Abstract
Histology is often underappreciated for the detection of the amphibian pathogenic fungus Batrachochytrium dendrobatidis, the cause of the potentially lethal skin disease chytridiomycosis. We evaluated the sensitivity of histology to detect chytrids in 20 wild specimens of 2 frog species from Uruguay that were clinically normal, but confirmed by PCR to be infected by B. dendrobatidis. We detected maturing and sporulated sporangia in 15 of 20 (75%) frogs, which is more sensitive than previously reported for histology. The effort needed to identify chytrids in histologic skin sections of Physalaemus henselii and Pleurodema bibroni required examination of 3.2 and 8.7 mm of skin sections for each frog species, respectively.
Chytridiomycosis, the skin disease of anuran amphibians caused by Batrachochytrium dendrobatidis, is potentially lethal in postmetamorphic life stages.3,14 It has been considered an emerging disease for the past 2 decades, given its apparent range expansion and increasing deleterious impact on several amphibians from temperate and tropical areas.12,13,19 Amphibian chytrids have a simple lifecycle with flagellated spores that develop small sporangia in the skin of adult frogs. 5 Common signs in postmetamorphic diseased amphibians are excessive ecdysis, cutaneous erythema and discoloration, mainly in ventral parts of the abdomen and thighs, and eventually death as a result of osmotic imbalance.4,20 Chytrid infection can be detected by histologic examination of the skin in non-larval stages, or the keratinized oral structures of larvae. 4 Defining histologic features are the presence of sporangia in the stratum corneum, measuring 6–15 µm, usually containing 4–10 basophilic spores of ~1–2 µm, and the observation of spore discharge tubes or the internal septa of empty sporangia. 4
Histology is viewed as a low-sensitivity method for the detection of B. dendrobatidis in amphibians. 11 Its value for the study of chytrid infection has been underappreciated, especially since the development of specific endpoint PCR protocols. 2 Further standardization of more sensitive, real-time PCR (rtPCR) has led some authors to consider rtPCR as the first choice in the study of B. dendrobatidis and chytridiomycosis because of both low detection threshold and cost-efficiency of the method. 11 Remarkably, amphibian pathogen surveys sometimes do not distinguish between B. dendrobatidis detection (i.e., subclinical infection) and the occurrence of disease. 18 Although clinical signs and histopathology are of key importance in making that distinction, only a few studies have compared the efficiency of histology and molecular methods to detect amphibian chytrids.6,9,11 We studied the sensitivity of conventional histology to identify B. dendrobatidis in wild specimens of 2 endangered frog species from Uruguay. We estimated the sampling effort needed to achieve positive detection of infection, and quantification of chytrids in microscopic skin sections.
We studied 20 adult frogs of species Physalaemus henselii and Pleurodema bibroni (anuran family Leptodactylidae, subfamily Leiuperinae). Specimens were captured in southern Uruguay between 2009 and 2012 during winter (June–September). Collection permits were issued by División Fauna/MGAP (Res. 18/09 and 01/10). Authorizations for implementation of research experimental protocols with live vertebrates were provided by Comisión Honoraria de Experimentación Animal, Universidad de la República.
Study specimens were captured while in reproductive activity, as part of other taxonomic and ecological studies on Uruguayan amphibians, and did not exhibit anomalous behavior or signs of disease. All 20 frogs were positive for B. dendrobatidis infection by means of PCR testing of skin swabs. Swabs were made individually after capture, by gently rolling a polyvinyl tipped swab (Puritan Medical Products, Guilford, ME) 5 times over the ventral surface of the abdomen and feet. 5 Samples were stored in dry tubes at −20°C, until DNA was extracted (DNeasy blood & tissue kit, Qiagen, Valencia, CA). Extracts were checked with B. dendrobatidis–specific endpoint PCR with primers Bd1a (5’-CAGTGTGCCATATGTCACG-3’) and Bd2a (5’-CATGGTTCATATCTGTCCAG-3’) designed from the first and second ribosomal internal transcribed spacer regions (ITS1 and ITS2), respectively. 2 We analyzed reactions by electrophoresis in polyacrylamide gels, stained with AgNO3. 17 The positive control was DNA extracted from an ethanol-fixed sample of B. dendrobatidis pure culture (strain JEL423, School of Biology & Ecology, University of Maine, Orono, ME). Serial dilutions of the positive control were used in triplicate to assess PCR sensitivity, which was ~10 pg of DNA, as described previously. 2 Reactions with 0.001, 0.002, 0.005, 0.01, 0.1, 0.2, 0.5, and 1 pg of template DNA were all negative, as well as one reaction with 10 pg; remaining reactions with 10, 20, 50, and 100 pg were positive. We further tested the quality of reactions with a random sample of amplicons that were cleaned (QIAquick PCR purification kit, Qiagen), and submitted to the Institut Pasteur at Montevideo for sequencing in both senses; GenBank accessions MH644192–MH644195, MH644199, and MH644196–MH644198 were submitted for B. dendrobatidis samples of P. henselii and Pl. bibroni, respectively.
After swabbing, we euthanized the frogs by cutaneous application of 20% lidocaine gel, 7 and fixed the frogs for 5–10 d in 10% neutral-buffered formalin. For histologic analysis, we dissected a small rectangular skin strip of ~1–5 × 2–3 mm from the caudal abdominal region (pelvic patch) of each specimen. Fixed samples were processed routinely, sectioned at 5 µm, and stained with hematoxylin and eosin (H&E). We prepared 2 or 3 slides from each specimen, taking from a given block 1 slide every 4 tissue sections, which were separated from each other at least 15 µm, to produce independent samples. We analyzed 344 skin sections under a binocular microscope (E100, Nikon, Tokyo, Japan); each section was photographed (Micrometrics LE, ACCU-SCOPE, Commack, NY). Because histologic detection of chytrids may target the stratum corneum, which is usually only a few cells thick, the length of analyzed skin preparations was measured on image files (Image J 1.45, https://imagej.nih.gov/ij/download.html). 1 Skin sections measured on slides were 0.8–4.6 mm (x– = 2.6 ± 0.8), accounting for 424.0 and 478.5 mm of skin from P. henselii and Pl. bibroni, respectively. The same observer (C Borteiro) conducted randomized observations of slides at 1,000×, recording for each specimen the number of skin sections and the corresponding mm of skin needed to detect B. dendrobatidis sporulated sporangia, and also the total number of B. dendrobatidis regardless of maturity stage. Although usually regarded as indicative of infection, B. dendrobatidis empty sporangia were not considered, to avoid possible misidentification of tissue structures or artifacts (Fig. 1). We calculated proportion confidence interval (CI) for the frequency of infection, 21 and minimum sample number to achieve detection. 8
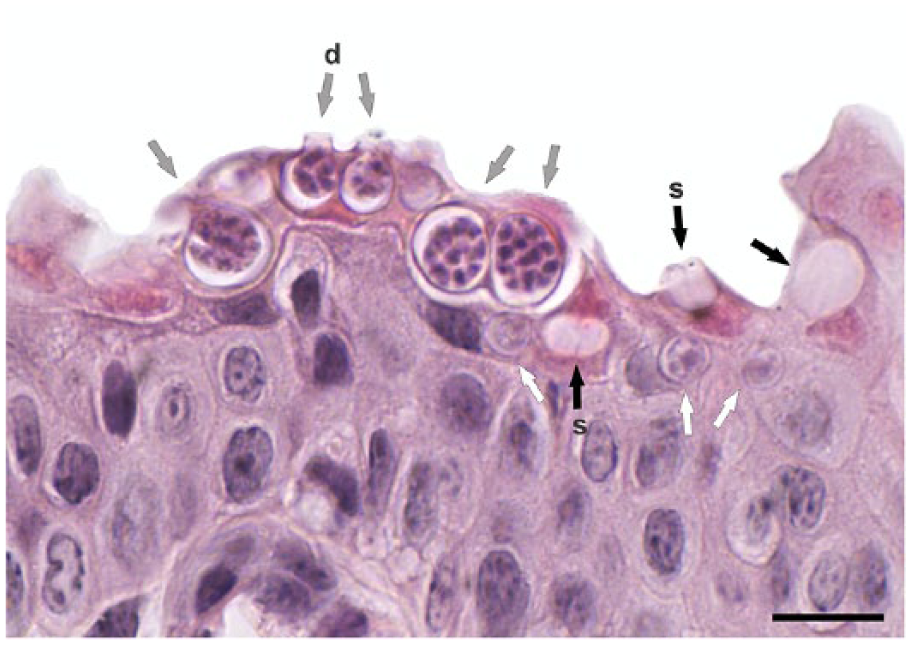
Skin infection by the amphibian chytrid fungus Batrachochytrium dendrobatidis in Physalaemus henselii. H&E. Arrows indicate sporangia at different developmental stages: immature (white), sporulated (gray), and empty (black). Note the spore discharge tubes (d) and septum (s). Bar = 20 μm.
Histology revealed B. dendrobatidis sporulated sporangia in 15 of 20 frogs: 7 P. henselii and 8 Pl. bibroni (p = 0.75, 95% CI: 0.51–0.91; Table 1). In most specimens, we identified infection after analyzing just 1 or 2 skin sections, corresponding on average to 6.1 mm of skin. Six P. henselii (all males) were positive when the first skin section was analyzed, which involved on average 3.2 mm of skin. Similarly, 5 Pl. bibroni (1 of them female) were positive at inspection of the first or second skin section, but representing on average 8.7 mm of skin. Nearly half of the skin sections were positive in both frog species (156 of 346), accounting for a detection probability of 0.45 in the whole sample. The estimated minimum number of sections required to identify B. dendrobatidis infection are 4 and 6, for 90% and 95% CIs, respectively. A sampling effort of 6 sections would imply the preparation of at least 13.2 mm of skin for P. henselii and 19.0 mm for P. bibroni.
Histologic detection of Batrachochytrium dendrobatidis infection (+) in the frogs Physalaemus henselii (Ph) and Pleurodema bibroni (Plb).
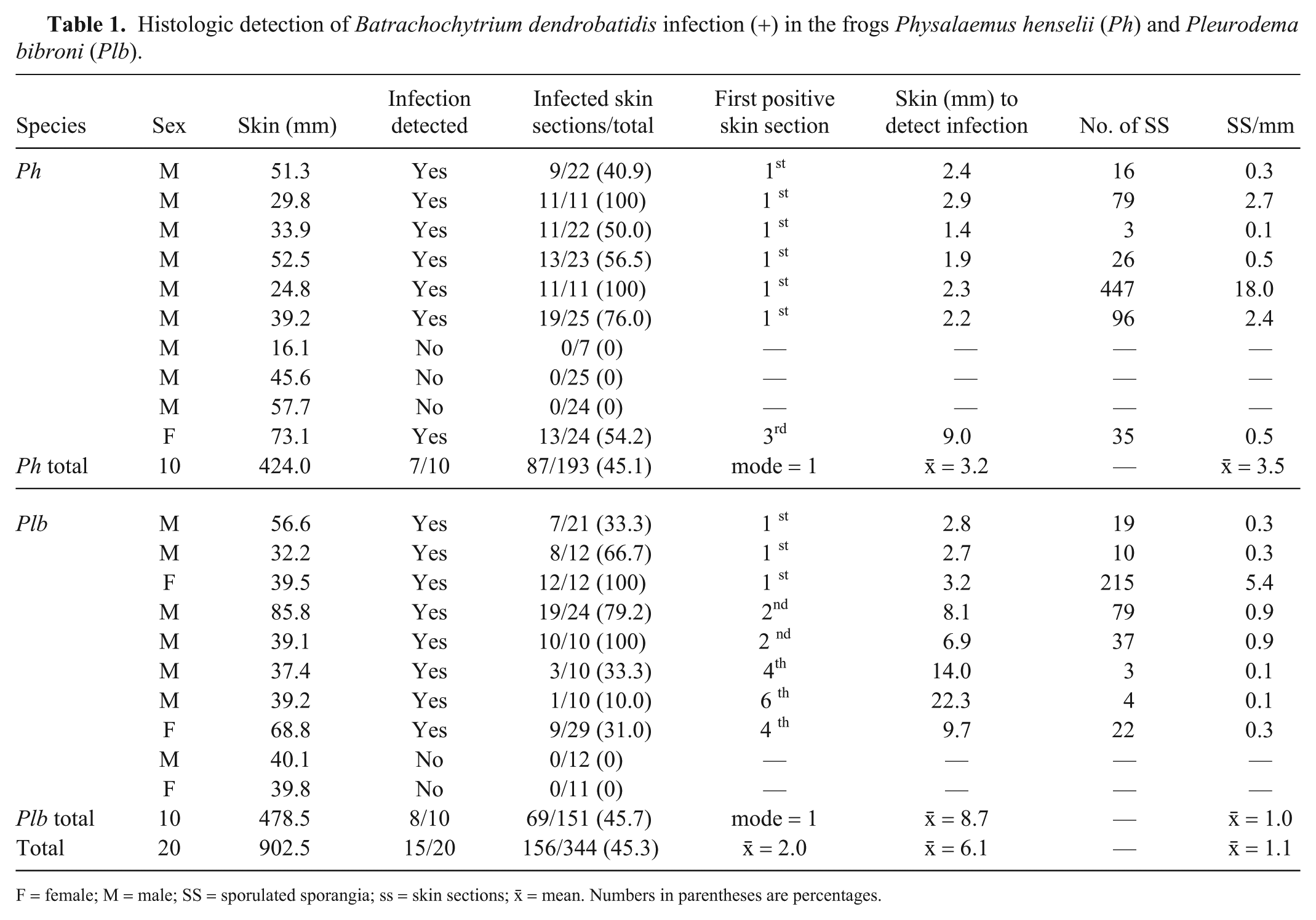
F = female; M = male; SS = sporulated sporangia; ss = skin sections; x̄ = mean. Numbers in parentheses are percentages.
Both frog species studied herein have declined in Uruguay since the mid-1970s, and chytridiomycosis is one of the possible factors involved. 10 However, the studied specimens did not have clinical signs of disease or severe skin disruption on histologic examination, even at higher pathogen loads. The higher relative counts of sporulated sporangia, and the lower extent of skin necessary to detect them in P. henselii, could be the result of behavioral differences between these pond breeders. Reproductive activity of Pl. bibroni is mostly explosive after heavy rains, whereas males of P. henselii call to attract females semi-submerged in shallow ponds throughout the reproductive season, likely enhancing their encounter probability with zoospores.
We compared B. dendrobatidis detection using histology and conventional endpoint PCR; further comparison with rtPCR was out of the scope of our work. Furthermore, high B. dendrobatidis load rtPCR results may present a variable correlate with positive histologic detection. 6 Proper interpretation of quantitative chytrid load in amphibian skin provided by rtPCR would imply laborious B. dendrobatidis characterization, given that the number of ITS copies varies greatly among strains. 15 Sensitivity tests of histology to detect infection by B. dendrobatidis have only been available from work on frogs in Australia, which also included comparison with rtPCR.6,9,11 In a survey of 101 wild juveniles of Mixophyes iteratus, B. dendrobatidis infection was identified by H&E in only 1 frog, which was negative on rtPCR, whereas rtPCR detected the pathogen in 23 (22.8%) individuals. 11 Another study found that, after 28 d of experimental infection, all but one toe clips of 27 Mixophyes fasciolatus were positive on rtPCR, but only 14 (54%) frogs were infected using the immunoperoxidase (IPX) histologic method. 6 In a third study with this same frog species, by 42 d of B. dendrobatidis inoculation, the sensitivity of rtPCR detection on toe clips was 87% and that of histology accounted for 57% and 50% when using IPX or H&E, respectively. 9
The sensitivity of our histologic examination to detect chytrid infection in P. henselii and Pl. bibroni was far higher than that of previous reports using H&E or IPX. We achieved 75% sensitivity targeting live sporangia, although we avoided the consideration of “suspicious” chytrid images, a difficulty already noted for the histologic identification of B. dendrobatidis.11,16 Our results indicate that conventional histology could be a sensitive method to detect B. dendrobatidis infection in wild frog populations. Amphibian chytrids are widespread in Uruguay, and high prevalences are common during winter, which would facilitate detection by histology. For instance, in a previous study, the frequency of infection in adults of the studied species tested by endpoint PCR were 33.3% (n = 30) and 53.2% (n = 47), in P. henselii and Pl. bibroni, respectively, in 2009–2012 (Borteiro C. [Skin diseases of amphibians from Uruguay and southeastern Brazil: new diagnoses and potential effects] [PhD dissertation]. Facultad de Ciencias, Universidad de la República, Montevideo, Uruguay, 2016. Spanish).
Our sampling method involved the dissection of ventral skin strips, avoiding the processing delay associated with demineralization needed for toe clips. 4 However, it would be useful only in dead specimens. Histology would be a valuable tool when PCR methodology is not available. Future studies should consider the histologic quantification of sporulated sporangia to distinguish between asymptomatic B. dendrobatidis infection (as in our study), and the occurrence of chytridiomycosis.
Footnotes
Acknowledgements
We thank Mailén Arleo, Antonio Moraña, and María Jesús Sabalsagaray for their valuable support at the laboratory, and Joyce E. Longcore who kindly provided the sample of strain JEL423. The suggestions of the editor and 2 anonymous reviewers greatly improved our work.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was funded in part by PEDECIBA, Universidad de la República, and Agencia Nacional de Investigación e Innovación (ANII), Uruguay.
