Abstract
Seventy-seven feline transplant kidney specimens, obtained from 1 to 3,183 days (9 years) after transplantation, were reevaluated histologically and classified on the basis of the Banff '97 guidelines for human renal transplant kidneys. Overall, this classification system appeared useful in detecting rejection reactions and confirmed the finding in humans that biopsies can diagnose subclinical rejection and therefore are an important diagnostic tool for the follow up of renal transplants. However, on the basis of serum creatinine values, the severity of the acute or active and chronic lesions was not accurately reflected by this scoring system. This is thought to be due to the significant differences in histologic rejection patterns, especially in acute or active rejection, in cats when compared with humans. Tubulitis, lymphocytic glomerulitis, and vasculitis, which are the main pillars of the Banff '97 acute or active rejection scoring system, are either rare or not found in cats. The presence of significant necrotizing glomerulitis and vasculitis in feline renal transplants might imply that the rejection is complicated by acute antibody-mediated rejection. Alternatively, cyclosporine toxicity also should be considered because some of these kidneys show other signs of cyclosporine toxicity. Finally, the significance of subcapsular and interlobular phlebitis, rarely described in human rejection reactions but a distinct entity in cats, is unknown. From this study, it is clear that there are significant differences in the histology of acute or active rejection between humans and cats and that a better understanding of the histologic appearance of renal allografts will be especially beneficial for treatment and prognostic purposes.
Renal transplantation has become an established treatment for end-stage kidney disease in cats. 15,16 In human renal transplant patients, histologic assessment of renal biopsies remains the gold standard for the diagnosis of acute and chronic renal allograft rejection. 18,23 In the 50-year history of human renal transplantations, it has become clear that standardization of renal allograft biopsy interpretation is necessary to guide therapy in transplant patients and to establish an objective end point for clinical trials of new antirejection agents. 4,18,23,28 The classification most commonly used in human renal transplant pathology originated in a meeting held in Banff, Canada, in 1991. 28 Since then, the classification has been updated regularly. 1,4,18,22–24 The latest detailed Banff '97 working classification includes information based on the analysis of data from clinical trials and observation of clinical practice in use for the classification worldwide. 23 Although renal transplantations in cats have been performed for at least a decade, surprisingly, there are neither descriptions of the microscopic appearance of renal allografts in cats nor is there any guideline for the interpretation of renal transplant histology.
At the University of California, Davis, we have performed 166 renal transplants in cats since 1987 and have collected a unique repository of 77 transplant kidney specimens ranging from 1 to 3,183 days (9 years) after transplantation. In this study, the transplant renal material was reevaluated histologically and classified on the basis of the Banff '97 guidelines for human renal transplant kidneys. The aims of this study were to evaluate whether the Banff '97 scheme adequately describes the microscopic picture observed in cats and to relate the Banff '97 scores and diagnostic categories to the clinical features observed at the time the specimen was collected. This study provides valuable information on the histologic lesions in biopsy specimens from cat kidney transplants and hopes to encourage the appropriate use of biopsies, which will improve the outcome and management of feline kidney transplant patients in the future.
Materials and Methods
Specimens
Seventy-seven renal allograft specimens were obtained from 64 cats; 51 cats had a single specimen, and 13 cats had two specimens. Renal allograft specimens were obtained by renal punch biopsy in 18% of cats (14 of 77), and the entire transplanted kidney was available in 82% (63 of 77) of the specimens. Because it has been shown in humans that in transplant rejection one biopsy is representative of the whole kidney, we decided to include renal punch biopsies in this study. 5,20 For histologic evaluation, 2-µm-thick sections were made from all available tissue blocks and stained with hematoxylin and eosin and periodic acid–Schiff (PAS).
Graft recipients
All cats involved in this study had end-stage kidney disease but had no other organic or infectious disease at the time of surgery. The cats received the transplant kidneys from young, healthy colony or shelter-source cats. Various surgical techniques for both vessels and ureter were used. Owners of the recipients adopted the donors. Postoperatively, the cats were maintained on an immunosuppressive protocol of cyclosporine, either oil-based formulation (Sandimmune) or microemulsified formulation (Neoral)—target trough concentration (C12) of 500 ng/ml for 1 month, then 300 ng/ml combined with prednisone. Cyclosporine was administered orally twice daily. The clinical background information is summarized in Table 1. Breeds of the cats included Domestic Shorthair (47 cats), Domestic Longhair (11), Domestic Mediumhair (4), Siamese (6), Siamese cross breed (4), Himalayan (2), and Abyssinian, Manx/Siamese cross, and Persian cross (1 each). The median age of the cats was 9 years (range 1–17). The mean survival time of the renal allograft was 270 days (± 532 days; minimum 1 day, maximum 3,183 days). On the basis of clinical data at the time of biopsy/necropsy, 34 cats had azotemia as their only diagnosis. Other clinical diagnoses included sepsis secondary to Mycobacterium avium, Toxoplasma gondii, or Actinobacillus spp. (nine cats); neoplasia (seven cats); and miscellaneous causes (eight cats). Nineteen cats died in the immediate postoperative period, associated with neurologic signs of undetermined etiology (five cats), renal torsion (three), hemoabdomen (four), renal artery thrombosis (two), ureteral obstruction (two), and cardiac arrest (three). Both BUN and creatinine concentrations were measured at the time of biopsy/necropsy in 49 cats, creatinine concentration alone was measured in four cats, and three cats were reported to have normal renal parameters, although the values were not available; 21 cats did not have renal parameters measured at the time of biopsy/necropsy. The mean (± SD) creatinine concentration in blood was 4.8 mg/dl (± 2.4 mg/dl; minimum 1.2 mg/dl, and maximum 10.4 mg/dl), and the mean BUN value was 97.1 mg/dl (± 72.9 mg/dl; minimum 15.7 mg/dl, and maximum 277.0 mg/dl).
Overview of the clinical information of the 64 cats used in this study.∗
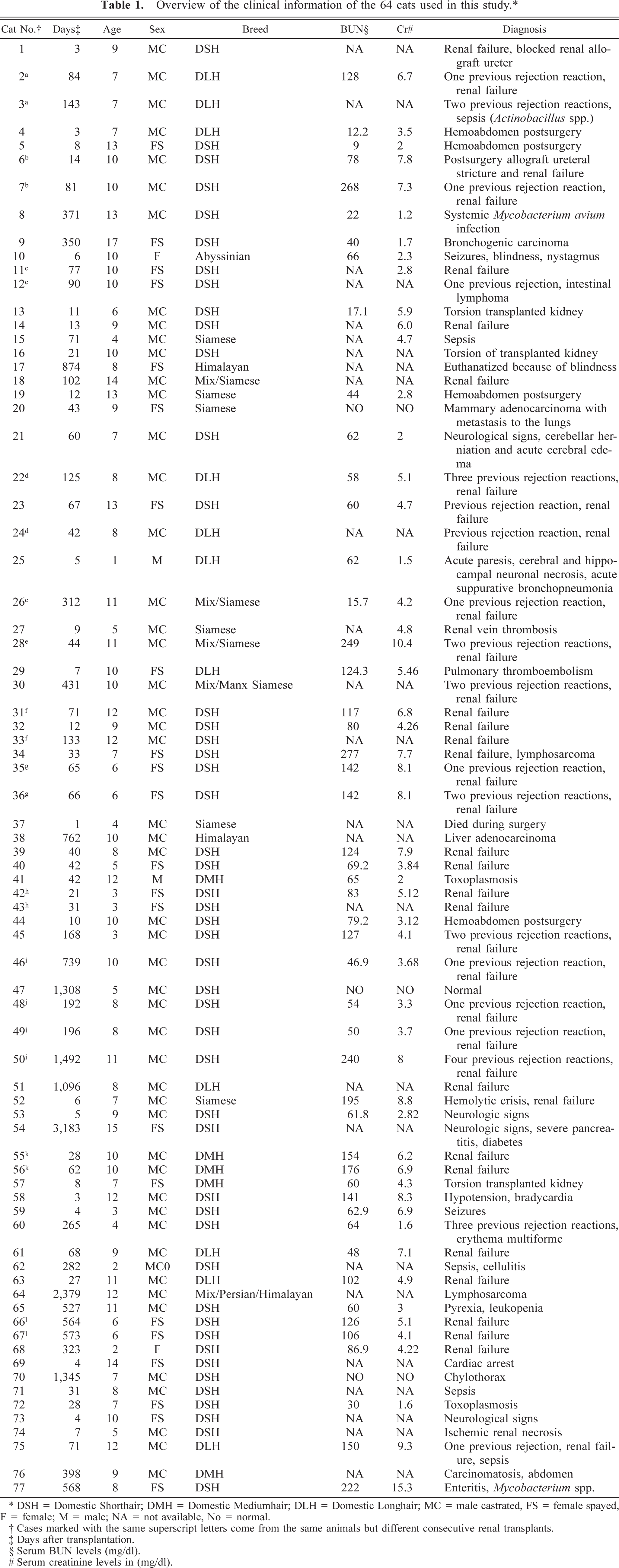
∗ DSH = Domestic Shorthair; DMH = Domestic Mediumhair; DLH = Domestic Longhair; MC = male castrated, FS = female spayed, F = female; M = male; NA = not available, No = normal.
† Cases marked with the same superscript letters come from the same animals but different consecutive renal transplants.
‡ Days after transplantation.
§ Serum BUN levels (mg/dl).
# Serum creatinine levels in (mg/dl).
Scoring
All sections were examined by a board certified veterinary pathologist (H. E. V. De Cock) unaware of the clinical or clinicopathologic findings in the cats and without access to the previous histology report. The pathologist evaluated all specimens using the Banff '97 working classification for human renal transplant histology. 23 This classification divides renal allograft biopsies into five categories of rejection: normal, antibody-mediated rejection, borderline changes (suspicious for acute rejection), acute or active rejection, and chronic or sclerosing allograft nephropathy. The threshold for a minimal acceptable sample was set at 20 glomeruli and one artery. In this classification system, individual parenchymal components, the glomeruli, interstitium, tubules, and vessels are scored on a scale of 0–3.
The basic features used to diagnose acute or active rejection are tubulitis and arteritis, but a minimal threshold for interstitial inflammation must be reached before acute or active rejection is diagnosed. Tubular inflammation or tubulitis is defined as a distinct infiltration of tubular intraluminal or intraepithelial lymphocytes and graded in the t-score. For the arterial and interstitial inflammation, graded in, respectively, the v- and i-score, only the mononuclear inflammation is considered. Glomerulitis (g-score) is not considered a specific criterion for rejection, but is graded in humans because it has implications for late graft function. The combined semiquantitave scoring of the i-, t-, and v-grades provides the morphologic basis for the acute/active rejection classification. When there was contradiction between the i- and the t-scores, in agreement with the Banff '97 system, the t-score was considered decisive.
On the basis of these scores, acute rejection is divided as follows. KDAB, acute antibody-mediated rejection (g1–3). KDARB, borderline change (i0–2, t0–2, v0). KDAR, acute or active rejection divided into the following categories based on i-, t-, and v-scores: KDAR1A, Banff type 1A (i1–3, t2, v0); KDAR1B, Banff type 1B (i2–3, t3, v0); KDAR2A, Banff type 2A (i1–3, t0–3, v1); KDAR2B, Banff type 2B (i1–3, t0–3, v2); KDAR3, Banff type 3 (i1–3, t0–3, v3).
Chronic sclerosing changes develop in the renal allograft after infectious, ischemic, and drug-related insults and secondary to chronic recurring immune reaction to the graft. In human graft pathology, chronic changes are described in glomeruli, interstitium, tubules, and vessels. Similar to the acute or active rejection, the chronic or sclerosing allograft nephropathy changes are defined on the basis of the presence of interstitial fibrosis (ci-score), tubular atrophy (ct-score), allograft glomerulopathy (cg-score), vascular fibrous intimal thickening (cv-score), mesangial matrix increase (mm-score), and arteriolar hyaline thickening (ah-score). In the Banff '97 classification system, tubular (ct) and interstitial changes (ci) are used as the basis of the grading of severity of chronic allograft nephropathy. On the basis of these scores, chronic or sclerosing allograft nephropathy (KDCR) is divided into mild (KDCR1), moderate (KDCR2), and severe (KDCR3). Because chronic changes are not always considered secondary to rejection, an ‘A’ is added to the final score if changes strongly suggestive of rejection are not found. If changes strongly suggestive of rejection are present, a ‘B’ is added. Feline allografts were included in group B when they showed signs of acute rejection, in other words, when the feline allografts already received a KDAR score. When the changes were categorized as KDARB (borderline rejection), the feline allografts were considered to have had insufficient changes to diagnose acute rejection.
The following grades for chronic allograft nephropathy are recognized: KDCR1A, mild, without specific changes suggesting chronic rejection; KDCR1B, mild, with specific changes suggesting chronic rejection; KDCR2A, moderate, without specific changes suggesting chronic rejection; KDCR2B, moderate, with specific changes suggesting chronic rejection; KDCR3A, severe, without specific changes suggesting chronic rejection; and KDCR3B, severe, with specific changes suggesting chronic rejection.
Because no guidelines are specified in the Banff '97 system for classification into the different categories, we used the following system: KDCR1 includes all specimens with ci-, ct-scores ≤ 1; KDCR2 includes all ci-, ct-scores = 2; and KDCR3 includes all ci-, ct-scores = 3.
In addition, all specimens were examined for histologic changes indicative of cyclosporine nephrotoxicity. Toxic effects of cyclosporine are described in human renal transplant rejections as isometric tubular vacuolation, arteriolar myocyte vacuolation, arteriolar endothelial cell necrosis, arteriolar subendothelial hyaline deposition, and glomerular microthrombi. 23
Statistical analysis
The significance of differences in acute rejection scores (KDAR-scores) and chronic allograft nephropathy scores (KDCR-scores) when compared with serum creatinine levels was tested with the Kruskall-Wallis nonparametric test (> two independent samples). For testing the correlation between histologic scores (i, t, ci, ct) and serum creatinine values, the Spearman rank correlation test was used. P values < 0.05 were considered significant. 12
Results
A summary of the scoring according to the Banff '97 classification of the feline renal transplant specimens is given in Table 2.
Banff'97 scoring system for the 77 feline renal transplants.
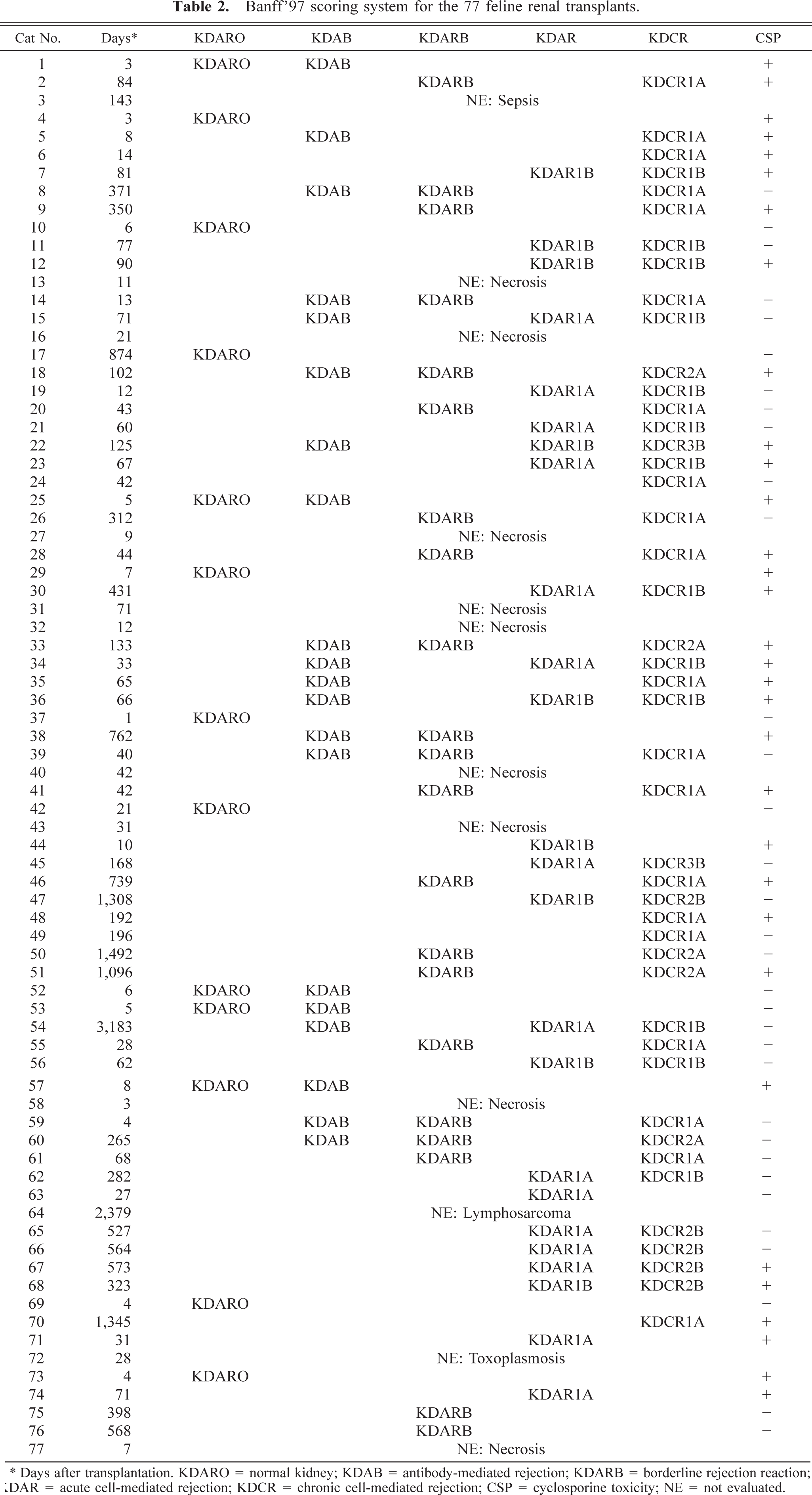
∗ Days after transplantation. KDARO = normal kidney; KDAB = antibody-mediated rejection; KDARB = borderline rejection reaction; KDAR = acute cell-mediated rejection; KDCR = chronic cell-mediated rejection; CSP = cyclosporine toxicity; NE = not evaluated.
Active or acute lesion scoring
Tubulitis and interstitial inflammation were evaluated in 65 specimens. Eleven specimens were excluded because of insufficient normal renal parenchyma secondary to a diffuse inflammatory infiltrate related to septicemia or diffuse parenchymal necrosis associated with infarction. One specimen was excluded because neoplastic lymphocytes obscured the parenchyma. Tubulitis, as described in human renal allografts, was difficult to appreciate in the feline transplanted kidneys. In most feline renal transplants, there was significant tubular fragmentation or interstitial inflammation that interfered with the evaluation for tubular inflammation. Destruction of the tubules took place early in the rejection so that a distinct tubulitis stage was not easily recognized (Fig. 1). In 22 of the 65 specimens (34%), no tubular infiltration with mononuclear cells could be determined. A tubulitis score of t1 (1–4 mononuclear cells/tubular cross section) was given to 19 specimens (30%), a t2-score (5–10 mononuclear cells/tubular cross section) in 15 specimens (23%), and a t3-score (10 mononuclear cells/cross section) in nine specimens (14%).
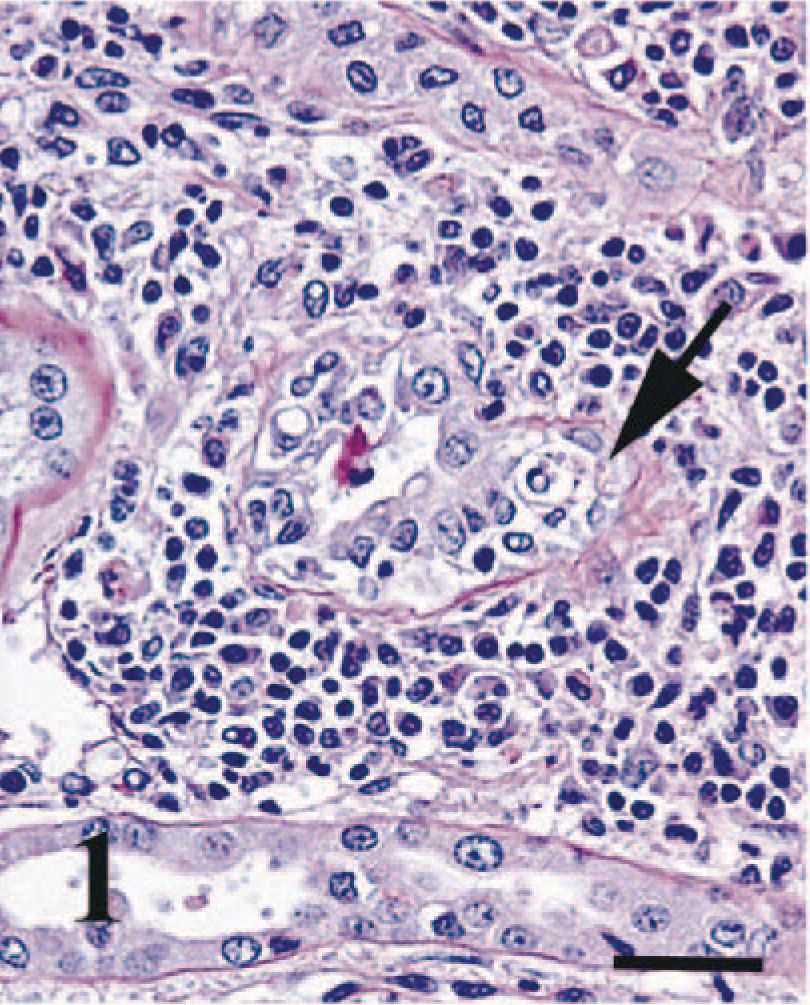
Transplanted kidney; cat. Marked tubular basement membrane fragmentation associated with interstitial and intratubular lymphocytic inflammation (arrow). PAS. Bar = 25 µm.
Lymphocytic interstitial inflammation was not found in 23 of the 65 specimens (35%), these specimens were scored as i0. Twenty-four (37%) of the 65 specimens had an i1-score (10–25% of parenchyma with lymphocytic inflammation). Ten specimens (15%) were given an i2-score (25–50% of parenchyma with lymphocytic inflammation), and eight specimens (12%) received an i3-score (> 50% of parenchyma with lymphocytic inflammation). The lymphocytic inflammation was always most prominent in the corticomedullary region and, as it increased in severity, spread in the cortex toward the capsule and within the renal papilla. In 32 of the 65 specimens (49%), eosinophils were also found. In some specimens, the eosinophils were present in prominent numbers admixed with the lymphocytes in the corticomedullary region; in other specimens, the eosinophils were more diffusely spread throughout the cortex. In the latter, the eosinophils were commonly accompanied by a neutrophilic and/or plasma cellular inflammation, also diffusely distributed throughout the cortex and medulla. Another often striking feature was the presence of interstitial edema usually also affecting the corticomedullary region and occasionally, when more severe, involving the entire cortex (Fig. 2).
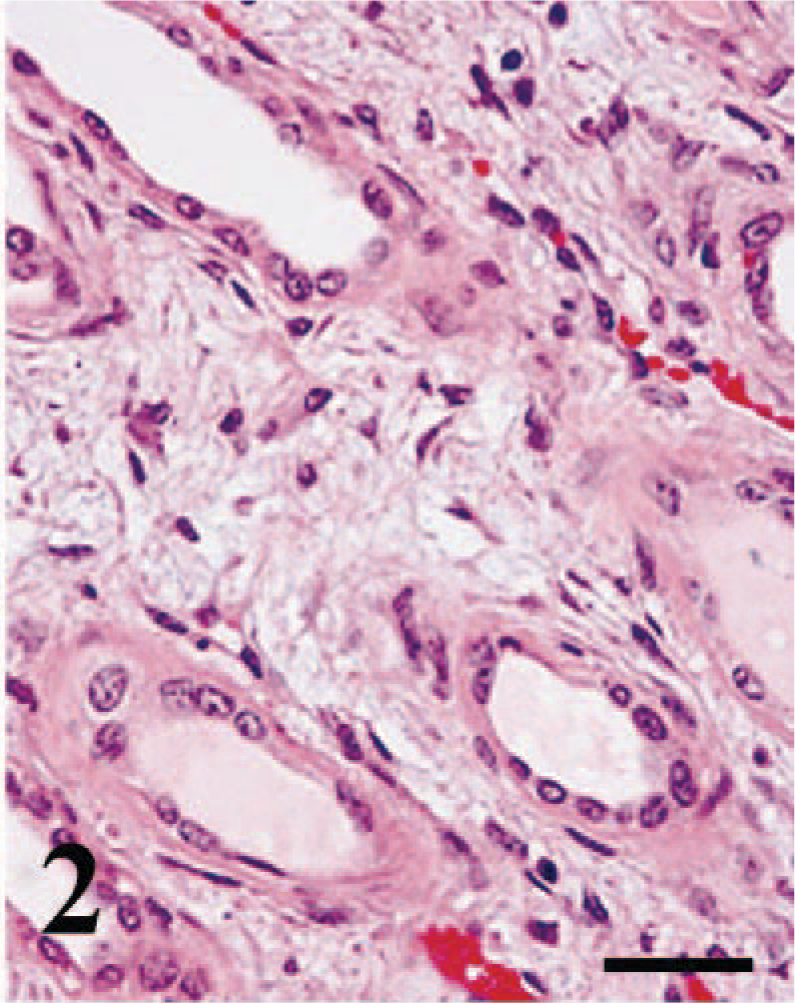
Transplanted kidney; cat. Marked separation of the tubuli by interstitial edema in the corticomedullary region. HE. Bar = 25 µm.
A total of 25 specimens had insufficient numbers of glomeruli within the available sections (< 20 glomeruli per section), including punch biopsy specimens and allografts with diffuse ischemic necrosis. Fifty-two specimens were examined for glomerular changes. Glomerulitis, as defined in the Banff '97 classification, which implies an infiltrate of mononuclear cells within the glomerulus was not found. However, in 13 specimens (25%), the glomeruli were infiltrated with neutrophils or eosinophils, or both. These lesions were given a g′-score to differentiate from the glomerulitis, as described in the Banff '97 system. On the basis of the presence of this neutrophilic glomerular infiltrate, a g1′-score (< 25% glomeruli affected) was found in six specimens, a g2′-score (25–75% glomeruli affected) in four specimens, and a g3′-score (> 75% glomeruli affected) in another three specimens. In the most severely affected glomeruli (with g3′-score), microthrombi, and in one specimen, mesangial necrosis was also found (Fig. 3).
Transplanted kidney; cat. Glomerular necrosis, neutrophilic inflammation, and thrombosis of capillary loops. HE. Bar = 10 µm.
The monocytic arteritis described in the Banff '97 classification and graded in the v-score was not found in any of the 77 specimens. Inflammation found in the wall of arteries was always neutrophilic (Fig. 4). Because polymorphonuclear arteritis is considered a sign of an acute antibody-mediated rejection, we graded the presence of a polymorphonuclear infiltrate in the tunica intima and tunica media and attributed this the v′-score. Nine of the 77 allografts (12%) showed clear evidence of neutrophilic arteritis in at least one medium-sized artery. A v′1-score (mild to moderate) was given to five specimens, a v′2-score (severe) in two specimens, and a v′3-score (transmural arteritis) in two specimens. In most specimens, this inflammation was associated with prominent vascular wall necrosis. In all kidneys with a vasculitis score of v′2 or higher, there was associated necrosis of the renal parenchyma.
Transplanted kidney; cat. Neutrophilic arteritis and fibrinoid necrosis of the vessel wall. HE. Bar = 20µm.
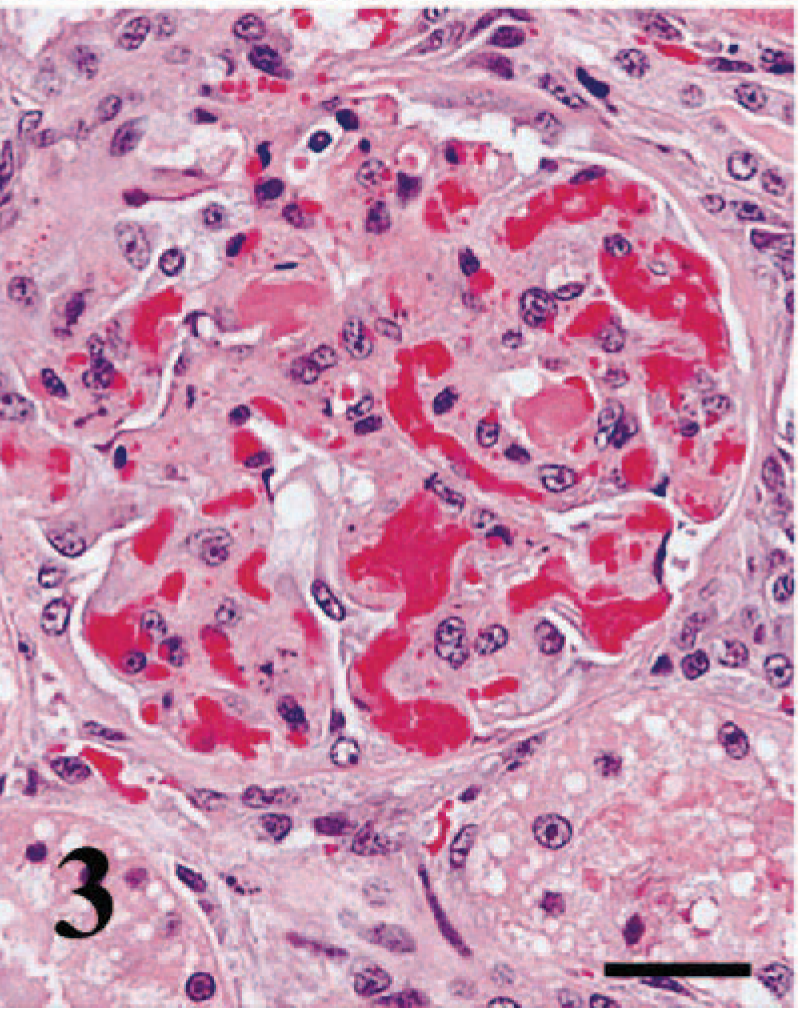
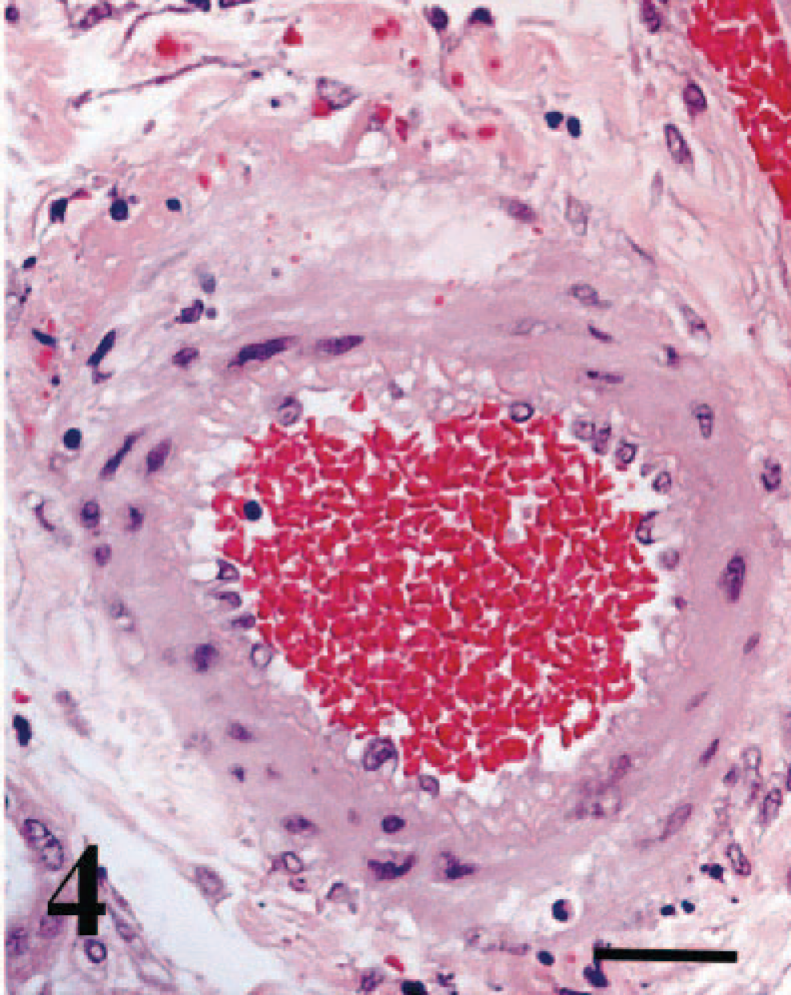
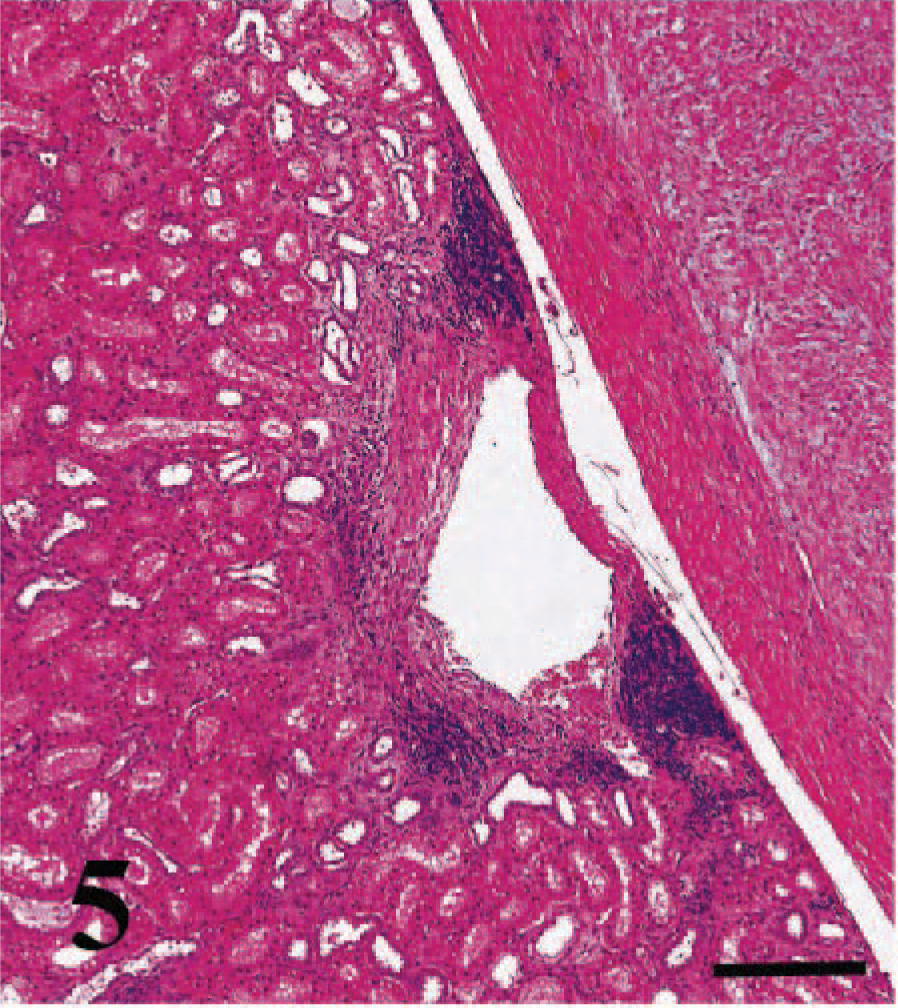
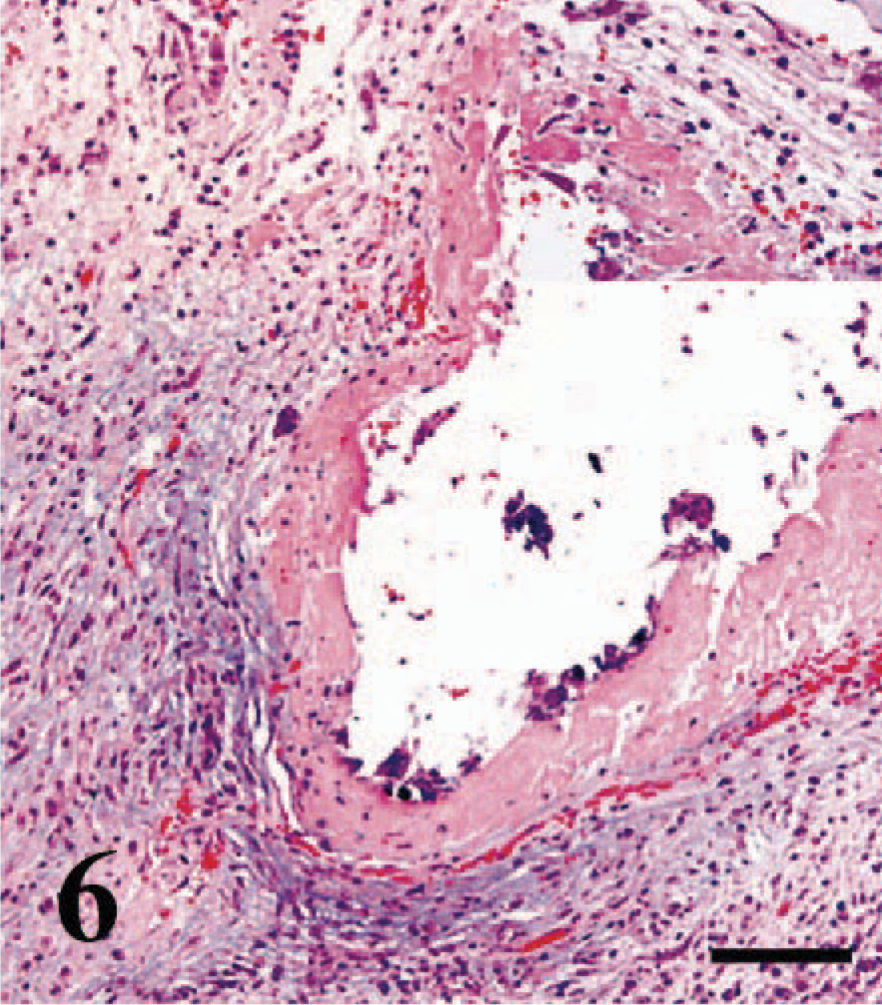
Another feature not considered in the Banff '97 classification but prominent in the cat renal allograft was phlebitis of the subcapsular, interlobular, and arcuate veins. In 37 of the 77 allografts (48%), there was a mild to severe lymphocytic inflammation surrounding the subcapsular veins or interlobular and arcuate veins, or all (Fig. 5). This phlebitis was commonly associated with significant perivascular fibrosis that sometimes caused up to 70% reduction of the venous lumen. Eosinophils and neutrophils were found within this perivenular inflammation in a minority of the specimens (Fig. 6). In four specimens, the inflamed veins contained well-organized thrombi (Fig. 6). One kidney also had multiple intravenous-organized thrombi in the absence of phlebitis.
Transplanted kidney; cat. Lymphocytic phlebitis of a subcapsular vein with moderate subendothelial fibrosis. HE. Bar = 100 µm.
Transplanted kidney; cat. Phlebitis of a subcapsular vein with intravascular thrombosis. Inset shows the mainly neutrophilic inflammation in the vessel wall. HE. Bar = 100 µm.
In 65 of the 77 specimens, all the scores could be obtained, and following categories were diagnosed: KDAR1A (Banff type 1A [i1–3; t2; v0]) in 15 (23%) of the renal transplant samples and KDAR1B (Banff type 1B [i2; t3; v0]) in nine (14%) of the renal allografts. KDAR2A (Banff type IIA [i1–3; t0–3; v1]), KDAR2B (Banff type 2B [i1–3; t0–3; v2]), and KAR3 (type III [i1–3; t0–3; v3]) were not represented in this study because no vasculitis, as defined in the Banff '97 system, was found.
KDAB or antibody-mediated rejection was diagnosed in 20 specimens (30%) on the basis of the presence of neutrophilic glomerulonephritis, glomerular thrombosis, glomerular necrosis, and neutrophilic arteritis. 3,30
Chronic or sclerosing lesion scoring
The 65 feline allografts used for the i- and t-scores were evaluated for interstitial fibrosis and tubular atrophy. No interstitial fibrosis (ci0) was found in 22 specimens (34%). A ci1-score (fibrosis in 6–25% of the cortical area) was given to 24 specimens (37%), a ci2-score (fibrosis in 26–50% of the cortical area) to 15 specimens (23%), and a ci3-score (fibrosis in > 50% of the cortical area) to four (6%) specimens (Fig. 7).
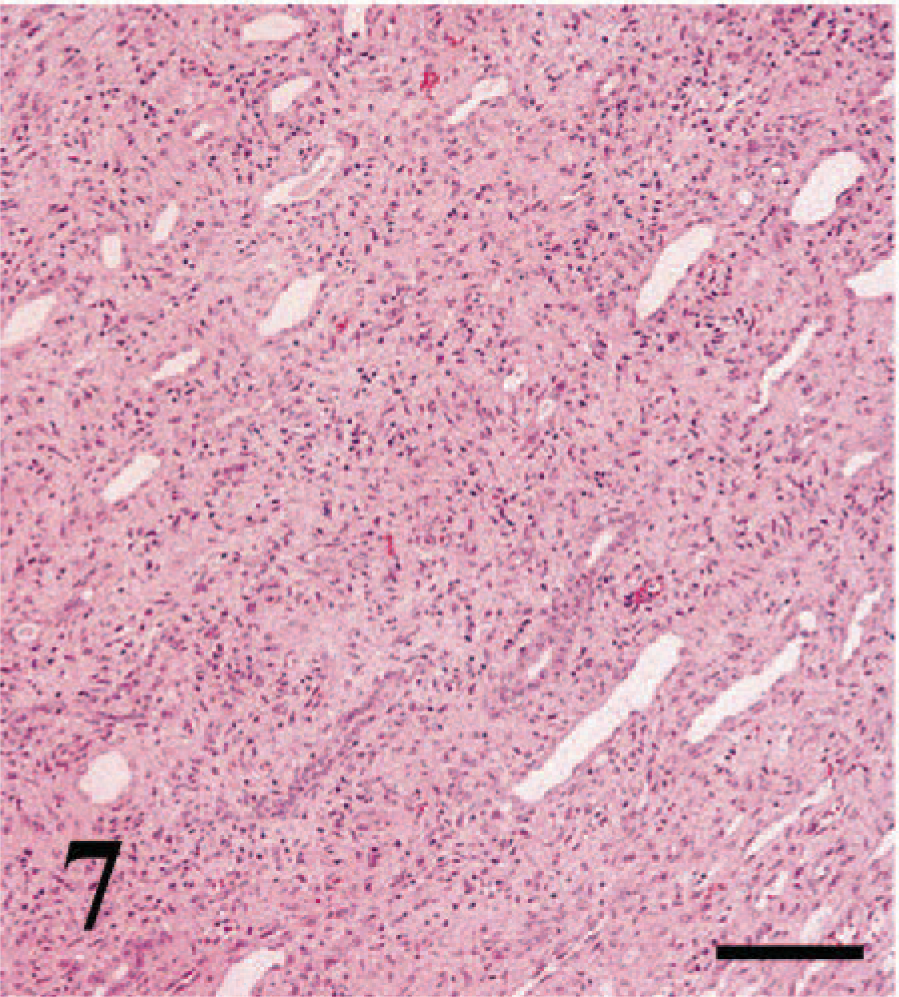
Transplanted kidney; cat. Diffuse interstitial fibrosis, and mild interstitial lymphocytic inflammation in the corticomedullary region. HE. Bar = 100 µm.
The tubular atrophy was scored as ct0 in 31 renal allografts (47%). A ct1-score (tubular atrophy in 25% of the cortical tubules) was found in 20 specimens (31%), a ct2-score (tubular atrophy in 26–50% of the cortical tubules) in nine specimens (14%), and a ct3-score (tubular atrophy in > 50% of the cortical tubules) in five specimens (8%).
A sufficient number of glomeruli were present to evaluate allograft glomerulopathy in 52 feline renal allografts. Double contours were found in three specimens only, and all were graded as cg1 (double contours affecting up to 25% of the peripheral capillary loops in the most affected nonsclerotic glomerulus). All other feline renal transplants were graded as cg0 (no glomerulopathy).
Mesangial matrix increase was scored as mm0-score no mesangial matrix increase in 13 feline allografts (25%). An mm1-score (up to 25% of nonsclerotic glomeruli affected) was found in 18 specimens (35%), an mm2-score (up to 26–50% of nonsclerotic glomeruli affected) in five specimens (10%), and an mm3-score (up to > 50% of nonsclerotic glomeruli affected) in 16 (31%) specimens.
As for the acute vascular changes, chronic arteriolar changes appeared to be very minimal in the evaluated feline renal allografts. Only two specimens were scored as ah1 (mild to moderate PAS-positive hyaline thickening in more than one arteriole).
In total, 45 (69%) feline allografts showed signs of chronic allograft nephropathy. The KDCR1A score was given to 21 specimens (31%) and KDCR2A to five specimens (8%); no specimen was scored as KDCR3A. Specimens with evidence of rejection included 13 with KDCR1B score (20%), five with KDCR2B score (8%), and one with KDCR3B score (2%).
Tubular isometric vacuolation was the most prominent finding suggestive of cyclosporine toxicity in the feline allograft kidneys (Fig. 8). On the basis of the tubular changes, cyclosporine toxicity was present in 33 (51%) feline renal allografts of the total of 65 specimens that could be evaluated for tubular changes. Arteriolar changes were present but not always very distinct and therefore were sometimes questionable. Glomerular microthrombosis, another reported 32 change with cyclosporine toxicity, was found in four specimens, two of which also had tubular isometric vacuolar changes. If the two additional specimens are included, the total number of feline renal allografts with evidence of cyclosporine nephrotoxicity was 35 (54%).
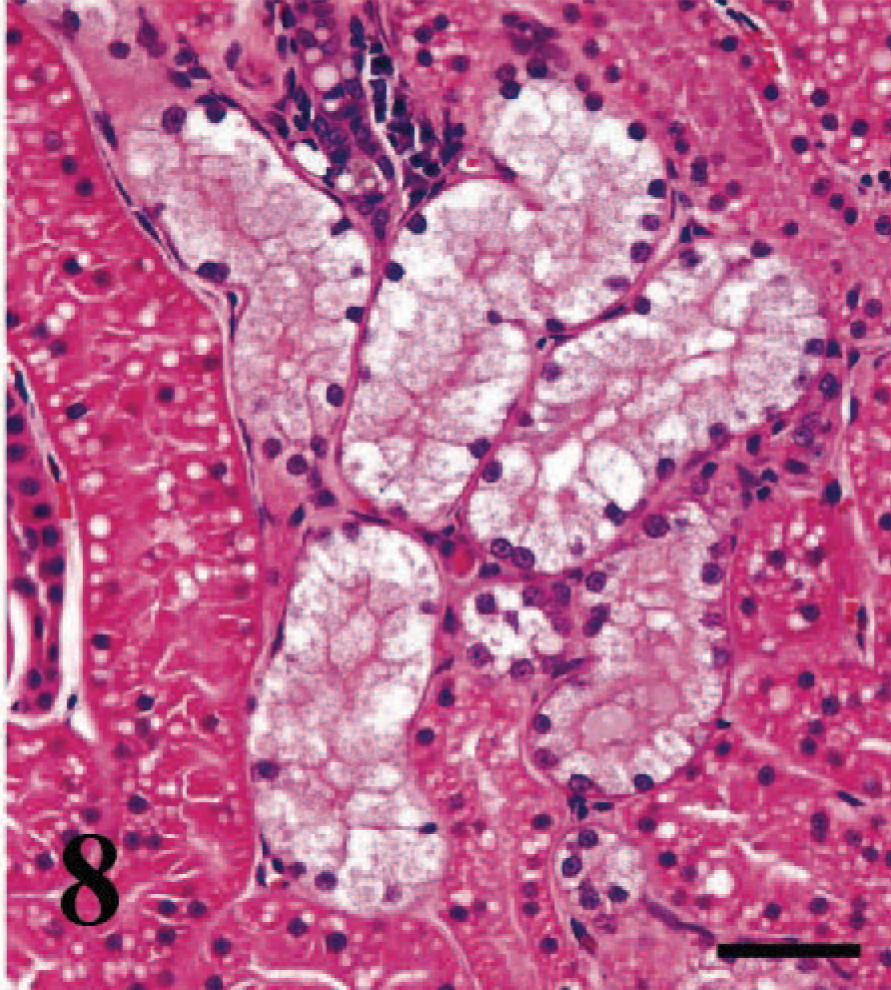
Transplanted kidney; cat. Isomorphometric tubular vacuolization suggestive for cyclosporine toxicity. HE. Bar = 20 µm.
Normal and borderline lesions
On the basis of the Banff '97 classification system, 13 of the 65 examined renal transplant samples (20%) had no signs of cell-mediated rejection (KDARO). Only one of these cats with no lesions had no clinical history of renal failure, survived for 874 days, and was euthanatized because of blindness. In two cats, one that died from pulmonary thromboembolism after 7 days and one that died during surgery, the only finding was mild vacuolation of arterial smooth muscle cells. In 10 cats with mean survival time of 6.5 days (range 3–21 days), the lesions were quite significant. However, none of their lesions were classified as rejection. One transplant biopsy had diffuse parenchymal necrosis consistent with an ischemic event that explained the clinical signs of renal failure. Eight renal transplants had a variable degree of acute tubular necrosis and proteinuria in association with intimal neutrophilic arteritis (3), phlebitis with thrombosis (3), or parenchymal edema (3). Changes consistent with cyclosporine toxicity were found in five of these eight renal transplants; in one, it was the only lesion found.
A KDARB score (borderline change [i0; t0–2; v0]) was given to 21 (32%) of the examined feline renal transplants.
Statistics
There was no statistically significant difference between the different groups of the acute/active rejection (P = 0.942) or chronic allograft nephropathy (P = 0.282) on the basis of serum creatinine levels. Table 3 shows the correlation coefficient (r) and probability (P) for the most important histologic scores used in the acute/active (i- and t-score) or chronic allograft nephropathy (ci- and ct-score), serum creatinine values, and days after transplantation. No significant correlation was found between any of the histologic scores and creatinine values. There was a positive correlation between the time after transplantation and severity of the t, i, ci, and ct histologic scores.
Correlation of histologic findings to serum creatinine and number of days after transplantation.
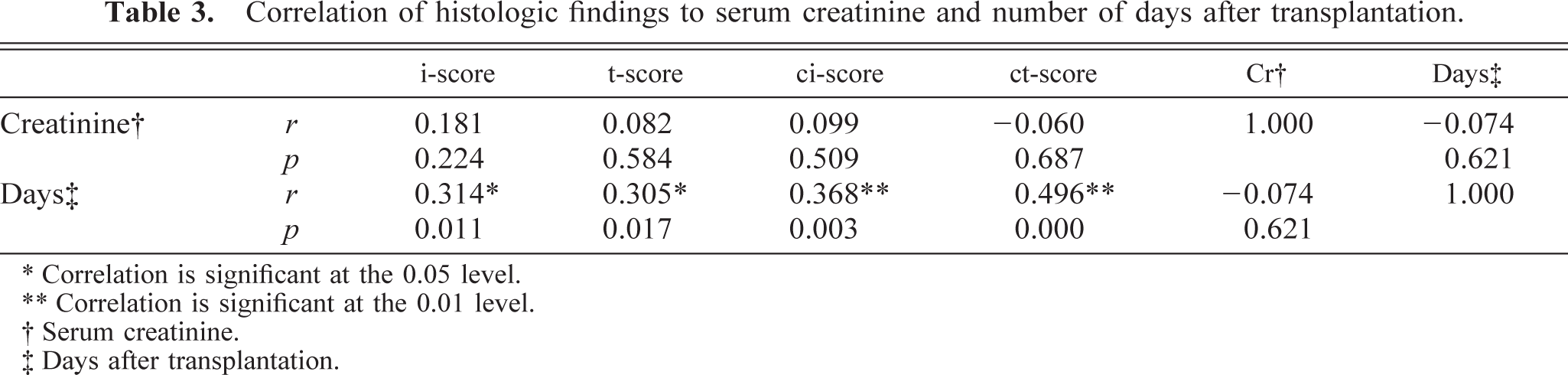
∗ Correlation is significant at the 0.05 level.
∗∗ Correlation is significant at the 0.01 level.
† Serum creatinine.
‡ Days after transplantation.
Discussion
In this histologic study, changes consistent with rejection were evaluated in feline-transplanted kidneys. Lesions were graded using the most commonly used grading system for human renal transplants, known as the Banff '97 classification. 23 Overall, this classification system appeared useful in detecting rejection reactions. Although only 44% of the 77 feline renal transplants had a history of renal failure at the time of biopsy, 80% showed lesions consistent with a certain degree of rejection on histology. Most feline renal transplants graded as KDARO (no signs of rejection) were obtained within the first 14 days after transplantation, which explains the absence of rejection. Of the 50 feline renal transplants that survived more than 1 month, 48 showed histologic evidence of rejection, whereas only 34 showed clinical evidence of renal failure. This discrepancy between clinical signs of renal failure and histologic findings of rejection confirms the finding in humans that renal allograft biopsies can diagnose subclinical rejection and are an important diagnostic tool for the follow up of renal transplants. 7,13,14,21,26,27
Although the Banff '97 classification was helpful for detecting a rejection, it did not reliably reflect the severity of the reaction. No significant differences were found in serum creatinine concentrations (used as measure for the severity of renal failure) in the different groups of the acute or active (P = 0.942) or chronic (P = 0.282) rejection reactions. Although not statistically significant, grading of the severity of chronic rejection on the basis of the Banff '97 classification system when applied on feline renal transplants seemed to better reflect the severity of the lesions. This is most likely because the parameters used for chronic rejection, interstitial fibrosis, and tubular atrophy corresponded well between cats and humans. In contrast, the usefulness of this system for grading the severity of the acute rejection appeared very limited. Although differentiation of acute rejection reactions from other lesions such as pyelonephritis, ischemic necrosis, toxic necrosis, and chronic rejection was adequate, the severity of the acute rejection reaction was not reflected in the final score. The main reason seems to be the differences that exist between acute rejection reactions in humans and cats. In feline renal transplants, the incidence of distinct tubulitis, in which individual lymphocytes are found in the tubular wall or lumen, is low and difficult to ascertain, whereas in the Banff '97 classification system, tubulitis is one of the principal indicators of an acute cellular rejection. 23,27 The severity of this tubular inflammation forms the basis for the differentiation between renal tubulointerstitial rejection and inflammation characterized as borderline changes (KDARB). Therefore, in this system, many feline renal transplants with distinct interstitial inflammation attributed to acute cellular rejection were not classified as such. Moreover, lymphocytic arteritis that upgrades acute cellular rejection to either moderate or severe, regardless of the severity of the interstitial inflammation, was never found. Arteritis in feline renal transplants was always neutrophilic and necrotizing as discussed below. The stress on lymphocytic arteritis in human rejection reactions is based on several studies that identified intimal lymphocytic arteritis as pathognomonic for acute cellular rejection and evidence that the biological behavior of this vascular rejection is quite different from that of pure interstitial rejection. 1,8,18 In this way, the feline renal transplants never obtained a score higher than KDAR1B, and this seemed to result in significant underscoring of the acute cellular rejection. In addition, lymphocytic glomerulitis, which is not used as a criterion but believed to be a significant sign of acute rejection, was also never found in feline renal transplants. Three major criteria used for grading acute rejection in the Banff '97 system, therefore, were either not found (vasculitis, glomerulitis) or rare (tubulitis) in feline renal transplants. Acute cellular rejection of renal transplants in cats was mainly characterized by interstitial inflammation. This inflammatory infiltrate was predominantly lymphocytic and often very prominent. It predominantly started at the corticomedullary region, spreading gradually into the surrounding parenchyma as it increased in severity. The absence of distinct tubulitis seemed largely a result of an early destruction of the tubules. On the basis of the PAS stains, it appeared that, even with minor lymphocytic inflammation, tubular basement membranes were already fragmented. Although not a common feature, “vanished” tubules, as this phenomenon is called in the human literature, also is described in renal cellular rejections in humans. 2,22 It is attributed to tubules being overrun and destroyed by inflammatory cells. 22
Besides a different reaction pattern in acute cellular rejections, the differences also may reflect the predominance of a different type of rejection reaction in both species. Neutrophilic arteritis 3,10,11 and neutrophilic glomerulitis, 3,30 two important findings in the feline allografts, have been described as signs of antibody-mediated rejection. Supportive of the presence of an antibody-mediated rejection is the finding of eosinophils admixed within the lymphocytic inflammation. 3,25 This is an interesting observation because antibody-mediated rejection is increasingly considered to be an important cause for ongoing renal allograft damage in humans. 9,17
It should be kept in mind that all cats were treated with cyclosporine, a potential nephrotoxin. 19,31 Cyclosporine intoxication in the form of isomorphometric tubular vacuolization 19 was found in a fair number of cats. Because glomerular thrombosis and vascular necrosis have been associated with cyclosporine toxicity in humans, this might also be the underlying etiology for these lesions in cats. 19,29,32
That a vascular component plays a distinctive role in the feline allograft rejection either due to antibody-mediated rejection or cyclosporine toxicity is clear from the prominent number of transplanted kidneys with distinct interstitial edema (37/65; 57%). 6
From this study, it is clear that there is a high need for better characterization of the type of rejection reaction found in feline renal transplants. Differentiation between acute cellular and antibody-mediated rejection might be valuable because it has been proved in humans that the type of rejection reaction not only dictates the treatment regimen but also has significant implications for prognosis. On the basis of the findings of this study, it is also clear that a significant part of the lesions in feline renal transplants might be cyclosporine induced. Because cyclosporine is the most commonly used immunosuppressive drug in cats with a renal transplant, better characterization of its potential toxicity might prove very useful in protecting feline renal allografts.
