Abstract
Paired samples of formalin-fixed, paraffin-embedded ileum and lymph node from 204 culled dairy cows were investigated for evidence of infection by Mycobacterium avium subsp. paratuberculosis. Of the samples, 151 were from animals that were tissue-culture positive for M. avium subsp. paratuberculosis, and 53 were from animals that were tissue and fecal culture negative. From the culture-positive animals, M. avium subsp. paratuberculosis was isolated from 78 samples of ileum and from 107 samples of lymph node. Ziehl-Neelsen acid-fast and immunoperoxidase stained slides were examined for 15 minutes each. Acid-fast organisms were identified in 7 of 78 (8.97%) and 6 of 106 (5.61%) culture-positive ileum and lymph node samples, respectively. Immunohistochemical (IHC) analysis of the same tissues identified infection in the ileum of 9 of 78 (11.54%) and in the lymph node of 5 of 106 (4.67%) culture-positive tissues. All tissues from culture-negative animals tested negative when using acid-fast and IHC staining. The sensitivity of these 2 tests in detecting M. avium subsp. paratuberculosis in culled dairy cows was not significantly different, and the tests exhibited substantial to almost perfect agreement. Both tests were much less sensitive than bacterial culture, detecting less than 6% of tissues positive compared with culture.
Introduction
Johne's disease, or paratuberculosis, is a chronic infectious disease caused by Mycobacterium avium subsp. paratuberculosis (MAP), a slow-growing, intracellular acid-fast bacillus. A variety of animals can be infected, however, ruminants are the primary hosts. 25 There is a higher prevalence of disease in dairy cattle compared with beef cattle, likely reflecting differences in management practices, including the close housing of dairy cattle, which may lead to easier transmission of the organism from animal to animal. 17 Clinical disease in cattle is characterized by chronic or intermittent diarrhea, chronic progressive weight loss, decreased milk production, and decreased fertility, in association with granulomatous enteritis and lymphadenitis. 6,13,21
The primary route of MAP transmission is thought to be fecal-oral, however, in utero infection also occurs. 4,26,29 Once infected, animals may move through 4 main stages of disease. These are classified as the following: 1) silent infection, 2) subclinical disease, 3) clinical disease, and 4) advanced clinical disease. 32 Susceptibility to infection is highest in calves less than 30 days old; however, clinical disease is not usually apparent in cattle until 2–5 years of age, and only 10–15% of infected animals develop clinical signs of disease. 6,21
Commonly described gross pathologic findings in cattle include mucosal thickening, predominantly in the distal small intestine; dilated serosal and mesenteric lymphatic vessels; and enlargement of mesenteric lymph nodes. Other less common findings include alopecia, and endocardial and aortic calcification. 6 Histopathologic findings range from mild to marked granulomatous inflammation of the intestine and mesenteric lymph nodes as related in a previous study. 6
Johne's disease is of significant economic importance to the dairy industry because of reduced milk production, premature culling, and low body weights at slaughter. In infected herds with 10% of cull cows that demonstrate clinical signs of disease, economic losses were estimated to be more than US$200 per cow. 22 In addition, the controversial role of MAP in Crohn's disease has led to an increased focus on paratuberculosis and further emphasizes the importance of this bacterium. 8,20
A major obstacle in the control of this disease is the difficulty of identifying infected animals, especially those in the subclinical stage of infection. 21,27 For every cow with advanced Johne's disease on a farm, it is estimated that 15–25 others on that farm are infected, with less than 30% of these detectible by diagnostic tests. 32 As recently reviewed, 7,31 several tests for the diagnosis of MAP infection exist, and they can be broadly classified into 2 categories: those that detect specific serum humoral/cell-mediated immune responses and those that detect the bacterial agent. Serologic tests for Johne's disease include enzyme-linked immunosorbent assay, agar gel immunodiffusion, and the complement fixation test. These are the most widely used tests for the presumptive diagnosis of Johne's disease; however, none of these tests are particularly sensitive when compared with fecal culture, especially for the detection of low-shedding subclinically infected animals. 21,31
Fecal or tissue culture for organism detection is considered the gold standard for MAP diagnosis, however, the organism is fastidious and slow growing, and bacteriologic culture may take as little as 4–8 weeks or as long as 16 weeks, depending on the culture methodology. 19 MAP detection may also be accomplished by histologic examination in conjunction with Ziehl-Neelsen (ZN) acid-fast staining of target tissues, immunohistochemistry (IHC), and polymerase chain reaction (PCR). These tests are much more rapid than culture and conveniently can be applied to formalin-fixed, paraffin-embedded (FFPE) tissues, which are readily available to pathologists. The sensitivities of these tests vary greatly, with several reports supporting the superior performance of each of these tests compared with the others and compared with tissue/fecal culture. 2,9,12,23,27,28,30
The aim of this study was to evaluate and compare histopathology in conjunction with acid-fast staining and IHC in the diagnosis of Johne's disease in tissues from a large sample of dairy cattle collected at slaughter. Tests were compared by using bacteriologic culture as the gold standard. An additional goal was to determine if prolonged examination would improve the detection of MAP in FFPE tissues when using ZN acid-fast staining.
Materials and methods
Collection of samples
For a recent study on the prevalence of Johne's disease in culled dairy cows in Atlantic Canada and Maine, paired samples of mesenteric lymph node and terminal ileum were collected from 984 randomly selected cows at an abbatoir. 16 Briefly, tissue sections and fecal samples were cultured for MAP as previously described. 16 Of these, 85 samples of ileum, 110 samples of lymph node, and 33 fecal samples were positive for growth of MAP on culture (total 165 culture-positive animals). Sections of the collected tissues were fixed in 10% buffered formalin, embedded in paraffin, sectioned at 5 μm, and stained with ZN acid-fast stain. Each slide was examined for approximately 5 min, and acid-fast bacilli were identified in tissues from 7 animals. 16
Ziehl-Neelsen acid-fast examination
For this study, tissues from a subset of the 984 animals were used. The samples consisted of ZN acid-fast slides, and FFPE sections of ileum and lymph node from 151 of the 165 culture-positive animals, and from 53 randomly selected culture-negative animals. Tissue sections were unavailable for 10 of the tissue-culture-positive animals, and 4 animals were positive on fecal culture alone; these samples were not included in the study.
Each ZN acid-fast slide was examined microscopically for the presence of acid-fast bacilli within macrophages or multinucleate giant cells for a period of 15 min, with the majority of time spent carefully scanning the slide at low magnification (10×). The examiner was blinded to the culture status and to the previous acid-fast results of the samples.
Immunohistochemistry
Immunohistochemistry was performed on serial sections of the ileum and mesenteric lymph node from the same subset of animals for the localization of mycobacterial antigens by using an indirect streptavidin-biotin method with a commercially available peroxidase kit. a Briefly, tissue sections were mounted on glass slides, deparaffinized with xylene and alcohol rinses, and endogenous peroxidase activity was blocked via immersion in 3% H2O2. After rinsing, the slides were incubated with either polyclonal rabbit anti-Mycobacteria bovis antibody or a negative control reagent for 10 min. This polyclonal antibody is marketed for the detection of M. bovis and reacts with soluble and insoluble bacterial antigens, many of which are common to other mycobacterial species, including MAP. 2 The slides were then incubated with peroxidase-labeled goat anti-rabbit antibody for 10 min, followed by the application of freshly prepared substrate-chromogen solution. The peroxidase activity was detected with buffered substrate solution and 3,3′-diaminobenzidine. Enzymatic activity was allowed to continue for 5 min, and the slides were rinsed, counterstained with hematoxylin, rehydrated with ethanol and xylene, and cover-slipped. Control sections from other cattle that were positive for infection with MAP were prepared in the same manner. Similar to the acid-fast slides, these slides were examined for 15 min each, with the majority of time spent carefully scanning the slide at low magnification (10×); the examiner was blinded to the culture status of the samples.
Statistical analysis
Statistical analysis was performed with Stata 9.2 b and Minitab 14. c Confidence intervals (CI) for sensitivity of the ZN acid-fast and IHC tests were determined by using the Wilson estimate. 18 The McNemar test was used to compare paired population proportions for the ZN acid-fast and IHC tests, and for ZN acid-fast results obtained with 5-min and 15-min scanning times. A significant McNemar test indicates serious disagreement between tests, whereas a nonsignificant test indicates that the 2 proportions do not differ. P values below 0.05 were considered significant. Agreement among the results obtained for these tests was determined by calculating the kappa statistic. Because the kappa statistic becomes difficult to interpret when prevalence is low, 10 this value was calculated for a subset of the culture-positive animals to improve accuracy. The agreement was considered moderate, substantial, or almost perfect if values were estimated at 0.4 ≤ κ < 0.6, 0.6 ≤ κ < 0.8, or 0.8 ≤ κ < 1.0, respectively.
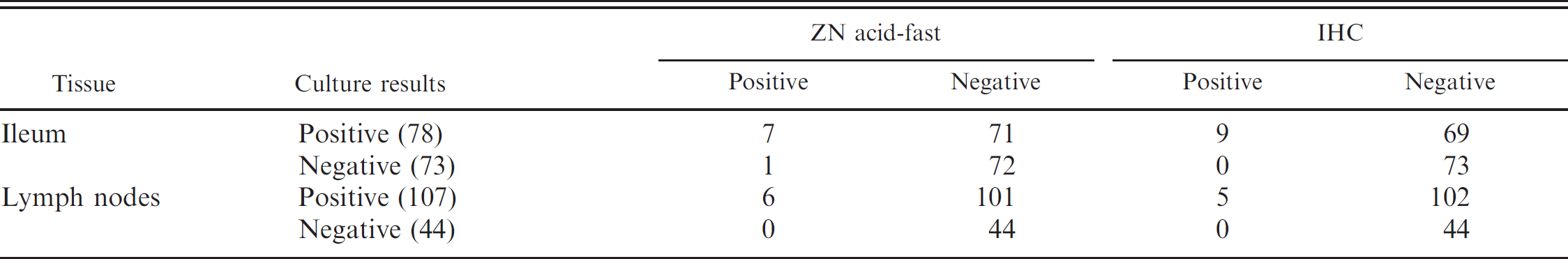
Results for tissue culture, Ziehl-Neelsen (ZN) acid-fast staining, and immunohistochemistry (IHC) for the detection of Mycobacterium avium subsp. paratuberculosis in ileum and mesenteric lymph node from tissue culture-positive cows.
Results
Acid-fast bacilli were identified in 7 of the 78 culture-positive samples of ileum, giving a test sensitivity of 8.97% (95% CI: 4.21–17.74%), and from 6 of the 107 culture-positive samples of lymph node, giving a test sensitivity of 5.61% (95% CI: 2.40–12.02%) when culture of tissues is used as the gold standard for detection (Table 1). A small number of acid-fast bacilli were detected in 1 sample of culture-negative ileum; this animal, however, was detected as positive for MAP on lymph-node culture. In total, acid-fast bacilli were identified in 8 (or 5.30%) of the 151 tissue-culture-positive animals.
Lesions varied from mild to marked, with mild lesions that consisted of small aggregates of macrophages or multinucleated giant cells that contained small numbers (2–4) of acid-fast bacilli infiltrating the villous lamina propria of the ileum or the paracortical and cortical tissue of the lymph node. Marked lesions consisted of sheet-like infiltrates of macrophages and multinucleated cells that contained large numbers of acid-fast bacilli, admixed with lymphocytes and plasma cells that expanded the lamina propria and submucosa of the ileum, and the paracortex and medullary sinuses in the lymph node.
No acid-fast organisms were identified in tissues from the 53 culture-negative animals. Occasionally, nonspecific acid-fast staining was noted within macrophages in the medullary sinuses of lymph nodes from the culture-positive and culture-negative animals.
In a previous study, 16 examination of each of the ZN acid-fast slides for 5 minutes revealed positive results in 7 samples of ileum and 6 samples of lymph node from a total of 7 animals. Extending the searching time from 5 to 15 minutes in the present study resulted in the identification of the same 7 infected animals identified previously, with a single additional positive result. These 2 tests were found to show almost perfect agreement, with a kappa value of 0.93, and a nonsignificant McNemar test result (P = 0.3173), indicating that the 2 proportions do not differ significantly.
Examination of immunoperoxidase-stained slides resulted in the identification of positive staining in 9 of the 78 culture-positive samples of ileum, giving a test sensitivity of 11.54% (95% CI: 6.04–20.79%), and from 5 of the 107 culture-positive samples of lymph node, giving a test sensitivity of 4.76% (95% CI: 1.78–10.83%) when culture of tissues is used as the gold standard for detection (Table 1). In total, positive IHC staining was identified in 9 (or 5.92%) of the 151 tissue-culture-positive animals.
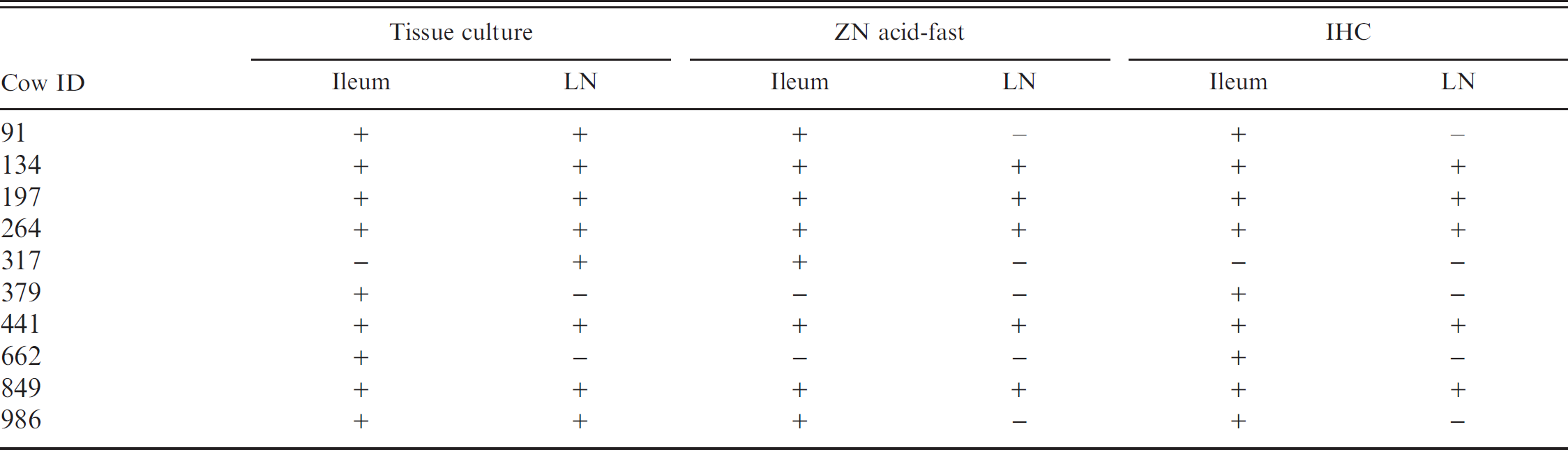
Positive results are seen as distinct, granular, brown staining within macrophages and multinucleated giant cells (as depicted in Fig. 1). Positive staining within these cells was most commonly noted in lesions as described for the ZN acid-fast staining. In a small number of cases, positive staining was identified focally within the Peyer's Patches of tissues in which acid-fast bacilli were not detected. When compared with acid-fast staining, IHC identified 7 of 8 of the same animals and an additional 2 animals (Table 2). No significant difference is noted in the sensitivity of IHC compared with ZN acid-fast staining for the identification of infected animals. These 2 tests were found to show substantial to almost perfect agreement, with a kappa value of 0.81, and a nonsignificant McNemar test result (P = 0.5637).
Discussion
Several tests exist for the diagnosis of Johne's disease in cattle; however, considerable debate exists over which is most effective, and this varies with the stage of disease. Although advanced clinical disease is relatively easily diagnosed, subclinically affected animals pose a diagnostic challenge. The tissues for the present study were collected from dairy cows that were randomly selected at slaughter, and it is likely, therefore, that the population includes uninfected animals, subclinically infected animals, and clinically infected animals. This is in contrast to many studies that compared diagnostic tests for Johne's disease in which only clinically or experimentally infected animals are examined. In addition, the disease exposure in this case is natural, and a large sample population was tested.
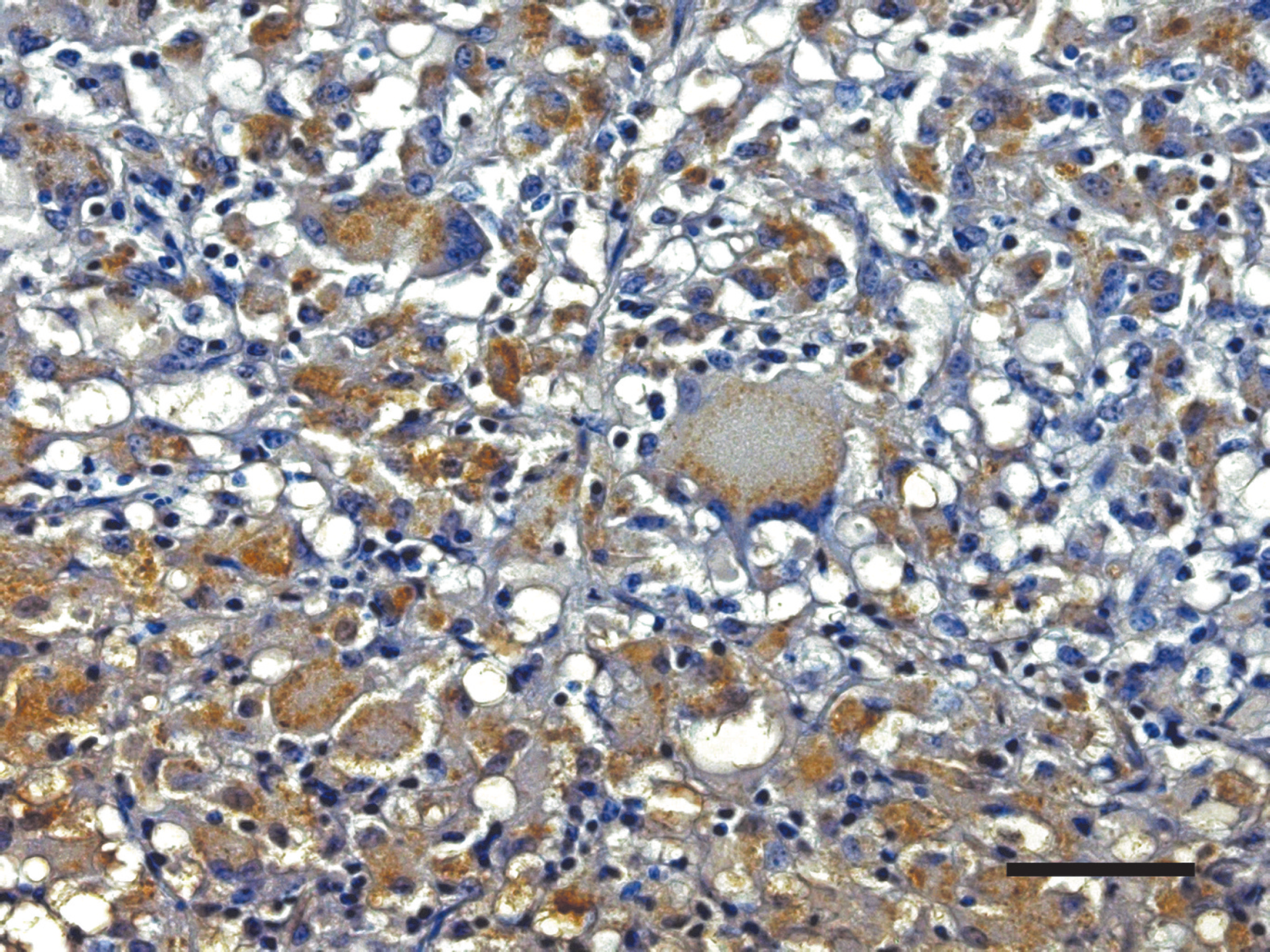
Tissue section of ileum immunostained with polyclonal rabbit anti-Mycobacterium bovis antibody. Positive reactions appear as distinct granular brown staining within macrophages and multinucleated giant cells. Bar = 50 μm.
Relationship of tissue culture to Ziehl-Neelsen (ZN) acid-fast staining and immunohistochemistry (IHC) for the detection of Mycobacterium avium subsp. paratuberculosis in ileum and mesenteric lymph node (LN).
Bacterial culture has long been considered the gold standard for paratuberculosis diagnosis, but it is costly and time consuming, and, when gross lesions are not apparent during necropsy, fresh tissues may not be submitted for culture. Therefore, the most convenient and rapid tests for pathologists are those that can be applied to FFPE tissues, including ZN acid-fast and IHC techniques.
Histopathologic analysis of tissues from infected animals, in conjunction with ZN acid-fast staining has been found in some studies to be the most sensitive method of confirmation of Johne's disease. 9,11 In 1 study, fecal and tissue cultures were shown to have lower sensitivities (15% and 30%, respectively) in experimentally infected lambs compared with histologic demonstration (80%) of ZN acid-fast bacilli. 14 Other studies demonstrated these 2 tests to perform equally well in identifying infected cattle. 12 In this study, histopathology was found to have a low sensitivity (5.30%) compared with bacteriologic culture, identifying only 8 of 151 culture-positive animals. This is similar to other studies that showed that tissue culture of lymph nodes, in particular, detected more infected cattle than histologic examination. 1 Several factors affect the sensitivity of this test, including the multifocal distribution of small granulomas, which may prevent histopathologic identification. 27 As well, the ZN acid-fast technique only stains bacteria with an intact cell wall; therefore, the absence of acid-fast bacteria may be because of degradation of bacteria, scarcity of bacteria in lesions, or the presence of only cell-wall deficient forms of MAP. 21 A larger amount of tissue was tested via culture (2 g of tissue 16 ) when compared with ZN acid-fast and IHC testing in which only a 5-μm thick section of tissue that averaged less than 10 mg in weight was examined microscopically. Because the same piece of tissue cannot be used for both culture and microscopy, the examined tissues are often millimeters to centimeters apart from the tissues submitted for culture. Therefore, it is possible that the examined sections were from uninfected areas, while cultures were taken from areas of infection. 34 It has been suggested that tissue samples from animals with subclinical paratuberculosis should be processed for both histopathologic examination and tissue culture to enhance diagnostic sensitivity. 12,15,33
Preliminary examination of histologic sections by pathologists may fail to detect subclinical Johne's disease lesions. In 1 study, it was found that prolonged examination of tissues with negative initial findings detected small focal lesions with acid-fast bacteria in 44% of slides reexamined. 9 It has been found that even small isolated lesions were readily recognized by scanning sections at low magnification (4×) and that extended high-power searching of ZN acid-fast slides was ineffective in increasing the accuracy of a histopathologic diagnosis of Johne's disease. 5 It, therefore, was decided that, in the present study, the majority of the 15 minutes would be spent carefully scanning the slide at low magnification (10×). No significant increase in sensitivity was gained by increasing the scanning period from 5 to 15 minutes. Increasing the time spent examining the slides resulted in the identification of only 1 additional positive animal, and most of the positive results obtained were identified within the first 2–7 minutes of examination. There are reports that emphasize the importance of examining large numbers of sections of intestine and lymph node, rather than spending prolonged periods scanning fewer slides. 5,9
Immunohistochemical techniques are able to detect not only intact viable organisms but also soluble MAP, cell fragments, and altered microorganisms with defective cell walls, and, therefore, are potentially more sensitive and specific than ZN acid-fast staining. 23 Immunoperoxidase staining has been found to be better than acid-fast staining for detection of low numbers of mycobacteria in FFPE bovine tissue sections from culture-positive animals. 2 In the present study, no significant difference in the sensitivities of these 2 tests was detected, with only 1 additional positive result obtained when using IHC compared with ZN acid-fast staining. When compared with a kappa estimator, these tests showed substantial to almost perfect agreement, identifying most of the same infected animals.
It has been found that histopathologic examination and histochemical staining produced evidence of infection in more goats than did a bacteriologic examination. 27,30 In cattle, IHC has been shown to perform equally as well as tissue culture for the diagnosis of Johne's disease. 12 However, in the present study, IHC had a low sensitivity when compared with culture, detecting positive staining in the lymph node or ileum of only 5.96% of the culture-positive animals. As for acid-fast staining, the low sensitivity of IHC relative to culture may be because of the multifocal nature of the lesions and because the same section of tissue cannot be used for both culture and microscopy. Another potential cause of false-negative results when using IHC is antigenic alteration because of formalin fixation. 24 Ideally, tissues to be used for IHC testing should be processed after a maximum of 24 hour's fixation in formalin. 23 Additional potential problems encountered when using immunoperoxidase staining include a reduced signal-to-noise ratio because of the presence of melanin and hemosiderin-laden macrophages within the lymph node and Peyer's Patches, 24 and because of the presence of “garbage” macrophages, which are frequently described in these tissues. 3,11,12
Slaughter and postmortem examination provide the opportunity to assess the histopathologic diagnosis of Johne's disease in clinically normal animals. In the present study, bacteriologic culture proved to be far superior to ZN acid-fast and IHC in the diagnosis of Johne's disease in tissues collected from culled dairy cows. ZN acid-fast staining and IHC exhibited almost perfect agreement, with no significant difference in sensitivity. In addition, prolonging the scanning time did not improve the sensitivity of the histopathologic diagnosis of Johne's disease when using FFPE tissues. Recently, PCR techniques were used for identification of MAP in FFPE tissues. 23 This test is potentially more sensitive and specific than ZN staining and IHC, and future work may include applying this test to these samples.
Acknowledgements
This project was supported by the Atlantic Veterinary College Internal Research Fund. The authors would like to thank Darlene Jones for her excellent technical assistance. Drs. Alfonso Lopez and David Sims for their assistance with digital photography, and Dr. Henrik Stryhn for his guidance with the statistical analysis.
Footnotes
a.
Dako LSAB 2 System, Peroxidase, DAKO Corp., Carpentaria, CA.
b.
Stata Corp., College Station, TX.
c.
Minitab, Inc., State College, PA.
