Abstract
Osteosarcoma (OS), the most common bone tumor, includes OS of the head (OSH) and appendicular OS (OSA). In dogs, it is classified into 6 histologic subtypes: osteoblastic, chondroblastic, fibroblastic, telangiectatic, giant cell, and poorly differentiated. This study investigated the significance of the histologic classification relevant to clinical outcome and the histologic and immunohistochemical relationships between pleomorphism and expression of cytoskeletal proteins in 60 cases each of OSH and OSA. Most neoplasms exhibited histologic diversity, and 64% of OS contained multiple subtypes. In addition to the above 6 subtypes, myxoid, round cell, and epithelioid subtypes were observed. Although the epithelioid subtypes were observed in only OSH, no significant difference in the frequency of other subtypes was observed. Also, no significant relevance was observed between the clinical outcome and histologic subtypes. Cytokeratin (CK) was expressed in both epithelioid and sarcomatoid tumor cells in various subtypes, and all CK-positive tumor cells also expressed vimentin. Vimentin and α-smooth muscle actin (SMA) were expressed in all subtypes. A few SMA-positive spindle-shaped tumor cells exhibited desmin expression. Glial fibrillary acidic protein–positive tumor cells were observed in many subtypes, and some of these cells showed neurofilament expression. Although OSH exhibited significantly stronger immunoreactivity for SMA than OSA, no significant difference in other cytoskeletal proteins was observed. Some tumor cells had cytoskeletal protein expression compatible with the corresponding histologic subtypes, such as CK in the epithelioid subtype and SMA in the fibroblastic subtype. Thus, canine skeletal OS is composed of pleomorphic and heterogenous tumor cells as is reflected in the diversity of histologic patterns and expression of cytoskeletal proteins.
Osteosarcoma (OS) is the most common primary malignant bone neoplasm of dogs, 39,41 and it shows aggressive local invasiveness and highly metastatic behavior. 5 Several clinical and histologic criteria have been reported as prognostic factors, including breed, tumor location, metastases to the lungs, completeness of surgical margins, adjuvant chemotherapy, and histologic subtype.* Between 20% and 25% of canine skeletal OS originates from bones of the axial skeleton. 41 Of these, approximately 50% of axial skeletal OS occurs in the head and 50% in the ribs, vertebrae, and pelvis. 41 OS of the head (OSH) is a comparatively rare tumor that has been reported in the mandible, maxilla, skull, and paranasal sinuses in dogs. 41 The appendicular skeleton is affected 3 to 4 times more often than the axial skeleton and most often in the metaphyseal region of long bones. 41 The proximal humerus and the distal radius in the forelimb—along with the distal femur, proximal tibia, and distal tibia in the hindlimb—are the common sites of OS of the appendicular skeleton (OSA). 41
OS within the medullary cavity of bone is histologically classified into conventional OS, telangiectatic OS, small cell OS, and low-grade central OS, according to the WHO Classification of Tumours of Soft Tissue and Bone in human medicine. 17,31,33 Conventional OS has a broad spectrum of morphology and is divided into 8 histologic subtypes: osteoblastic, chondroblastic, fibroblastic, giant cell-rich, osteoblastoma-like, epithelioid, clear cell, and chondroblastic. 33 Many immunohistochemical examinations have been performed to characterize human OS. OS has a broad immunoprofile that lacks diagnostic specificity, and the commonly expressed antigens include osteocalcin, osteonectin, S-100 protein, actin, α-smooth muscle actin (SMA), neuron-specific enolase, and CD99. 33 Importantly, as it is a diagnostic pitfall, OS may also express cytokeratin (CK) and EMA detected using immunohistochemistry. 33
OS is defined as a primary malignant bone tumor that arises in the medullary cavity, with production of osteoid and/or immature bone by malignant osteoblasts. 39,41 Canine OS shows various morphologies and arrangements of neoplastic cells, and it is divided into 6 histologic subtypes according to the World Health Organization’s histologic classification of tumors of domestic animals: osteoblastic, chondroblastic, fibroblastic, telangiectatic, giant cell, and poorly differentiated. 39 These subtypes are frequently mixed in the same neoplasm. 41 Immunohistochemical studies of canine OS using antibodies for p53, β-catenin, survivin, heat shock protein, epidermal growth factor receptor, and COX-2 have been performed to try to identify prognostic markers. 4,25,26,36 –38,40 However, no immunohistochemical analysis of cytoskeletal elements with correlation of the results to histologic subtype has been done for canine OS.
For this study, the histologic classification system of canine skeletal OS was compared to the clinical outcome. Also, we determined whether there was an association between the histologic subtypes of canine OS and immunohistochemical expression of the cytoskeletal proteins CK, vimentin, SMA, desmin, glial fibrillary acidic protein (GFAP), and neurofilament (NF).
Material and Methods
Animals and Clinical Data
Biopsy were collected from 120 dogs (60 OSH and 60 OSA). Clinical data of these dogs were evaluated, including age, sex, breed, and affected bone. Of these 120 dogs, 95 were lost to follow-up, and a definite date of death was obtained for the remaining 25 dogs. These dogs, including 12 with OSH and 13 with OSA, were used to calculate the survival times, and were evaluated for metastases, chemotherapy, surgical margin, and histologic subtype.
Histology
Biopsy samples were removed by mandibulectomy, maxillectomy, limb amputation surgery, incisional biopsy, and needle biopsy. They were fixed in 10% neutral formalin, embedded in paraffin, cut at 4-µm thickness, and stained with hematoxylin and eosin. The neoplasms, including osseous tissues, were decalcified using 10% formic acid after fixation.
Immunohistochemistry
Histologic sections of OS were immunohistochemically examined using the avidin-biotin-peroxidase complex (ABC) procedure (Vectastain Elite ABC Kit; Vector Laboratories, Burlingame, CA) on paraffin sections. To remove endogenous peroxidase, sections were immersed in 0.5% periodic acid solution at room temperature for 10 minutes. The sections were incubated with primary antibodies at 4°C for 16 hours, with biotinylated secondary antibody for 30 minutes at room temperature, and with ABC for an additional 30 minutes at room temperature. Visualization was accomplished using 0.05% 3,3′-diaminobenzidine solution. Mayer’s hematoxylin stain was used as a counterstain. Details of primary antibodies used in the present study are summarized in Supplemental Table 1. Immunoreactivity of the neoplasm cells was classified into 4 grades, according to whether 0%, 1%–25%, 26%–50%, or 51%–100% of neoplastic cells showed immunolabeling (allocated scores of 0, 1, 2, and 3, respectively).
Statistical Analysis
The survival times and prevalence of pulmonary metastasis were compared between OSH and OSA using the Kaplan Meier method, log-rank test, χ2 test, and Fisher exact probability test. The prognostic significance of chemotherapy and surgical margins were analyzed using the Kaplan-Meier method. The association between the survival times or pulmonary metastasis and the frequency of each histologic subtype was investigated using the Kaplan Meier method, log-rank test, χ2 test, and Fisher exact probability test.
The proportions of each subtype between OSH and OSA were compared using the χ2 test or Fisher exact probability test. Spearman rank correlation coefficient by rank test was used to investigate the potential correlation among the immunoreactivity scores of each of the cytoskeletal proteins. Also, immunoreactivity scores from each cytoskeletal protein among subtypes were compared using the Kruskal-Wallis test and Mann–Whitney U test. Furthermore, the relationships of immunoreactivity of each cytoskeletal protein between OSH and OSA were compared using the Mann–Whitney U test. A P value <0.05 was considered statistically significant.
Results
Clinical Data
The mean age of the dogs examined was 10 years (range, 6 weeks–18 years). There was a female:male ratio of 1.1:1, including 63 females and 57 males. Thirty breeds were represented, the most common ones being mongrels (31%), Golden Retrievers (13%) and Labrador Retrievers (10%). In the OSH cases, tumors were in the maxilla (22%), mandible (47%), and zygomatic bone (3%), while in the appendicular cases, OSA were in the scapula (15%), humerus (27%), radius (2%), ulna (5%), carpal bones (3%), phalanges of the manus (3%), femur (12%), and tibia (2%).
The survival data of 25 dogs, including 12 with OSH and 13 with OSA, are summarized in Supplemental Tables 2 and 3. The median ST of OSH was 525 days (range, 117–1486 days), and that of OSA was 238 days (range, 40–466). Metastases to the lungs were observed in 2 OSH and 10 OSA. Seven dogs received adjuvant chemotherapy using carboplatin, and the surgical margins of 3 dogs were dirty (1 OSH, 2 OSA).
Histologic Findings
The neoplasms were classified into 6 subtypes depending on the histologic classification of domestic animals. In some OSH and OSA cases, the neoplasms could not be classified into any of these 6 types, but were instead subclassified into myxoid, round cell, and epithelioid subtypes. Although the classification of subtype was determined depending on the predominant histologic findings, the neoplasms were categorized as a mixed subtype if multiple subtypes were present and a single subtype was not predominant. The frequency of the histologic subtypes of OSH and OSA is summarized in Supplemental Table 4.
In OSH, the neoplasms were composed of very pleomorphic tumor cells, and 63% of neoplasms were categorized as mixed subtypes since they contained multiple histologic subtypes. In the osteoblastic subtypes found in 45 OSH, the polyhedral to spindle tumor cells produced osteoid with calcification and formation of immature bone trabeculae (Fig. 1). In the chondroblastic subtypes found in 15 OSH, the polyhedral tumor cells produced hyaline cartilage (Fig. 2). In the fibroblastic subtypes found in 20 OSH, the spindle-shaped tumor cells were arranged in a wavelike fashion or a storiform pattern (Fig. 3). In the telangiectatic subtypes found in 16 OSH, the polyhedral tumor cells formed many large or small, blood-filled or empty cystic spaces, and the septa exhibited a variable thickness (Fig. 4). In the giant cell subtypes found in 14 OSH, the aggregated nests of multinucleate giant cells were sporadically present, mixed in with the components of other subtypes (Fig. 5). In the poorly differentiated subtypes found in 7 OSH, the neoplasms consisted of malignant mesenchymal cells exhibiting various morphologies, such as large pleomorphic cells and small cells resembling the reticular cells of bone marrow (Fig. 6). In the myxoid subtypes found in 6 OSH, the spindle-shaped tumor cells were loosely arranged in an abundant myxoid matrix (Fig. 7). In the round cell subtypes found in 6 OSH, the neoplasms consisted of round tumor cells with scant cytoplasms and round to oval nuclei, and the tumor cells proliferated in a sheet with the production of lacelike osteoid (Fig. 8). In the epithelioid subtypes found in 5 OSH, the tumor cells showed cytologic features such as large polyhedral morphology, a large nucleus, abundant eosinophilic cytoplasm, and shhet-like arrangment of cells (Fig. 9).
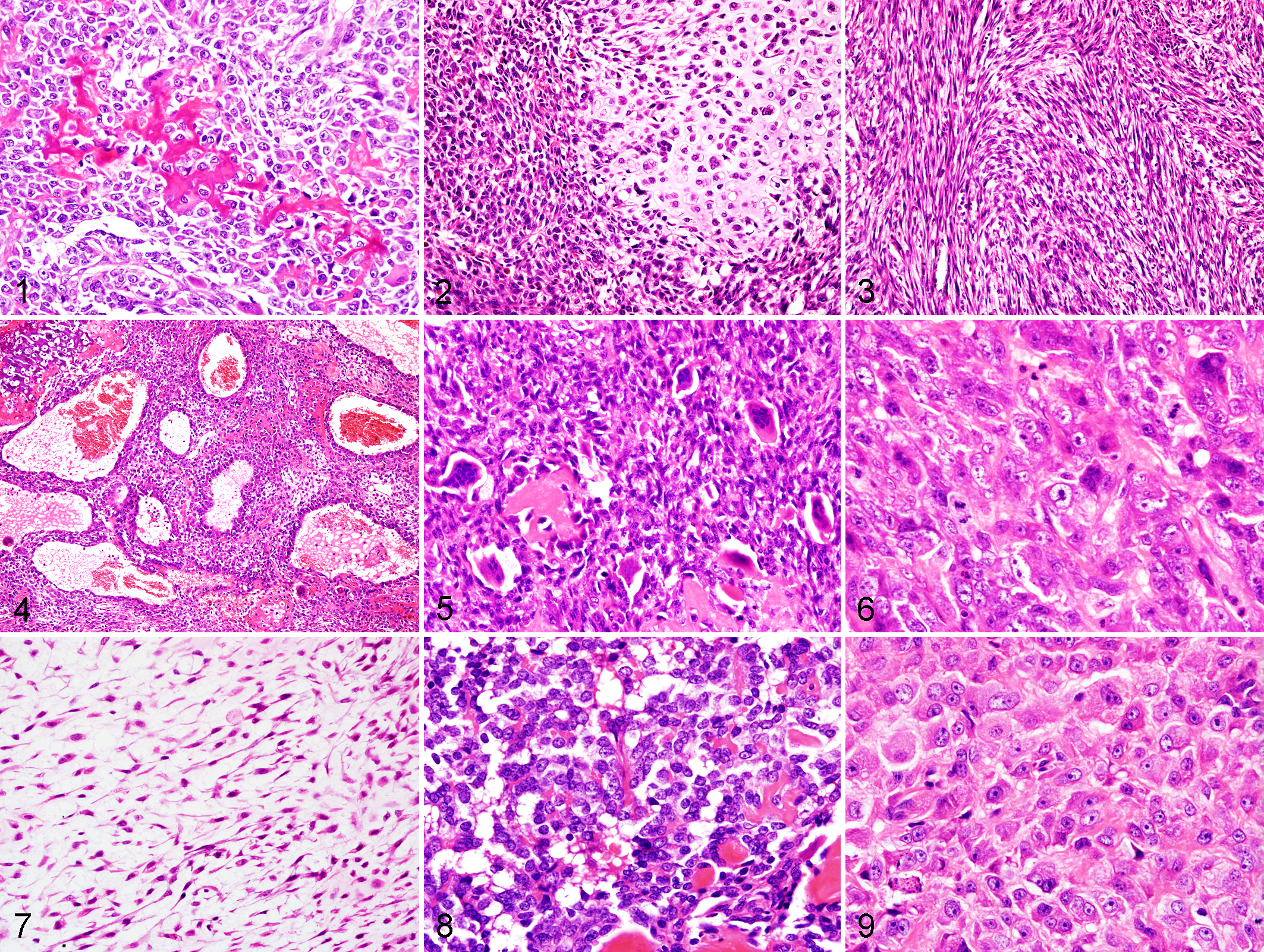
In OSA, 65% of neoplasms were categorized as mixed subtypes since they contained multiple histologic subtypes as with OSH. Subtypes observed in OSA included osteoblastic (n = 44), chondroblastic (n = 16), fibroblastic (n = 20), telangiectatic (n = 16), giant cell (n = 13), poorly differentiated (n = 7), myxoid (n = 3), and round cell (n = 8). The epithelioid subtype was not detected in OSA. The histologic features of each subtype of OSA were similar to those of OSH (Fig. 1–8), and significant differences in the frequency of each subtype were not observed between OSH and OSA.
In 25 dogs in which the survival data were available, 7 subtypes were found in OSH—including osteoblastic (n = 11), chondroblastic (n = 3), fibroblastic (n = 6), telangiectatic (n = 1), poorly differentiated (n = 1), myxoid (n = 1), and epithelioid (n = 1)—while 6 subtypes were found in OSA: osteoblastic (n = 11), chondroblastic (n = 6), fibroblastic (n = 6), telangiectatic (n = 1), poorly differentiated (n = 4), and myxoid (n = 1).
Immunohistochemical Findings
Both OSH and OSA showed various relationships between hitologic subtypes and immunohistochemical detection of cytoskeletal proteins. Immunohistochemical results of the OSH and OSA are summarized in Table 1.
Expression of Cytoskeletal Proteins for Each Subtype of Osteosarcoma (Percentage of Cases).
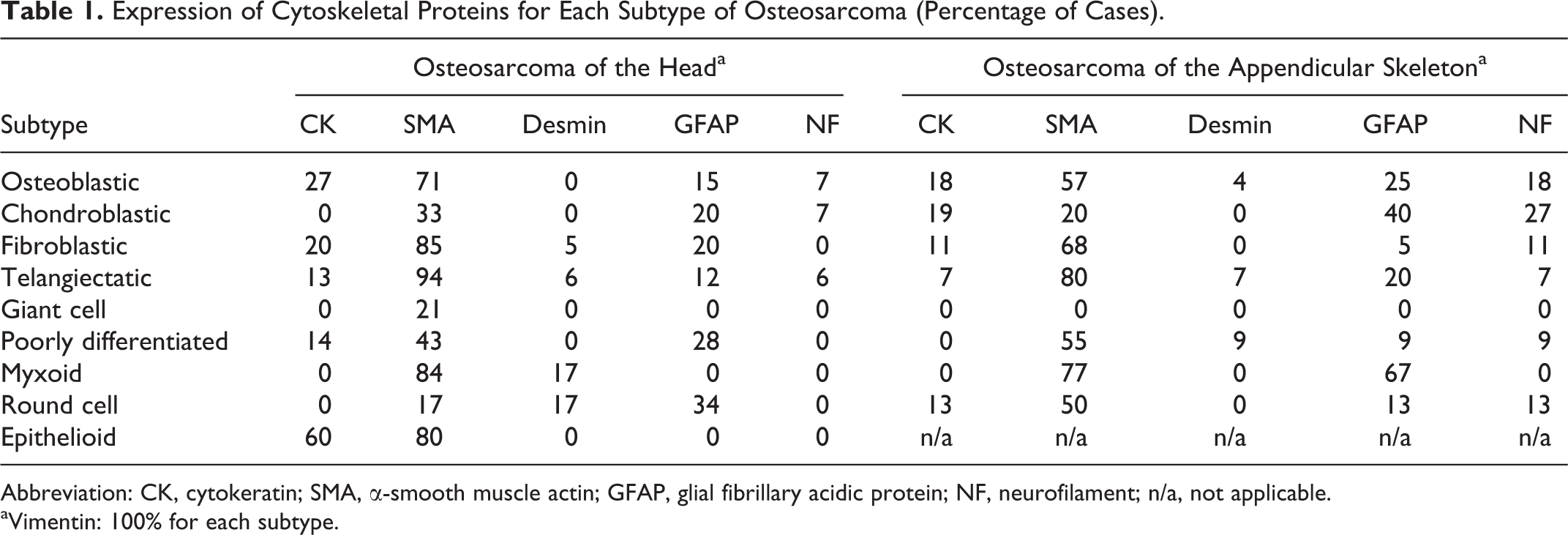
Abbreviation: CK, cytokeratin; SMA, α-smooth muscle actin; GFAP, glial fibrillary acidic protein; NF, neurofilament; n/a, not applicable.
aVimentin: 100% for each subtype.
In OSH, CK-positive tumor cells were present in 27% of osteoblastic (Fig. 10a), 20% of fibroblastic, 13% of telangiectatic, 14% of poorly differentiated, and 60% of epithelioid subtypes (Fig. 13a). All CK-positive tumor cells were immunoreactive for vimentin, and some of them were also immunoreactive for SMA (Figs. 10b, 13b). SMA-positive tumor cells were present in 71% of osteoblastic (Fig. 10b), 33% of chondroblastic, 85% of fibroblastic (Fig. 11b), 94% of telangiectatic (Fig. 12b), 21% of giant cell, 43% of poorly differentiated, 84% of myxoid, 17% of round cell, and 80% of epithelioid subtypes (Fig. 13b). Some of the SMA-positive tumor cells were also immunoreactive for desmin (Figs. 11c, 12c).
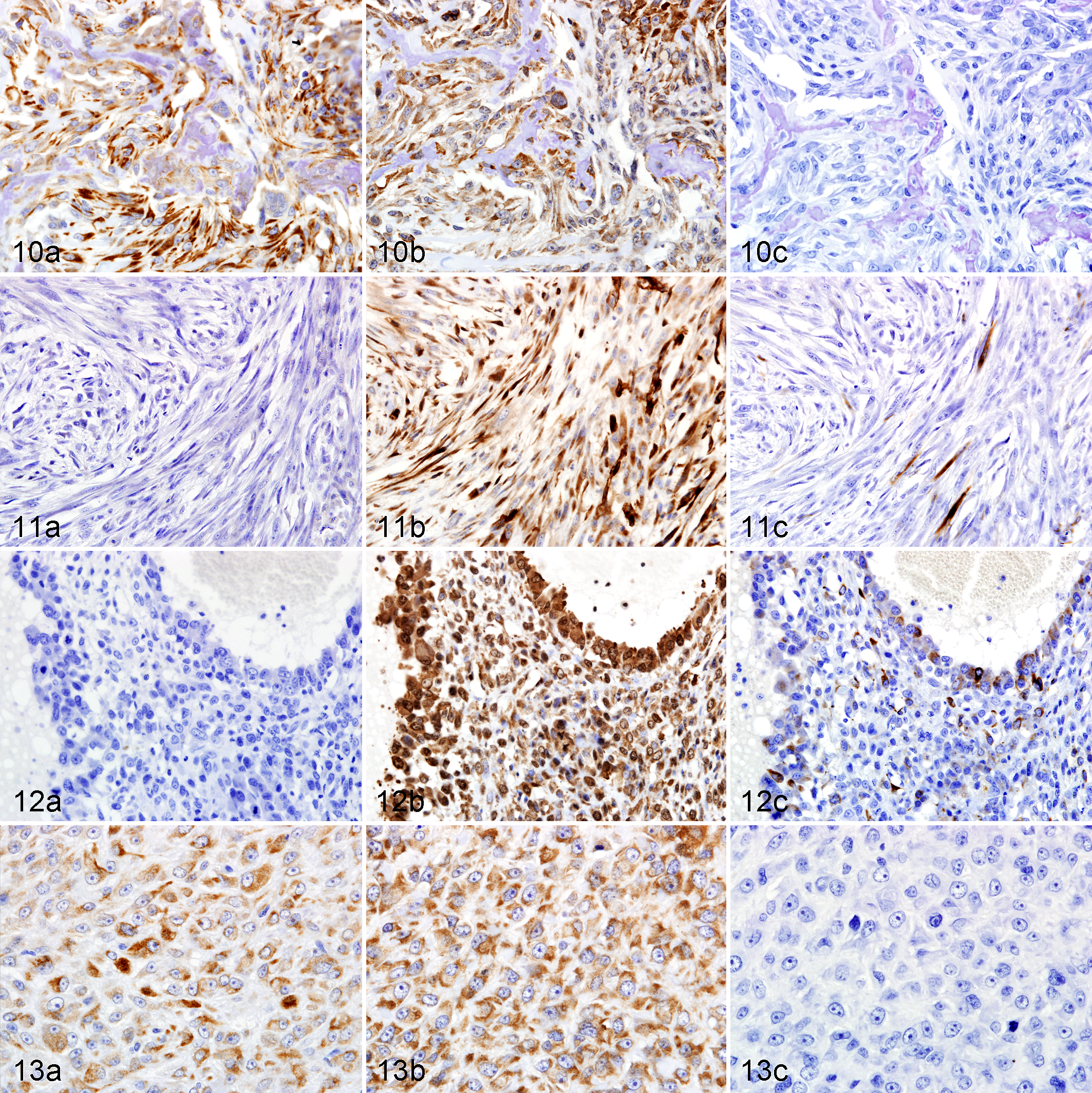
In OSA, GFAP-positive tumor cells were present in 25% of osteoblastic, 40% of chondroblastic, 5% of fibroblastic, 20% of telangiectatic, 9% of poorly differentiated, 67% of myxoid, and 13% of round cell subtypes. Some of the GFAP-positive tumor cells were also immunoreactive for NF. NF-positive tumor cells were present in 18% of osteoblastic, 27% of chondroblastic, 11% of fibroblastic, 7% of telangiectatic, 9% of poorly differentiated, and 13% of round cell subtypes.
Statistical Analysis
The survival times of OSH were significantly longer than those of OSA (P < 0.05), and prevalence of pulmonary metastasis was significantly higher for OSA than for OSH (P < 0.05). The survival times were not different regardless of whether chemotherapy was received or surgical margins were dirty (P > 0.05). No significant association was observed between the survival times or prevalence of pulmonary metastasis and the frequency of each histologic subtype (P > 0.05).
Although no significant difference between OSH and OSA was observed in the frequency of each histologic subtype (P > 0.05), the epithelioid subtype was detected only in OSH. Immunoreactivity for CK was significantly stronger for, the osteoblastic and epithelioid subtypes than for other subtypes (P < 0.05). Immunoreactivity for SMA was significantly stronger for the fibroblastic and telangiectatic subtypes than for other subtypes (P < 0.05), and the immunoreactivity of SMA of OSH was significantly stronger than SMA of OSA (P < 0.05). For cytoskeletal proteins other than CK and SMA, a significant difference in immunoreactivity was not observed among subtypes (P > 0.05). Also, except for SMA, significant differences in immunoreactivity for cytoskeletal proteins between OSH and OSA (P > 0.05). The immunoreactivity of NF and GFAP was significantly correlated (P < 0.05).
Discussion
The clinical data, including the age, sex, and breed, were comparable to those of previous studies. 11,20,23,24,27,41 The mandible was affected more than twice as often as the maxilla in OSH. Although the prevalence in the scapula and humerus was higher than in previous studies, the prevalence in the ulna and radius was lower. 16,20,21,27,41 Significant differences of the survival times and the frequency of pulmonary metastasis indicate that OSH shows a better prognosis than OSA. This finding supports previous studies that the survival times or metastases with OS of the axial skeleton are marginally better than those with appendicular tumors. 11,16,24,41
Canine skeletal OS, including OSH and OSA, were composed of pleomorphic and heterogenous tumor cells, and 64% of OSH and OSA contained multiple histologic subtypes in the same neoplasm. The mixed subtypes were more frequent than those of the previous studies. 11,20,23,24,27 Canine skeletal OS is considered to originate from multipotential mesenchymal cells in the medullary cavity, and these results suggest that this origin leads to its histologic diversity and heterogeneity. A significant difference between OSH and OSA in the proportion of each subtype was not observed indicating that canine skeletal OS may have diverse morphology regardless of the involved bone.
No correlation was observed between the survival times or prevalence of pulmonary metastasis and the frequency of each histologic subtype. Although several previous studies reported that fibroblastic OS has a relatively favorable prognosis and the telangiectatic OS has a very poor prognosis, 11,27 other studies including the present study concluded that the histologic subtypes were not associated with prognosis. 20,23 To evaluate the usefulness of histologic classification as a prognostic indicator for canine OS, the accumulation of data from large numbers of cases and the construction of new histologic classification system that evaluates the mixed subtype and the new subtypes may be warranted.
We classified 3 histologic subtypes—myxoid, round cell, and epithelioid—in addition to the subtypes previously used in domestic animals. The myxoid subtype was not observed as a component of chondroblastic OS, but instead was a predominant histologic subtype. This finding does not concur with previous reports that the myxoid morphology occasionally appears in a part of chondroblastic OS in humans. 33 The round cell subtype consisted of small round tumor cells similar to those described in a report of canine small cell osteogenic sarcoma. 10 Some OSH exhibited epithelioid features in the present study. In human medicine, small cell and epithelioid OS have been classified on the basis of their histologic features but these have not been described in canine OS. 17,33 Although it will be necessary to study more of these cases, we consider that the myxoid, round cell, and epithelioid subtypes should be distinguished as separate histologic subtypes in canine skeletal OS, because they exhibited predominant histologic features in some neoplasms.
Immunohistochemically, although the intermediate filaments have been popularly known as useful markers to understand the property of tumor cells and to diagnose their origin, some neoplasms show relationships between patterns of intermediate filament expression and histologic patterns. In general, intratumor heterogeneity –in which neoplasms consist of areas with different phenotypes–has been recognized for many years. 9 In the present study of canine skeletal OS, the expression of various cytoskeletal proteins including CK, vimentin, SMA, desmin, GFAP, and NF had some conformity with histologic morphology. We consider that these immunohistochemical characteristics are attributable to tumor heterogeneity of canine skeletal OS.
The tumor cells of both OSH and OSA showed expression of CK in the osteoblastic, chondroblastic, fibroblastic, telangiectatic, poorly differentiated, and round cell subtypes, but CK expression was especially prevalent in the epithelioid subtype. All CK-positive tumor cells coexpressed vimentin. These results revealed that epithelioid tumor cells have a tendency to express CK, but retain expression of mesenchymal cytoskeletal protein (vimentin). Recently, there have been several case reports of epithelioid OS in human medicine, with or without the expression of CK. † Okada et al reported that the expression of CK had been observed in some tumor cells of osteoblastic, chondroblastic, and fibroblastic OS with epithelioid features. 29 Hasegawa et al reported that osteoblastic and epithelioid OS showed the coexpression of CK and vimentin. 12,13 These reports support the view that canine OS also has the potential to exhibit epithelioid morphology and express CK. Furthermore, the results of the present study suggest that canine OS shows more histologic and immunohistochemical diversity than human OS. Specifically, unlike human OS, CK expression was detected in various histologic subtypes of canine OS including poorly differentiated and telangiectatic subtypes. To the best of our knowledge, the present study is the first report of canine skeletal OS with an epithelioid morphology and with expression of CK.
SMA-positive tumor cells were observed in all subtypes. In particular, the fibroblastic and telangiectatic tumor cells showed significantly stronger immunoreactivity for SMA than other subtypes, and OSH exhibited significantly stronger immunoreactivity for SMA than OSA. A few desmin-positive tumor cells were observed in the osteoblastic, fibroblastic, telangiectatic, poorly differentiated, and myxoid subtypes, and these tumor cells correspondingly showed immunoreactivity for SMA. Most SMA-and desmin-positive tumor cells exhibited a spindle-shaped and fibroblast-like morphology, and some of them showed the expression of CK and vimentin. Although it has been reported that OS with various histologic subtypes had immunoreactivity for SMA and desmin in human medicine, 13,15,42 statistical analysis concerning the correlation between the expression of cytoskeletal proteins and its histologic subtype has not been performed. The results of the present study suggest that SMA- and desmin-positive tumor cells, especially those of the fibroblastic and telangiectatic subtypes of OSH, have the property of myofibroblasts or smooth muscle cells. Although it is unclear why SMA expression of OSH was stronger than OSA, the difference in the origin of the primary bone of OS may be one of the factors.
GFAP-positive tumor cells were observed not only in the osteoblastic and chondroblastic subtypes but also in fibroblastic, telangiectatic, poorly differentiated, myxoid, and round cell subtypes. In addition, NF-positive tumor cells were observed in osteoblastic, chondroblastic, fibroblastic, telangiectatic, poorly differentiated, and round cell subtypes. Kasantikul and Shuangshoti reported that, in humans, mature osteocytes and chondrocytes had cytoskeletal intermediate filaments identical to or associated with GFAP. 18 Also, osteoblast-like tumor cells reportedly showed immunoreactivity for GFAP in human gliosarcoma with differentiation of osteoblastic and chondroblastic OS, 1,2 while human osteoblastic and chondroblastic OS exhibited expression of GFAP. 35 OS with the expression of NF has not been reported in both humans and dogs. The present study revealed that GFAP was preserved in canine OS not only with osteocartilaginous differentiation but also with other histologic morphologies.
In conclusion, canine skeletal OS is composed of highly pleomorphic and heterogenous tumor cells, and frequently shows various histologic subtypes in the same neoplasm. Some are of myxoid, round cell, or epithelioid subtype that are not included in the current classification of OS in domestic animals. Histologic classification into subtypes does not appear to be a useful prognostic indicator. No significant difference was observed between OSH and OSA with respect to the proportion of each subtype or the expression of cytoskeletal proteins other than SMA. The heterogeneity of canine skeletal OS is reflected in their expression of various cytokeletal proteins, and the potential to differentiate into epithelioid cells and various mesenchymal cells such as myofibroblasts and smooth muscle cells.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
