Abstract
Apparently synchronous, aggressive, mixed mesenchymal tumors in the right tibia, right femur, left femur, and rib cage produced multiple microscopic metastases in the lungs and macroscopic metastases in the liver, kidney, and spleen in a 1.5-year-old, neutered male, mixed-breed dog. No primary soft tissue tumor mass was present. Microscopically, the neoplasm exhibited osteosarcomatous, chondrosarcomatous, liposarcomatous, leiomyosarcomatous, fibrosarcomatous, angiosarcomatous, and leukocytic differentiation and was diagnosed as a multipotential osteosarcoma with various mesenchymal differentiation. Immunohistochemically, the neoplasm was cytoplasmically immunoreactive for vimentin, osteonectin, osteocalcin, CD 18, CD 31, desmin, and muscle-specific actin. Oil Red O staining was positive within liposarcomatous areas. Skeletal metastases from a primary bone tumor are exceedingly rare in human and veterinary medicine. However, the history, clinical signs, location, microscopic and immunohistochemical features were similar to those described in aggressive, poorly differentiated osteosarcomas of children. In addition, the wide range of mesenchymal tissue differentiation of this neoplasm was unusual, and to the authors' knowledge, an osteosarcoma with this degree of multiple differentiation has not been previously reported in the dog.
Keywords
Malignant mesenchymal soft tissue tumors and primary sarcomas of the skeleton belong to a large and heterogeneous group of neoplasms. Classification of these neoplasms is typically based on cell type and observed products of proliferating cells. Yet, histomorphologic, immunohistochemical, and experimental data suggest that most, if not all, sarcomas arise from primitive multipotential mesenchymal cells, which may differentiate along one or more lines during the course of neoplastic transformation. 16,17,20 Mesenchymal tumors arise from mesodermal-derived precursors of fibrous tissue, smooth and striated muscle, adipose tissue, nerve sheets, bone, cartilage, endothelium, and hematopoietic tissue. 15,20
Primary bone sarcomas that include osteosarcomas are divided into two categories, central and peripheral, with central tumors being higher in incidence and involvement. 17 Tumor types are typically named for the matrix they produce; however, when tumors contain multiple cell types and matrices, determination of histologic classification may be problematic. According to the World Health Organization classification of bone and joint tumors, an osteosarcoma is a primary malignant neoplasm of mesenchymal tissue that gives rise to a variety of patterns but always includes the production of bone by malignant osteoblasts. 17
Metastatic components may impart a differing histologic appearance in cell type and matrix formation than the primary tumor. 9,17 Secondary or metastatic tumors may affect bone either by direct extension or by hematogenous spread. We present a case of osteosarcoma apparently arising synchronously in multiple long bones in a 1.5-year-old dog. The tumor exhibited multipotential mesenchymal lines of differentiation including osteosarcoma, chondrosarcoma, liposarcoma, leiomyosarcoma, fibrosarcoma, and hemangiosarcoma and produced multiple visceral and possible skeletal metastases.
A 34-kg, 1.5-year-old, male castrated, mixed-breed dog was presented to the Michigan State University Small Animal Hospital on 8 July 2002 with a 4-month history of progressive right hind limb lameness. There was visible swelling of the hock joint of the right rear leg and muscle atrophy of right hip. On palpation, the leg was warm to the touch and painful, and there was a hard swelling of the distal tibia on the medial aspect of the hock joint.
Initial diagnostic testing included radiographic examination of the pelvic limbs and thorax. Multifocal, primarily destructive bone lesions of nearly similar size were present on the right distal femur and the proximal and distal tibia (Fig. 1), and a somewhat smaller destructive lesion was present in the left distal femur. Tumor tissue had effaced the metaphyseal spongiosa, penetrated the cortical compacta, and extended through the periosteal cuff of reactive bone bordering each of the long-bone lesions. Similar aggressive and destructive bone lesions were present in the metaphyses of the right sixth and ninth ribs and left ninth rib. The findings on radiographic examination were suggestive of a metastatic or multicentric bony neoplasm, and at this time, euthanasia was elected, and a postmortem examination was performed.
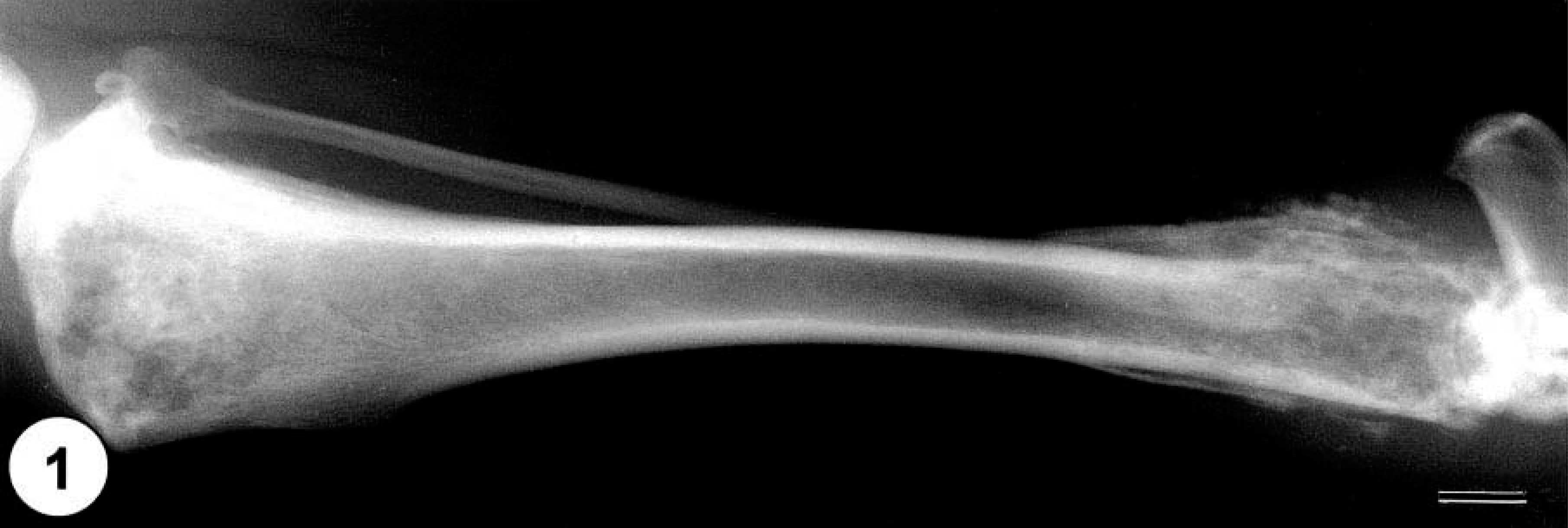
Lateral radiograph of right tibia; multifocal, large, osteolytic lesions present in the proximal and distal tibia. Bar = 2 cm.
Gross examination of affected long bones found that multiple areas of cortical and cancellous bone in the metaphyses and epiphyses of the distal femur and both the proximal and distal tibia had been destroyed and replaced by mottled, red to creamy yellow, partially necrotic tissue containing focal areas of irregular ossification (Fig. 2). Tumor metastases in the costochondral junctions of the right sixth and ninth ribs and left ninth rib destroyed local bone architecture and induced the formation of smoothly contoured periosteal cuffs. Although no gross metastatic lesions were found in the lungs, pulmonary metastases were found microscopically. Multifocal metastatic tumor nodules ranging from 0.5 to 5 cm in greatest dimension were found in the liver, spleen, and both kidneys. No other significant gross lesions were observed.

Right tibia; multiple neoplastic masses within the proximal and distal tibia that extend multifocally from the medulla through cortex and into the epiphyses. Bar = 2 cm.
Tissue samples were fixed in 10% neutral buffered formalin and processed for histopathologic examination. Immunohistochemistry was performed using commercially available antibodies. Antibodies to chromogranin A + B (Progen, Heidelberg, Germany), protein gene product 9.5 (PGP 9.5, R&D system, Minneapolis, MN), and S-100, neuron-specific enolase (NSE), and epithelial membrane antigen (EMA) (all Dako, Carpenteria, CA) were used to evaluate for neuroendocrine differentiation. Pancytokeratin (MNF116 and AE1/AE3) and laminin were used to evaluate for epithelial differentiation. Vimentin immunostain was used to document mesenchymal differentiation, and mesenchymal components were evaluated with CD 18 and CD 45 (courtesy of Peter Moore, University of California Davis) for round cell differentiation, desmin and muscle-specific actin (MSA) for muscle cell differentiation, and factor 8 and CD 31 for angioformative differentiation. Osteonectin and osteocalcin (Biogenex, San Ramon, CA) were used to document the osteosarcomatous population. A labeled streptavidin–biotin–peroxidase complex system was used to visualize all immune reactions.
Histopathologic examination of the bone masses revealed an invasive proliferation of poorly differentiated neoplastic mesenchymal cells forming streams, bundles, and nests, which resulted in destruction of normal bone (Fig. 3). Neoplastic cells exhibited random variation in cellular appearance and arrangement, as well as matrix composition and abundance. Haphazard and often intermingling patterns of osteogenic, chondroblastic, lipoblastic, and angiogenic tissue were present. Larger areas of neoplastic bone and hyaline cartilage contained multiple large areas of hemorrhage and necrosis bordered by neoplastic cells.
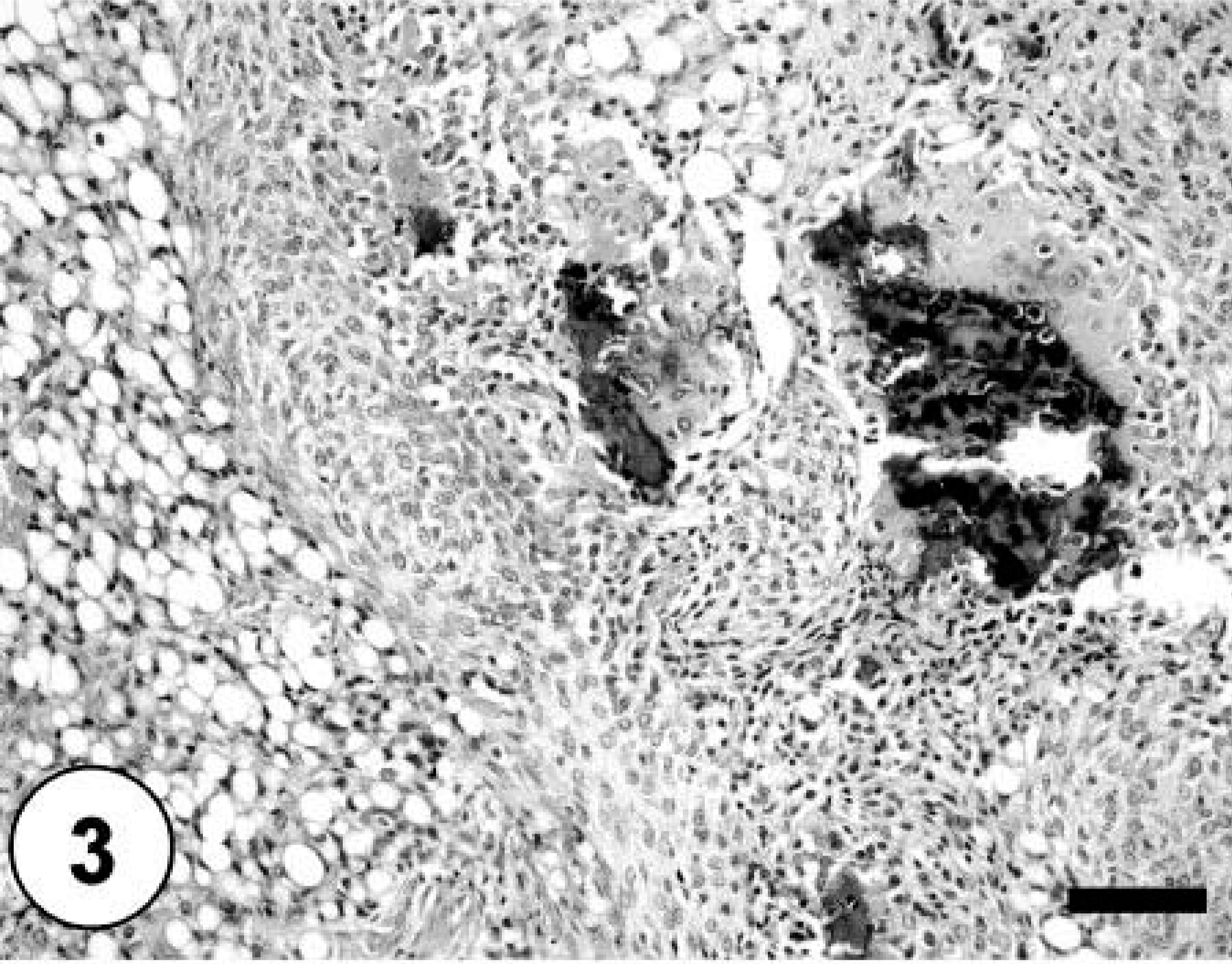
Bone; poorly differentiated neoplastic mesenchymal cells forming streams, bundles, and nests and causing destruction of normal bone. Note the multiple foci of chondromatous and lipoformative differentiation. HE staining. Bar = 100 µm.
Sections of lung, liver, spleen, and kidney contained multiple, random metastatic nodules of the malignant mixed mesenchymal sarcoma, infarction, and focal necrosis associated with vaso-occlusive neoplastic emboli. Microscopic metastases in the lung, liver, kidney and spleen consisted of neoplastic cells with varying proportions of lipoblastic (Fig. 4), angioblastic, fibroblastic, and leukocytic differentiation (Table 1). There was no evidence of osteoid production in metastatic nodules. Immunohistochemical staining of tumor tissue demonstrated cytoplasmic immunoreactivity with antibodies against vimentin, CD 18, CD 31, desmin (Fig. 5), MSA (Fig. 6), osteocalcin, and osteonectin (Fig. 7). Cells did not stain with chromogranin A + B, S-100, PGP 9.5, pancytokeratin (MNF116 and AE1/AE3), EMA, CD 45, or NSE. There was positive Oil Red O staining within the vacuolated cytoplasm of lipoblastic neoplastic cells (Fig. 8).
Various mesenchymal differentiations of primary bony neoplasms and hepatic, pulmonary, splenic, and renal metastases.
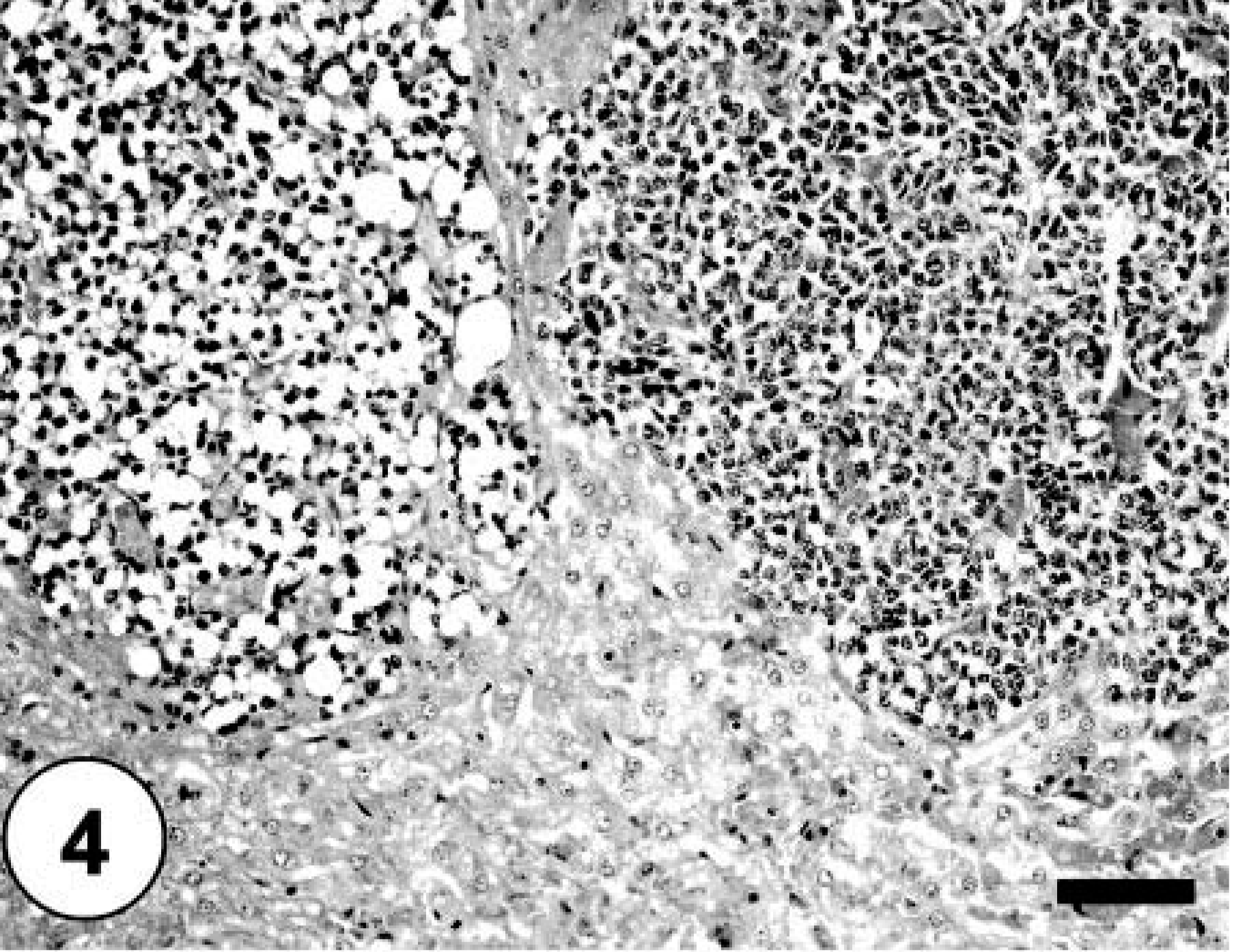
Liver; hepatic metastases composed of neoplastic adipocytes. HE staining. Bar = 100 µm.
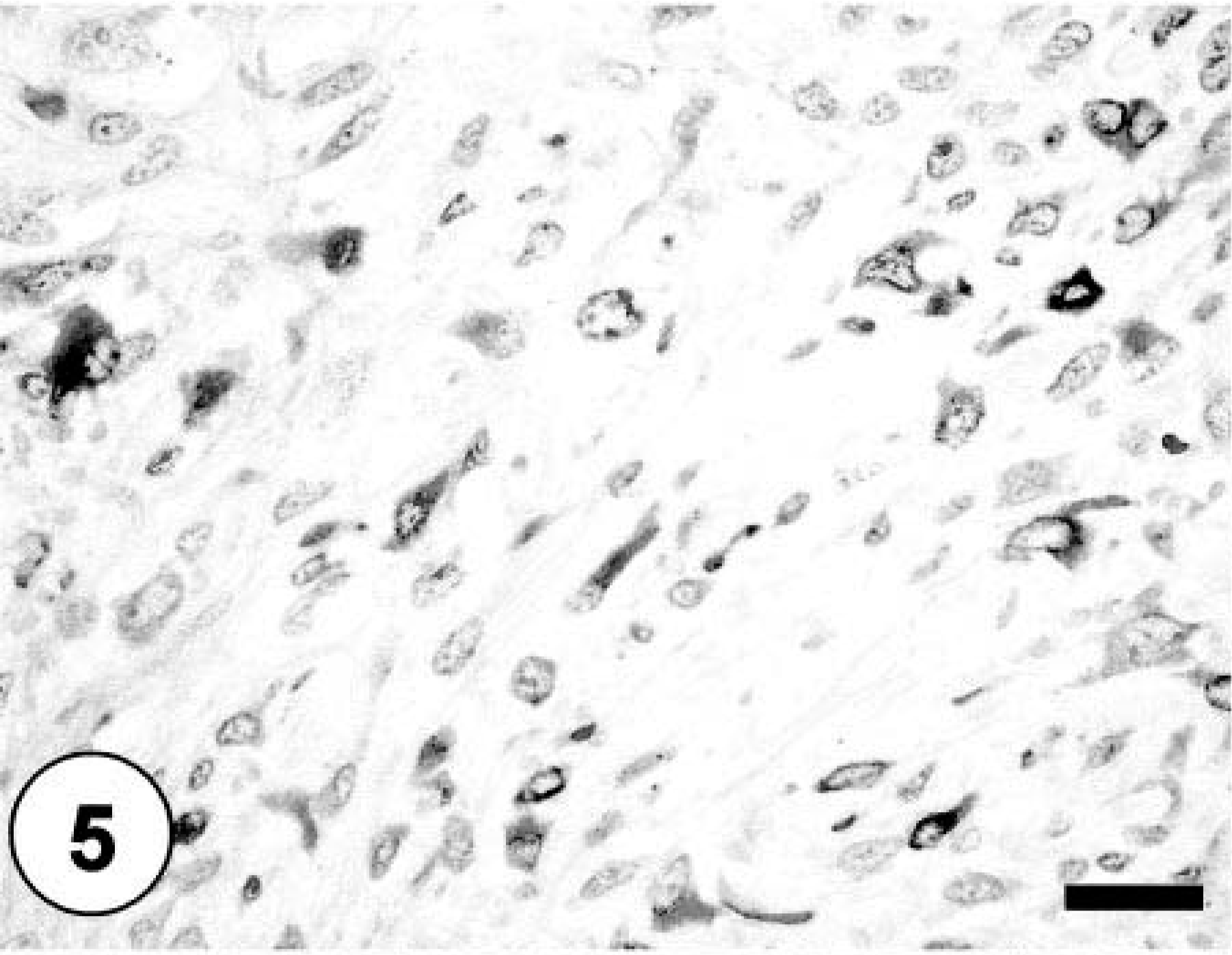
Bone; sheets and clusters of neoplastic cells demonstrating cytoplasmic staining against desmin. Labeled streptavidin–biotin–peroxidase complex method, hematoxylin counterstain. Bar = 40 µm.
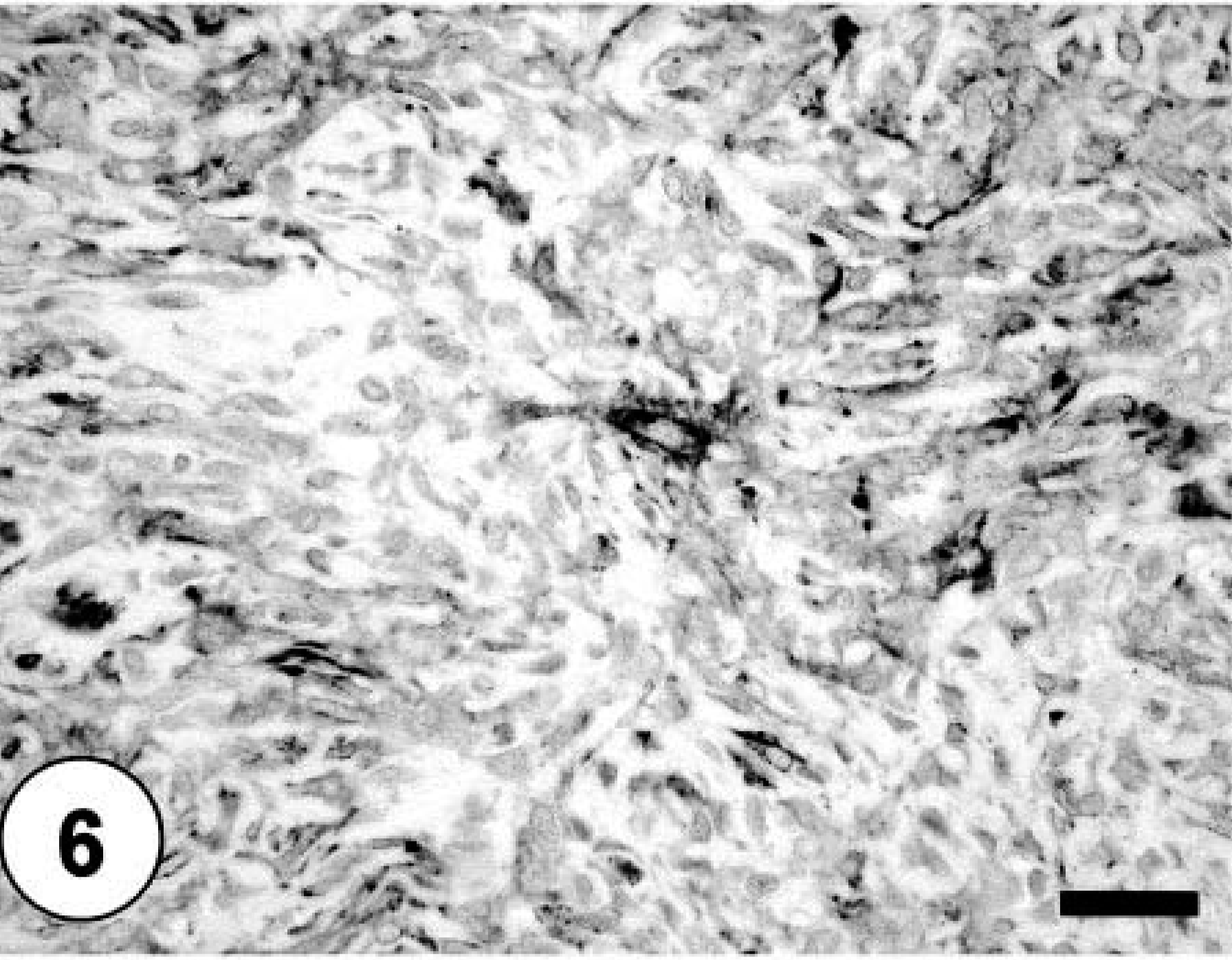
Bone; neoplastic cells exhibiting cytoplasmic immunoreactivity against MSA. Labeled streptavidin–biotin–peroxidase complex method, hematoxylin counterstain. Bar = 40 µm.
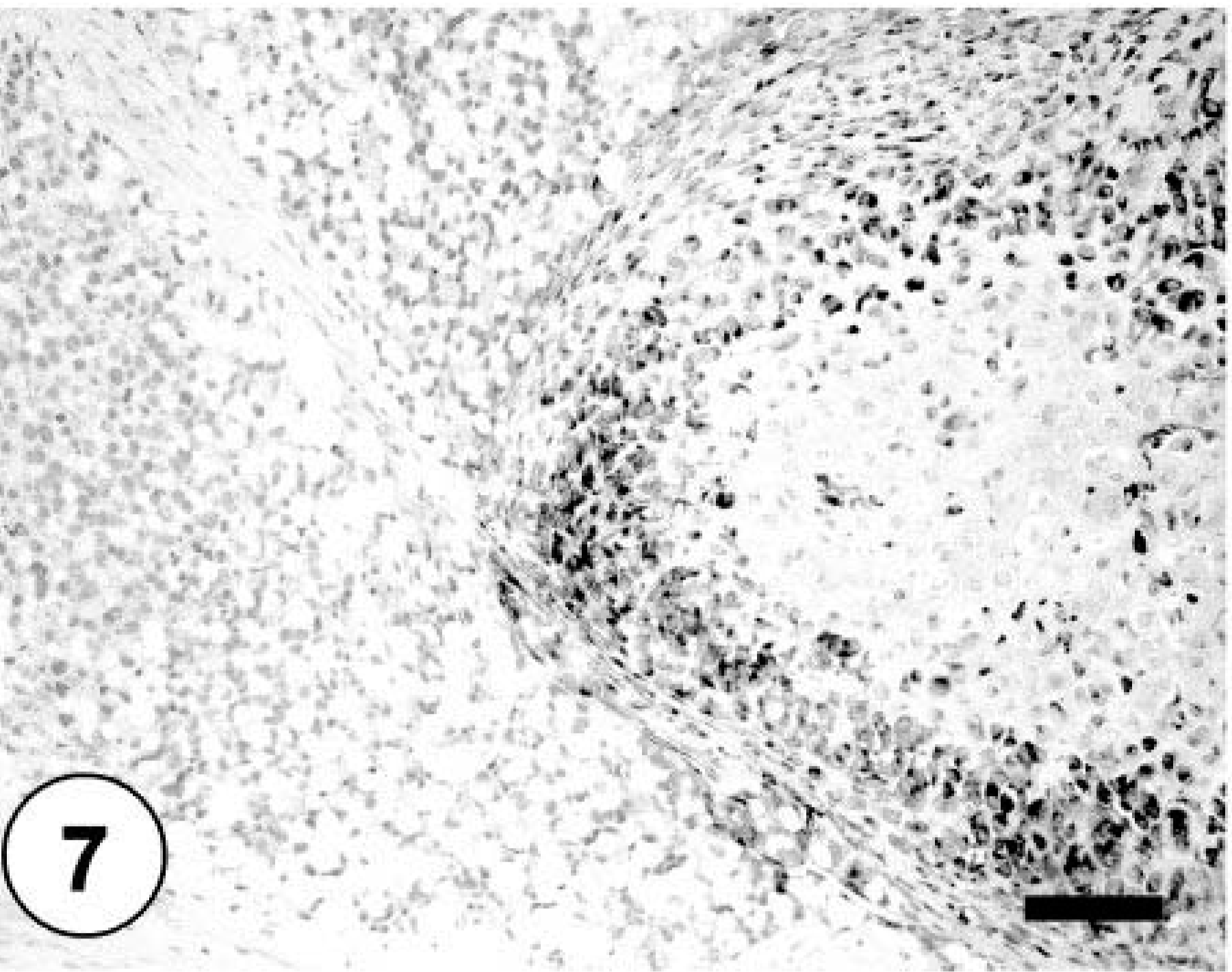
Bone; neoplastic cells exhibiting cytoplasmic immunoreactivity against osteonectin. Labeled streptavidin–biotin–peroxidase complex method, hematoxylin counterstain. Bar = 40 µm.
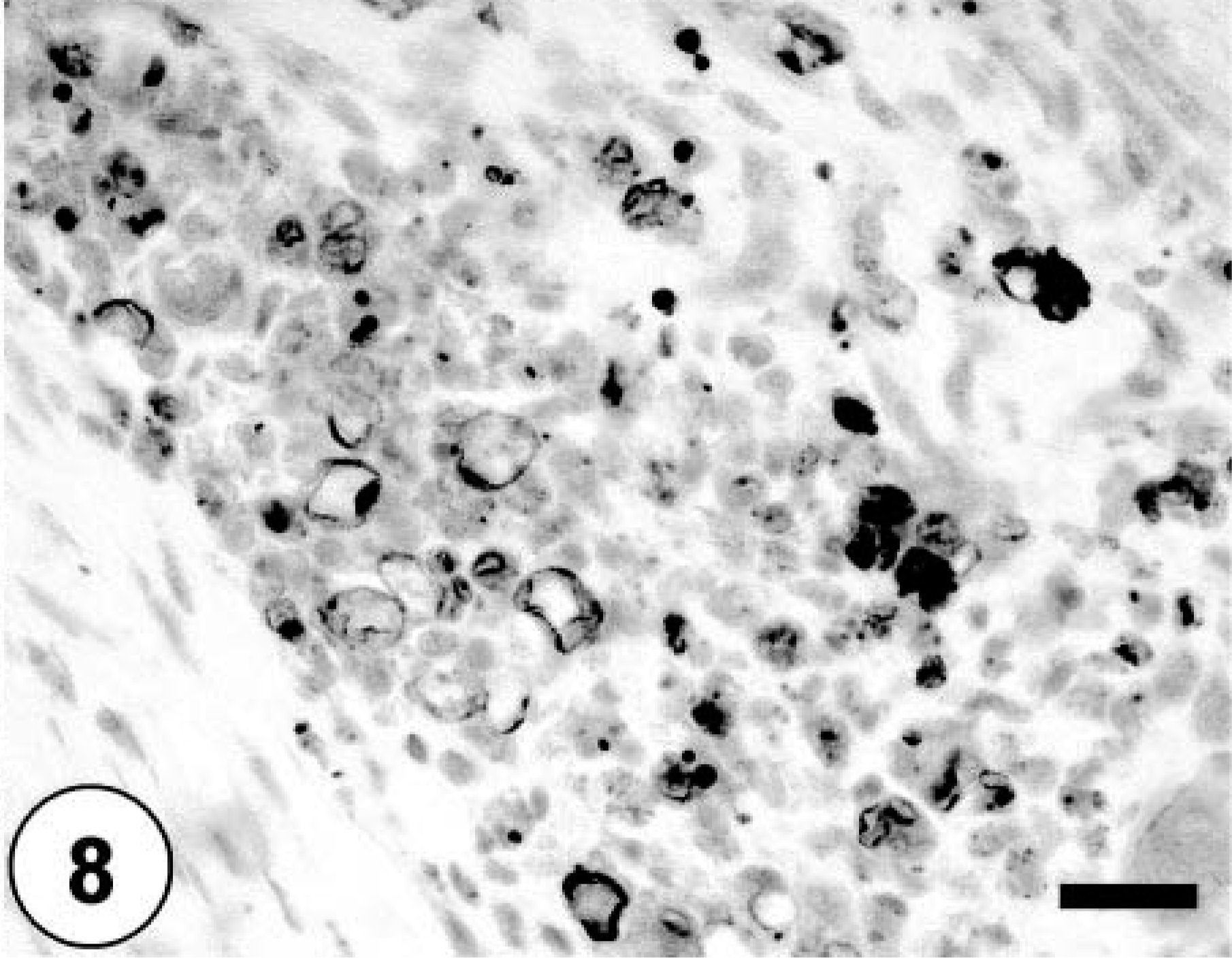
Bone; neoplastic cells exhibiting multifocal cytoplasmic staining with Oil Red O stain. Oil Red O. Bar = 40 µm.
This tumor was determined to be a highly aggressive and poorly differentiated malignant primary bone tumor having a significant component of osteoblastic and chondroblastic tumor tissue accompanied by variable amounts of lipoblastic, angioformative, myogenic, fibroblastic, and leukocytic differentiation with widespread local bone destruction and systemic metastases. The lack of osteoid in metastatic lesions is interesting and may be due to either the extremely poor differentiation of this tumor or the multipotential differentiation to nonosteoid-producing mesenchymal tissue.
Osteosarcomas are the most common primary bone tumor in dogs, accounting for more than 80% of malignant bone neoplasms. 2,10,13,20,24 They are most common in middle-aged to older large-breed dogs, with a higher incidence in male dogs. 10,12,13,20,24 Most osteosarcomas produce metastatic disease within 6 months to 1 year of diagnosis. 2,10,17 Osteosarcomas arise commonly in the proximal humerus, distal radius, distal femur, and proximal and distal tibia in the dog. 13,14,20,21,24 Primary osteosarcomas rarely cross joints or invade adjacent bones during early tumorogenesis, but as the tumor increases in size, both events occur more commonly. 8,21,23 Rare cases of multicentric osteosarcoma have been reported in humans and dogs. 3,8,10,14 An osteosarcoma that develops after successful treatment of another osteosarcoma lesion is termed a metachronous osteogenic sarcoma. 1,8,10 In human medicine, synchronous multicentric osteogenic neoplasms occur most commonly in children, usually under 15 years of age and carry a grave prognosis. 1,6,8,10 They commonly arise in the metaphyses of long bones, especially the distal femur and proximal tibia. 1,8,14,23 In such cases, it may be difficult to determine whether the neoplasm is multifocal in origin or if the lesions are caused by metastatic disease. Symmetrical and synchronous involvement with distal metaphyseal lesions of identical radiographic character in long bones and sparing of the visceral organs are suggestive of multicentric origin. 8,10,14,23 In this case, the three bone tumors in the right hind limb were larger and had more aggressive radiographic features than the bone tumor located in the left distal femur. Although the smaller size and less aggressive features of the lesion in the left distal femur could possibly reflect a later onset of a fourth primary bone tumor, the smaller size more likely reflects its origin as a metastatic neoplasm that arose at about the same time as that in the rib cage. The possibility of three synchronous tumors as separate primary tumors should be considered because of similarities in size of the right hind limb tumors.
In domestic animals, osteosarcomas can be classified as poorly differentiated, osteoblastic, chondroblastic, fibroblastic, telangiectatic, and giant cell types. 9,17,20,22 Mesenchymal tissue is known to possess high differentiation potential, and this explains the multipotential characteristics of the neoplasm in this case. In humans, although osseous neoplasms containing malignant chondroblastic or fibroblastic populations have been associated with a better prognosis, 3,12,20,23 there is still much debate in human and veterinary medicine whether histologic features of these neoplasms truly correlate with their prognosis. 3,9,20 The multipotential nature of skeletal osteosarcomas is evident in the abundant fibrosarcomatous or chrondrosarcomatous matrix in many human cases. 3,23,25 Approximately 50% of all human osteosarcomas are predominantly osteoblastic; the remainder may have a primarily chondroblastic, fibroblastic, small cell, or giant cell–rich pattern. 3,23 Osteosarcoma is one of the most common malignant neoplasms of adolescence in humans, exceeded by only leukemias, brain tumors, and lymphomas, 19,23 and the most common neoplasm of bone after myeloma. 3 Most are high-grade neoplasms, although occasionally a low-grade neoplasm is encountered. 3,23,25 Central high-grade osteosarcoma is the most common variant and usually affects children before the closure of growth plates. 3,23 Approximately 70–90% of tumors occur at the ends of long bones, particularly around the knee joint. 3,6,8,23
In the present case, the neoplasms that arose in the metaphyses of multiple long bones in the hind limbs were aggressive sarcomas that exhibited many lines of mesenchymal differentiation, making it difficult to determine the histogenetic classification of this tumor. Immunohistochemistry has been of limited use for the diagnosis of osteosarcoma in human medicine and is used primarily to exclude other neoplastic diseases. 4 Osteosarcomas are consistently immunoreactive for vimentin, and a majority of osteosarcomas express alpha smooth muscle actin focally, with a subgroup of these also concurrently expressing desmin. 4,5,7 Epithelial markers may rarely be weakly and focally reactive. 4,7,11 Glial fibrillary acidic protein and neurofilament are typically absent. 4 Osteosarcomas are reported to express immunoreactivity to antibodies to the noncollagenous proteins of bone matrix, osteocalcin, 4,7 osteopontin, 4,18 and osteonectin, 4,15 as well as the potent angiogenic protein, vascular endothelial growth factor. 4,18 Osteonectin, a 32-kd calcium-binding glycoprotein, is found in a variety of cell types, including active osteoblasts and young osteocytes. 4,5 It is involved in the early steps of mineralization of skeletal tissue and is thus a differentiation marker of normal osteogenic cells. 4,5 Osteosarcomas typically are strongly immunoreactive for osteonectin. 4,5 As expected, osteonectin immunoreactivity was diffusely present within osteosarcomatous bundles and streams of the neoplasm presented in this study. Osteocalcin is one of the most abundant noncollagenous proteins found in adult bone. 4,5 It is a highly conserved gamma-carboxyglutamic acid containing protein that is produced by osteoblasts and incorporated into the extracellular matrix of mature bone; it has been regarded as a more specific marker for bone-forming tumors than osteonectin. 4,5 Osteocalcin immunoreactivity was diffusely present within osteosarcomatous elements, especially those surrounding newly formed osteoid, and absent in liposarcomatous and other sarcomatous forms in this case. The majority of neoplastic cells demonstrated positive vimentin immunoreactivity, characterizing them as of mesenchymal origin. Desmin and MSA were expressed multifocally, as has been reported in the osteosarcoma of humans. 7 The liposarcomatous component was evident with Oil Red O staining, and angioformative bundles were immunoreactive for CD 31 and factor 8. These data taken together suggest the multipotent differentiation of this neoplasm into liposarcoma, leiomyosarcoma, fibrosarcoma, angiosarcoma, and round cell tumor in addition to its predominantly osteosarcomatous and chondrosarcomatous elements. To the authors' knowledge, an osteosarcoma with this degree of multiple differentiation has not been previously reported in the dog.
