Abstract
Testicular atrophy associated with biliary obstruction in chickens, produced by the ligation of both extrahepatic bile ducts, was examined grossly, histologically, immunohistochemically, and ultrastructurally. Grossly, reduction in testicular size and volume was evident in chickens that underwent bile duct ligation (BDL). Histologically, there was marked reduction in tubular diameter, peritubular fibrosis, loss of spermatogenic cells, and tubules lined only by Sertoli cells. In addition, Leydig cells, which accumulated in the inter-stitium of the testes, contained numerous large lipid vacuoles, as determined by electron microscopy. These features suggest that BDL in chickens causes hypogonadism and low serum testosterone.
In humans, complications of liver cirrhosis and chronic cholestatic liver disease lead to hypogonadism, metabolic bone loss, loss of muscle bulk, and alterations in thyroid hormone regulation and metabolism. 2,4,6,12 The pathogenesis of these changes is not completely understood.
Bile duct ligation (BDL) has been used in animal models to cause liver fibrosis and cirrhosis. Histologically, BDL can induce chronic cholestatic liver dysfunction, jaundice, proliferation of the intrahepatic bile ducts (ductular reaction), and liver fibrosis. 11 Prepubertal male rats that underwent BDL showed a reduction in testis size. 13 We found no reports on bile-duct–ligated chickens. In the present study, testicular atrophy associated with BDL in chickens was examined grossly, histologically, immunohistochemically, and ultra-structurally.
Nine 1-year-old male broiler chickens were used in this study. In six chickens, both extrahepatic bile ducts were ligated. 3 At 5 (n = 3) and 10 (n = 3) weeks after BDL, the chickens were bled and killed under ether anesthesia. The remaining three, in which bile ducts were left intact as controls, were also killed under ether anesthesia at the same time as were the chickens 10 weeks after BDL. Just before the killing, sera samples were collected from the heart under ether anesthesia, and tissue samples were collected from the control chickens at 5 and 10 weeks after BDL in the experimental groups. The serum was stored at −80 C until assayed for testosterone levels. 8 After surgery, controls and BDL chickens were housed in individual cages under standard temperature of 18 ± 1 C, lighting conditions of 12 hours light-dark cycle, and light intensity of 10 lux, and were fed a standard chicken chow ad libitum and given free access to water.
Testicular specimens were fixed with 10% neutral-buffered formalin, routinely processed, and embedded in paraffin. Tissue sections, 4–5 µm thick, were cut, stained by routine methods with hematoxylin and eosin, with Azan stain to detect collagen, and with immunohistochemistry. For Azan stain, sections were deparaffinized in xylene, dehydrated, rehydrated in distilled water, immersed in 5% potassium dichromate solution for 30 minutes, and stained with azocarmin G for 30 minutes. Sections were then immersed in 3% 12 tungsto(IV)phosphoric acid n -hydrate solution for 1 hour and stained with aniline blue–orange G for 20 minutes. For immunohistochemistry, peroxidase-labeled antibody methods were performed using anti–α-smooth muscle actin (α-SMA) monoclonal mouse serum (Nichirei Co., Tokyo, Japan) as the primary antibody. Sections were mounted on silanized slides (Dako Cytomation Co., Kyoto, Japan) and dried at 37 C for 24 hours and were deparaffinized in xylene, dehydrated, and rehydrated in distilled water for 5 minutes. Endogenous peroxidase activity was blocked in 3% hydrogen peroxide diluted in methanol for 30 minutes followed by three 5-minute rinses with 0.1 M phosphate buffer (pH 7.4). Sections were incubated with an undiluted primary antibody, described above, for 12 hours at 4 C and then using peroxidase-conjugated anti-mouse IgG (Nichirei Co., Tokyo, Japan) as secondary antibody for 1 hour at 37 C. Diaminobenzidine (Nichirei Co.) was used as a chromagen. Sections were counterstained with Mayer's hematoxylin and cover-slipped. For electron microscopy, tissues were fixed with 0.1 M phosphate-buffered glutaraldehyde for 24 hours, postfixed with 1% osmic acid for 2 hours, dehydrated, and embedded in epoxy resin. The embedded specimens were sectioned at 1 µm and stained with toluidine blue for light microscopic observation. Ultrathin sections were stained with uranyl acetate and lead citrate and were examined using a transmission electron microscope (H-7000, Hitachi Ltd., Tokyo, Japan). For morphometric analyses, diameter of the seminiferous tubules was calculated, using a software (Motic Images 2000, Shimadzu Co., Tokyo, Japan), as the average of 100 tubules that were randomly selected on light microscopical sections in each experimental and control group. The Student's t -test (two tailed) was used for all statistical comparisons in serum testosterone level and seminiferous tubular diameter. A P value less than 0.05 was considered significant.
After surgery, all chickens in the experimental groups lost weight, had jaundice in the skin and diarrhea, but intake of feed or water was not different compared with controls. Gross lesions consisted of reduction in testicular size and volume in BDL chickens as compared with controls (Fig. 1). At 10 weeks after BDL, atrophic testes were pale and sclerotic. Serum testosterone levels were significantly reduced (P < 0.05) in BDL chickens compared with controls (Table 1).
Serum testosterone in the three experimental groups.

∗ Statistically significant (P < 0.05) compared with the control group.
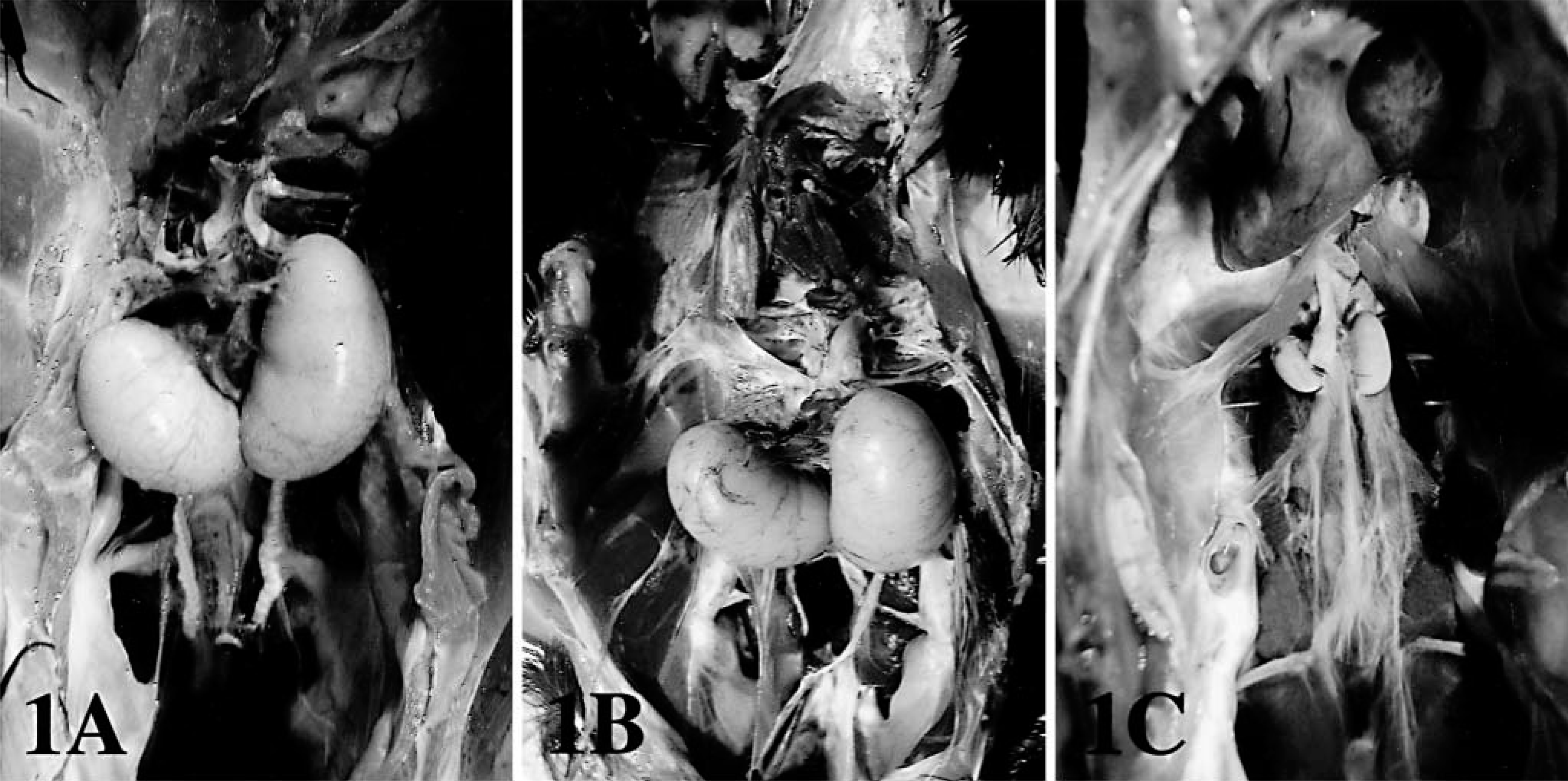
Testes; chicken.
Histopathologically, BDL chickens showed liver fibrosis with proliferation of the intrahepatic bile ducts and obstructive cholestasis. There was testicular atrophy, with a marked decrease in tubular diameter (P < 0.05) and interstitial tissue expansion (Table 2, Fig. 2). Spermatogenesis was absent in BDL chickens, and only Sertoli cells were present in seminiferous tubules. Spermatogenic cells (spermatogonia, spermatocytes, spermatid, and spermatozoa) were absent. In the interstitium of the testis, accumulation of Leydig cells and collagen fibers that stained blue with Azan were observed. In addition, the myoid cells, which were positive to α-SMA antibody by immunohistochemistry, were observed around the seminiferous tubules. Ultrastructurally, various stages of spermatogenesis were observed in the seminiferous tubules of controls. Oval spermatogonia were connected to the basement membrane and contained few cytoplasmic mitochondria and free ribosomes. Leydig cells were scattered in the interstitium and contained numerous cytoplasmic organelles, including mitochondria with tubular cristae and smooth endoplasmic reticulum. In the BDL chickens, spermatogonia were detached from the basement membrane, pyknotic, and contained various-sized cytoplasmic vacuoles (Fig. 3). Leydig cells were vacuolated, and cytoplasmic organelles were scarce (Fig. 4).
Seminiferous tubular diameter in the three experimental groups of chickens.

∗ Statistically significant (P < 0.05) compared with the control group.
∗∗ Statistically significant (P < 0.05) compared with the 5-weeks after BDL group.
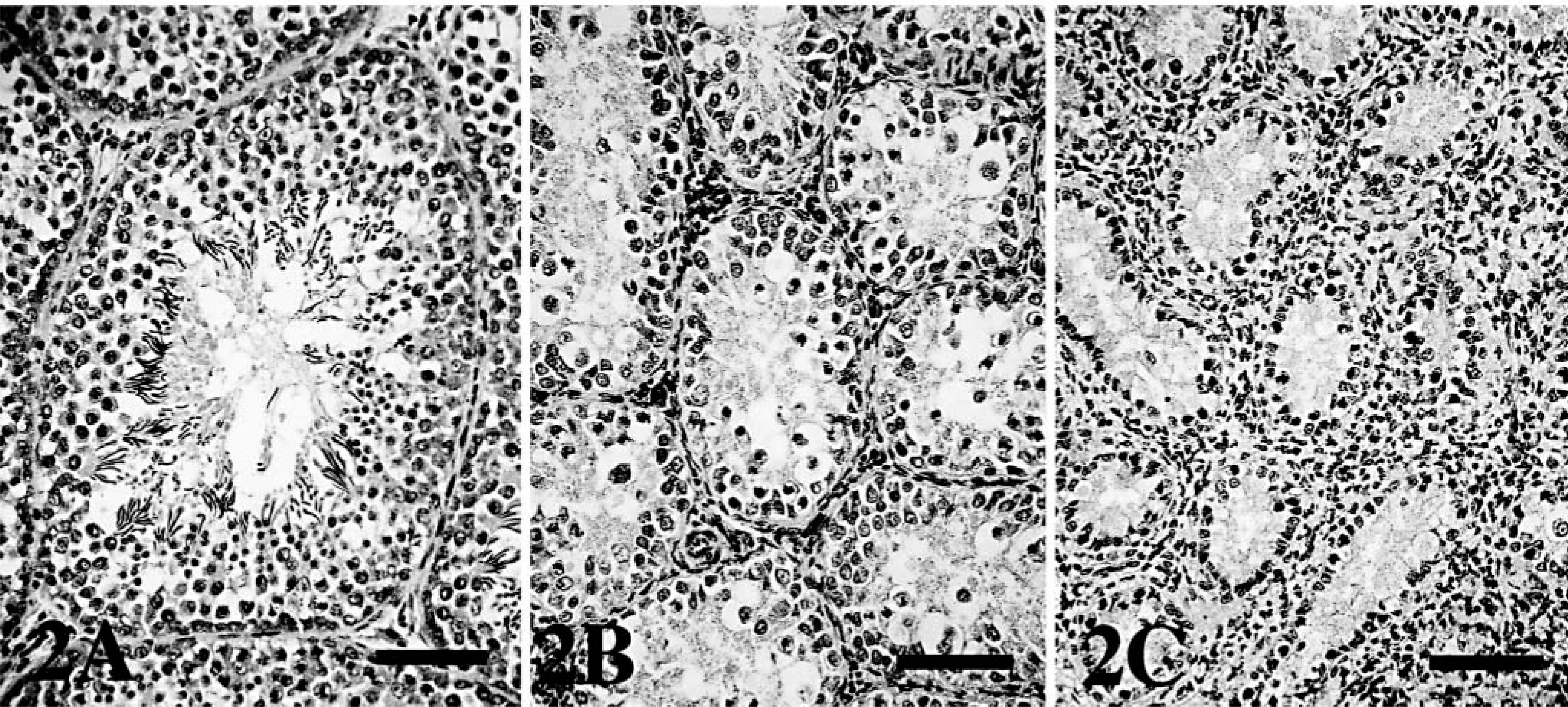
Testes; chicken.
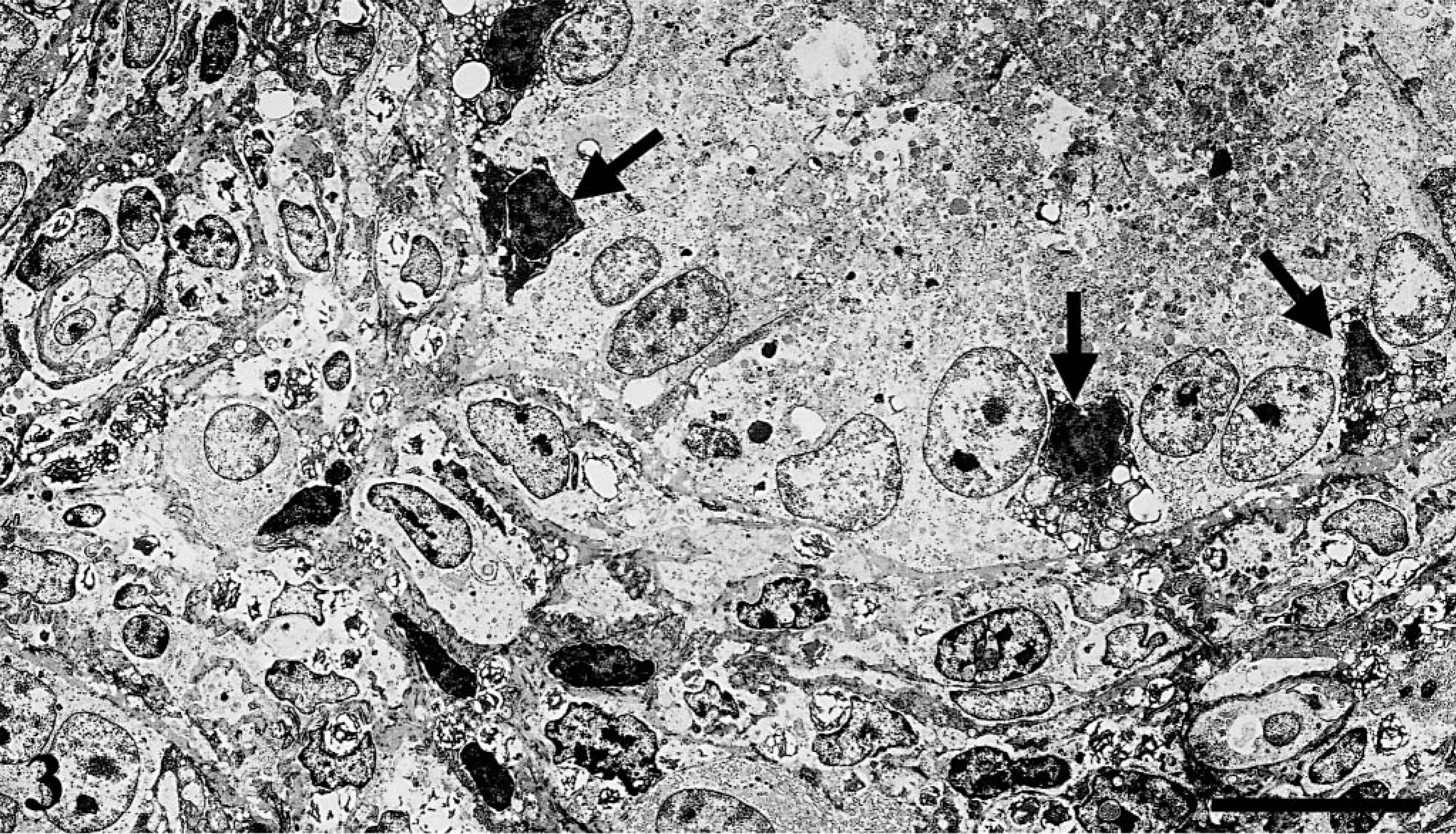
Electron micrograph, seminiferous tubules; chicken. Ten weeks after BDL, degenerative spermatogonia (arrows) are pyknotic. Bar = 10 µm.
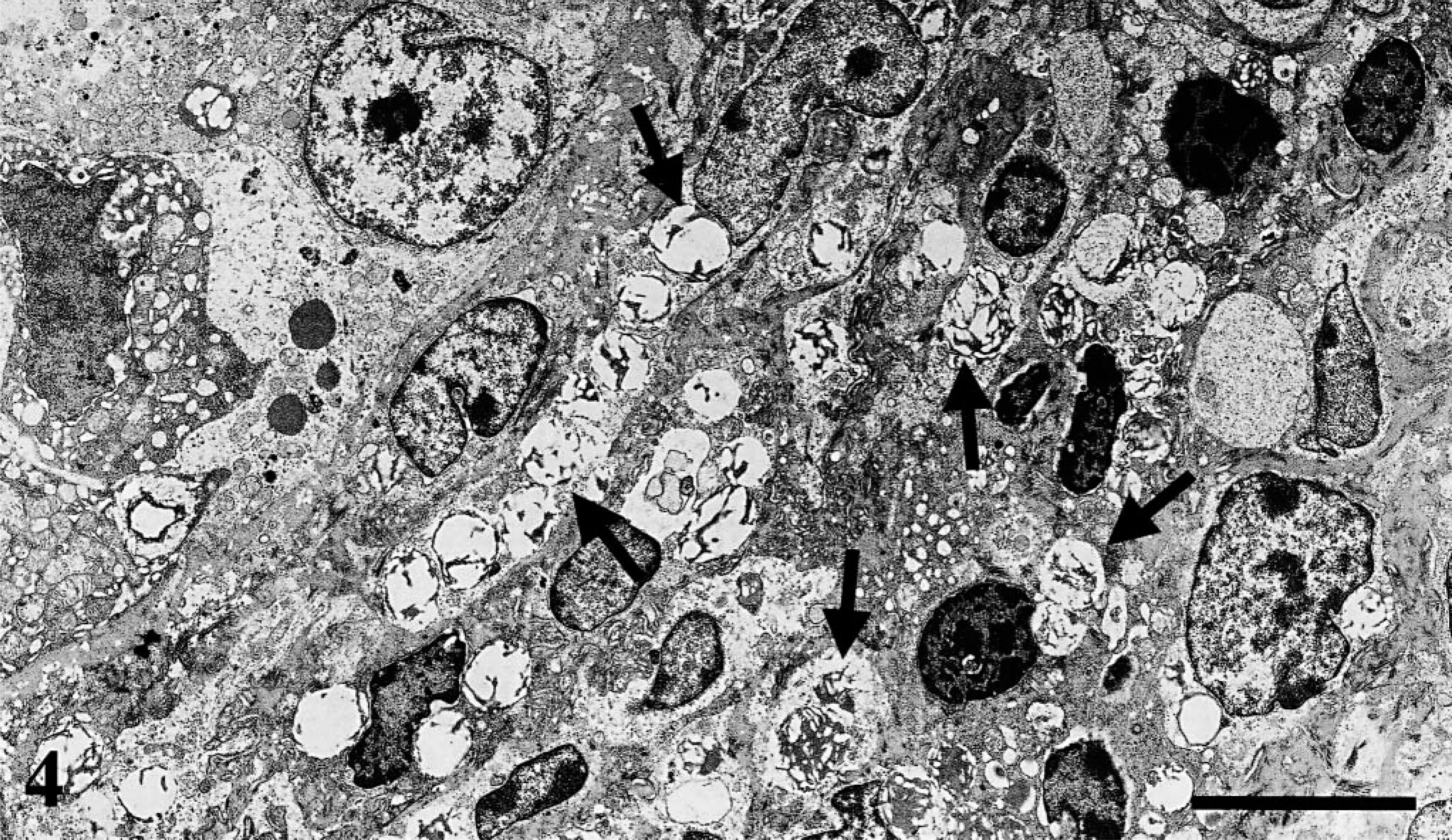
Electron micrograph, testis; chicken. Ten weeks after BDL, Leydig cells contain numerous vacuoles (arrows). Bar = 5 µm.
Our study demonstrated that chickens with liver fibrosis secondary to biliary ligation had severe testicular atrophy. Histopathologic observations suggest that the testis, after BDL, shows hypogonadism, and were similar to those observed in cirrhosis and portocaval shunt. 12,14 Testicular atrophy and hypogonadism as a result of advanced cirrhosis is well documented in humans. However, experimental models of hypogonadism associated with liver injury are few. Testicular atrophy has been noted in one report 13 in which pre-pubertal male rats that underwent BDL had a reduction in testis size. These data suggest that chronic cholestasis contributes, at least in part, to the pubertal and maturational failure that occurs with the chronic cholestatic diseases of childhood. Portocaval shunts in rats are reported to cause testicular atrophy. 14 This atrophy, manifested histologically by the loss of germinal epithelium, is due to decreased mitosis and increased apoptosis, loss of spermatogonia and spermatozoa in the lumen of the seminiferous tubules, and eventual atrophy of the seminiferous tubules leaving them lined only by Sertoli cells.
In this study, accumulation of Leydig cells in the interstitium of the testis was observed in the BDL chickens. Ultra-structurally, the cells contained numerous vacuoles, consistent with lipid. This feature has been reported in Leydig cells in Klinefelter's syndrome in humans, and ultrastructural studies revealed four Leydig cell types. 9 The multivacuolated Leydig cells that accumulate testosterone precursors in the form of lipid droplets are considered to be involuted Leydig cells incapable of synthesizing testosterone. 9 In this study, we suggest that the vacuolated cells were involuted Leydig cells, which caused the low serum testosterone. Multivacuolated Leydig cells have not been reported previously in hypogonadism associated with BDL.
Regarding the relationship between testicular atrophy and liver injuries, administration of insulin-like growth factor-I (IGF-I) reverses testicular atrophy and improves testicular function in a carbon tetrachloride model of cirrhosis in the rat. 1 IGF-I is a member of a family of insulin-related peptides that mediate the growth-promoting actions of growth hormone. 5 The liver is the principal source of circulating IGF-I and circulating IGF-I–binding proteins, which stimulate testosterone synthesis and spermatogenesis. 7,10 Perhaps, IGF-I plays a role in the pathogenesis of the hypogonadism in chickens after BDL.
In conclusion, chickens with liver fibrosis after BDL showed testicular atrophy and hypogonadism, and Leydig cell involution caused low serum testosterone.
