Abstract
Diagnostic records from 338 canine oral melanomas in 338 dogs received at the Veterinary Medical Diagnostic Laboratory (1992-1999) were reviewed. Of these tumors, 122 plus an additional 7 metastatic melanomas of unknown origin were selected for clinical follow-up, histologic review, and immunohistochemistry. Chow Chow, Golden Retriever, and Pekingese/Poodle mix breeds were overrepresented, whereas Boxer and German Shepherd breeds were underrepresented. There was no gender predisposition and the average age at presentation was 11.4 years. Forty-nine dogs were euthanized due to recurrence or metastasis. The average postsurgical survival time was 173 days. The gingiva and the labial mucosa were the most common sites. Most tumors were composed of either polygonal cells (27 cases, 20.9%), spindle cells (44 cases, 34.1%), or a mixture of the two (polygonal and spindle) (54 cases, 41.9%). Clear cell (3 cases, 2.3%) and adenoid/papillary (1 case, 0.8%) patterns were uncommon. The metastases of 6/6 oral melanomas had morphologic and immunohistochemical features similar to those of the primary tumors. Immunohistochemically, Melan A was detected in 113/122 oral (92.6%) and 5/7 (71.9%) metastatic melanomas. Only 4/163 nonmelanocytic tumors were focally and weakly positive for Melan A. Antibodies against vimentin, S100 protein, and neuron-specific enolase stained 129 (100%), 98 (76%), and 115 (89.1%) of 129 melanomas, respectively. Antibodies against other melanocyticassociated antigens (tyrosinase, glycoprotein 100) did not yield adequate staining. We conclude that Melan A is a specific and sensitive marker for canine melanomas.
Melanomas are one of the most frequently diagnosed malignant neoplasms of the canine oral cavity. 2 7 9 10 16 19 21 29 40 56 60 Oral melanoma is most common in dogs older than 10 years. 7 10 16 21 56 60 Small breeds, especially the Cocker Spaniel, and dogs with heavily pigmented oral mucosa are at greater risk of developing oral melanoma. 10 16 19 21 29 This tumor can be located in any part of the oral cavity, but the gingiva is the most common site. 7 10 16 19 21 56 The usual behavior of oral melanomas is focal infiltration, recurrence, and metastasis to regional lymph nodes and less frequently to lungs and other organs. 2 7 10 21 29 41 56 60 Therefore, nearly all oral melanomas are considered malignant. 8 59 The diagnosis of oral melanomas may be challenging because there is variation in the degree of pigmentation and tumors can be completely unpigmented. 10 21 27 29 Another complicating factor is the considerable repertoire of microscopic appearances; 21 59 melanomas may resemble carcinomas, sarcomas, lymphomas, and osteogenic tumors. 14 42 Thus, immunohistochemical confirmation of the diagnosis of melanoma is frequently necessary to establish a prognosis and therapeutic plan.
Histochemically, melanocytic tumors are positive by the dihydroxyphenylalanine oxidase (DOPA) reaction and the Masson-Fontana stain. However, the DOPA reaction is best performed in frozen tissues, and the Masson-Fontana stain reacts with substances other than melanin such as lipofuscin and argentaffin granules of neurosecretory cells. 52 Immunohistochemical detection of human melanocytic markers employs antibodies to S100 protein, neuron specific enolase, glycoprotein 100, tyrosinase, melanosoma associated antigens, and Melan A protein. 6 11 12 18 20 23 30 31 35 47 54 57 Antibodies to microphthalmia transcription factor (Mitf), a melanocytic nuclear protein critical for embryonic development and postnatal viability of melanocytes, 28 58 have been recently used in the diagnosis of human melanocytic tumors. 37
The marker most widely used in immunohistochemical diagnosis of canine melanomas is S100 protein, which is demonstrated in more than 50% of melanomas. 14 44 48 49 However, antibodies against S100 protein stain a variety of normal tissues and nonmelanocytic neoplasms. 54 Antibodies directed against specific melanocytic antigens used in canine melanomas are HMB-45 (against glycoprotein 100 of premelanosomes 1–3), 4 51 53 NKI/C3 (against premelanosomal vesicles), 57 and HMSA-1 and HMSA-5 (against human melanosome-specific antigens). 1 4 32 A monoclonal antibody against a canine melanoma cell line has been also used for immunohistochemical detection of melanomas. 43
The purposes of this study were 1) to report epidemiologic data from a large collection of oral melanomas; 2) to review tumor behavior and examine microscopic characteristics of 129 oral and 7 metastatic melanomas; 3) to report immunohistochemical results using antibodies to vimentin, neuron-specific enolase, S100 protein, and Melan A in 129 canine oral and metastatic melanomas; and 4) to determine the presence of Melan A in normal tissues and nonmelanocytic tumors. To our knowledge, this is the most comprehensive study of canine oral melanomas including epidemiologic, microscopic, and immunohistochemical data. This is the first report in which immunohistochemistry for Melan A has been used to characterize canine melanocytic neoplasms.
Materials and Methods
Selection of cases
The database of the University of Missouri–Veterinary Medical Diagnostic Laboratory was searched for cases of canine melanomas in the digestive tract. Of 338 canine oral melanomas diagnosed from January, 1992, through December, 1999, 135 cases were initially selected and reviewed by one author (JAR) to determine their suitability for the study. Cases with small samples or no tissue remaining were excluded. Those cases in which the morphologic diagnosis was not definitive were reviewed by another three pathologists (MAM, GCJ, LWP) to establish consensus. This review process resulted in selection of 122 oral melanomas for detailed study. An additional seven metastatic melanomas (any melanoma with unknown primary site located in lymph nodes or internal organs) were included in the study. Metastases of six oral melanomas already included in this study were also available. One dog had an oral melanoma and a dermal melanoma on its hip. The representation of each breed, age, and sex in the 338 dogs with oral melanoma was compared with the proportion of that breed, age, and sex in the population (all canine necropsy and surgical biopsy accessions from 1992–1999) by chi-square analysis of contingency tables using SigmaStat, version 2.03 (Access Softek, Inc.). The Mann–Whitney rank sum test was used to determine the significance of the differences in median survival time between 18 dogs with gingival and 18 dogs with nongingival melanocytic tumors for which survival time was known. This test was also used to determine the significance of the differences in median survival time by mitotic index among 40 dogs for which survival time was known. Values were considered significantly different when P < 0.05. The following characteristics were recorded for the 129 melanomas studied in detail: breed; sex; age; survival time; tumor size (only for oral masses); location of the primary tumor (gingiva, lip, tongue, or other oral site) including metastases if present; morphology of the neoplastic cells (epithelioid, spindle, mixed, adenoid, and clear); degree of pigmentation using a subjective scale from nonexistent, scant (pigment in fewer than 30% of cells), moderate (pigmentation in 31–80% of cells), and abundant (pigment in more than 80% of cells); junctional activity (present or absent); number of mitoses/10 high-power fields (hpf); amount of stroma (scant, moderate, or abundant); intravascular tumor cells; completeness of excision (complete or incomplete).
Reagents
Three monoclonal antibodies to melanocytic antigens, a mouse monoclonal antibody melanoma Ab-3 (Lab Vision, MS-688, clones HMB-45 and HMB-50), a mouse monoclonal antibody against tyrosinase (Lab Vision, MS-800, clone T311), and a mouse monoclonal antibody against Melan A (Novocastra Laboratories, NCL-Melan A, clone A103), were initially tested in five canine melanomas to determine reactivity and establish the best immunohistochemical protocol. Antibodies to the following markers were also tested in the 129 oral melanomas and metastases selected: S-100 protein (Novocastra Laboratories, NCL-S100p, rabbit polyclonal, 4 µg/ml); neuron-specific enolase (NSE) (Dako Corporation, A0587, rabbit polyclonal, 14.6 µg/ml), and vimentin (Dako Corporation, M7020, mouse monoclonal, clone Vim 3B4, 0.65 µg/ml). In eight cases, staining with a monoclonal antibody broadly reactive with cytokeratins (Dako Corporation, M0821, mouse monoclonal, clone MNF116, 0.16 µg/ml) was also performed.
Immunohistochemical staining procedure
Formalin-fixed, paraffin-embedded tissue sections were used. Four-micrometer sections on positive-charged slides were processed according to four different protocols: 1) no pretreatment; 2) pretreatment with proteinase K (Dako corporation) according to the manufacturer's specfications; 3) pretreatment with a steamer, heating the slides in antigen retrieval citrate buffer solution (Dako corporation) at pH 6.0; or 4) pretreatment with a steamer, heating the slides in antigen retrieval EDTA buffer solution (Lab Vision) at pH 8.0. Detailed protocols of antigen retrieval have been reported. 45 Two-fold dilutions (starting at 1/10) of the primary antibody were used to determine the optimal concentration. We tested two signal-generating systems, including labeled streptavidin-peroxidase (LSAB+, Dako Corporation) and a two-step, non–avidin-biotin method (ENVISION+, Dako Corporation). Different incubation times (30, 60, and 90 minutes) at room temperature (RT) were used for the primary antibodies. Other layers of antibodies or reagents were incubated for 30 minutes at RT. 45 Based on this initial testing, the only antibody that gave consistent staining of melanoma cells without background was that directed against Melan A. The optimal pretreatment was heating the slides in EDTA buffer (pH 8.0) with a steamer, although citrate buffer also revealed reactivity but less intensely and in fewer cells. Not only did tyrosinase antibody react strongly with melanoma cells, but also it stained nonmelanocytic cells and tissues including mucosal epithelium, epidermis, adnexa, and skeletal muscle. HMB-45/50 antibody did not react under the conditions tested. Thus, we used Melan A antibody to test the following canine samples: 122 canine oral melanomas (including six metastatic sites), 7 metastatic melanomas, and 163 nonmelanocytic and normal tissues (Table 2). Melan A antibody was used at a concentration of 1.15 µg/ml and was incubated for 90 minutes at RT. Incubation protocols for S100 protein, NSE, and vimentin antibodies have been described. 45 Antigen retrieval for S-100 immunohistochemistry was done heating the slides in citrate buffer.
Breed distribution in 338 canine oral melanomas.
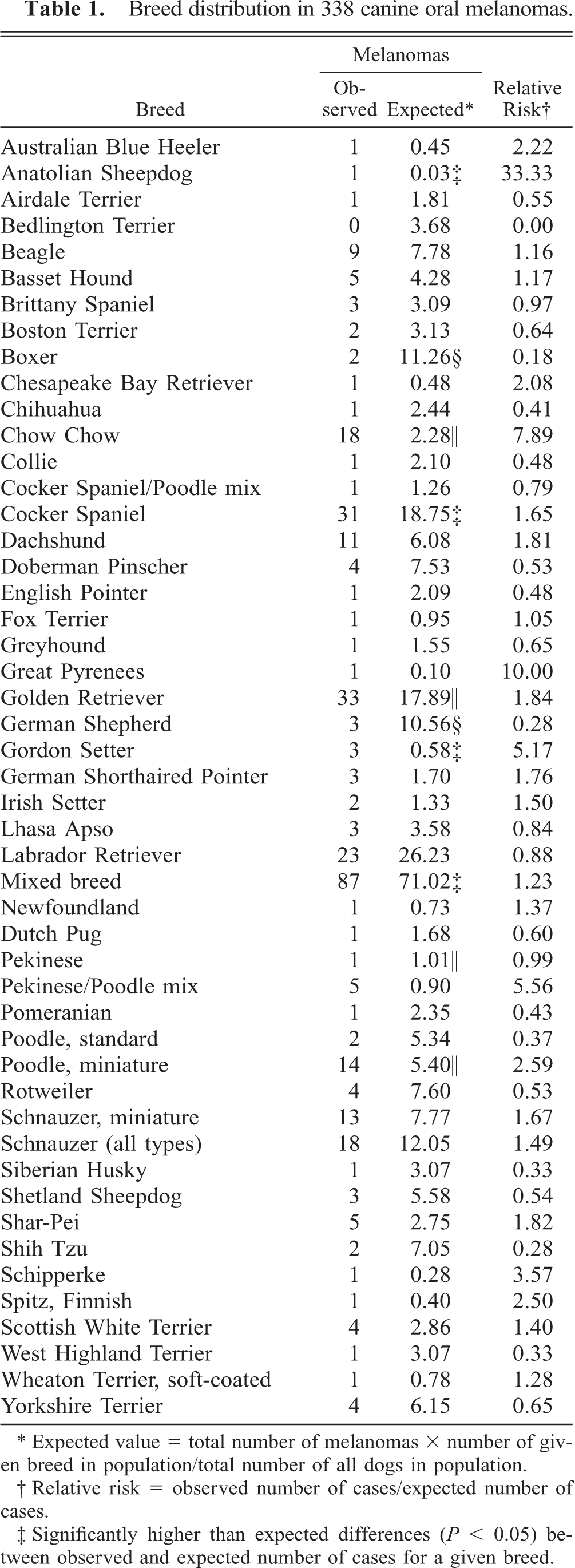
Expected value = total number of melanomas × number of given breed in population/total number of all dogs in population.
Relative risk = observed number of cases/expected number of cases.
Significantly higher than expected differences (P < 0.05) between observed and expected number of cases for a given breed.
Significantly lower than expected differences (P < 0.05) between observed and expected number of cases for a given breed.
Significantly higher than expected differences (P < 0.001) between observed and expected number of cases for a given breed.
Immunohistochemical staining results in formalin-fixed, paraffin-embedded nonmelanocytic canine tumors and normal tissues with antibody to Melan A.

Including bronchogenic and bronchoalveolar carcinomas and adenocarcinomas.
The reaction in the positive tumors was weak and focal.
Respiratory and alveolar epithelium.
Weak reactivity.
An unrelated monoclonal antibody of the same isotype and similar protein concentration replaced the primary monoclonal antibody for negative control. For polyclonal antibodies, normal rabbit serum replaced the primary antibody. In cases in which the amount of melanin obscured the immunologic reaction, tissues were counterstained with Azure B. 34
Scoring of immunohistochemical results with Melan A
A semiquantitative evaluation of the percentage of positive tumor cells was used: 1 (5–10% of positive cells), 2 (11–50%), 3 (51–80%), and 4 (>80%). A subjective grading system was used to determine the intensity of the reaction for Melan A, in which 0 indicated no reaction, 1 indicates weak reaction, 2 indicates moderate reaction, and 3 indicates intense reaction. Due to the cellular heterogeneity of many melanomas, a cell type reactivity (epithelioid, spindloid, etc.) to Melan A was also recorded for each melanoma as well as reactivity of junctional melanocytes and individual intraepithelial melanocytes.
Effect of prolonged fixation for detection of Melan A
Since the fixation time of most tumors was unknown, one oral melanoma was fixed for 24 hours to determine the effects of prolonged fixation on immunohistochemistry. The tumor stained positive for Melan A, vimentin, neuron-specific enolase, and S100 protein. Additional samples from this melanoma were fixed for 3, 6, 11, 19, 26, 35, and 41 days before processing. Another oral melanoma was tested with fixation times of 1, 5, 14, and 20 days. Intensity of immunostaining was compared among samples by semiquantitative evaluation of percentage of positive cells and subjective grading of intensity for Melan A, S100 protein, NSE, and vimentin. Fixation time for normal tissues collected from healthy adult dogs was 24–48 hours.
Results
Epidemiologic analysis
The 338 oral melanomas comprised 0.99% of the 33,826 canine pathology accessions received from 1992 through 1999. Anatolian Sheepdog, Cocker Spaniel, Gordon Setter, mixed breed dogs (P < 0.05), Chow Chow, Golden Retriever, Pekingese/Poodle cross, and Miniature Poodle breeds (P < 0.001) were significantly overrepresented, whereas Boxer and German Shepherd breeds were underrepresented in the oral melanoma population (Table 1). Ages ranged from 1 to 19 years (mean 11.4 years; median 4,380 days). There were 167 (49.4%) females, 163 (48.22%) males, and 8 animals with unreported sex. No significant differences for breed, age, or sex were found between all canine oral melanoma accessions and the subset of 129 melanomas examined microscopically and immunohistochemically.
Additional epidemiologic information was obtained by a phone survey to the referring veterinarians on 100/129 (77.5%) cases. Two animals died of natural causes, and 49 (49%) dogs were euthanized. The reason for euthanasia was usually dysphagia or evidence of metastasis. Five animals were euthanized at the time of surgery. The average survival time for the 46 animals that were euthanized (not including those euthanized at the time of surgery) or died of natural causes was 173 days (range 5–869 days). At the time of writing this paper, 33 dogs had survived 45 days to 3.9 years after diagnosis.
The maximum dimension of 122 oral melanomas, as measured in histologic sections, was less than 1 cm in 23 (18.8%) cases, 1.1–2 cm in 63 (51.7%) cases, 2.1–3 cm in 32 (26.2%) cases, and greater than 3.1 cm in four cases (3.3%).
Slightly more than half (65 cases, 53%) of 122 oral tumors involved the labial mucosa or mandibular gingiva. Less common sites included the cheeks, the palate/pharynx, the tongue, the maxillary gingiva, the tonsils, and a nonspecified gingival location. The exact location of 15 (12.3%) tumors was not recorded.
Recurrences were reported in 31/72 (43%) oral melanomas and 41/72 (57%) cases did not have evidence of recurrence; this information was not available in the remaining cases. Although the Kaplan–Meier survival curve showed differences among different locations (results not shown), they were not significantly different. Metastatic status was reported for 66/122 oral melanomas; 46 (69.7%) cases did not have evidence of metastases and 20 (30.3%) had metastases, usually to regional (submandibular) lymph nodes.
Microscopic features
Most of the 129 tumors reviewed microscopically were classified in three groups by their preponderant cell type: 27 (20.9%) tumors had polygonal (epithelioid) cells, 44 (34.1%) had spindle cells (Fig. 1), and 54 (41.9%) tumors had mixed (polygonal and spindle) cell types. In addition, three (2.3%) cases consisted mainly of large, clear cells (Fig. 2) and one case (0.8%) had cords of polygonal cells with an adenomatous/papillary appearance. Metastases from six oral melanomas were morphologically similar to their primary tumors and included three spindle, two polygonal, and one mixed cell type. One dog had a dermal (hip) melanoma with spindle cell morphology and an oral melanoma with mixed morphology. The degree of cellular pleomorphism was moderate in 81 (62.8%) and high in 48 (37.2%) cases. The cytoplasm of melanocytic cells was usually abundant and had variable tinctorial characteristics (from eosinophilic to basophilic). Nuclei were oval to round and usually had reticulated chromatin with prominent single or multiple nucleoli. The degree of pigmentation ranged from nonexistent (41 cases, 32%) to scant (32 cases, 24.5%), moderate (26 cases, 19.5%), and abundant (30 cases, 24%). Melanophages (epithelioid macrophages containing abundant coarse melanin granules) were present in moderate or abundant numbers in 68 cases. The number of mitoses/10 high-power fields (hpf) were 0 (9 cases, 22.5%), 1–10 (29 cases, 22.5%), 11–30 (41 cases, 31.7%), 31–50 (24 cases, 18.6%), 51–70 (17 cases, 13.2%), 71–90 (4 cases, 3.1%), and more than 90 mitoses (5 cases, 3.9%). The average mitotic index/10 hpf was 31.4, the standard deviation was 25.6, and the median was 25.0. The highest mitotic index was 103/10 hpf. Although the Kaplan–Meier survival curve showed differences among different mitotic indexes, these results were not statistically significant. Junctional activity was present in 80 (64.5%) cases and absent in 20 (16.1%). Due to the size of the biopsy or the degree of ulceration, we could not evaluate this feature in 22 cases. Stroma was scant in 67 (51.8%) cases, moderate in 48 (37.2%) cases, and abundant in 14 (11%) cases. Vascular invasion was only apparent in 8 (6.45%) cases. Complete excision was documented histologically in only 8 of 122 (6.5%) cases.
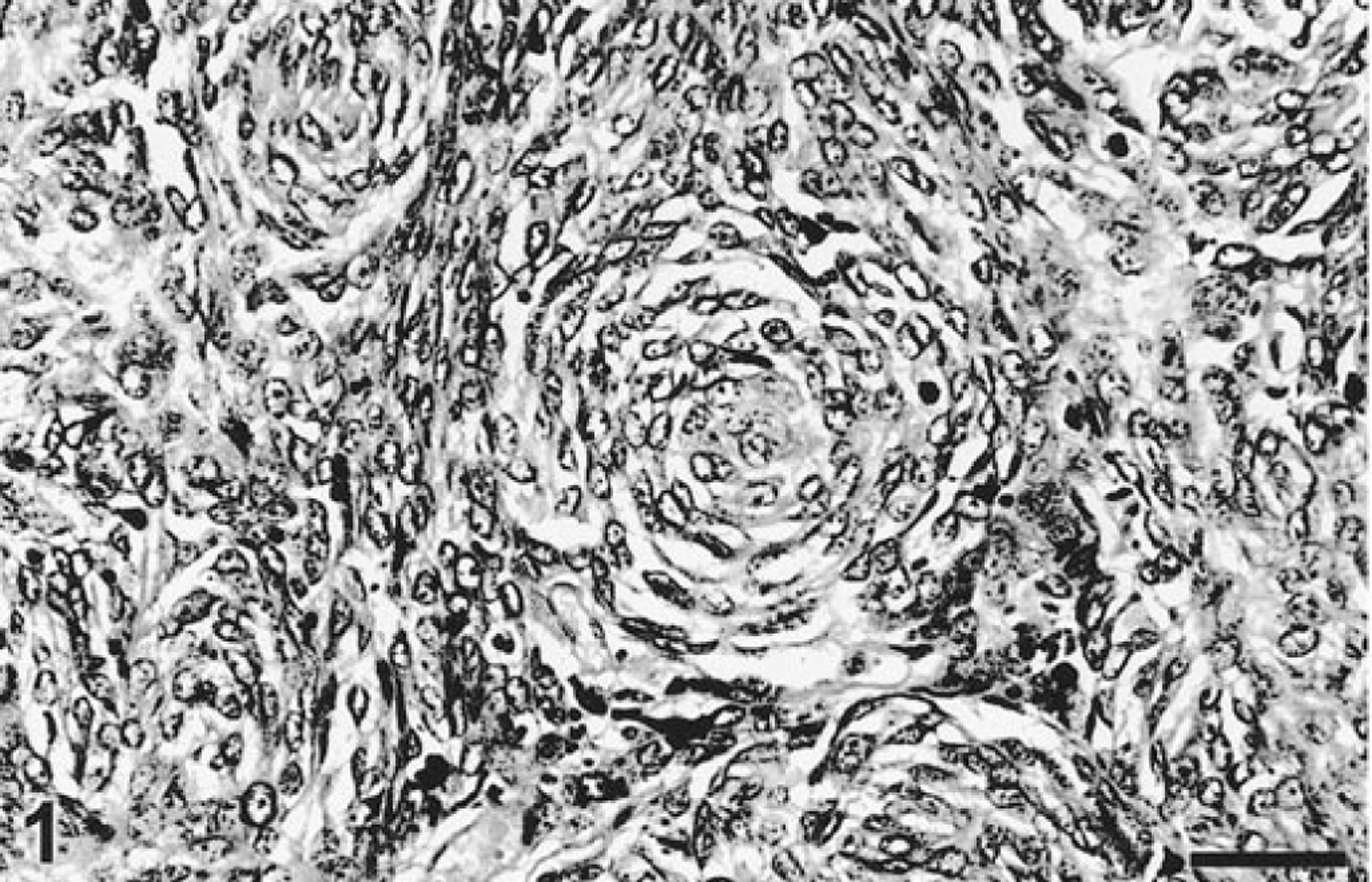
Gingiva; dog. Melanoma. Whorls of spindle cells. HE. Bar = 76 µm.
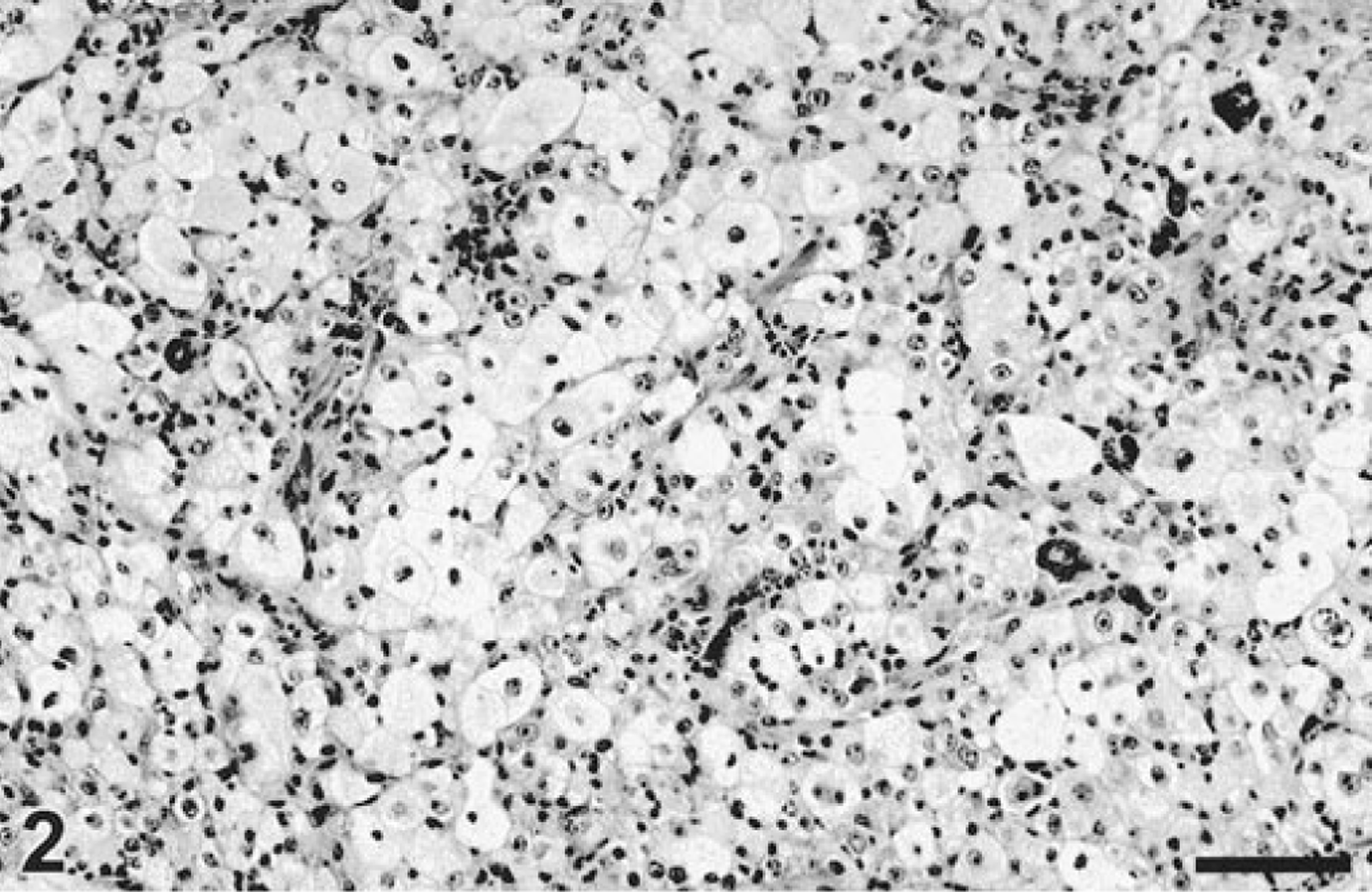
Gingiva; dog. Melanoma. Large polygonal cells with clear cytoplasm. HE. Bar = 76 µm.
Immunohistochemical results
Melan A staining in normal tissues (Table 2)
The epithelium of the interlobular and intralobular ducts of salivary glands had strong nuclear and less intense cytoplasmic staining. Although intralobular ducts reacted less intensely than interlobular ducts, intralobular staining was still prominent, whereas other salivary gland tissues were negative. Frequently, intraepidermal melanocytes of the skin and more rarely melanocytes at the hair bulb were positive. Prostatic acinar cells and urethral epithelium were weakly positive (cytoplasmic). The epithelial cells of the eyelid were multifocally positive (nuclear and cytoplasmic). Numerous intrapulmonary airway cells reacted (strongly nuclear and weak cytoplasmic). Many stained cells were ciliated, but some nonciliated cells were also positive. Melan A staining in the lung was heterogeneous, with some airways not showing any reactivity. Cells lining alveolar walls were strongly and consistently positive. The staining predominantly involved the nucleus of squamous and cuboidal cells (most likely both Type I and Type II alveolar epithelium). However, in many areas, there was a distinct thin line of immunoreactivity along the alveolar surface, which suggested cytoplasmic staining of Type I alveolar epithelium. Free round cells in alveolar lumina were negative. Unlike other tissues, in which reactivity varied among samples, reactivity of pulmonary cells was consistent in the majority of normal samples. The pancreas had moderate cytoplasmic and nuclear staining of the intralobular ductal epithelium only. The cytoplasm of adrenal cortical epithelial cells, especially the zonae fasciculata and reticularis, stained strongly and diffusely in all samples. Adrenal medullary cells were negative. In the testis, only interstitial cells consistently yielded a strong reaction. This reactivity was intense in the nucleus and moderate in the cytoplasm. Other tissues (parathyroid, liver, kidney, spleen, lymph node, stomach, small and large intestine, esophagus, skeletal muscle, peripheral nerve, spinal cord, and brain) were negative.
Immunoreactivity for Melan A in melanomas
Melan A reactivity was demonstrated in 118 (91.5%) of the 129 melanomas examined, including 113 oral and 5 metastatic melanomas. Metastases of Melan A-negative primary sites were also negative (2/2 cases), whereas 5/5 primary oral melanomas with metastases had positive staining for Melan A in both the primary and metastatic sites; however, the staining was somewhat weaker in the metastases. Thirteen (11%) tumors had 5–10% positive cells, 34 (28.8%) had 11–50% positive cells, 37 (31.4%) had 51–80% positive cells, and 34 (28.8%) had more than 80% positive cells. The intensity of staining was high in 101 (78.3%) cases, moderate in 11 (8.6%) cases, weak in 6 (4.6%) cases, and absent in 11 (8.5%) cases. Nuclear staining was not visible in any melanoma. All tumor cell types demonstrated reactivity for Melan A (Figs. 3, 4). The staining in the majority of cases was heterogeneous with areas not stained at all and others strongly stained in the same section. There was no obvious relationship between the location of tumor cells (superficial or deep within the mass) and reactivity for Melan A. Although there was a tendency for staining of fewer spindle than polygonal cells, some spindle cell tumors reacted strongly and diffusely. Two of three clear cell melanomas (Fig. 5) reacted strongly and one was negative. The adenoid/papillary cell type was also strongly and diffusely positive (Fig. 6). Melan A was detected in the cytoplasm in three patterns: diffuse (87 cases, 67.4%), polar or punctate (5 cases, 3.9%), and both diffuse and polar (26 cases, 20.1%) (Fig. 7). Junctional cells and individual atypical melanocytes were usually stained (Fig. 8). There was no staining in the negative controls.
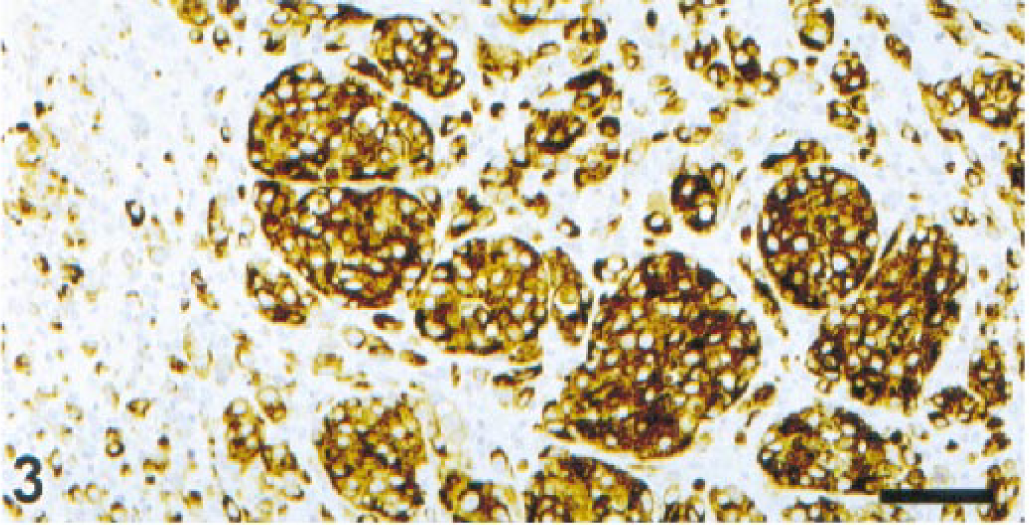
Lip; dog. Packets of polygonal cells strongly positive for Melan A. ENVISION+ peroxidase stain with Mayer's hematoxylin counterstain. Bar = 76 µm.
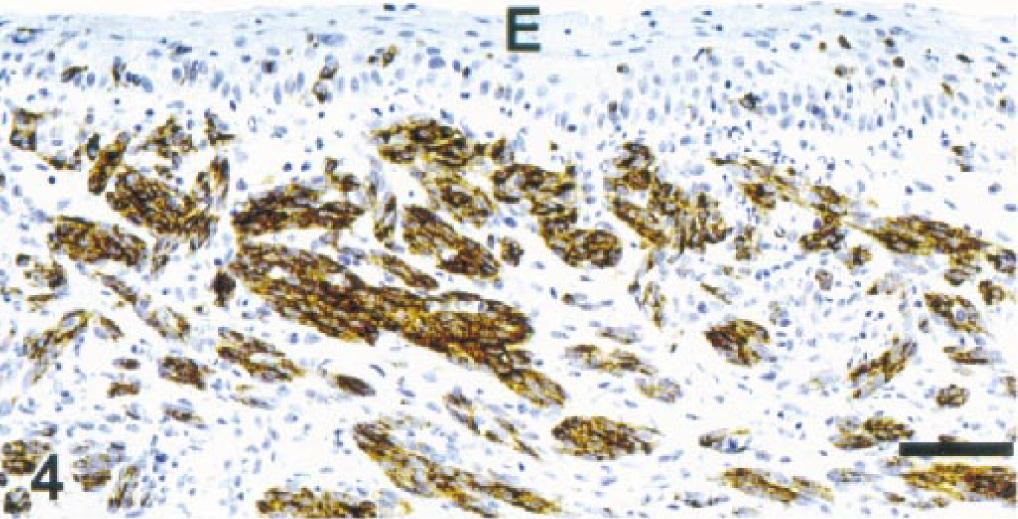
Gingiva, dog. Melanoma. Spindle cells in the submucosa. Melan A. E = mucosal epithelium. ENVISION+ peroxidase stain with Mayer's hematoxylin counterstain. Bar = 76 µm.
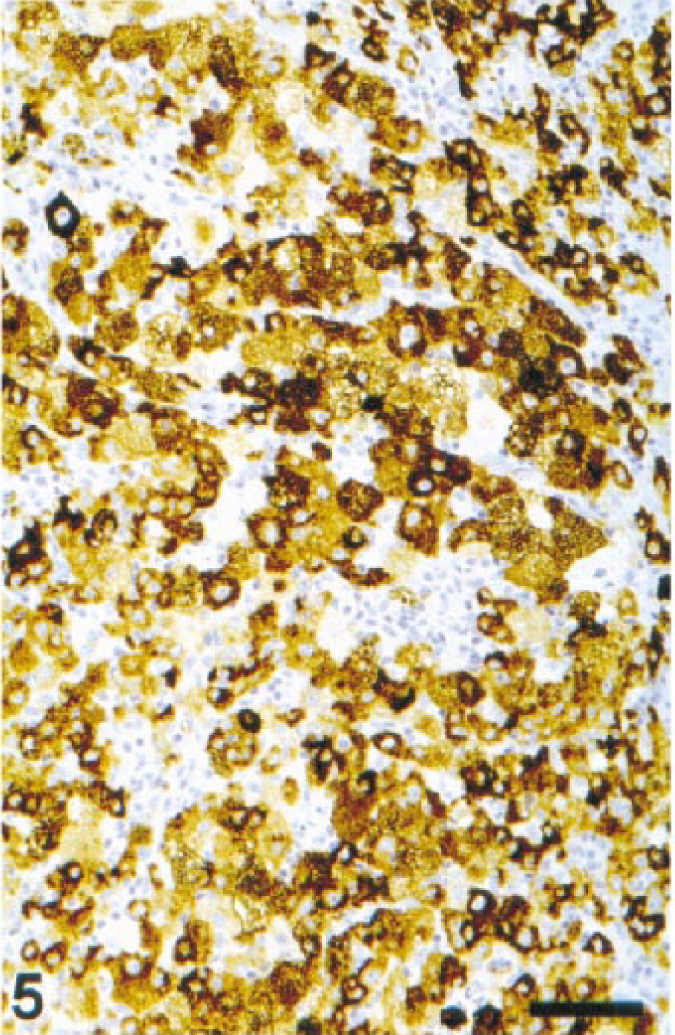
Cheek; dog. Melanoma. Large, clear cells strongly positive for Melan A. ENVISION+ peroxidase stain with Mayer's hematoxylin counterstain. Bar = 76 µm.
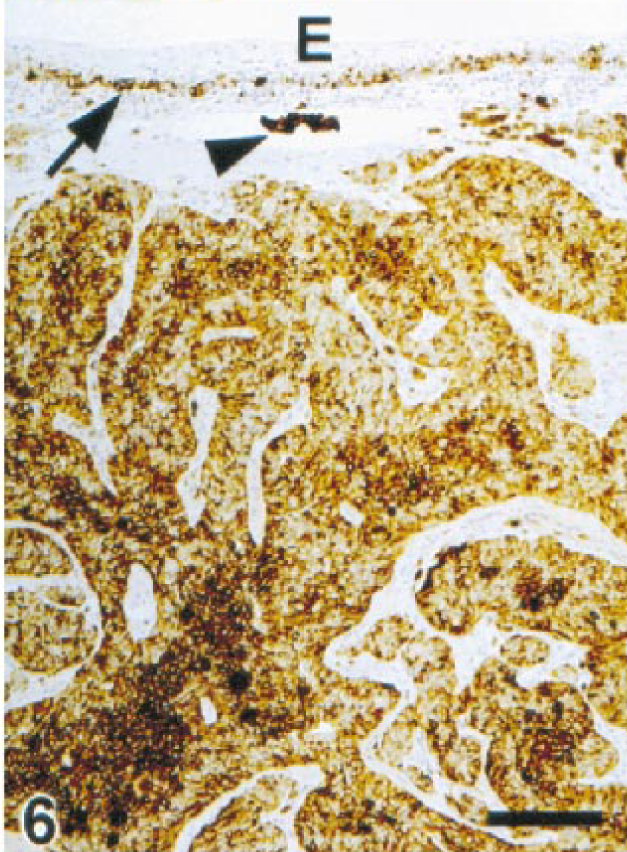
Gingiva; dog. Melanoma. Adenoid/papillary growth in the submucosa positive for Melan A. Intravascular cluster of melanoma cells (arrowhead). Intraepithelial melanocytes (arrow) are also positive for melan A. E = mucosal epithelium. ENVISION+ peroxidase stain with Mayer's hematoxylin counterstain. Bar = 153 µm.
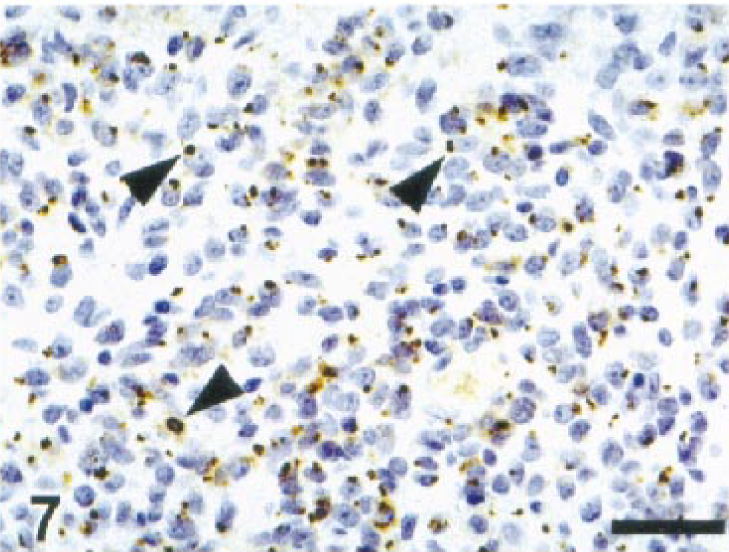
Lip; dog. Melanoma. Numerous melanoma cells are positive for Melan A with a punctate pattern of staining (arrowheads). ENVISION+ peroxidase stain with Mayer's hematoxylin counterstain. Bar = 38 µm.
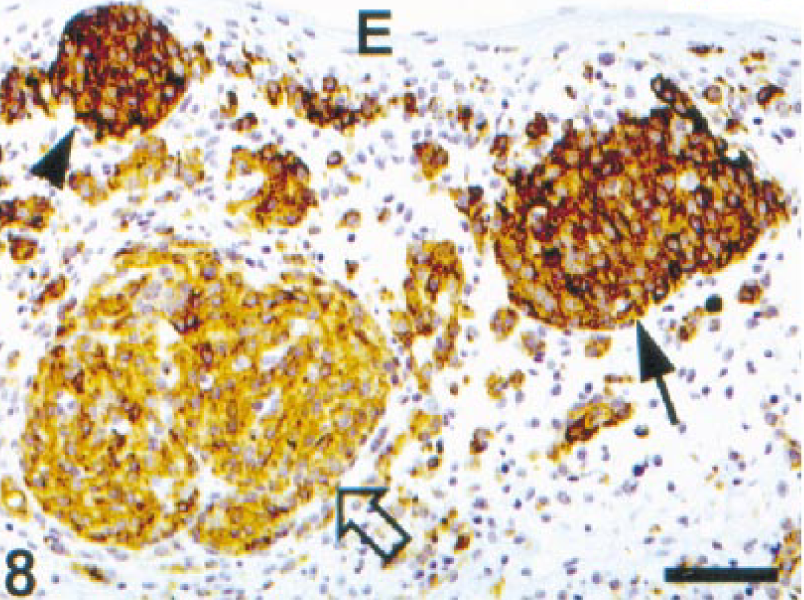
Lip; dog. Melanoma. Junctional cells (arrowhead), an early invading cluster of cells (arrow), and another cluster of cells already in the submucosa (open arrow) are strongly positive for Melan A. ENVISION+ peroxidase stain with Mayer's hematoxylin counterstain. Bar = 76 µm.
Melan A staining of nonmelanocytic neoplasms
One hundred sixty-three nonmelanocytic neoplasms were tested (Table 2). Only four tumors (2.45%), two salivary carcinomas and two transitional cell carcinomas, reacted with Melan A antibody (Table 2). This reaction was focal and weak in all four cases (Fig. 9). To confirm immunoreactivity for Melan A, four heavily pigmented melanomas were counterstained with Azure B. Melanin stained blue-green and DAB remained brown, although it darkened. Cells containing both melanin and Melan A stained dark green-brown to black and were easily distinguished from those with only melanin or DAB (Fig. 10). Azure B did not stain hemosiderin.
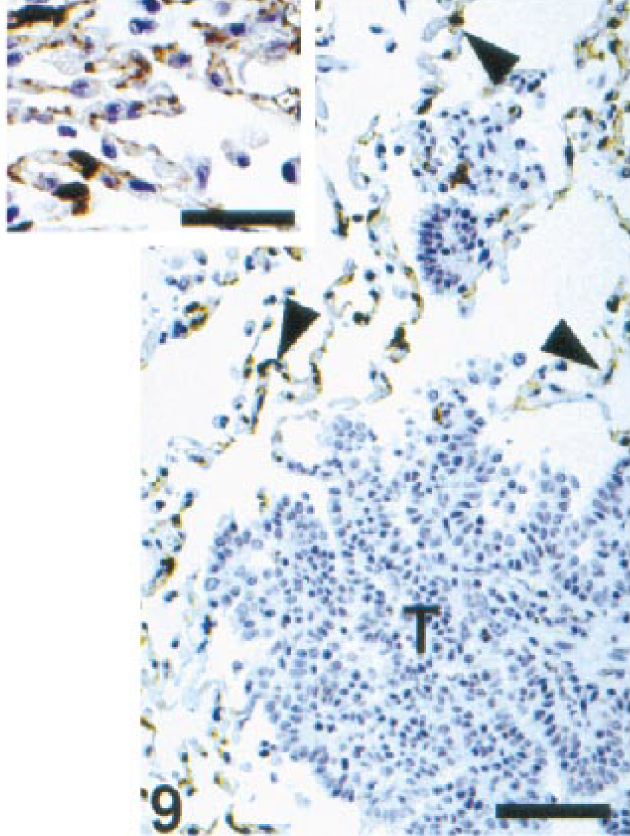
Lung; dog. Pulmonary adenocarcinoma. Neoplastic cells (T) do not react with Melan A antibody. Alveolar cells (arrowheads) are strongly positive for Melan A. ENVISION+ peroxidase stain with Mayer's hematoxylin counterstain. Bar = 153 µm. Inset: Detail of alveolar staining. Bar = 38 µm.
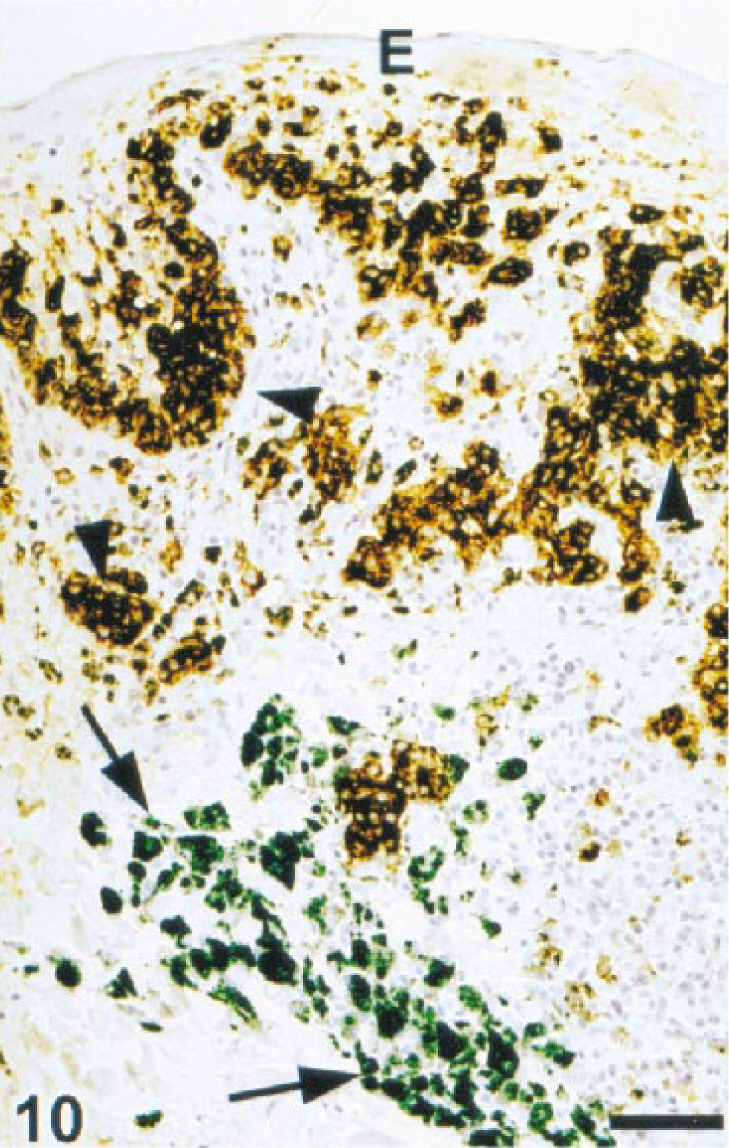
Gingiva; dog. Melanoma. Melan A-positive melanoma cells are dark brown when peroxidase and DAB are used (arrowheads). Melanin within some neoplastic cells and melanophages is blue-green after counterstaining with Azure B (arrows). ENVISION+ peroxidase stain with Azure B counterstain. Bar = 76 µm.
Immunostaining of melanomas with vimentin, S100 protein, and NSE
The staining was cytoplasmic for vimentin, cytoplasmic and nuclear for S100 protein, and cytoplasmic with occasional nuclear staining for NSE. All melanomas (100%) were positive for vimentin, 98 (76%) were positive for S100 protein, and 115 (89.1%) were positive for NSE. Of the 118 (91.5%) Melan A-positive cases, all were also positive for vimentin (100%), S100 protein (92 cases, 71.3%), and NSE (106 cases, 82.2%). Only 6/11 (54.5%) cases negative for Melan A were positive for S100. Nine of 11 (81.8%) cases negative for Melan A were positive for NSE. Five of 129 (3.8%) melanomas were negative for both Melan A and S100; 2/129 (1.5%) cases were negative for both Melan A and NSE. Twenty-six of 129 (20%) melanomas were positive for Melan A and negative for S100 protein, and 12/129 (9.3%) cases were positive for Melan A and negative for NSE. The agreement of positive Melan A immunohistochemical results with vimentin, S100 protein and NSE reactivity was 91.5, 73.6, and 83.7%, respectively. Eight of eight cases (including three clear cell and one adenoid/papillary tumors) were negative by immunohistochemistry for cytokeratins.
Effects of fixation on antigenicity
The stain for Melan A was strong with 1–7 days fixation, then diminished progressively as indicated by number of positive cells and intensity of reaction. However, positive cells were still found after 41 days of fixation (last day tested). For S100 protein, tissues fixed for 19 or 26 days reacted more strongly than tissues fixed for shorter periods. After 26 days, the staining intensity dropped dramatically, and staining was barely visible by 41 days of fixation. The staining for NSE was weak at 1 day fixation, strong at 6–26 days; it then declined slowly but was still fairly intense at day 41 of fixation. Tissues of an additional case fixed for 1 day were negative for NSE, whereas tissues fixed for 5, 14, and 20 days yielded moderate to strong reactivity. Staining for vimentin was weak at day 1, moderate at day 6, and strong through day 41 of fixation.
Discussion
Anatolian Sheepdog, Cocker Spaniel, Gordon Setter, mixed breed dogs (P < 0.05), Chow Chow, Golden retriever, Pekingese/Poodle cross, and miniature Poodle (P < 0.001) breeds were significantly overrepresented in the oral melanoma population. Although results obtained for Anatolian Sheepdog and mixed dog breeds were statistically significant, the power of the chi-square test (0.643 and 0.528, respectively) was below the desired power (0.800). Our results were in accordance with those of other reports that reveal a higher prevalence of oral melanomas in Cocker Spaniels, 10 16 29 56 small breeds, 21 29 56 and animals with pigmented mucosa. 10 29 56 However, there were discrepancies, such as the underrepresentation in our study of Boxers and German Shepherds (P < 0.05). In other studies, Boxers 16 and German Shepherds 56 were reportedly predisposed to oral melanomas.
In our series, melanoma was the second most common oral tumor, following squamous cell carcinoma, and was more common than acanthomatous epulis. These results are in accordance with previous reports, in which melanoma is one of the most commonly diagnosed tumors in the oral cavity. 7 9 10 16 19 29 56 Oral melanomas are tumors of adult/aging dogs. 7 10 16 21 29 56 The average age of presentation of oral melanomas in our study was 11.4 years (range 1–19 years). The average age for squamous cell carcinomas and acanthomatous epulis was 9.5 and 9.2 years, respectively. Also, sex distribution of oral melanomas was almost equal in our series, although in the general population of dogs, there were slightly more females (55%) than males (45%). This result contrasts with most reports of oral melanomas, in which males are significantly overrepresented. 7 10 29 56 However, one other paper reports equal representation of sexes in oral melanoma. 16 In our series, oral squamous cell carcinoma and acanthomatous epulis had a gender distribution similar to oral melanoma (50 and 52.6% were females, respectively). The location of the tumor in 107/122 oral melanomas was specified; the gingiva (35.2%) was the most common site. Other locations were the labial mucosa (32%), cheeks (7.4%), and tongue (5.7%). These results are in agreement with other reports. 7 10 16 19 29 56 The tongue and tonsils are uncommon locations for melanomas. 2 56 Interestingly, in our series, most of the gingival tumors in which the exact location (maxilla or mandible) was recorded were in the mandible (26/28, 93%). Other reports do not indicate significant differences between the upper and lower arcade. 10 56 Most oral melanomas are considered malignant, whereas most cutaneous melanomas are reported to be benign. 8 27 59 Some tumors have benign microscopic features but malignant biologic behavior. 8 Metastases of oral melanomas are frequent (14–74% of tumors), with regional lymph nodes and the lungs most commonly affected. 29 56 We documented 13 metastases microscopically, and another 7 metastases were reported by the referring veterinarians in the follow-up study. Although the percentage of metastatic melanomas in our study is within the expected range, we did not obtain information about metastasis in 63 (48.8%) of the cases. The prognosis for canine oral melanomas is poor 29 and unrelated to sex, location, 25 volume of the tumor, mitotic index, microscopic appearance, or amount of pigment. 8 25 In this respect, we did not find statistically significant differences in survival among different sites or mitotic indices. Reported survival time after surgical resection ranges from 3 to 45 months. 2 8 25 56 In our series, average survival time was 5.7 months. Bone invasion is frequent in some reports. 29 Bone tissue was not available for the oral melanomas examined.
The microscopic appearance of the oral melanomas in our study was similar to that reported, 27 with a preponderance of the mixed (polygonal and spindle) type. Other authors have reported that the epithelioid (polygonal) type is the most common cell type of oral melanoma. 21 Three of four cases consisted of large, clear cells, and one case had adenoid/papillary structures. To our knowledge, the adenoid/papillary type has not been reported in canine melanomas and the clear cell type has been previously reported only in cutaneous melanomas. 24 Some human melanomas have an adenoid/papillary pattern. 42 Two of our cases had chondroid differentiation with Melan A-positive cells close to cartilagenous cells. Chondroid differentiation is uncommon in cutaneous melanomas 22 24 and, to our knowledge, has not been reported in oral melanomas. Melanomas with osteoid formation have been described in human and animal cases. 14 22 39 Although this theory is not widely accepted, it has been suggested that melanoma cells can deposit osteoid. 39 We have observed heterotopic cartilage and bone in other nonosteogenic neoplasms. 46
Antibodies to tyrosinase and melanosomal proteins (e.g., HMB-45) are used for immunohistochemical diagnosis of human melanomas. 5 12 17 26 35 38 47 50 In our preliminary study, we did not find positive reactivity with antibody HMB-45/50. Also, we found that tyrosinase antibody reacted with other tissues. However, a combination of high temperature antigen retrieval and bleaching with potassium permanganate and oxalic acid reportedly restores immunostaining with HMB-45 in canine melanomas. 53 We did not test this novel procedure, and we used only Melan A as a melanoma-specific marker for the entire study. Melan A is involved in the recognition of melanomas by cytotoxic lymphocytes. 15 36 Our results indicate that canine oral melanocytes and melanoma cells, but not melanophages, react with antibody to Melan A. The percentage of canine melanomas stained with Melan A was similar to that reported in human melanomas, in which more than 80% of primary or metastatic tumors were positive. 11 13 17 35 In our study, there was complete agreement for Melan A immunostaining between primary tumors and their metastases, although the number of metastatic cases examined was too low. The pattern of staining with Melan A was granular diffuse, polar/punctate, and mixed (punctate and diffuse). In human melanomas, Melan A staining is usually granular and diffuse. 13 35
None of the other antibodies tested in our series except vimentin yielded a higher proportion of positive results than Melan A. Vimentin is a general marker for mesenchymal cells and consistently stains human melanomas, 42 although few can be negative. 3 38 Therefore, although vimentin is a sensitive marker, its specificity is low and it is suitable only for preliminary tumor screening (epithelial versus mesenchymal). The percentage (76%) of melanomas positive for S100 in our study was similar to that in other reports. 14 44 48 49 S100 is present in practically all human melanocytic lesions. 47 However, S100 protein is also demonstrated in a variety of nonmelanocytic cells, including glial cells, neurons, Schwann cells, Langerhans cells, macrophages, myoepithelium, and chondrocytes. 47 54 NSE is a protein in neurons, peripheral and central neuroendocrine cells, and melanocytes and tumors derived from them, 47 which makes the specificity of this marker for melanomas very low. In our experience (results not included), the NSE antibody used also consistently stains smooth muscle. Although the staining with NSE in many occasions was diffuse and involved many neoplastic cells, it was dull and sometimes difficult to distinguish from background staining.
Antibody to Melan A (clone A103) reacts with human adrenal cortical cells, ovarian granulosa cells, and testicular Leydig cells and their neoplastic counterparts. 11 33 By molecular analysis, no Melan A RNA exists in these tissues, so the antigens that bind anti-Melan A in these organs most likely represent immunologically cross-reacting molecules. 33 We observed a similar staining pattern with Melan A in normal and neoplastic canine adrenal, ovarian, and testicular tissues (data not included). In addition, and not previously reported in human literature, salivary gland and pancreatic ducts, airway and alveolar epithelium of the lung, prostatic epithelium, and transitional epithelium of the urinary bladder were variably positive. Whether the epitope recognized by anti-Melan A in these tissues was Melan A antigen or a cross-reacting antigen with similar amino acid sequence was not determined. Four nonmelanocytic tumors were weakly reactive for melan A, including 2/8 salivary adenocarcinomas and 2/10 transitional carcinomas. The likelihood of confusing melanoma with these two tumor types is low. In addition, immunostaining with keratin and vimentin antibodies should help differentiate these tumors. Immunohistochemistry for various melanoma-associated antigens has also been used in canine melanomas, with a positive reaction in not more than 70% of cases. 4 43 Using a combination of antibodies increased the percentage of positive tumors to 83%. 4 Unfortunately, these antibodies stained a variety of nonmelanocytic tumors including squamous cell carcinomas, mammary neoplasms, epulides, lymphosarcoma, 43 granular cell tumor, plasmacytoma, 4 and basal cell tumors. 4 43 In our study, these nonmelanocytic tumors were consistently negative for Melan A, which was not evaluated in the previous studies. One report also demonstrates staining of salivary gland epithelia, although it does not indicate the type of cell stained. 4
It is usually accepted that antigenicity of many proteins is lost progressively during fixation. 55 In our study, prolonged fixation moderately reduced the intensity and number of cells stained with Melan A after the first week. Although duration of fixation was not known for most specimens, we presumed that most samples were processed within 1 week of fixation; therefore, fixation should not have significantly influenced Melan A reactivity. On the other hand, staining for NSE and S100 protein (and to a lesser extent for vimentin) was weak or nonexistent in tissues fixed for less than 1 week. This finding might explain, at least in part, the lower number of positive cases obtained with NSE and S100 protein when compared with Melan A but probably did not affect vimentin reactivity (100% of positive cases).
Our findings indicate that 1) Cocker Spaniels, miniature Poodles, Pekingese/Poodles, and Chow Chows are predisposed to oral melanomas, and Boxers and German Shepherds are not predisposed; 2) the most common location of oral melanoma is the gingiva; 3) survival time is not related to mitotic index or tumor location; 4) Melan A is a highly sensitive and specific marker for melanocytic tumors.
Footnotes
Acknowledgements
We thank all referring veterinarians who contributed epidemiologic information in this report and Howard Wilson for assistance in preparation of illustrations.
