Abstract
In 2009, a virulent strain of Aeromonas hydrophila caused severe disease and high mortalities (motile aeromonad septicemia variant [MASv]) in farmed channel catfish (Ictalurus punctatus) and hybrid catfish (I. punctatus × I. furcatus) in eastern Mississippi and Alabama. As is common in MAS, there is severe hemorrhagic dermatitis with ulceration, as well as abdominal hyperemia, petechiation, and mild ascites. Additional findings in MASv cases include panophthalmitis and orbital cellulitis, leading to ocular rupture, and brains are often hyperemic with mild random acute hemorrhage. In MASv infections, there is consistently also marked hemorrhage and edema in the submucosa and muscularis of the stomach, with lymphangitis and a few bacteria, plus splenomegaly with infarcts. Microscopically, spleens have necrosis of ellipsoids with macrophage infiltration and small numbers of bacteria; however, large infarcts are filled with bacteria. Other organs, such as liver, kidneys, and intestine, which are typically associated with MAS caused by various aeromonad species, are less affected. The findings in the stomach have not been reported in MAS in farmed catfish, to our knowledge, and the splenic changes are highly characteristic of MAS compared to infection with other gram-negative bacteria, including Edwardsiella ictaluri and other aeromonad species and strains.
Keywords
Aeromonas hydrophila is a ubiquitous gram-negative bacterial inhabitant of freshwater environments and is a well-known pathogen of many species of cultured and wild fish, including minnows, baitfish, carp (Cyprinus carpio), channel catfish (Ictalurus punctatus), striped bass (Morone saxatilis), largemouth bass (Micropterus salmoides), and tilapia (Oreochromis spp.), often associated with disease outbreaks secondary to stress or infection with a primary pathogen. 2 Motile aeromonad septicemia (MAS) is caused by infection with aeromonad bacteria, most commonly applied to A. hydrophila, but it may include species such as Aeromonas sobria and Aeromonas caviae. A diverse pattern of lesions can be elicited in diseased fish, depending on the species affected, the strain of bacteria, and the method of infection. Dermal necrosis, ulcerations, petechiation or hyperemia, and ocular disease are frequent external manifestations. Hepatic and renal necrosis are common, often with hemorrhage and necrosis in the intestine; splenic necrosis is variable. 2 Natural infection in channel catfish has been described as taking 4 forms: septicemia, focal visceral lesions (liver, kidney, muscle, skull), cutaneous, and asymptomatic. 7 In the cutaneous form, bacteria may not be culturable from internal organs. 10
Although typically considered to be an opportunistic pathogen in farmed catfish (Ictalurus sp.), in 2009 in Mississippi and Alabama a virulent strain of A. hydrophila (VAh) became a major cause of disease in farm-raised catfish. This particular strain may have come from an Asian source, as it is highly similar to an isolate from Asian grass carp and is unusually virulent to catfish. 5 Typically in farmed catfish ponds, MAS occurs in conjunction with severe environmental stress or coinfection with a primary pathogen, such as Flavobacterium columnare. At the aquatic diagnostic laboratory at Mississippi State University (Starkville, MS), this variant (MASv) has been by far the most common diagnosis made for the past 3 y, and the disease is most common in the summer months. In ponds that experience outbreaks, mortalities in ponds often range from 4,000 to 10,000 pounds lost (~8,000–15,000 fish, or ~ 5–15%), but can be very high (up to 100% loss); fish typically lack evidence of concurrent pathogens, further confirming that VAh is very different from previous catfish isolates.
We describe the findings from 15 outbreaks of MASv in ponds of channel catfish or hybrid catfish (channel × blue catfish crosses); VAh isolates from all cases were verified by standard microbial methods (growth on blood agar plates at 30°C, with biochemical testing [Sensititre system, Thermo Fisher Scientific, Waltham, MA]) and published PCR methods. 4 All fish from the outbreaks had A. hydrophila identified (pure cultures) from the kidney (mesonephros), and histopathology of all major visceral organs was performed on all fish.
All of the MASv cases seen by the primary author during 2014 and 2015 (n = 18) were examined; 15 cases were included and 3 were rejected because of decomposition (1) or lack of bacterial isolates for PCR testing (2). In all cases included here, fish were received freshly dead with minimal autolysis. In 8 cases, 1 fish was submitted, in 7 cases 2 fish were submitted, for a total of 22 fish. Eight cases were from channel catfish ponds; 7 were from hybrid catfish ponds. Ponds contained 90,000–150,000 fish and were ~2.2–4.5 hectares (5–10 acres). Both adult and juvenile catfish were examined, showing typical external signs of disease, varying from generalized petechiation (17 of 22 fish) to dermal necrosis (11 of 22 fish; Fig. 1A), periocular and ophthalmic necrosis (Fig. 1B), and occasionally even endomeningitis (teleost equivalent of leptomeningitis) 8 with necrotizing cellulitis associated with the foramen between frontal bones, which is characteristically described in Edwardsiella ictaluri infections 3 (Fig. 1B, 1C).
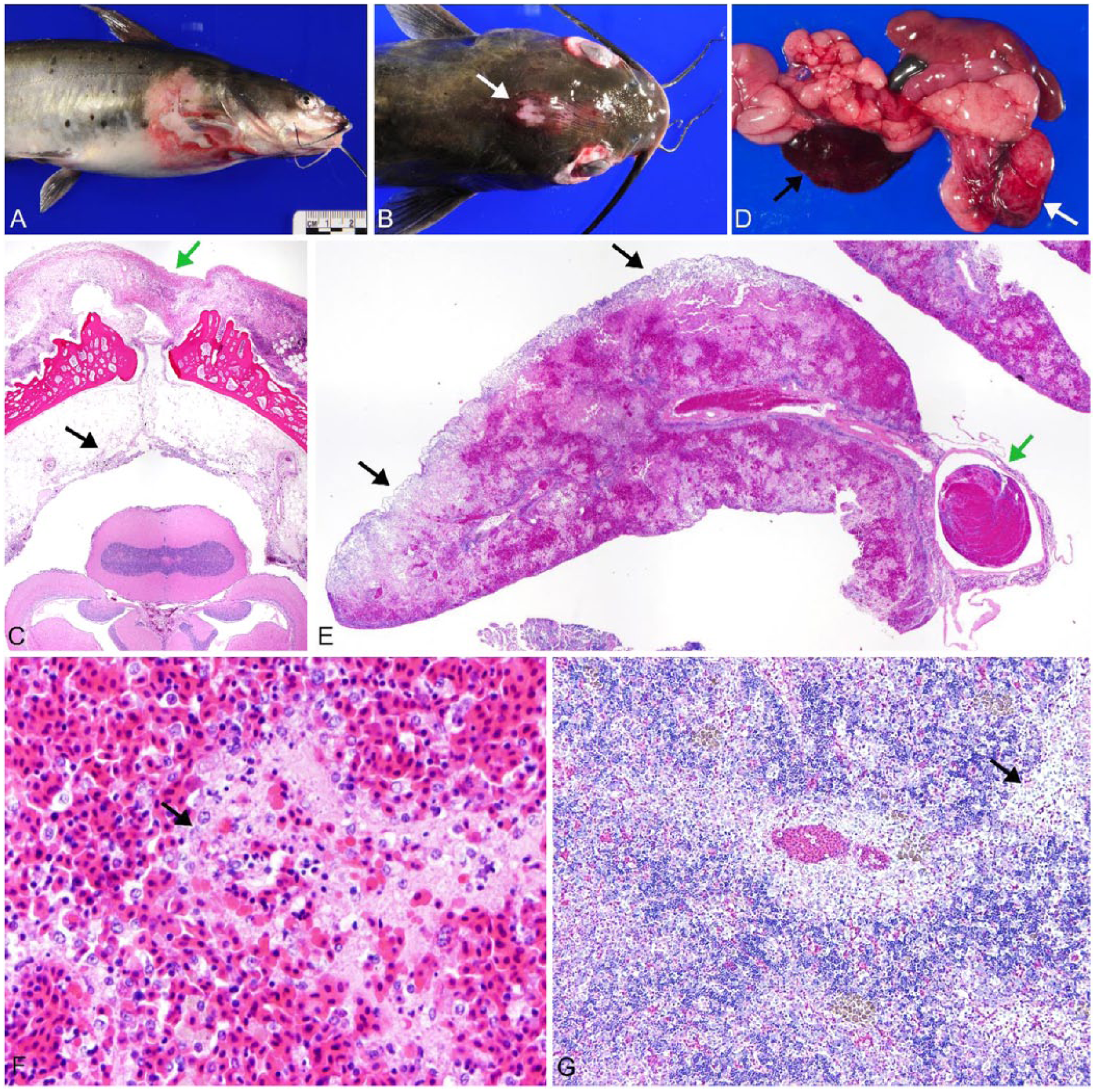
Channel catfish (Ictalurus punctatus) infected by virulent Aeromonas hydrophila.
Internally, there was often hyperemia, ecchymosis, a small amount of mildly hemorrhagic exudate, and marked splenomegaly (20 fish; Fig. 1D), as well as striking gastric hemorrhage and edema (20 fish; Fig. 1D). Spleens were estimated to be 5–10 times normal size, friable, and extremely congested. In some cases, infarcts predominated and were tan grossly and necrotic microscopically (Fig. 1E). Rod bacteria were abundant in the large infarcts. However, in most cases the spleen lacked these rarefied areas, and bacteria were few. Microscopically, all ellipsoids were necrotic (22 fish), with karyorrhectic debris, erythrocyte fragments, fibrin, and amorphous protein accumulation; the red pulp was markedly congested (Fig. 1F) in all but 2 fish. In 12 fish, large numbers of macrophages surrounded ellipsoids in 2–5 layers; bacteria were rarely evident. The head kidney (pronephros) was also commonly affected (14 fish), most often with accumulations of large macrophages and pyknotic cells in vascular sinuses and occasional immature granulomas (Fig. 1G). The trunk kidney (mesonephros) had mild granulomatous inflammation in 3 fish. Mild hyperplasia of the hematopoietic cells in both the pronephros and mesonephros occurred in 2 fish.
Gastric hemorrhage was associated with severe mural edema (22 fish; Fig. 2A) with vasculitis (21 fish). Microscopically, the submucosa was severely edematous with acute multifocal hemorrhage in the submucosa and muscularis (Fig. 2B). Blood vessels often showed little change aside from endothelial hypertrophy; however, lymphangitis was common. Lumens contained macrophages, pyknotic cells, proteinaceous debris, and a few rod bacteria; inflammation was often mild to moderate, and occasionally severe (Fig. 2C). Multiple small necrotic foci were present in the muscularis, and where lesions centered on vessels, numerous bacteria were seen in association with vascular necrosis (Fig. 2D). The gastric mucosa was not ulcerated grossly, and microscopically the only changes seen were variable numbers of apoptotic cells (McKnight cells) between the epithelial cells, as well as mild glandular atrophy. McKnight cells are commonly seen in the stomachs of diseased, pond-reared catfish in association with nonspecific gastroenteric disease. Changes in the intestine included submucosal edema and mild hemorrhage (18 fish) with mild inflammation (10 fish; Fig. 2E) and occasional multifocal epithelial necrosis.
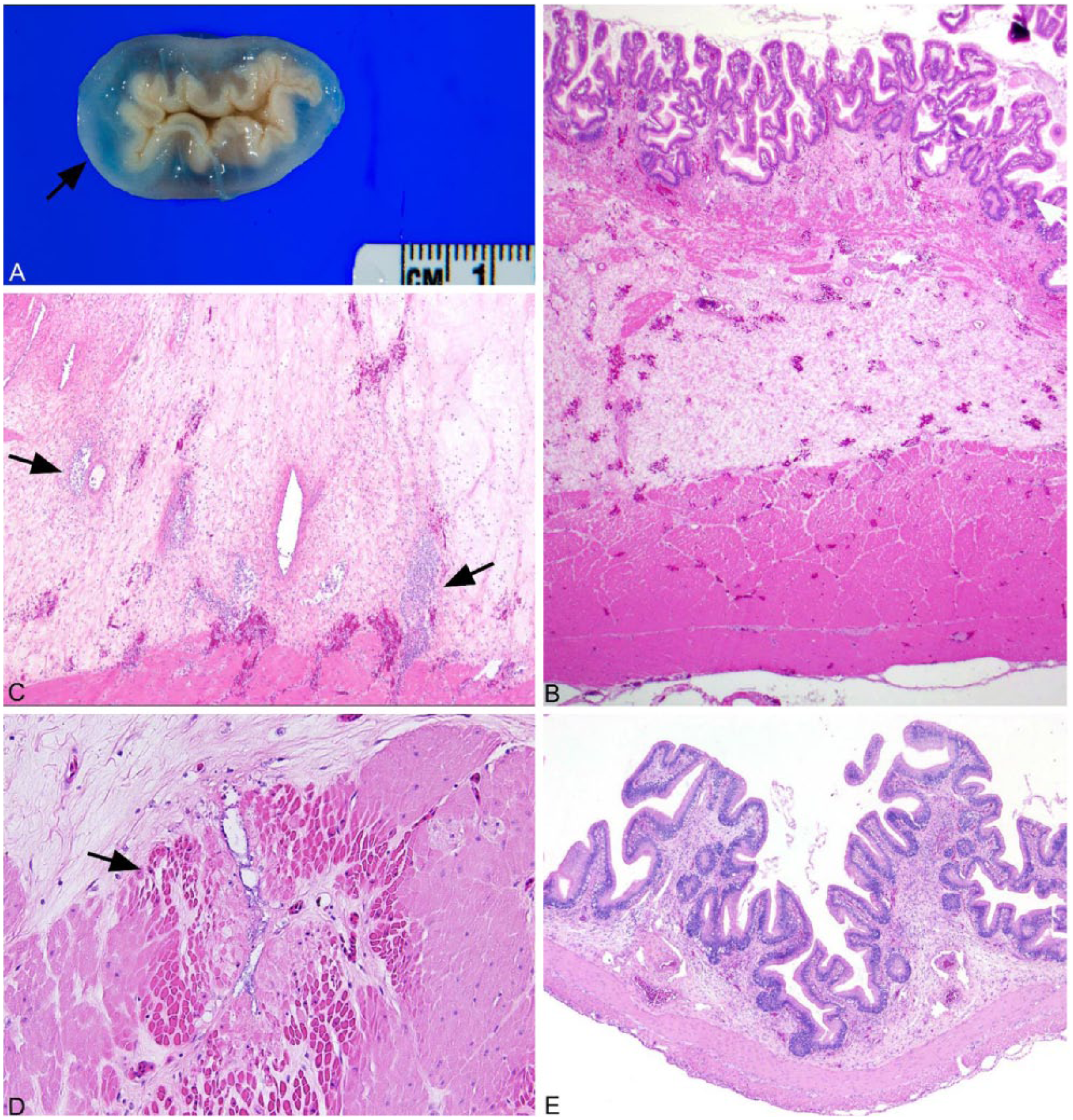
Channel catfish (Ictalurus punctatus) infected by virulent Aeromonas hydrophila.
In 3 fish, mild random multifocal hepatic hemorrhage without inflammation was seen; very mild random hepatitis and hepatocellular necrosis were seen in 2 fish. Various degrees of intrahepatic pancreatic exocrine cell necrosis, which is common with gram-negative bacterial sepsis in catfish, were also evident. Gills examined were within normal limits.
Catfish with MAS may lack external signs of disease; 7 most fish with MASv had external signs of sepsis. One fish that lacked external lesions had severe splenic and gastric changes, with mild enteritis and petechiation. Overall, the gastric and splenic changes were some of the most consistent findings in the cases examined; we found no reference to hemorrhagic gastritis or lymphangitis in association with MAS in fish of any type. The most consistent absence of findings was in the liver and mesonephros, where inflammation was uncommon. This is remarkable given that the liver and kidneys are considered target organs for A. hydrophila infections. 2
Splenic necrosis, varying from multifocal to severe, is a reported characteristic of A. hydrophila infection in channel catfish, both naturally and experimentally.10,11 Infection experiments have shown that the splenic ellipsoids are a primary area of bacterial sequestration, with marked macrophage infiltration and necrosis. 1 In cases of catfish infected with A. sobria (an opportunistic pathogen), there are often numerous bacteria with macrophage infiltration in the splenic ellipsoids, but in MASv, bacteria are rarely evident and inflammation occurs in only half of the cases. The most prominent finding in MASv-infected spleens was hemorrhage and necrosis. Given the degree of tissue damage, the relative paucity of bacteria is somewhat surprising; however, when splenic cultures were performed, MASv were isolated consistently. In VAh-infected spleens that have infarcts and have progressed well into decomposition (grossly tan and friable), large numbers of bacteria fill the dead tissue, indicating that A. hydrophila thrives in anaerobic tissue environments.
Case materials submitted from farm pond outbreaks are typically 1–2 catfish from a population of 90,000–150,000 fish, with clinical signs or mortality for 1–2 d, and only small numbers of live fish can be seen at the pond side. It is difficult to deduce reliably the progression of disease in this setting. Infection models for MAS in fish have been notoriously problematic, which has hampered efforts to resolve hypotheses about MAS disease progression. Use of a previously published MASv infection model based on fin clipping of catfish may address some of these etiologic questions. 12
A. hydrophila produces a wide variety of toxins, giving rise to a characteristic toxemia with abundant necrosis. 2 The VAh strain, which has an expanded arsenal of potential virulence genes, may produce an array of toxins different from isolates previously examined, and thus may also have a different mechanism of disease. 5 Experimental infections in channel catfish using A. hydrophila or VAh by immersion exposure have found that stress, skin injury, or a combination of both can induce mortality.9,12 The bacterium can be recovered from the gills, skin, and stomach or intestine throughout the first 24 h post-exposure, providing ample opportunity for infection by any of these routes.9,12 In the VAh immersion model, aside from the gill, bacterial numbers were highest in the intestine compared to all other tissues in the first hour post-exposure. Further, over the first 2 h post-exposure, bacterial numbers were higher in the intestine than in the skin (intact and fin-clip site). 13 By 4 h post-exposure, the spleen had significantly higher numbers of bacteria compared to other viscera. 13
By comparison, the cases described herein often had skin and gastrointestinal lesions. Mortalities in ponds range from 10% to 90%, which is similar to that seen experimentally.9,12,13 The course of disease in pond outbreaks was also probably rapid in these cases (within 48 h), as evidenced by a lack of a reactive renal hematopoietic hyperplasia in the affected fish; hyperplasia is relatively common in other gram-negative septicemias. The conspicuous involvement of the stomach, which has not been reported in MAS in catfish, may have some relation to the route of infection in MASv pond outbreaks. Although gross signs of gastric mucosal damage were not seen, microscopic injury could easily be missed, particularly because the cases examined had been presumably infected at least a day prior to presentation. Further, it is not uncommon to examine fish dying of gram-negative sepsis where the route of entry is unclear (personal observation). In our experience, the gastric changes seen in these cases are not characteristic of gram-negative sepsis in catfish. If infection did not occur through the stomach, perhaps the bacterium associates with the gastric vasculature via organ-specific ligands and/or receptors. The possibility of gastrointestinal infection in MAS in catfish has previously been established 9 and is an active area of research in fish species.6,14 Splenic changes were predominantly those of vascular necrosis and hemorrhage, with a relatively mild inflammatory component and few bacteria. These changes suggest a toxin-induced necrosis, which seems reasonable given the spleen’s role in blood filtration. It has been reported that, in naturally occurring MAS cutaneous disease in catfish, bacteria were not isolated from visceral organs, even though multiple necrotic foci were seen in the spleen. 11 The lack of lesions in the liver and mesonephros may also be the result of a lack of receptors for the bacterium or its toxins in these organs.
Deductions from the case materials here are limited; a relatively small number of fish were examined from outbreaks where the time course and route of infection were not known. Thus, the true prevalence of these lesions cannot be determined, nor can we confidently establish the true route of infection, whether by skin, gill, or gastrointestinal tract. Stress and injury in various forms are daily occurrences in catfish pond aquaculture, and more than one route of VAh infection may occur in any pond outbreak. However, our findings show that there are some differences between MASv and previous descriptions of MAS in experimental models and pond outbreaks, and these differences may have important implications for further research.
Footnotes
Acknowledgements
We thank Drs. Mark Lawrence and Lora Petrie-Hanson, as well as Beth and Mark Peterman for their assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
