Abstract
The pathogenicity induced by co-challenge with the rB strain of very virulent Infectious bursal disease virus (vvIBDV) and IBDV pathotypes endemic in the United States was evaluated in specific pathogen–free chickens. Four- and 6-week-old birds were simultaneously challenged with a 105 50% egg infectious dose (EID50) of rB mixed with a 105 EID50 of one of the following viruses: standard classic (STC), subclinical variant (Del-E), subclinical variant (T1), or avirulent serotype 2 (OH). Each challenge group consisted of 5 chickens. The severity of disease was assessed by comparing the 5-day mortality rates, bursal lesions (mean bursal lesion scores), and mean bursal-to-body weight ratios in each of the challenged groups. A mortality of 100% (10/10 and 5/5) was observed in birds inoculated with only the vvIBDV (rB) strain at 4 weeks and 6 weeks of age, respectively. Although the sample sizes were low, a significant reduction in mortality and severity of disease, based on mean bursal lesion scores, was observed in groups co-challenged with rB and the less virulent pathotypes Del-E, T1, or OH at 4 weeks of age. Co-challenge with rB and the antigenically similar STC strain did not result in a significant decrease in mortality compared to challenge with the pathogenic rB strain at 4 weeks of age, but a significant reduction in the mean bursa lesion score was observed. At 6 weeks of age, a significant decrease in mortality and mean bursa lesion score was observed in the rB groups co-challenged with STC, Del-E, or T1 but not OH.
Introduction
Infectious bursal disease virus (IBDV), an immunosuppressive virus targeting primarily B-lymphocytes in the bursa of Fabricius of chickens, are nonenveloped, bisegmented RNA virus strains belonging to the Avibirnavirus genus within the Birnaviridae family. 24 Of the 2 distinct serotypes of IBDV distinguishable by virus neutralization and cross-protection studies, only serotype 1 strains are pathogenic in chickens. 14
A range of pathogenicity is observed with IBDV with the most virulent strains being designated as “very virulent.” 26 IBDV strains are generally subdivided as avirulent, subclinical, classical virulent (also referred to as standard), and very virulent (vv) pathotypes. 16
Criteria for designation of IBDV strains as the vvIBDV pathotype are based on a combination of molecular, antigenic, and phenotypic characteristics. 27 Phylogenetic analyses indicate that vvIBDV strains evolved from a common lineage and exhibit a high degree of genetic and antigenic homogeneity. 8 The vvIBDV designation is usually reserved for viruses causing higher mortality than classic strains. The reported mortality induced by vvIBDV can range between 40–100% in fully susceptible specific pathogen–free (SPF) chickens, 60% in layers, and 30% in broilers.17,25,26 Some, but not all, of the genetic determinants of pathogenicity have been elucidated. Both segments A and B of the IBDV genome are thought to influence pathogenicity.5,9,10 Highly conserved sequences have been observed in the hypervariable (hv) region of the viral protein 2 (VP2) of vvIBDV strains. Four amino acids in the hvVP2 at positions 222 (Ala), 256 (Ile), 294 (Ile), and 299 (Ser) are highly correlated with the vvIBDV pathotype.3,27 Using monoclonal antibody panels, vvIBDV strains tested were antigenically similar to classic serotype 1 strains with only minor variations noted.17,22
The vvIBDV pathotype was first reported in Belgium in the late 1980s and has since rapidly spread throughout Europe, Middle East, Asia, and South America. Previous to the emergence of vvIBDV, less virulent strains of IBDV were well controlled by vaccination, and mortality of less than 2% was typically observed in the field. 23 The vv pathotype of IBDV, as exemplified by the rA and rB strains, was first isolated in the United States in commercial poultry in California in December 2008. 21 The rA and rB vvIBDV strains resulted in spiking mortality of 26% and 34%, respectively, in affected layer flocks. 21 Despite ongoing continuous surveillance for vvIBDV in California poultry, detection of vvIBDV has only been found in distinct geographical areas in California since 2008 (B. Crossley, personal communication, 2012). The distinct geographical separation is contradictory to the typical explosive expansion and establishment of the vvIBDV strain in other geographical regions. 23 The purpose of the current investigation was to determine if coinfection with other endemic IBDV strains could alter the severity of clinical disease typically associated with the vvIBDV pathotype. The present in vivo challenge study investigated the pathogenicity associated with coinfection of SPF chickens with vvIBDV (rB strain) and one of the non-vvIBDV strains, namely, standard classic (STC strain), subclinical Delaware E (Del-E), variant strain (T1), or avirulent serotype 2 Ohio strain (OH), in 4- and 6-week-old SPF chickens.
Materials and methods
Viruses
The vvIBDV (rB strain) was initially isolated from commercial pullets in California from the first reported case of vvIBDV in the United States in December 2008.17,21 The standard classic strain, avirulent serotype 2 OH strain, and subclinical variants Del-E and T1 were previously described. 15 These viruses were selected for co-challenge with vvIBDV (rB strain) based on their endemicity in U.S. poultry flocks.
Experimental design
The 50% egg infectious dose (EID50) for viruses used for challenge was determined by viral titrations in 9-day-old embryonated SPF chicken eggs via the chorioallantoic membrane with 0.1 ml of bursal homogenate as previously described. 17 The SPF chickens were divided into 5 challenge groups (1a–e) and 1 uninoculated negative control group, 1f (Table 1). Five birds were allotted to each group except for positive control group 1e, which consisted of 10 birds (Table 1). Four-week-old birds were simultaneously challenged via the oral-nasal route, as the oral cavity in poultry is connected to the nasal cavity by the choanal slit. A 105 EID50 dose of vvIBDV (rB strain) mixed with a 105 EID50 dose of one of the following viruses was used: standard classic IBDV (STC strain), subclinical variant IBDV (Del-E strain), subclinical variant IBDV (T1 strain), or avirulent serotype 2 IBDV (OH strain). Each dual challenge group consisted of 5 chickens each. The experiment was repeated in 6-week-old SPF birds divided into 5 challenge groups (2a–e) and 1 negative control group, 2f. The experimental protocol was similar to that used for the 4-week-old birds except that 5 birds were placed in the positive control group 2e and 3 birds in the negative control group 2f (Table 2). Each experimental group was housed separately in environmentally controlled isolation units a and given feed and water ad libitum.
Pathology observed with 4-week-old specific pathogen–free chickens co-challenged with very virulent Infectious bursal disease virus (vvIBDV; rB strain) and a second IBDV pathotype as compared to vvIBDV (rB) alone.*
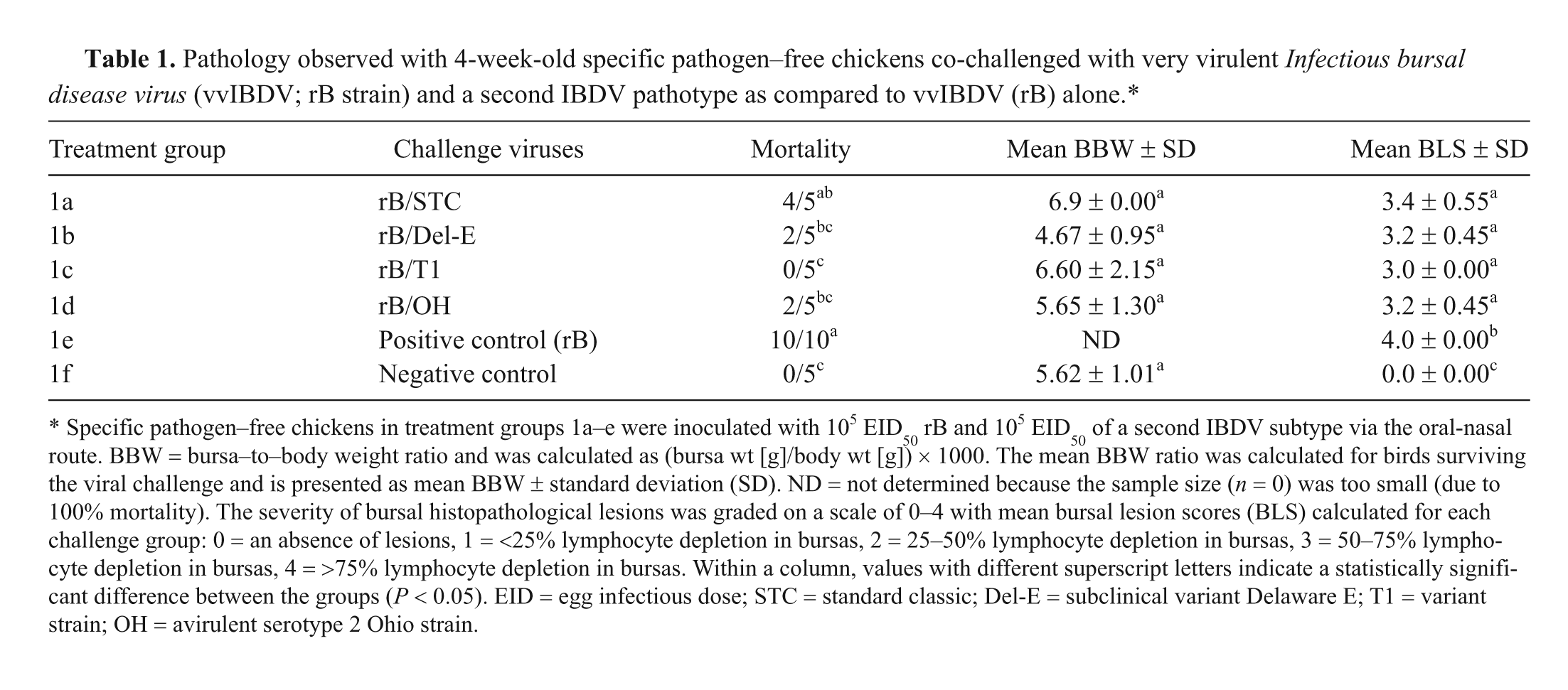
Specific pathogen–free chickens in treatment groups 1a–e were inoculated with 105 EID50 rB and 105 EID50 of a second IBDV subtype via the oral-nasal route. BBW = bursa–to–body weight ratio and was calculated as (bursa wt [g]/body wt [g]) × 1000. The mean BBW ratio was calculated for birds surviving the viral challenge and is presented as mean BBW ± standard deviation (SD). ND = not determined because the sample size (n = 0) was too small (due to 100% mortality). The severity of bursal histopathological lesions was graded on a scale of 0–4 with mean bursal lesion scores (BLS) calculated for each challenge group: 0 = an absence of lesions, 1 = <25% lymphocyte depletion in bursas, 2 = 25–50% lymphocyte depletion in bursas, 3 = 50–75% lymphocyte depletion in bursas, 4 = >75% lymphocyte depletion in bursas. Within a column, values with different superscript letters indicate a statistically significant difference between the groups (P < 0.05). EID = egg infectious dose; STC = standard classic; Del-E = subclinical variant Delaware E; T1 = variant strain; OH = avirulent serotype 2 Ohio strain.
Pathology observed with 6-week-old specific pathogen–free chickens co-challenged with very virulent Infectious bursal disease virus (vvIBDV; rB strain) and a second IBDV pathotype as compared to vvIBDV (rB) alone.*
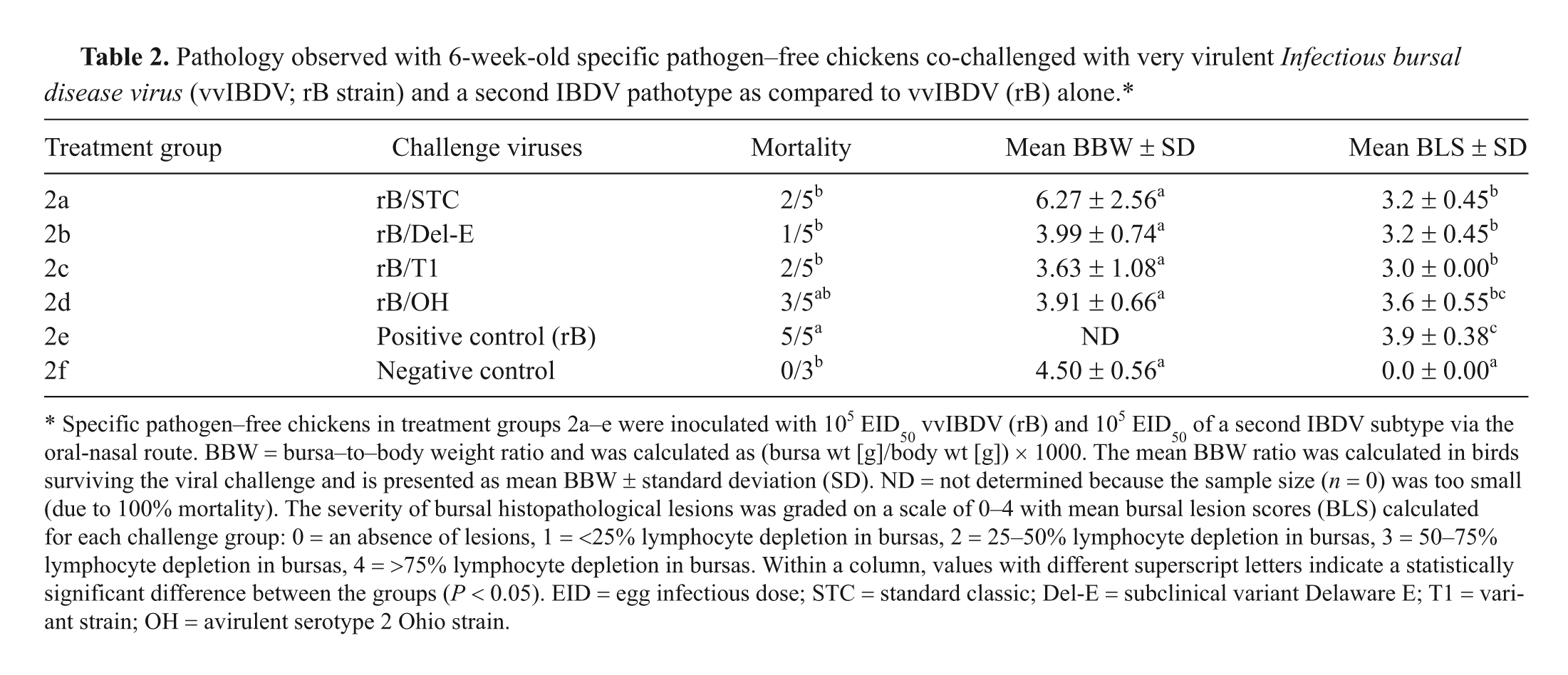
Specific pathogen–free chickens in treatment groups 2a–e were inoculated with 105 EID50 vvIBDV (rB) and 105 EID50 of a second IBDV subtype via the oral-nasal route. BBW = bursa–to–body weight ratio and was calculated as (bursa wt [g]/body wt [g]) × 1000. The mean BBW ratio was calculated in birds surviving the viral challenge and is presented as mean BBW ± standard deviation (SD). ND = not determined because the sample size (n = 0) was too small (due to 100% mortality). The severity of bursal histological lesions was graded on a scale of 0–4 with mean bursal lesion scores (BLS) calculated for each challenge group: 0 = an absence of lesions, 1 = <25% lymphocyte depletion in bursas, 2 = 25–50% lymphocyte depletion in bursas, 3 = 50–75% lymphocyte depletion in bursas, 4 = >75% lymphocyte depletion in bursas. Within a column, values with different superscript letters indicate a statistically significant difference between the groups (P < 0.05). EID = egg infectious dose; STC = standard classic; Del-E = subclinical variant Delaware E; T1 = variant strain; OH = avirulent serotype 2 Ohio strain.
Birds were monitored daily, and morbidity and mortality were recorded. At 5 days postchallenge, surviving birds from all experimental groups were humanely euthanized and necropsied. At necropsy, gross lesions were recorded along with bursa and body weights from euthanized birds. The bursa–to–body weight (BBW) ratio ([bursal weight (g)/body weight (g)] × 1000) was calculated from euthanized birds only and not birds that died during the course of the experiment, as dehydration of carcasses from birds that died would have altered the validity of the results. Tissue sections from the bursa, spleen, and proventriculus were taken from all birds for histology. Bursa tissue sections from all birds were also taken for molecular detection of IBDV.
Histology
Tissue sections from bursa, spleen, and proventriculus were fixed in 10% neutral buffered formalin, sectioned at 4 μm, stained with hematoxylin and eosin, and examined by light microscopy. The severity of lymphocyte depletion in bursal follicles was quantified by assigning bursal lesion scores (BLS) ranging from 0 to 4: 0 = an absence of lesions; 1 = <25% lymphocyte depletion in bursas, 2 = 25–50% lymphocyte depletion in bursas, 3 = 50–75% lymphocyte depletion in bursas, and 4 = >75% lymphocyte depletion in bursas.
RNA extraction from bursas
Approximately half of the bursa of Fabricius of each bird was collected at necropsy and homogenized using an equal volume (v/v) of TNE buffer (10 mM of Tris–HCl [pH 8.0], 100 mM of NaCl, 1 mM of ethylenediamine tetra-acetic acid) added to the bursa samples. A 300-μl volume was used to extract viral RNA with a commercial reagent b using a standard procedure previously described. 18 The RNA extracted from each sample was suspended in a 35-μl volume of 90% dimethyl sulfoxide c and used for reverse transcription polymerase chain reaction (RT-PCR) assays.
Real-time RT-PCR amplification
Real-time RT-PCR assays were performed using a real-time instrument d and a commercial RT-PCR kit. e A real-time RT-PCR assay was used for the detection of a portion of genome segment A of serotype 1 IBDV strains. 18 Briefly, 1.0 μl of RNA extracted from the bursa was used for the assay. A 743-bp region of hvVP2 from nucleotides 737–1479 was amplified using primers 743-F (5’-GCCCAGAGTCT ACACCAT-3’) and 743-R (5’-CCCGGATTATGTCTT TGA-3’). Two probes f were used to identify and differentiate the amplified RT-PCR products. One probe was specific to vvIBDV (FAM-5’-CTCAGCTAATATCGATGC-3’), and the other probe was specific for non-vvIBDV (VIC-5’-CAGCCAACATTGATGC-3’). The complementary DNA (cDNA) synthesis and pre-denaturation were performed at 48°C for 10 min and at 95°C for 10 min, respectively. This was followed by 40 cycles of PCR at 95°C (denaturation) for 15 sec, 58°C (annealing) for 90 sec, and 72°C (extension) for 90 sec. After completion of the 40 cycles, a final extension at 72°C for 7 min was performed.
For detection of serotype 2 segment A, the hvVP2 gene was amplified in a real-time RT-PCR assay using primers S2-F (5’-AAGTTGATGGCCACGTGCG-3’) and S2-2 (5’-CTCGGATTATGTCCTTAAAACC-3’). 16 These primers amplify a 778-bp region of the serotype 2 VP2 gene from nucleotides 686–1,463 and do not amplify genome segment A of serotype 1 IBDV. The amplified serotype 2 segment A product was detected using probe f FAM-5’-GTCGCAACTCATCCCAAGT-3’. The cDNA synthesis, pre-denaturation, and PCR conditions were as described previously.
A real-time RT-PCR was also performed for partial sequence amplification of a 722-bp region of segment B of both serotype 1 and serotype 2 IBDV using primers B-168AF (5’-CATAAAGCCTACAGCTGGAC-3’) and B-889R (5’-GTCCACTTGATGACTTGAGG-3’). 16 The amplified segment B product was detected using probe f FAM-5’-TACTCAAGCAGATGAT-3’. The cDNA synthesis, pre-denaturation, and PCR conditions were as described previously.
Restriction fragment length polymorphism
The restriction fragment length polymorphism (RFLP) analysis was performed to further determine if dual IBDV strains were present in samples. While PCR only detects specific (vv or non-vv) strains, the restriction enzyme pattern would show clearly if any other strains were detected. RFLP analysis was performed on the PCR-amplified 743-bp fragment of hvVP2 and the 722-bp fragment of segment B. Segment A PCR products were digested using restriction enzymes g BstNI and MboI. Segment B PCR products were digested using BamHI and EcoRI as previously described. 13 Restriction enzyme–digested DNA was visualized on 2.0% agarose h gels stained with ethidium bromide.
Nucleotide sequence and predicted amino acid analysis
The RT-PCR products were prepared for sequencing using a commercial kit. i Sanger sequencing was conducted at a specialized laboratory. j Nucleotide sequence results were downloaded using commercial software. k Sequence analysis and ClustalW alignments were conducted using commercial software. l
Statistical analysis
Mortality, mean BLS, and BBW ratios were calculated for each group. Statistical differences were determined using a one-way analysis of variance, and multiple comparisons were conducted using Fisher least significant difference test. Statistical differences among the groups were measured at P < 0.05.
Results
Pathogenicity in SPF chickens
The mortality observed in birds in the positive control groups 1e and 2e challenged with only the rB strain was 100% in both the 4- (10/10) and 6-week-old (5/5) inoculated birds. At 4 weeks of age, a significant reduction in mortality was observed when the birds were co-challenged with rB and the Del-E, T1, and OH viruses (Table 1). A significant reduction in mortality was not observed at 4 weeks of age when birds were co-challenged with rB and the antigenically similar STC strain, but at 6 weeks of age, the mortality was significantly lower for this group (Table 2). The other groups with a significant reduction in mortality at 6 weeks of age were those co-challenged with rB and either Del-E or T1. Although a reduction in the mortality was observed for the group co-challenged with rB and OH, the difference was not significant.
Clinical signs observed in birds that succumbed to infection included depression, inappetence, ruffled feathers, and diarrhea. Macroscopic bursal lesions observed included yellow caseous cores and bursal edema with petechial hemorrhages on the mucosa and submucosa in a few birds at 4 and 6 weeks of age. No gross or microscopic lesions were observed in any of the tissues of the negative control birds at 4 and 6 weeks of age. Mean BBW ratios of the 4 dual challenged groups were not statistically different from the uninoculated negative control group in both 4- and 6-week-old birds. The mean BBW ratio was not determined for the rB-positive control groups 1e and 2e because no birds survived 5 days postchallenge.
The mean BLS was significantly higher in the rB-challenged positive control group 1e compared to groups co-challenged with rB and a second IBDV strain (1a–d) in 4-week-old birds. While the mean BLS was higher in 6-week-old birds challenged with rB alone, it was not statistically different from the BLS of the group co-challenged with rB and the OH strain (Table 2). The mean BLS was significantly lower for the birds co-challenged with rB and the STC, Del-E, and T1 viruses at 6 weeks of age compared to the birds that received rB alone (Table 2). The BLS was 0 in both control groups 1f and 2f, and no microscopic abnormalities were observed in the controls.
Microscopic bursal lesions observed in all viral challenge groups (1a–e, 2a–e) included lymphocyte depletion, inflammatory cell infiltration, and stromal edema. Microscopic hemorrhages were observed in a few bursal tissue sections in both 4- and 6-week-old birds in all viral challenge groups. Lymphocyte depletion in the white pulp of the spleen was observed from a few birds from the rB/STC, rB/T1, and rB/OH groups in 4-week-old birds and from the rB/STC groups in the 6-week-old birds. Mild hemorrhage was observed in the mucosa of the proventriculus in a few sections from the rB/STC, rB/Del-E, and rB/OH groups in 4-week-old birds and from the rB/STC and rB/OH groups in the 6-week-old birds.
Molecular detection of IBDV pathotypes
The real-time RT-PCR assay using the probe specific to segment A of vvIBDV strains was positive in all 5 IBDV viral challenge groups in 4- and 6-week-old birds. All 6 groups were negative for non-vvIBDV segment A using the real-time RT-PCR assay in both 4- and 6-week-old birds. The real-time RT-PCR assay with the segment A serotype 2 probe was also negative in all 6 challenge groups in both 4- and 6-week-old birds. The real-time RT-PCR and segment B IBDV probe was positive in both 4- and 6-week-old birds. This probe detects both serotype 1 and serotype 2 of IBDV. All real-time RT-PCR assays were negative for IBDV in the 4- and 6-week-old negative control groups.
Sequencing
The PCR-amplified segment A products from groups challenged with rB alone (1e, 2e) as well as groups challenged with rB and a second IBDV strain (1a–d, 2a–d) matched the 743-bp fragment of the hvVP2 of rB (vvIBDV: GQ221683) in both 4- and 6-week-old birds. The real-time PCR assays targeting the 722-bp fragment of segment B yielded fragments that were homologous with the partial segment B sequence of rB (GQ221685) in all viral challenge groups (1a–e, 2a–e). Non-vvIBDV sequences were not detected in any of the experimental groups in 4- and 6-week-old birds.
RFLP analysis
The 743-bp amplicon of segment A was digested using BstNI into 3 bands of 424 bp, 172 bp, and 119 bp for all IBDV-challenged groups (1a–e, 2a–e). The MboI restriction enzyme digested the segment A amplicon into 362-bp and 229-bp fragments for all IBDV-challenged groups (1a–e, 2a–e). The segment A RFLP patterns using BstNI and MboI are synonymous with those seen with digestion of the 743-bp fragment of VP2 of vvIBDV. The 722-bp amplified fragment of segment B was digested using BamHI into a 655 bp visible band for all treatment groups for all IBDV challenged groups (1a–e, 2a–e). This pattern is synonymous with BamHI digestion of the 722 VP1 fragment of vvIBDV. The smaller 67 bp band was not visible on the gel.
Discussion
A major objective of the current study was to determine if co-challenge with endemic IBDV pathotypes would alter the severity of clinical disease typically associated with vvIBDV infection. The standard classic STC, variant Del-E, variant T1, and avirulent OH strains were chosen for co-challenge with the rB strain based on their endemicity in chickens in the United States. The severity of disease was assessed by evaluating the mortality rates, BBW ratios, and the severity of bursal lesions, which was quantified using the mean BLS.
The T1 IBDV variant was initially isolated from chickens in the state of Georgia. 15 The T1 variant was reported as a subclinical strain of IBDV capable of causing bursal lesions in chickens with maternal immunity against both classic and variant Del-E IBDV strains. 15 The T1 variant was reported to induce bursal atrophy and mild bursal inflammation in challenged chickens, but no mortality or morbidity was reported. 15 The variant Del-E typically induces bursal atrophy and lymphocyte depletion, but no mortality or clinical disease is typically associated with Del-E infection in chickens. 29 The STC strain typically induces mortality between 0–30% in SPF chickens.17,29 The serotype 2 OH strain has been previously described. 11 While serotype 2 strains of IBDV can be infectious, they are typically nonpathogenic in chickens. 3 In addition, there is no antibody cross-protection between serotype 1 and serotype 2 strains. 4 The exact mechanisms by which IBDV pathotypes induce mortality and disease have not been fully elucidated.
The 100% mortality observed in 4- and 6-week-old SPF chickens challenged with vvIBDV (rB strain) correlated with mortality reported by other investigators.17,21 Mortality of 91–100% in 4-week-old SPF chickens challenged with 102.0–105.5 EID50 of the rB vvIBDV strain has been previously reported. 17 Mortality of 90–100% was also reported in SPF flocks inoculated with vvIBDV strains originating from Europe.6,25
In the current experiment, the mortality rates observed in SPF chickens simultaneously challenged with vvIBDV (rB strain) and either the classic STC, variant (Del-E, T1), or serotype 2 OH was lower than the mortality observed in groups challenged with rB alone at both 4 and 6 weeks of age. Although animal care and use guidelines required the use of low numbers of birds in each group, a statically significant difference in the mortality was observed in 4-week-old birds co-challenged with rB/Del-E, rB/T1, and rB/OH. At 6 weeks of age, a significant reduction in mortality was observed in birds co-challenged with rB/STC, rB/Del-E, and rB/T1. There was also a statistically significant reduction in mean BLS in the dual challenged groups compared to the rB group in both the 4- and 6-week-old birds. The only group that did not have a statistically significant reduction in BLS compared to the rB group was the rB/OH group at 6 weeks of age. In general, there was a reduction in the severity of lymphocyte depletion in bursas from birds challenged with the 2 IBDV strains compared to groups challenged with vvIBDV (rB strain) alone.
The normal weight of the chicken bursa is approximately 0.3% of body weight (BBW of 3.0). 1 Pathogenic strains of IBDV result in bursal atrophy and are associated with a statistically significant reduction in BBW compared to uninoculated controls. 19 BBW ratios below 1.0 are highly suggestive of bursal atrophy and infection with pathogenic IBDV strains. 19 Interpretation of mean BBW ratios on its own is considered inadequate to evaluate bursal atrophy due to the lack of sensitivity of the results. 1 In the current study, mean BBW ratios were recorded only in euthanized birds and not birds that died during the course of infection, as the dehydration of dead carcasses alters the validity of result interpretation. No statistical differences in mean BBW ratios were identified between any of the treatment and control groups at both 4 and 6 weeks of age. In fact, the mean BBW ratios were higher in some treatment groups compared to the negative control groups. It was noted that the highest BBW ratios were recorded in groups challenged with rB/STC at both 4 weeks and 6 weeks of age. This result is not surprising because both very virulent and standard classic IBDV strains are correlated with bursal edema and enlargement, which is reflected in relatively high BBW ratios. 7 The nonbursal systemic lesions observed in the proventriculus and spleen in some of the challenge groups were not unexpected, as the pathogenicity of vvIBDV strains have been correlated with viral distribution in nonbursal lymphopoietic and hematopoietic organs.7,25
By evaluating the mortality rates and BLS, it was concluded that coinfection with rB and either STC, Del-E, T1, or OH strains can reduce the severity of disease compared to infection with rB alone. The PCR, RFLP, and sequencing tests detected the presence of only the rB strain by 5 days postchallenge in all viral challenge groups (1a–e, 2a–e) in both 4- and 6-week-old birds. Hence, based on molecular results, it can be inferred that vvIBDV (rB strain) was able to outcompete the less virulent pathotypes STC, Del-E, T1, and OH by 5 days postchallenge.
The results of this current investigation suggest that viral competition between vvIBDV and endemic serotype 1 STC, Del-E, or T1 strains can reduce the severity of disease in fully susceptible chickens. Similarly, the current study indicated that simultaneous challenge with serotype 1 (rB strain) and serotype 2 (OH strain) results in a reduction in mortality in fully susceptible chickens. The latter observation is interesting because serotype 2 IBDV strains do not replicate in chicken lymphoid cells. 20 However, binding studies show that the serotype 2 viruses are still capable of binding to lymphoid cells located in the bursa. 20 Saturation and competition experiments have shown that receptor sites common to both serotype 1 and 2 are present on lymphoid cells. 20 This suggests that viral interference between the rB and OH strains may be occurring at the extracellular level and that competition for B-lymphocyte cell surface receptors may be a mechanism for the reduced mortality observed in the rB and OH challenged birds.
In the co-challenged birds, it is also possible that the stimulation of nonspecific immunity may be responsible for inducing some degree of protection against vvIBDV. The stimulation of humoral immunity and antibody production against IBDV is not expected to have a significant influence on host protection at 5 days postchallenge. Hence, the antigenic similarities between rB and STC and induction of B-cell–mediated immunity are not believed to play a role in the decrease in disease severity in this experiment.
Viral interference related to coinfection with multiple strains of IBDV has also been reported by other researchers. A reduction in mortality was reported when 4-week-old SPF chicks were simultaneously inoculated with T1 and vvIBDV (rB). 12 In this prior experiment, only the vvIBDV RNA was recovered from the bursal tissues of dual challenged birds. 12 In vivo viral interference and persistence of the more pathogenic strain has also been reported when the pathogenic STC strain was given 24 hr after challenge with a live mild vaccine strain. 2 Viral competition between the coinfecting IBDV pathotypes may be influenced by factors such as competition for cell surface receptors and intracellular biosynthetic machinery required for viral replication.2,12,28 Host factors, such as the stimulation of nonspecific immunity, including cytokine production, may also act to decrease the severity of pathology. It is also possible that the presence of the non-vvIBDV pathotypes may be stimulating the infected cells to produce interferon proteins, which can render uninfected cells resistant to viral infection.
In conclusion, the current study supports the theory that coinfection with endemic IBDV strains may be a significant factor influencing the relatively low mortality currently associated with vvIBDV infection in the field in California. This makes it more plausible that vvIBDV can be present but unrecognized in commercial poultry flocks for prolonged periods. In addition, the finding of mean BLS scores of greater than 3 in the co-challenged groups indicates that bursal injury and the subsequent immune suppression can still be significant factors in birds infected with multiple IBDV pathotypes. The ability of vvIBDV to outcompete the endemic viruses could be a potential factor influencing the ability of vvIBDV to become endemic and established in new regions. These factors emphasize the need for continued active surveillance in the field. Further research is also needed to determine the efficacy of vaccination and cross-protection between vvIBDV and antigenically similar endemic classic IBDV strains. In addition, the current study highlights the importance of additional research to investigate the efficacy of vaccination during the face of an outbreak to reduce mortality.
Footnotes
a.
Horsfall-Bauer isolation units, Wooster, OH.
b.
Trizol, Invitrogen Corp., Carlsbad, CA.
c.
DMSO, Sigma-Aldrich, St. Louis, MO.
d.
LightCycler 480 Instrument, Roche Diagnostics Corp., Indianapolis, IN.
e.
GenAmp RNA PCR kit, Applied Biosystems, Roche Molecular Systems Inc., Branchburg, NJ.
f.
TaqMan Probes, Applied Biosystems, Foster City, CA.
g.
Restriction enzymes, Promega Corp., Madison, WI.
h
Agarose, Lonza Rockland Inc., Rockland, ME.
i.
Wizard SV Gel and PCR Clean-Up System, Promega Corp., Madison, WI
j.
University of Wisconsin, Biotechnology Center, DNA Sequence Laboratory, Madison, WI.
k.
Technelysium Pty. Ltd., Queensland, Australia.
l.
Accelrys, San Diego, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the Harold E. Ford US Poultry Foundation, U.S. Poultry and Egg Association, Tucker, GA.
