Abstract
Myogenin and MyoD regulate the development of skeletal muscle, and their expressions are specific to the stages of myogenesis. Therefore, these myogenic regulatory proteins could be considered as sensitive and specific markers for rhabdomyosarcoma. In this report we investigated the immunohistochemical reactivities of myogenin and MyoD in two canine bladder botryoid rhabdomyosarcomas that were different in the degree of differentiation. MyoD was stained in the Ki-67 antigen-positive undifferentiated mesenchymal cells, which had proliferative activity similar to myoblasts differentiated from mesoblasts. In contrast, multinucleated neoplastic cells were positive for myogenin and α-sarcomeric actin but not for Ki-67 antigen, similar to the myotubes differentiated from myoblastic cells. The expressions of myogenin and MyoD were closely correlated to the histologic features of myogenic neoplastic cells.
Myogenin and MyoD are myogenic transcriptional proteins expressed early in skeletal muscle development, and their expressions are specific to the stages of myogenesis. These myogenic regulatory proteins are considered as sensitive and specific markers for human rhabdomyosarcoma. 1,3,5 Canine botryoid rhabdomyosarcoma arises commonly in the urinary bladder of juvenile large-breed dogs, growing as grapelike polypoid masses. Histologically, the tumor is composed of neoplastic cells that mimic myogenic cells in embryonal myogenesis. 2 Therefore, it is interesting to investigate the relation between morphologic features and the expression of myogenic regulating factors. In this report we studied the expression of myogenic regulation factors myogenin and MyoD in botryoid rhabdomyosarcomas in the bladders of two dogs.
Dog No. 1, an 18-month-old female Golden Retriever, presented with hematuria. Cystography revealed a large mass in the area of the trigone. During surgery, a large (6.5 × 6 × 3 cm) reddish mass with severe necrosis was observed in the trigone. Partial resection of the bladder including the tumor was performed. However, the dog died 1 month after the surgical resection. A necropsy was not performed.
Dog No. 2 was a 12-month-old female Golden Retriever presenting with dysuria. There was a solid mass in the neck of the bladder, identified by palpation and cystography. During surgery the area between the bladder neck and the uterus was replaced by a firm white mass. The mass was not resectable, and the dog was euthanatized. A necropsy was performed immediately after euthanasia. Grossly, a dark reddish irregular mass was found throughout the bladder wall and extending into the uterus. Additional masses were observed in liver, lung, heart, kidney, ovary, and skin.
The tissue samples were fixed in 10% neutral buffered formalin and embedded in paraffin. Sections were cut to 3-µm thickness and stained with hematoxylin and eosin (HE). Selected sections were also stained with phosphotungstic acid–hematoxylin stain (PTAH) for detection of cross striations. Immunohistochemistry was performed by the avidin–biotin–peroxidase technique (Dako Cytomation, Glostrup, Denmark) with the following primary antibodies: mouse monoclonal antibody against human vimentin (clone V9), human desmin (clone D33), and rabbit α-sarcomeric actin (clone Alpha-Sr-1) and a rabbit polyclonal antibody against human myoglobin. In addition, two monoclonal antibodies against myogenic regulating factors, anti-rat myogenin (clone F5D) and anti-human MyoD1 (clone 5.8), were applied. For detection of proliferative cells, monoclonal antibody against Ki-67 (clone MIB-1) was used. All antibodies were provided by Dako Cytomation. All primary antibodies with the exception of myoglobin were applied to sections after heat-induced antigen-retrieval treatment with Target Retrieval Solution (Dako Cyomation; 121 C, 15 minutes). For detection of coexpression of myogenin and α-sarcomeric actin, selected sections were double-labeled for myogenin and α-sarcomeric actin (myogenin: peroxidase-diaminobenzidine, α-sarcomeric actin: alkaline phosphatase [Blue Alkaline Phosphatase Substrate Kit III]; Vector Laboratories, Burlingame, CA). After staining with myogenin, sections were incubated in 0.1 M glycine-HCl buffer (pH 2.2) for 2 hours at room temperature to eliminate antibodies.
In dog No. 1 the proliferating neoplasm elevated the mucosa and infiltrated into the smooth muscle layer of the urinary bladder. The neoplasm consisted of undifferentiated mesenchymal cells that were round, fusiform, or elongate with scant or moderate cytoplasm. The submucosa was edematous. Multinucleated neoplastic cells that had abundant eosinophilic cytoplasm were interspersed in the edematous area (Fig. 1). These cells had distinct cross striations stained by PTAH, indicating myotube differentiation. Two to three mitotic figures were observed per high-power field (HPF).
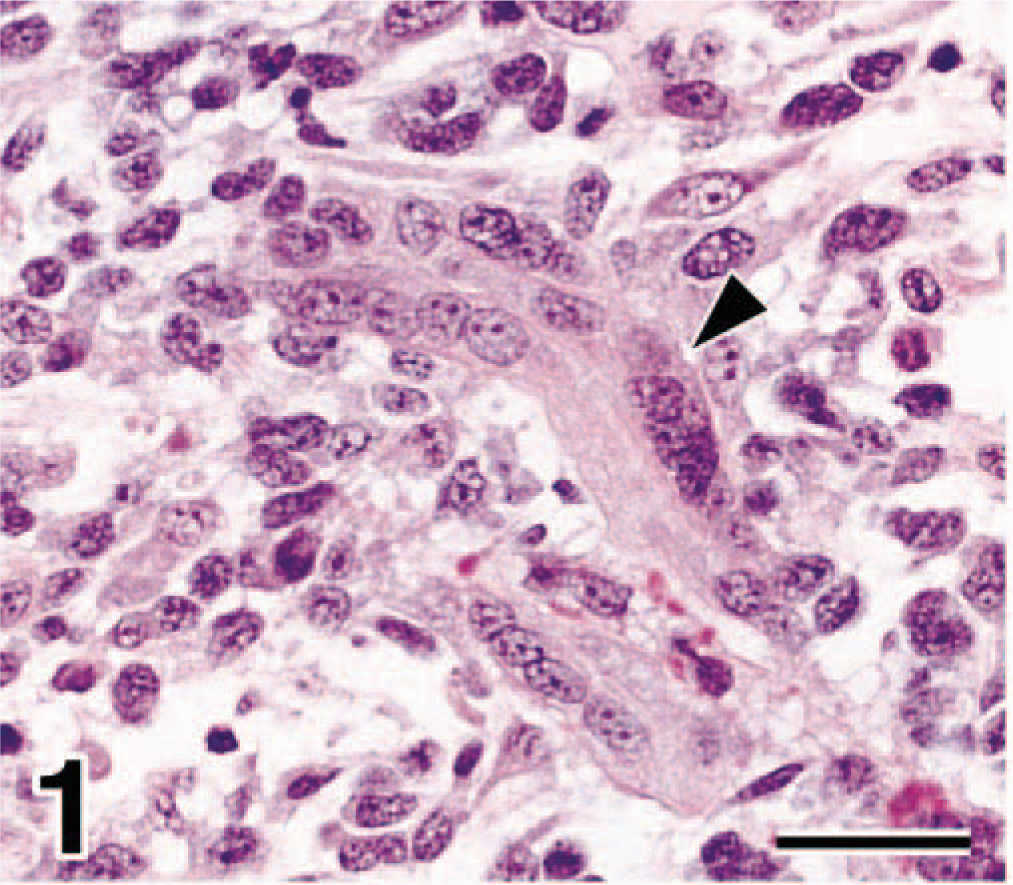
Bladder mass; dog No. 1. In edematous submucosa, multinucleated neoplastic cells (arrowheads) that had abundant eosinophilic cytoplasm were observed. HE. Bar = 29 µm.
In dog No. 2 the neoplasm effaced the submucosa and infiltrated into the smooth muscle layer. There was severe necrosis. The neoplasm consisted of undifferentiated mesenchymal cells similar to those in dog No. 1 (Fig. 2). However, in dog No. 2 myotube-like multinucleated cells were not present. Two to three mitotic figures were observed per HPF. Metastatic lesions consisted of undifferentiated mesenchymal cells with scant or moderate eosinophilic cytoplasm, similar to those in the primary lesion. In any metastatic lesion myotube-like multinucleated cells were not observed.
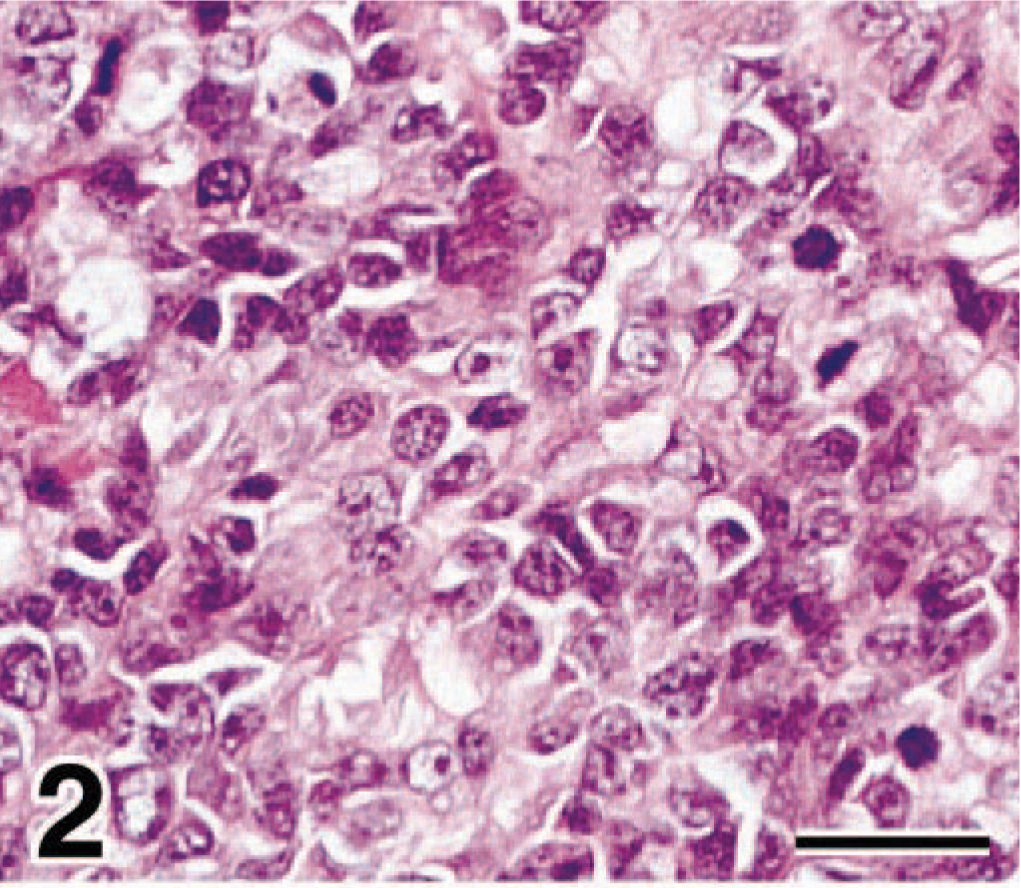
Bladder mass; dog No. 2. The neoplasm consisted of undifferentiated mesenchymal cells that were round or elongate with scanty or moderate cytoplasm. Mitotic figures were often observed (arrowhead). HE. Bar = 29 µm.
Regardless of the morphology of the neoplastic cells, the cytoplasm of almost all neoplastic cells was stained strongly positive for vimentin and desmin in both cases. However, the neoplastic cells had moderate to abundant cytoplasm stained positive for α-sarcomeric actin. In particular, myotube-like multinucleated cells in dog No. 1 were strongly positive. There were only a few positive cells in dog No. 2. Myotube-like cells in dog No. 1 were positive for antimyoglobin; in contrast, in dog No. 2 myoglobin-positive cells were not observed in all the tumors in the primary and metastatic lesions. In both dogs the nucleus of most neoplastic cells was positive for myogenin and MyoD; however, the expression was closely related to the degree of differentiation. In dog No. 1 myogenin was expressed intensely in the nuclei of differentiated cells that had abundant eosinophilic cytoplasm and myotube-like morphology. In contrast, MyoD was expressed in the nuclei of undifferentiated mesenchymal cells (Fig. 3A) but weakly or not at all in differentiated cells. In particular, the myotube-like cells were negative for MyoD (Fig. 3B). Thus, in dog No. 2 MyoD-positive cells dominated. Ki-67–positive reaction was shown mainly in undifferentiated mesenchymal cells (Fig. 3C) but not in differentiated myoblastic cells including multinucleated myotube-like cells (Fig. 3D). In double-immunostaining for myogenin and α-sarcomeric actin, myogenin-positive cells also expressed α-sarcomeric actin in their cytoplasm (Fig. 3E).
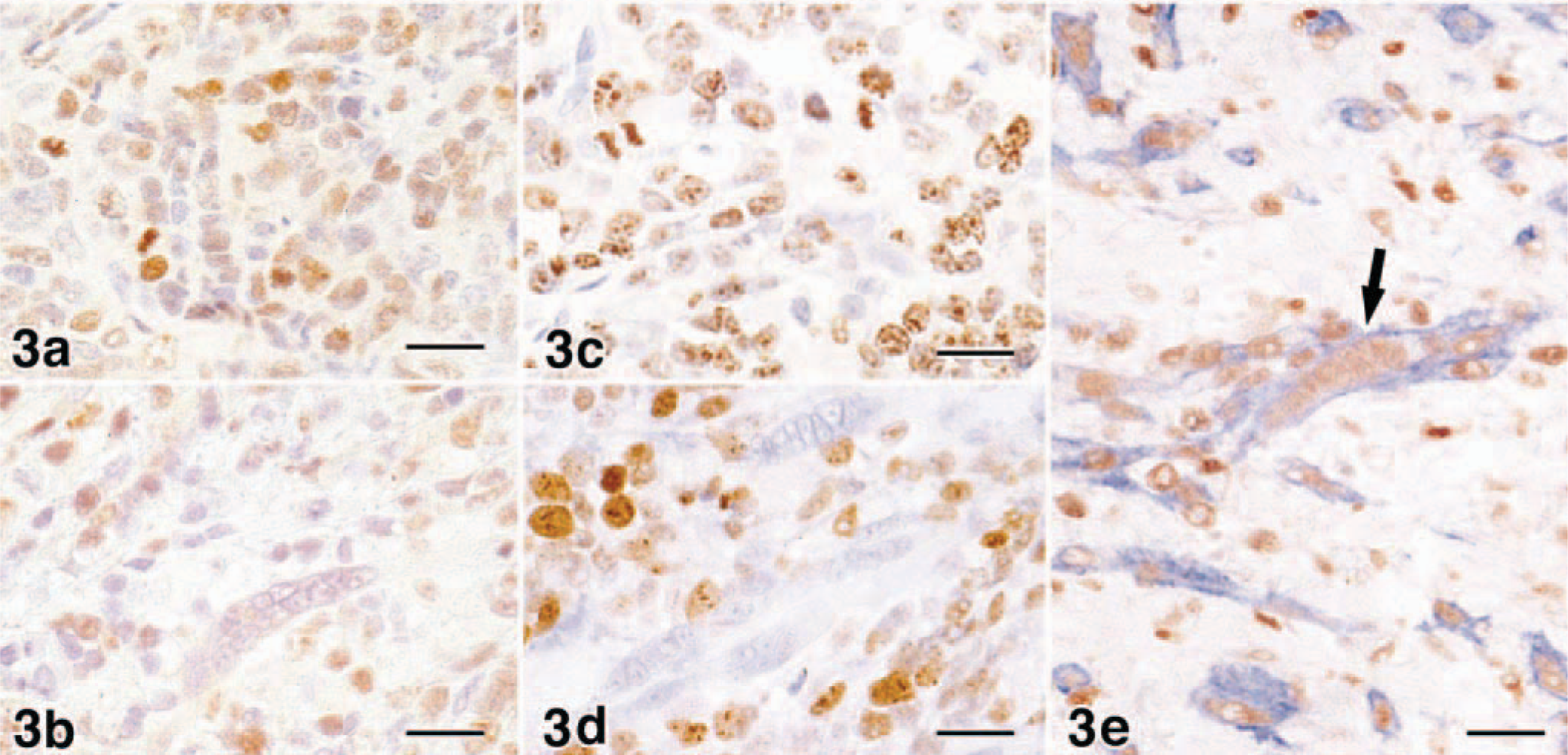
The expressions of myogenin and MyoD were closely related to the differentiation of neoplastic cells in these two cases. During myogenesis MyoD plays an essential role in differentiation to myoblast, which has proliferative activity 6 Subsequently, myogenin induces differentiation of the myoblast into the multinucleated myotube, which has abundant cytoplasm including myofibrils and is incapable of mitosis. 6,7 In both dogs, and most prominently in dog No. 2, there was strong expression of MyoD in the undifferentiated mesenchymal cells, which were negative for α-sarcomeric actin and myoglobin. Additionally, these cells expressed Ki-67 antigen, which indicates proliferation. These characteristics were similar to those of the myoblast derived from the mesoblast. In contrast, in dog No. 1 myotube-like cells that had distinct cross striations and myoglobin were strongly positive for myogenin but negative for Ki-67 antigen. These cells were also positive for α-sarcomeric actin, similar to skeletal muscle–specialized cells. Therefore, these cells were regulated by myogenin and differentiated from the myoblast to the myotube. These results suggested that in dog No. 1 some neoplastic cells differentiated to the stage of myotube formation regulated by myogenin; on the other hand, in dog No. 2 the tumor consisted of undifferentiated mesenchymal cells only, with no progression to the stage of myotube formation.
Myogenin and MyoD were found to be useful markers for canine rhabdomyosarcoma. Especially, MyoD is a positive marker for the rhabdomyosarcoma, distinguishing it from poorly differentiated myogenic neoplasms. However, it should be noted that the lack of immunostaining with MyoD and myogenin does not rule out myogenic origin for a neoplasm because expression is closely associated with degree of differentiation. Pleomorphic rhabdomyosarcoma, a phenotype more similar to adult than to embryonal skeletal muscle, may express lower or no MyoD and myogenin. 4 In addition, myogenin expression is also seen in nonneoplastic atrophy or regenerating skeletal muscle not infiltrated by tumor and in muscle adjacent to a region of tumor. 1
In conclusion, the expressions of myogenin and MyoD were closely correlated to the histologic features of myogenic neoplastic cells. And they may be useful markers considering the myogenic origin and differentiation of canine rhabdomyosarcoma. However, additional studies to determine the specificity of these two markers are needed.
