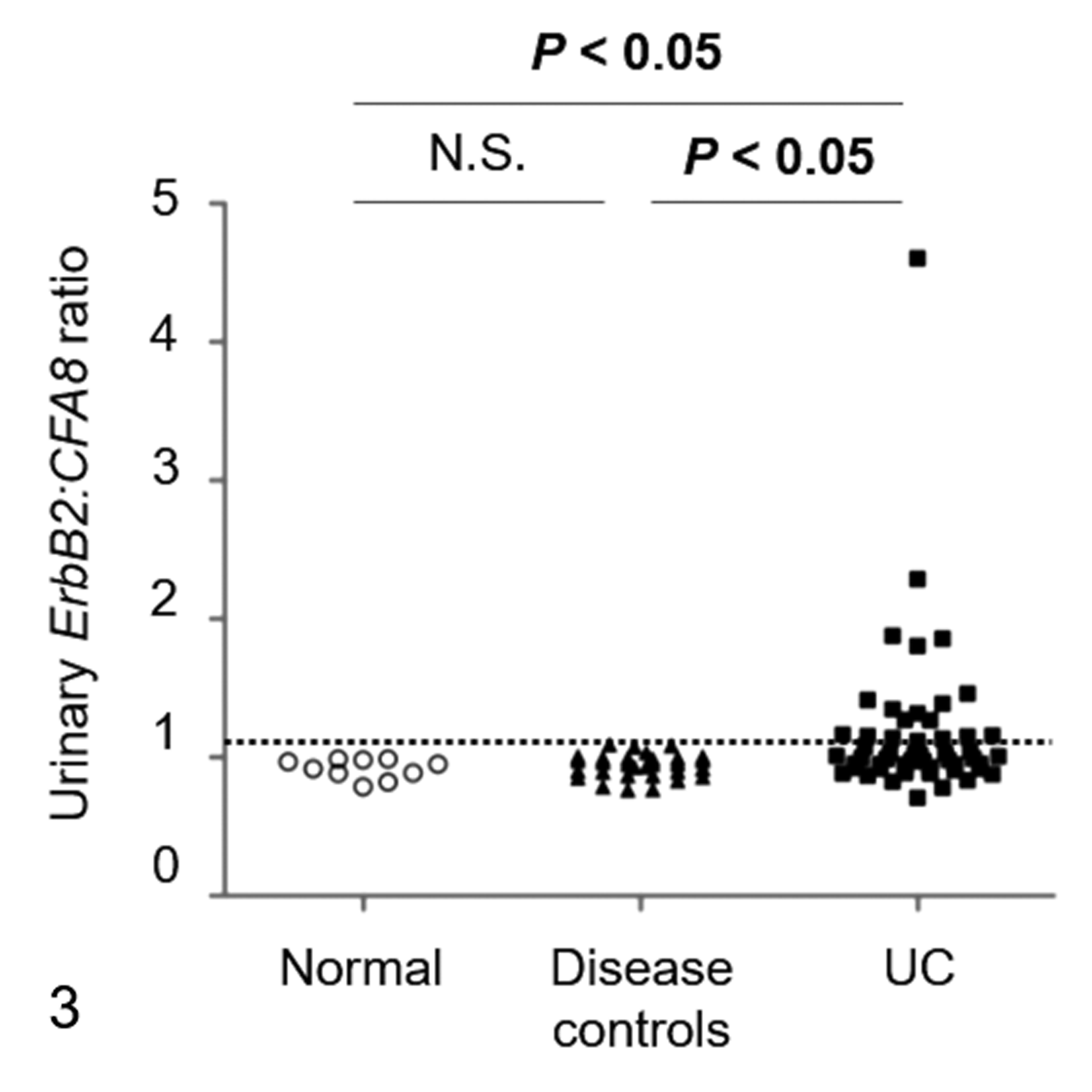
ErbB2:CFA8 ratios in urinary sediments of normal controls (n = 10), disease controls (n = 30), and urothelial carcinoma (UC, n = 54). The dotted line indicates a universal threshold (>1.1) for detecting ErbB2 copy number aberration. The disease controls included cystitis (n = 16), polypoid cystitis (n = 10), and cystolithiasis (n = 4). N.S., not significant.
Both tissue and urinary ErbB2:CFA8 ratios were evaluated in 10 dogs with UC and 1 dog with polypoid cystitis. There was significant correlation between tissue and urinary ErbB2:CFA8 ratios in these cases (r = .7182, P < .05; Fig. 4).
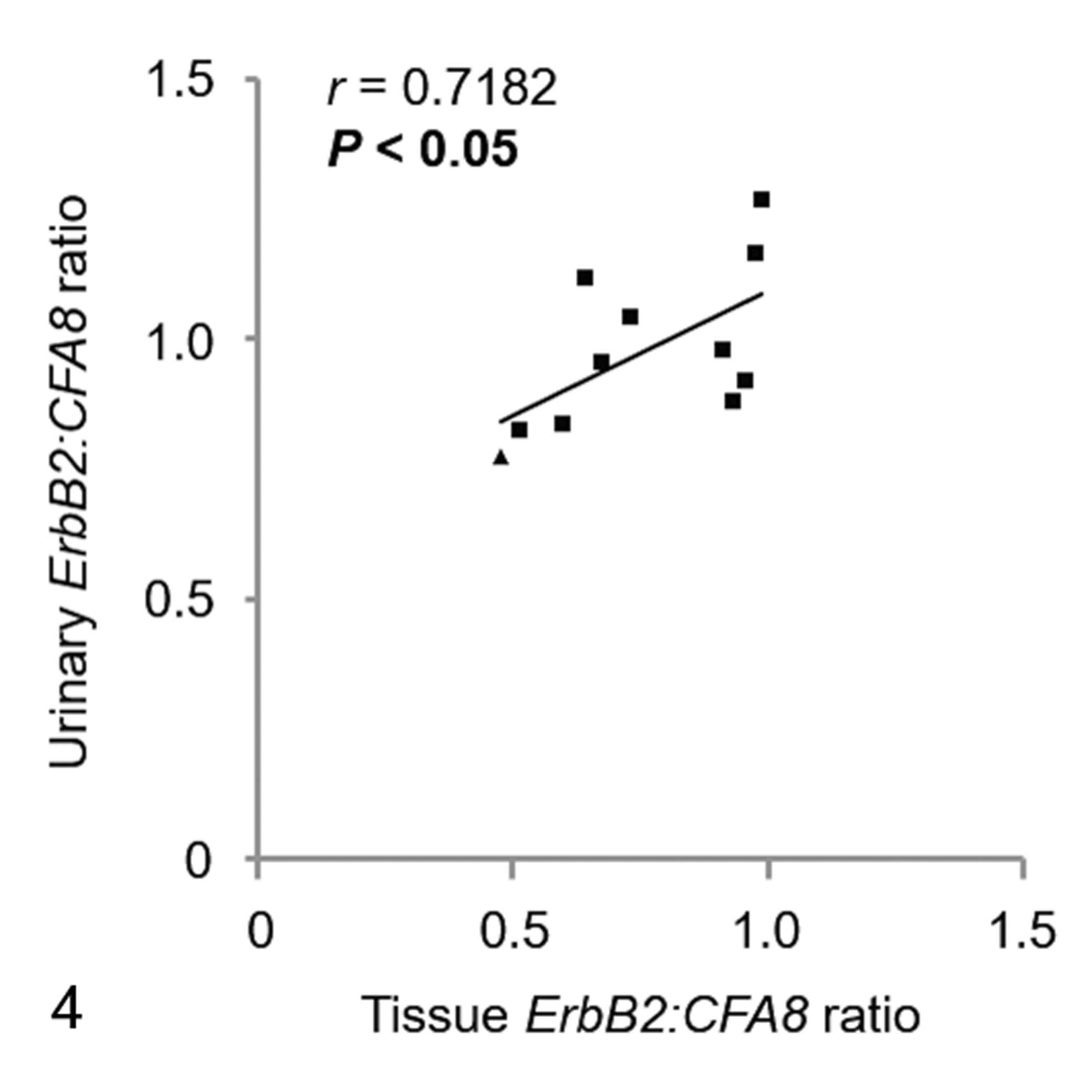
Correlation between ErbB2:CFA8 ratios of urinary bladder tissues and urinary sediments in 10 urothelial carcinomas (square) and 1 sample of polypoid cystitis (triangle).
Association With Clinical Outcomes
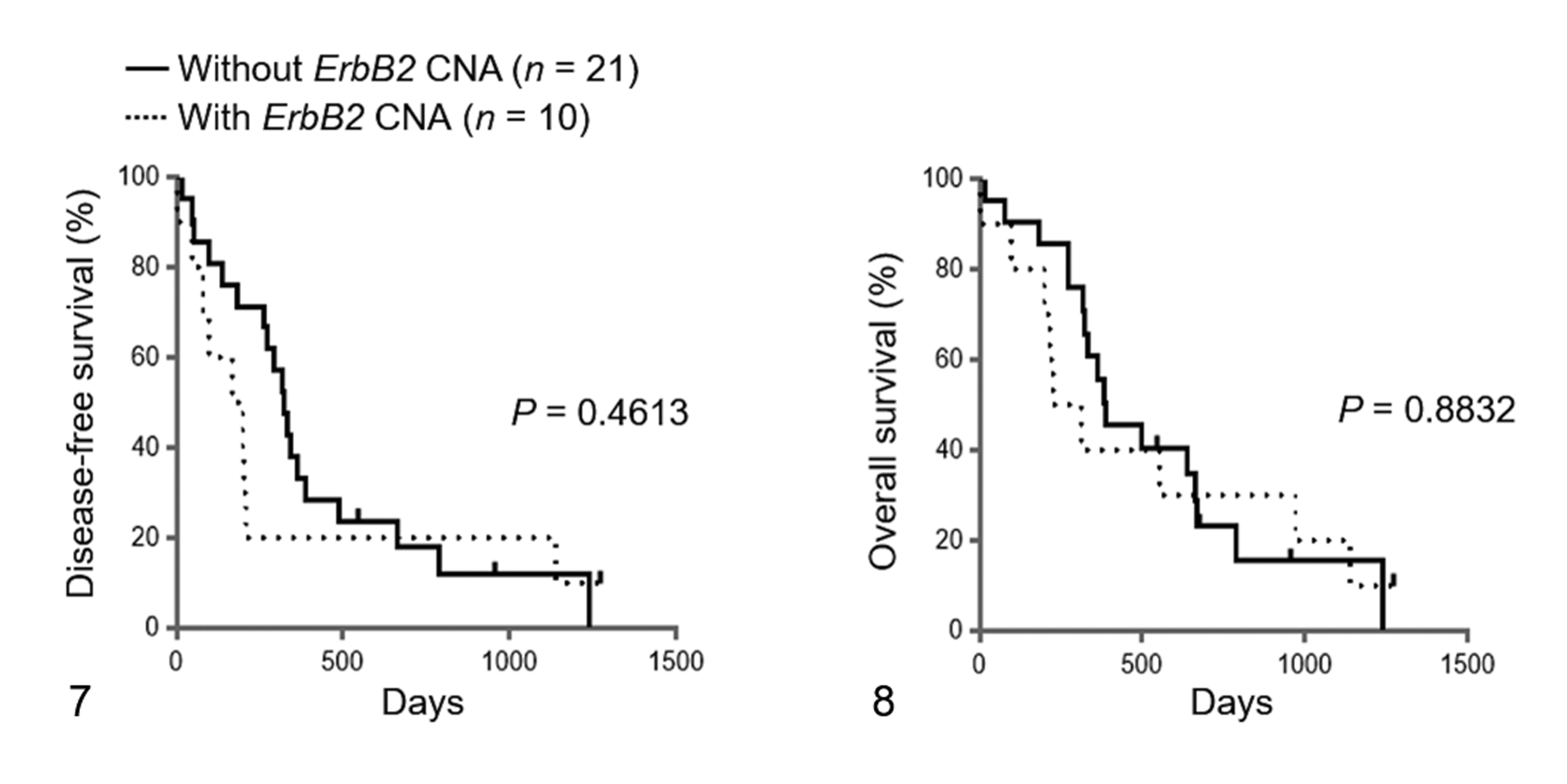
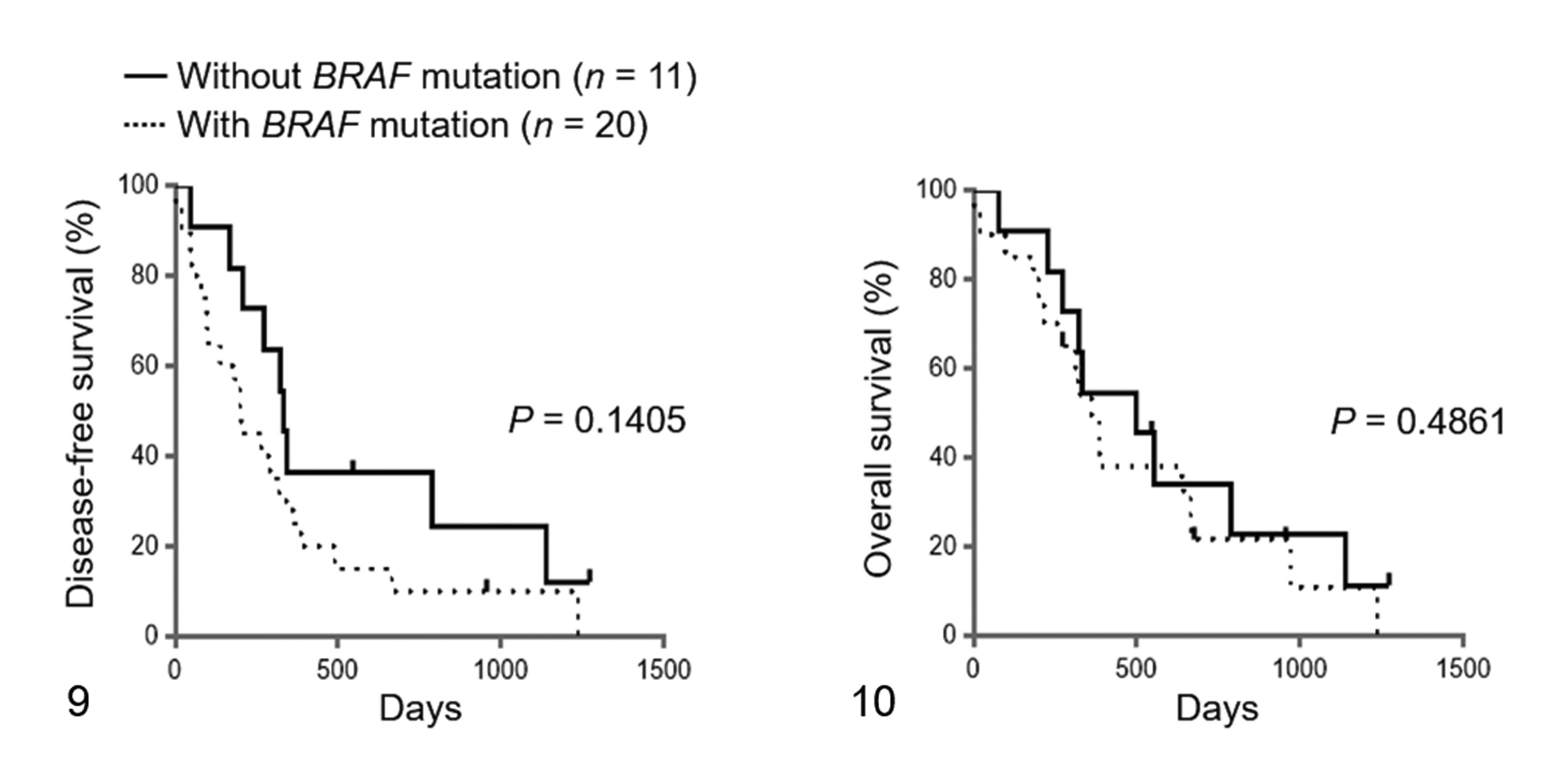
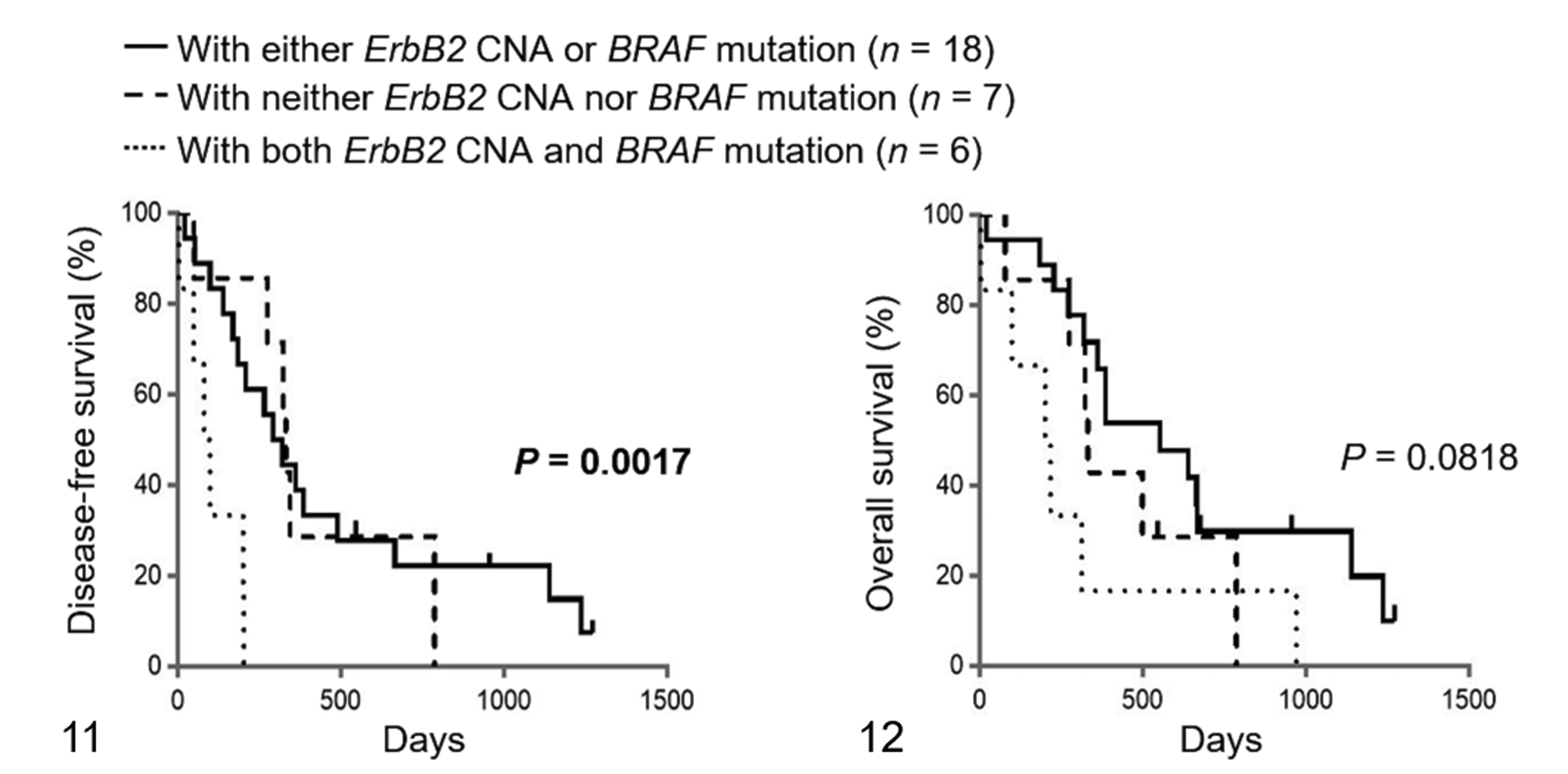
For the survival analyses, 31 dogs with UC that underwent total or partial cystectomy were used. These dogs included 6 cases with both ErbB2 CNA and BRAF mutation, 4 cases with only ErbB2 CNA, 14 cases with only BRAF mutation, and 7 cases with neither ErbB2 CNA nor BRAF mutation. There was no significant difference in age, sex, neutered status, TNM classification, and preoperative and postoperative therapies between the cases with and without ErbB2 CNA (Table 4). Of the 31 dogs with UC, 13 relapsed during the study period. The dogs exhibiting relapse included 5 cases with ErbB2 CNA and 8 cases without ErbB2 CNA. Twenty-five out of the 31 dogs with UC died during the study period. The cause of death is shown in Supplemental Table S1. No cases were euthanized. The dogs that died included 9 cases with ErbB2 CNA and 16 cases without ErbB2 CNA. The median DFS in the cases with and without ErbB2 CNA was 184 days (range, 3–1271 days) and 322 days (range, 20–1236 days), respectively. The median OS in the cases with and without ErbB2 CNA was 269 days (range, 3–1271 days) and 386 days (range, 20–1236 days), respectively. No significant difference was observed in the DFS and OS between the 2 groups (Figs. 7, 8). Similarly, there was no significant difference in the DFS and OS between the cases with and without BRAF mutation (Figs. 9, 10). Interestingly, there were significant differences in the DFS among the cases with both ErbB2 CNA and BRAF mutation, cases with either ErbB2 CNA or BRAF mutation, and cases with neither ErbB2 CNA nor BRAF mutation (DFS: P < .01; Fig. 11). The DFS in the cases with both ErbB2 CNA and BRAF mutation was significantly shorter than those observed in the cases with either ErbB2 CNA or BRAF mutation (P < .01) and those with neither ErbB2 CNA nor BRAF mutation (P < .01). On the other hand, no significant difference was observed in the OS among the 3 groups (Fig. 12). These results were similar to those analyzed only in dogs with UC that underwent total cystectomy (Supplemental Table S4 and Supplemental Figs. S1–S6).
Association of ErbB2 Copy Number Aberration (CNA) and Clinical Characteristics in Dogs With Urothelial Carcinoma.
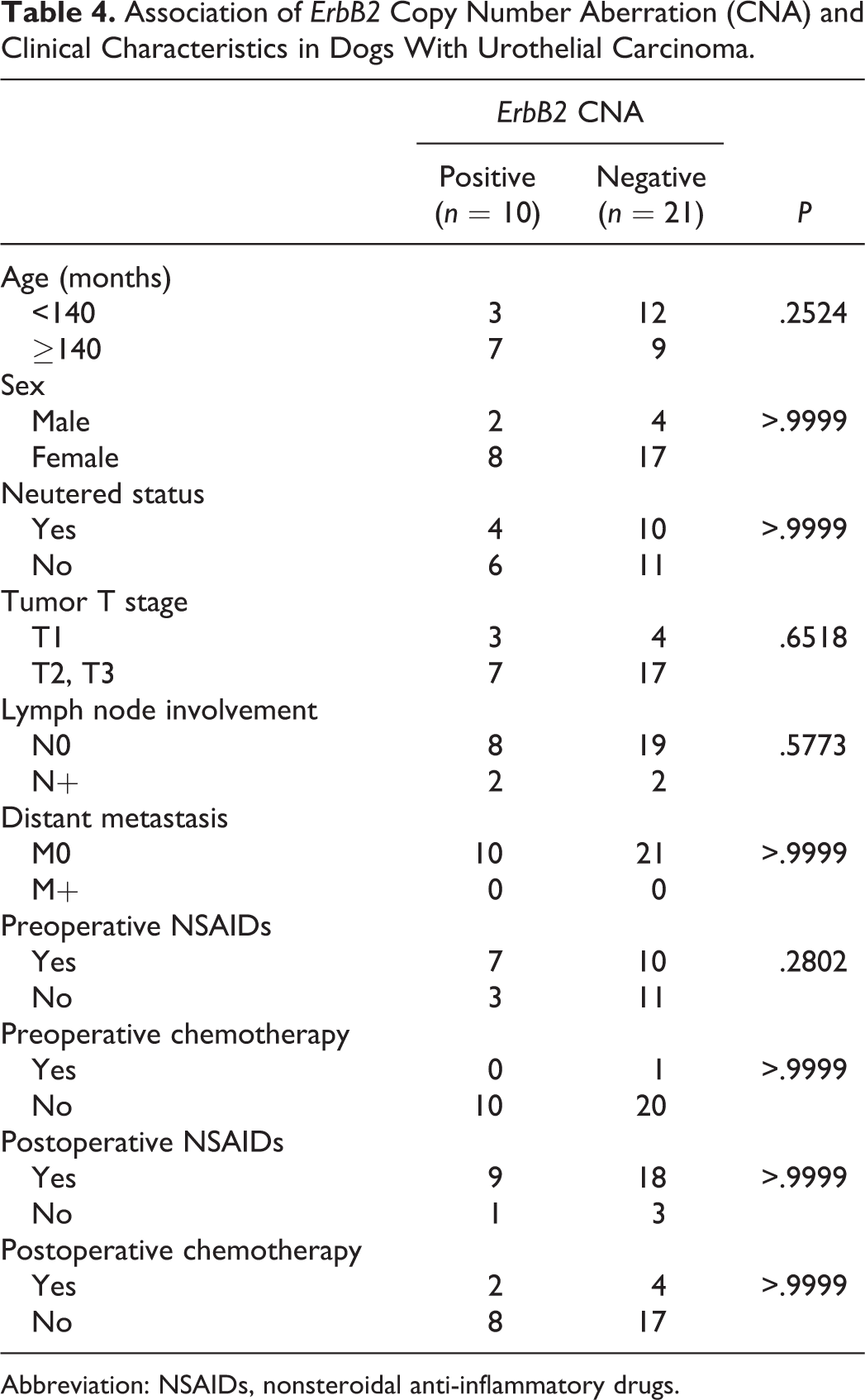
Abbreviation: NSAIDs, nonsteroidal anti-inflammatory drugs.
Discussion
In this study, we demonstrated that the tissue ErbB2:CFA8 ratios were significantly higher in UC than in the normal controls. Based on our established threshold, tissue ErbB2 CNA was detected in one-third of UC, suggesting that gene amplification and/or polysomy occur in dogs with UC. However, the prevalence of ErbB2 CNA was lower than that of ErbB2 overexpression (64%) in the same dogs with UC. ErbB2 CNA was detected in 21% of cases with ErbB2 overexpression. There are 2 explanations for this discrepancy. First, contamination with normal tissues might interfere with detection of ErbB2 CNA in dogs with UC. Second, ErbB2 overexpression in dogs with UC might be due to not only CNAs but also other mechanisms such as transcriptional or posttranscriptional mechanisms. In fact, it is known that ErbB2 overexpression is caused by increased transcription through high levels of transcriptional activators, such as action protein 2 and Yin Yang 1 in human breast cancer. 1 In human patients with ErbB2-overexpressing bladder cancer, ErbB2 CNA is detected at various rates (0%–92%), 4,6,15,20,31,32,40,41,50 suggesting that other mechanisms are involved in ErbB2 overexpression. Further studies are needed to investigate the detailed mechanisms of ErbB2 overexpression in both human bladder cancer and canine UC.
BRAF is a member of the RAF family (A-RAF, B-RAF, and C-RAF). This molecule is a component of mitogen-activated protein kinase/extracellular signal-regulated kinase (MAPK/ERK) pathway, which is included in downstream signaling of ErbB2. 36 A previous study has reported that mutated BRAF in canine UC activates the MAPK/ERK pathway more strongly compared to wild-type BRAF. 7 In this study, ErbB2 overexpression was significantly associated with BRAF mutation but not ErbB2 CNA in dogs with UC. BRAF mutation may be involved in the mechanism of ErbB2 overexpression in dogs with UC.
ErbB2 CNA is observed in 9% to 92% of human patients with bladder cancer and is significantly associated with disease aggressiveness and poor outcomes. 21,32,41,43 As in humans, ErbB2 CNA was also observed in dogs with UC. Although we failed to identify a significant correlation between ErbB2 CNA and prognosis in the dogs with UC, the BRAF mutation-positive cases with ErbB2 CNA had significantly shorter DFS and OS than the other cases. Considering the small sample size in this study, larger and ideally prospective studies are needed to further evaluate associations between ErbB2 CNA and prognosis in dogs with UC.
In the dPCR assay with urinary bladder tissues, ErbB2 CNA was detected in 2 polypoid cystitis cases. In humans, bladder polyps represent a preneoplastic condition, with urothelial dysplasia observed in 6% of patients. 11,39 In dogs, it is unclear whether polyps are capable of becoming malignant. However, a previous study has shown that bladder polyps in a dog developed into neoplastic lesions. 24 Thus, the ErbB2 CNA detected in polypoid cystitis might be associated with a preneoplastic condition. On the other hand, ErbB2 CNA was not detected in the urinary sediments obtained from the dogs with polypoid cystitis. Although the reason was unclear, we estimated that this might be because detachment of epithelial cells into urine is more likely to occur in UC than in polypoid cystitis. dPCR using urinary sediments may be more specific for the diagnosis of canine UC than using urinary bladder tissues.
Recent studies have identified high prevalence (67%–87%) of BRAF mutations in DNA samples isolated from tumor tissues of dogs with UC. 7,27,28 Moreover, BRAF mutations were detected in 83% of DNA samples obtained from urinary sediments of dogs with UC. 28 In this study, detection rates of BRAF mutations in the tissue and urine DNA samples of UC were 61% and 70%, respectively. The frequencies of BRAF mutations were lower than those reported in the previous studies. This variation might be due to small sample size in both the present and previous studies. Interestingly, ErbB2 CNA was detected in 29% of the tissue and 31% of the urine DNA samples in UC without BRAF mutation. By combining both the detection rates of ErbB2 CNA and BRAF mutation, the sensitivities for detecting UC were increased to 72% and 80% for the tissue and urine DNA samples, respectively. Therefore, detection of ErbB2 CNA and BRAF mutation in urinary sediments using dPCR assay might be useful for diagnosing canine UC before surgery.
In human breast cancer, CNA and protein overexpression of ErbB2 are observed in 10% to 25% of patients. 30 Treatment with trastuzumab, anti-ErbB2 humanized monoclonal antibody, has significantly improved the DFS (33%–52%) and OS (34%–41%) of human patients with ErbB2-overexpressing breast cancer. 34,44 Moreover, treatment with lapatinib, a small-molecule tyrosine kinase inhibitor of ErbB2, has shown to prolong progression-free survival in human patients with breast cancer that progressed on prior trastuzumab monotherapy. 2,3 As in humans, ErbB2 overexpression is also observed in 56% to 61% of dogs with UC. 26,46 Moreover, lapatinib has shown antitumor effect on canine UC cell lines and the cell line-engrafted mice, 38 suggesting that lapatinib has therapeutic potential for dogs with UC. The present dPCR assay for detecting ErbB2 CNA might be useful as a predictor for response to anti-ErbB2 therapy in dogs with UC. However, there was no evidence of ErbB2 protein overexpression in half of the UC cases with ErbB2 CNA. This finding suggests that just because there is detection of ErbB2 CNA, it does not mean this will translate to protein overexpression. This should be considered when interpreting ErbB2 CNA detected in dogs with UC.
There were some limitations to this study. First, fluorescence in situ hybridization analysis for detecting ErbB2 CNA was not performed in tumor tissue samples of the dogs with UC. However, previous studies have shown that dPCR assay had a high concordance with result of fluorescence in situ hybridization analysis in tissue samples of human breast cancer. 14,52 Second, histopathological evaluation for definitive diagnosis was not performed in 32 of 54 UC used by urinary ErbB2 testing. BRAF mutation was not observed in 8 of 32 UC without histopathological evaluation. However, tumor progression (invasion into bladder wall, lymph node involvement, or distant metastasis) was observed in all the cases later. Third, a previous study, using dPCR assay, has reported that gain of CFA8 was detected in 1 of 14 dogs with UC. 29 However, the CFA8 CNA was not confirmed by in situ hybridization. Further study will be needed to identify the stability of CFA8 in dogs with UC. Fourth, diagnosis of recurrence was based on ultrasonography and/or urine cytology. It may have been useful to obtain data on histopathology and BRAF mutation in the dogs with UC. Unfortunately, we could not obtain these data because this is a retrospective study. Finally, treatment after surgery in UC cases was not uniform in this study. Postoperative chemotherapy may have influenced the DFS and OS in each case. Further prospective studies with uniform treatment after surgery would be needed.
In conclusion, we demonstrated ErbB2 CNA in urinary bladder tissues and urinary sediments of dogs with UC using dPCR assay. Our findings provide insight into the molecular pathogenesis, diagnostics, prognostics, and therapeutic potential for dogs with UC.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Sakai_et_al - ErbB2 Copy Number Aberration in Canine Urothelial Carcinoma Detected by a Digital Polymerase Chain Reaction Assay
Supplemental Material, Combined_supplemental_materials-Sakai_et_al for ErbB2 Copy Number Aberration in Canine Urothelial Carcinoma Detected by a Digital Polymerase Chain Reaction Assay by Kosei Sakai, Shingo Maeda, Kohei Saeki, Ryohei Yoshitake, Yuko Goto-Koshino, Takayuki Nakagawa, Ryohei Nishimura, Tomohiro Yonezawa and Naoaki Matsuki in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the grants-in-aid for fellows of the Japan Society for the Promotion of Science (JSPS), Scientific Research (KAKENHI Grant Number 16H06208), and the Anicom Capital Research Grant (EVOLVE).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
