Abstract
Hibernomas are uncommon benign tumors of brown fat that occur in humans and various animal species. They have not been observed in the orbit of dogs, humans, or other animals. Here we report clinical, light and electron microscopic, and immunohistochemical features of a series of 7 hibernomas arising in the orbital region of dogs. These neoplasms occurred in adult dogs with no breed predilection. The mean age of the affected dogs was 10.4 years (range, 8–13 years). All neoplasms presented as soft lobular masses composed of predominantly round or polygonal neoplastic cells with granular eosinophilic and vacuolated cytoplasm resembling adipocytes. The cytoplasm contained large numbers of pleomorphic mitochondria with dense matrices and indistinct cristae. Immunohistochemical evaluation confirmed positive labeling of neoplastic cells from all cases with uncoupling protein 1 (UCP-1) consistent with brown fat differentiation. Interestingly, rare neoplastic cells also expressed myogenin and myoD, possibly suggesting a common progenitor cell for neoplastic brown adipose and skeletal muscle cells.
Keywords
Humans, neonates, and hibernating animals have 2 types of adipose tissues: white adipose tissue (WAT) and brown adipose tissue (BAT). 3,7 They have opposite physiological functions: WAT stores energy in the form of fat; conversely, BAT burns energy. 3,7,15 BAT is present primarily in the thoracic and abdominal cavities and in interscapular subcutaneous tissues in neonates, rodents, and humans. 3,7 BAT is of particular importance in rodents, neonates, small mammals in cold environments, and hibernating animals because of its ability to produce nonshivering thermogenesis during periods of cold-induced stress. 3,7,14,15 Hibernoma is a rare benign tumor of brown adipose tissue. It was first described in humans by Merkel in 1906 as a “pseudolipoma.” 17 Hibernomas have been reported in laboratory rodents 2 and in humans. 8,9,12,16 The distinctive microscopic features of hibernomas include (1) lobules of closely apposed large polygonal cells and capillaries, (2) 3 principal cell types (granular eosinophilic, multivacuolated, and univacuolated), (3) investment of each tumor cell by the basal lamina, (4) an inverse relationship between lipid droplet size and the number of mitochondria per unit of cytoplasm, and (5) pleomorphic mitochondria with dense matrices or large, round mitochondria with transverse lamellar cristae. 9 The diagnosis of hibernoma may be challenging, particularly differentiation from atypical lipomas and poorly differentiated liposarcomas. Immunohistochemistry using uncoupling protein 1 (UCP1) has been used to confirm the diagnosis of hibernomas in humans and rodents. 2,16,21,26,33,35 To our knowledge, there is a single report of an omental hibernoma in a dog and no reports of orbital hibernomas in either humans or animals. 22 Here, we report the clinical, microscopic, ultrastructural, and immunohistochemical features of a series of orbital hibernomas in 7 dogs.
Materials and Methods
The study included orbital neoplasms from 7 dogs that presented as solitary soft-tissue neoplasms. These specimens had been submitted to the Comparative Ocular Pathology Laboratory of Wisconsin (COPLOW) as routine diagnostic samples between 1994 and 2010. All tissues had been fixed in 10% neutral buffered formalin; representative tissue samples were routinely processed, embedded in paraffin, sectioned, and stained with hematoxylin and eosin (HE).
Transmission electron microscopy (TEM) was performed on a tissue available from 1 case (dog No. 4). For TEM, sections of formalin-fixed tissue were immersed in a solution of 2.5% glutaraldehyde and 2.0% paraformaldehyde in 0.1 M sodium phosphate buffer, pH 7.4, overnight at 4°C. Tissue samples were then postfixed in 1% osmium tetroxide in the same buffer for 2 hours at room temperature followed by dehydration in a graded ethanol series. After further dehydration in propylene oxide, samples were embedded in Epon or Durcupan epoxy resin. Ultra-thin sectioning was as follows: first, semi-thin (1-μm) sections were cut with a Leica EM UC6 ultra microtome (Leica Microsystems GmbH, Wetzlar, Germany) and observed under a light microscope to establish proper orientation. Ultra-thin sections of the selected areas were cut with the same microtome and collected on 200 mesh copper grids or pioloform (cat. 19244; Ted Pella, Redding, CA)–coated 1-hole slot grids. Sections were stained with Reynolds lead citrate and 8% uranyl acetate in 50% EtOH and observed with a Philips CM120 electron microscope (Hillsboro, Oregon). Images were captured with a MegaView III side mounted digital camera (Mega view centre valley, PA).
Immunohistochemistry for UCP1 was performed on sections of tissue from all 7 tumors. Deparaffinization, antigen retrieval, and immunostaining of formalin-fixed, paraffin-embedded tissues were performed on the Bond MAX Automated Staining System (Leica Microsystems GmbH) using the Bond Polymer Detection System (Leica Microsystems) and a rabbit polyclonal antibody against UCP1 (Acris Antibodies, San Diego, CA) at a dilution of 1:20. Antigen retrieval was achieved using the Bond Epitope Retrieval Solution, pH 8 (Leica Microsystems GmbH), for 20 min. The immunoreaction was visualized with 3,3′-diaminobenzidine (DAB) substrate (Leica Microsystems GmbH), and sections were counterstained with hematoxylin. Immunohistochemistry for MyoD1 and myogenin was performed on sections of all 7 tumors using the Autostainer Link 48 (Dako North America, Carpinteria, CA) automated stainer with 2 mouse monoclonal antibodies against MyoD1 and myogenin (Dako North America) at dilutions of 1:100 and 1:50, respectively. Deparaffinization and antigen retrieval of formalin-fixed, paraffin-embedded tissues were performed on the PT Link (Dako North America) using the Dako Target Retrieval Solution, citrate pH 6 (Dako North America). Detection of the target was achieved using the EnVision Flex+ detection system (Dako North America), and the immunoreaction was visualized with DAB substrate (Dako North America) counterstained with hematoxylin.
Positive immunohistochemical controls were canine fetal skeletal muscle and rodent brown fat to which appropriate antisera were added. For negative controls, the primary antibodies were replaced with homologous nonimmune sera.
Results
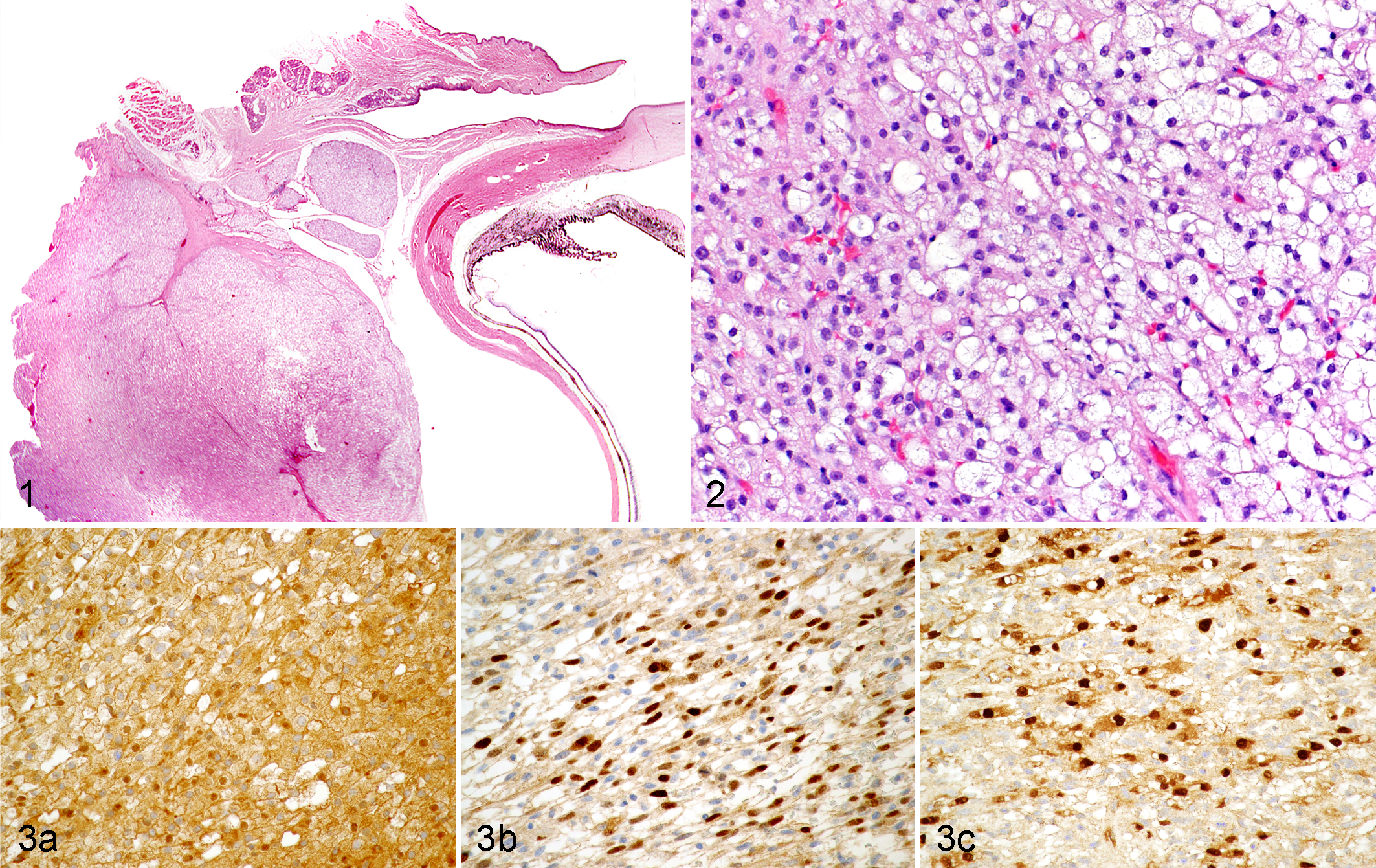
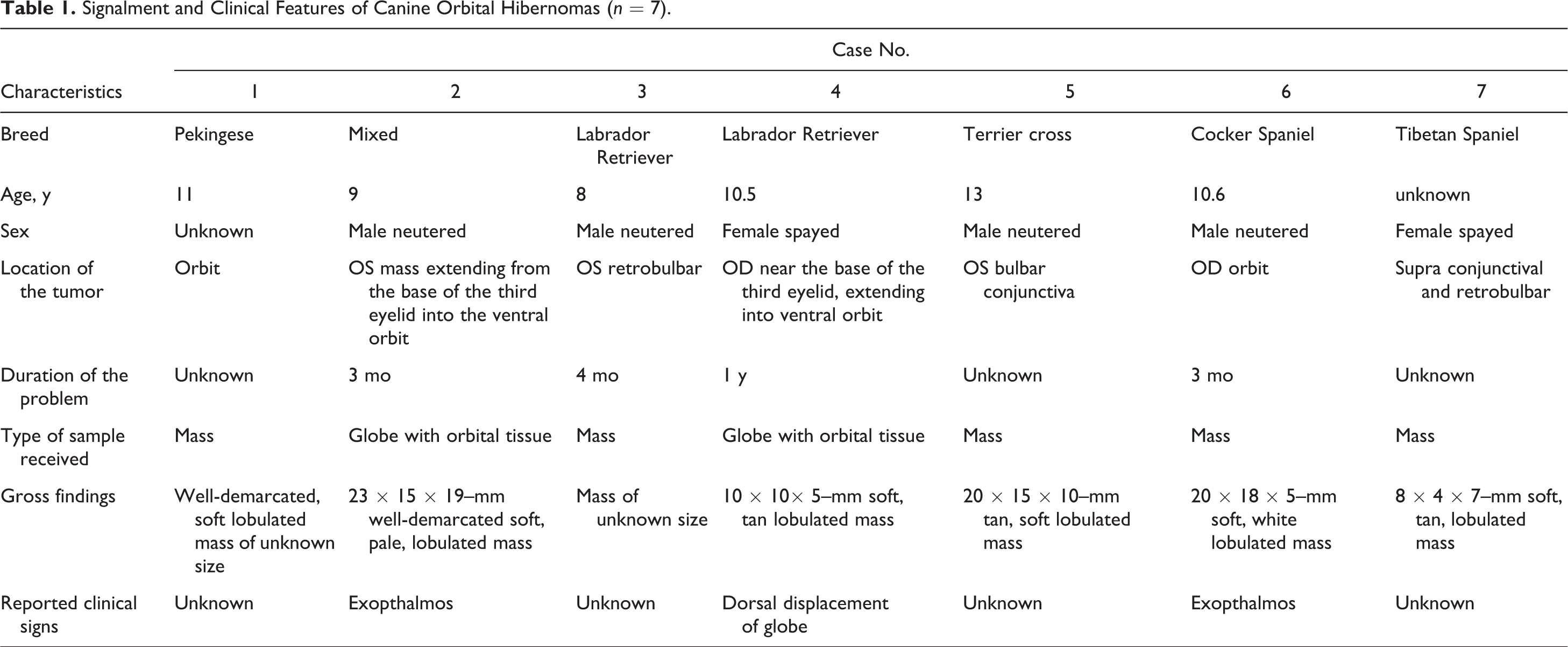
The signalment of the 7 dogs was obtained from the submission forms (Table 1). There was no breed predilection. The neoplasms were diagnosed as benign hibernomas based on low mitotic activity, minimal nuclear atypia, and lack of invasion. All neoplasms were completely or partially encapsulated and composed of lobules separated by thin or thick fibrovascular septae (Fig. 1). The neoplastic cells were large, round to polygonal and closely opposed to each other and to ramified small capillaries. The predominant neoplastic cells were round or polygonal with moderate amounts of granular, pale to deeply eosinophilic cytoplasm with or without vacuoles admixed with larger, multivacuolated or univacuolated neoplastic cells resembling adipocytes. The vacuoles were consistent with, but not proven to be, lipid (Fig. 2). Nuclei were round to oval and central to eccentrically placed with finely stippled chromatin and 1 to 3 nucleoli. There were mild anisocytosis and anisokaryosis, and the mitotic index varied from 0 to 3 per ten 400× fields. There was no evidence of vascular invasion.
Signalment and Clinical Features of Canine Orbital Hibernomas (n = 7).
In all cases, the neoplastic cells were diffusely and strongly positive for UCP1, similar to positive controls (Fig. 3a). In 1 case (dog No. 2), 40% to 60% of the neoplastic cells had nuclear labeling with MyoD1 (Fig. 3b) and myogenin (Fig. 3c), and the other neoplasms contained rare, individual, positive cells.
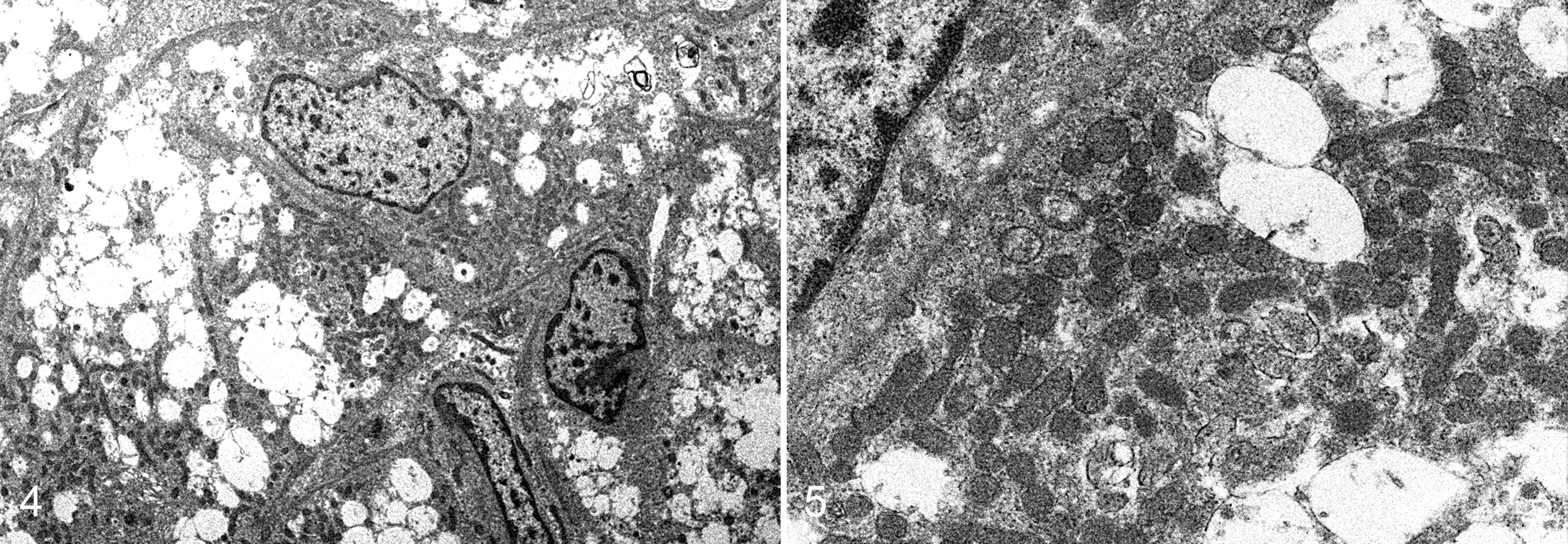
The orbital tumor from 1 animal (dog No. 4) was examined ultrastructurally. The neoplastic cells were separated by fairly distinct basal laminae, and the predominant cytoplasmic constituents were large numbers of mitochondria and lipid-like droplets (Fig. 4). The mitochondria in the eosinophilic granular cells were prominent, large round or rod shaped with electron-dense matrices and fairly distinct parallel cristae (Fig. 5). They were also present in low numbers between lipid droplets in multivacuolated cells. The nuclei of corresponding granular cells were paracentrally located and euchromatic, and they contained prominent nucleoli. Lipid vacuoles in the multivacuolated cells displaced nuclei eccentrically. There was variation in the size of lipid droplets in multivacuolated cells, and their numbers were inversely proportional to the number of mitochondria. Other cytoplasmic constituents included glycogen and lipofuscin granules.
Discussion
The name hibernoma is derived from its histomorphological similarity to brown adipose tissue commonly seen in hibernating animals and rodents. These neoplasms are very rare in companion animals, and to our knowledge, only a single case of a hibernoma in the omentum of a 9-month-old dog has been reported. 22 There are no reports of orbital hibernomas in either animals or humans. The orbital hibernomas presented here consistently exhibited histologic, ultrastructural, and immunohistochemical features similar to those of hibernomas reported in a dog, rodents, and humans. 2,8,9,16,22,24,25
Potential differentials for the orbital neoplasms reported here include lipomatous neoplasms, rhabdomyomas, granular cell tumors, oncocytomas, and xanthogranulomas. Lipomas differ from hibernomas as they are less vascularized and resemble mature white adipose tissue with neoplastic cells having characteristic, large, cytoplasmic, lipid vacuoles that eccentrically displace the nucleus. 28 Liposarcomas are more cellular than their benign counterparts but similarly composed of adipocytes with cellular atypia and high mitotic activity. 6 Rhabdomyomas are benign skeletal muscle neoplasms that are characterized by glycogen-rich, eosinophilic, striated muscle cells with numerous mitochondria, myofilaments, and electron-dense Z-lines. 18 Granular cell tumors are distinguished by cytoplasmic periodic acid–Schiff (PAS)–positive granules that correspond to heterogeneous lysosomes of variable size. 23,27 In oncocytomas, the abundant eosinophilic cytoplasmic granules correspond to large numbers of mitochondria. 29 Cytoplasmic vacuolation is not a feature of granular cell tumors and oncocytomas. Finally, xanthogranulomas are composed of lipid-laden foam cell macrophages admixed with birefringent, Alcian blue–positive crystals in stellate patterns. 34
Besides characteristic microscopic features, the orbital hibernomas presented here had large numbers of mitochondria in the neoplastic brown adipocytes. Large numbers of mitochondria are a consistent feature of brown adipocytes and enhance their ability to efficiently metabolize fatty acids into energy. 11,20 We are unable to do Oil Red O staining for lipids on paraffin-embedded tissues as the lipid is removed during processing. Immunohistochemistry for UCP1 has been used to differentiate BAT from WAT. 33 Brown adipocytes uniquely express inner mitochondrial UCP1 in contrast to other cells, including white adipocytes, which gives the cells’ large number of mitochondria an ability to uncouple oxidative phosphorylation and use substrates efficiently to generate heat rather than adenosine triphosphate. 7,11,20 In the present study, neoplastic cells in all the orbital tumors expressed UCP1, consistent with brown adipose tissue. Various studies found that white and brown adipocytes originate from distinct cell lineages. 5,30 Brown adipocytes not only express UCP1 but may also express myogenic factors such as myogenin, MyoD, and Myf5, as observed in this study, suggesting a common origin for brown adipocytes and myocytes. 32 This may explain why brown adipocytes ultimately specialize in lipid catabolism rather than fat storage, much like oxidative skeletal muscle.
In the present study, all dogs were mature adults. Similar to our study, hibernomas in humans tend to occur in adults (mean age, 38 years; range, 2–75 years) with an increased prevalence in males. 8 A recent study described spontaneous hibernomas in aging Sprague-Dawley rats with a higher prevalence in males and the majority originating in the thoracic cavity. 2
The cause for the unusual location in the orbital region of the hibernomas described here is unknown. Speculation exists about the mechanism(s) of development of hibernomas. A recent study demonstrated numerous, poorly differentiated cells resembling brown adipocyte precursors in a pericytic position around capillary walls, and the report suggested that brown adipocyte precursors arise in close association with vessel walls. 16 It was also postulated that prolonged exposure to cold, chronic hypoxemia, chronic sympathoadrenal stimulation, starvation, and chronic wasting diseases may cause hypertrophy and/or hyperplasia of BAT. 10,19 Cold exposure stimulates BAT growth by increasing the release of norepinephrine from sympathetic nerves, with this neurohormone activating mitoses in BAT precursor cells via β-adrenergic pathways. 10,13,19,31 The absence of histologically distinct brown fat in adult dogs does not mean that there are no specific brown fat stem cells in this species. Adrenergic stimulation of white fat taken from 5 locations in adult dogs has resulted in reactivation of WAT UCP expression characteristic of BAT. It is possible that tumors could arise from activated “dormant” BAT that has the morphological appearance of WAT but represents preadipocyte brown fat cells. 4 Norepinephrine (NE) agonists increase sympathetic stimulation and NE levels, which lead to elevated β-oxidation, proliferation, and increased UCP-1 expression with concurrent inhibition of apoptosis in BAT, which may ultimately lead to the development of hibernomas. 1
The present study has demonstrated the existence of orbital hibernomas in dogs. Hibernomas are rare benign neoplasms of brown adipose tissue, and treatment by complete excision should be curative. Hibernoma should be considered a differential in evaluating lipomatous neoplasms. Although microscopic features are helpful for the diagnosis of hibernoma, UCP1 expression distinguishes BAT tumors from other lipomatous neoplasms.
Footnotes
Acknowledgements
The authors would like acknowledge Dr. Gholam-Reza Hafez for helping with histological interpretation. Special thanks to Sandy Cheasty, Barb Reese, Stephanie Mayer and Kate Lieber for assistance with specimen processing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
