Abstract
The cell proliferation activity of proliferating bile ducts produced by bile duct ligation (BDL) in rats was examined histologically, immunohistochemically, and ultrastructurally. Proliferating bile ducts, which were similar to normal bile ducts, increased with time after BDL. The cell proliferation activity of proliferating bile ducts, measured using proliferating-cell nuclear antigen and 5-bromo-2′-deoxyuridine antibodies, tended to be high at 1 and 3 days after BDL and decreased progressively at 2 to 4 weeks after BDL. On the other hand, α-smooth muscle actin-positive myofibroblast-like cells increased continuously after BDL. These findings indicate that there is a negative correlation between the cell proliferation activity of proliferating bile ducts and that of myofibroblast-like cells.
Keywords
The proliferation of the bile ducts (ductular reaction) of the liver in response to various liver injuries is characterized by an increase in the number of intrahepatic bile ducts. 3, 11 It is usually classified as a ductular reaction to typical, atypical, or oval cell proliferation. 11 The typical ductular reaction involves a marked increase in the number of biliary epithelial cells. The highest labeling index (LI) for biliary epithelium was observed 48 hours after bile duct ligation (BDL) in a thymidine-labeling study. 13 So far, there have been only a few reports of cell proliferation activity in proliferating bile ducts. 5, 13 In this study, the cell proliferation activity of proliferating bile ducts produced by BDL in rats was examined histologically, immunohistochemically, and ultrastructurally.
Fifteen 10-week-old male Wistar rats were used in this study. In 12 rats, the extrahepatic bile duct was ligated (BDL). At 1 (n = 3) and 3 (n = 3) days and 2 (n = 3) and 4 (n = 3) weeks after BDL, the rats were bled and euthanatized under ether anesthesia. The remaining three control rats, in which the bile ducts were left intact, were euthanatized under ether anesthesia 4 weeks after BDL was performed on the other rats. After surgery, the control and BDL rats were housed in individual cages at a standard temperature of 25 ± 1° C and a 12 hour light/dark cycle and fed ad libitum on standard rat chow, with free access to water. 5-Bromo-2′-deoxyuridine (BrdU; Sigma, St. Louis, MO) was injected intraperitoneally (50 mg/kg body weight) into the BDL and control rats at 1 and 3 days and 2 and 4 weeks after BDL and 2 hours before the animals were euthanatized.
Liver specimens were fixed with 10% neutral buffered formalin, routinely processed, and embedded in paraffin. Tissue sections, 4–5 μm thick, were cut and stained by routine methods with hematoxylin and eosin (HE), with Azan stain to detect collagen, or by immunohistochemistry. For Azan staining, sections were deparaffinized in xylene, dehydrated, rehydrated in distilled water, immersed in 5% potassium dichromate solution for 30 minutes, and stained with azocarmine G for 30 minutes. Sections were then immersed in 3% 12 tungsto (IV) phosphoric acid n-hydrate solution for 1 hour and stained with aniline blue-orange G for 20 minutes. For immunohistochemistry, peroxidase-labeled antibody reactions were performed using anti-cytokeratin AE-1 monoclonal rabbit serum (Ylem SRL, Roma, Italy), which recognizes cytokeratins 10, 14, 16, and 19, anti–α-smooth muscle actin (α-SMA) monoclonal mouse serum (Nichirei Co., Tokyo, Japan), anti–proliferating-cell nuclear antigen (PCNA) monoclonal mouse serum (Nichirei), and anti-BrdU monoclonal mouse serum (Amersham Pharmacia Biotech Co., Tokyo, Japan) as the primary antibodies. Sections were mounted on silanized slides (Dako Cytomation Co., Kyoto, Japan), dried at 37° C for 24 hours, and then deparaffinized in xylene, dehydrated, and rehydrated in distilled water for 5 minutes. Endogenous peroxidase activity was blocked with 3% hydrogen peroxide diluted in methanol for 30 minutes followed by three 5-minute rinses in 0.1 M phosphate buffer (pH 7.4). Sections were then incubated with undiluted primary antibody (described above) for 12 hours at 4° C. Peroxidase-conjugated anti-mouse or anti-rabbit immunoglobulin G (Nichirei) was used as the secondary antibody and applied for 1 hour at 37° C; diaminobenzidine (Nichirei) was used as the chromogen. Sections were counterstained with Mayer's hematoxylin and covered with coverslips. For morphometric analyses, the PCNA LI and BrdU LI were calculated as the percentage of positive cells relative to the number of biliary epithelial cells by counting a total of at least 500 nuclei in bile ducts from each animal. Student's t-test (two-tailed) was used for all statistical comparisons of PCNA LI and BrdU LI. A P value < 0.05 was considered to be significant. Tissues were fixed with 0.1 M phosphate-buffered glutaraldehyde for 24 hours, postfixed with 1% osmic acid for 2 hours, dehydrated, and embedded in epoxy resin. The embedded specimens were sectioned to 1 μm and stained with toluidine blue for light microscopy. Ultrathin sections were stained with uranyl acetate and lead citrate and examined using a transmission electron microscope (H-7000, Hitachi Ltd., Tokyo, Japan).
In general, proliferation of bile ducts and infiltration of inflammatory cells such as neutrophils, eosinophils, and lymphocytes were commonly observed in the portal areas of all BDL rats. Proliferating bile ducts were similar to normal bile ducts, consisting of a well-defined lumen, basement membrane, and epithelial cells. The epithelial cells had hypochromatic nuclei and clear cytoplasm (Fig. 1). Immunohistochemically, the epithelial cells showed an intense positive cytoplasmic staining for cytokeratin AE-1, but the hepatocytes were negative. One day after BDL, slight proliferation of the bile ducts was seen in the portal areas. In addition to the increased number of bile ducts, proliferating bile ducts were surrounded by spindle cells 3 days after BDL in the portal areas. At 2 weeks after BDL, the proliferating bile ducts infiltrated the hepatic parenchyma. Hepatocytes in areas of proliferated bile ducts were atrophied at the 4 weeks time point after BDL. Ultrastructurally, most epithelial cells in the proliferating bile ducts had characteristics similar to those of normal bile ducts. The nuclei were round, and the scanty cytoplasm contained a few organelles, including mitochondria, rough endoplasmic reticulum, and free ribosomes. The epithelial cells were connected by desmosomes, forming a well-defined lumen with abundant microvilli. Basement membranes surrounded the proliferating bile ducts (Fig. 2).
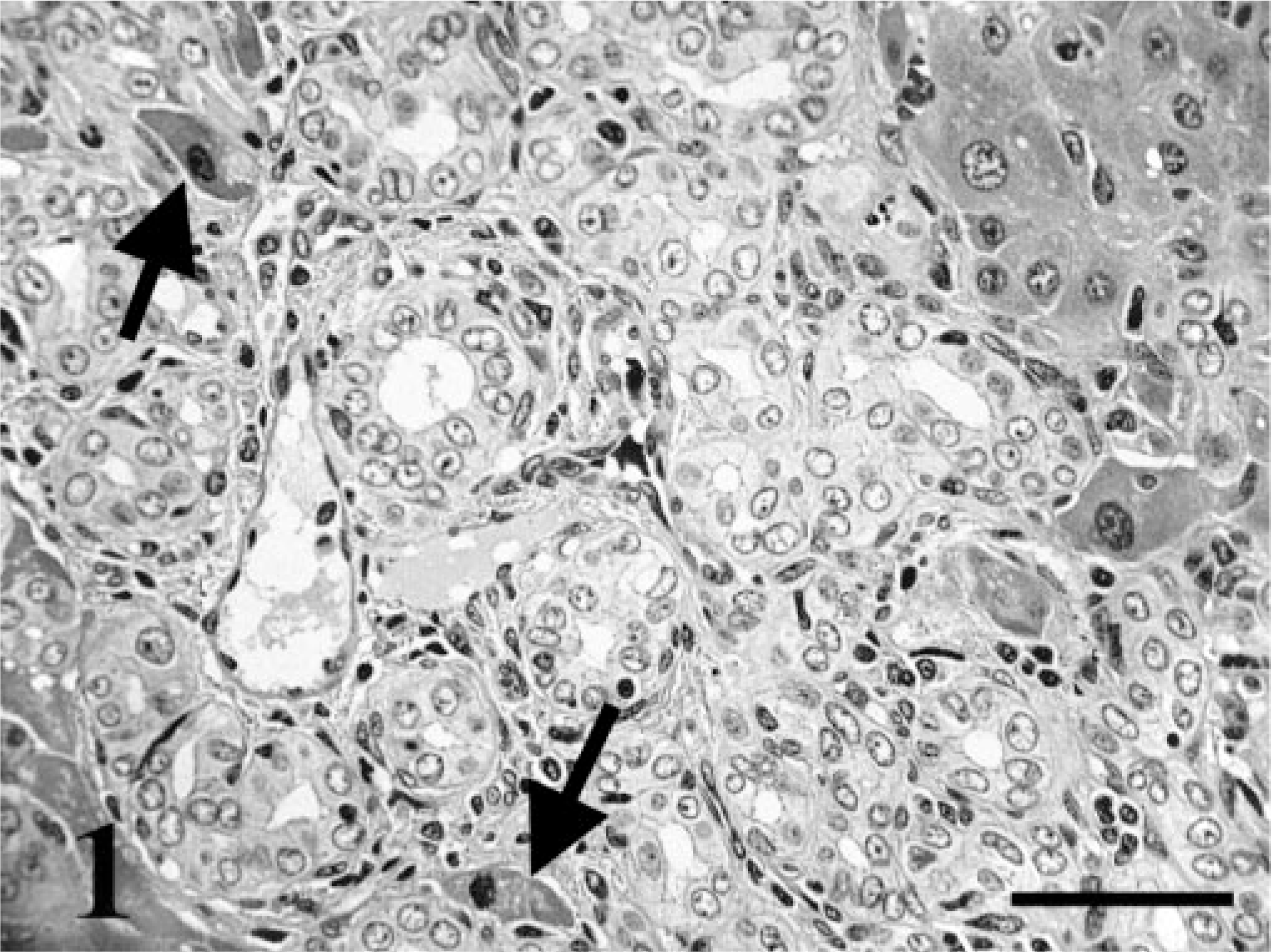
Liver; rats, 4 weeks after bile duct ligation. Proliferating bile ducts are similar to normal bile ducts. Atrophic hepatocytes (arrows). HE staining. Bar = 50 μm.
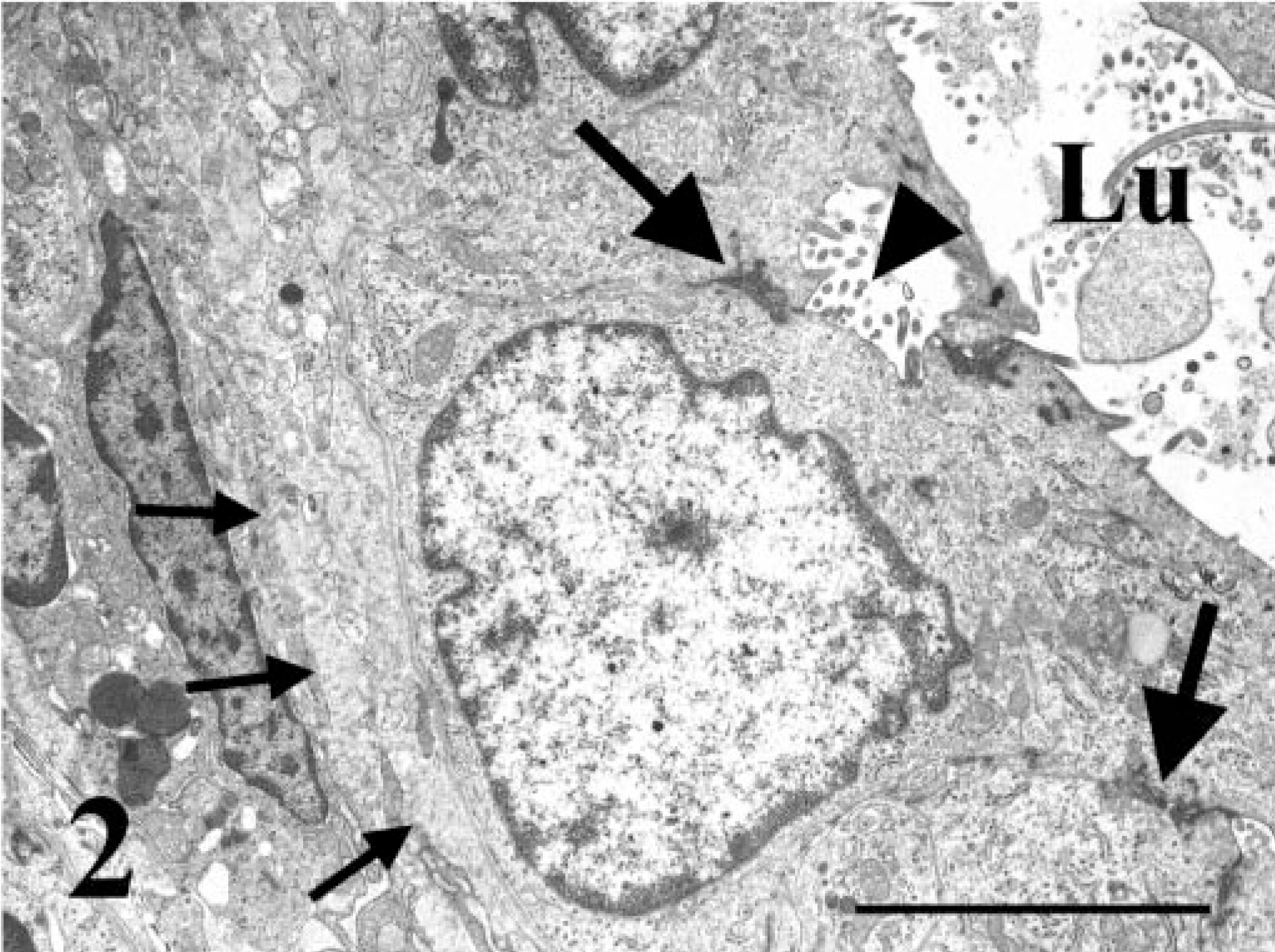
Liver; rat, 4 weeks after bile duct ligation. Ultrastructure of a proliferating bile duct. Basement membrane (small arrows), desmosomal junction (large arrows), well-defined lumen (Lu) with abundant microvilli (arrowhead). Electron micrograph. Bar = 5 μm.
Collagen fibers, staining blue with Azan stain, were observed around the bile ducts and blood vessels in the portal areas 1 day after BDL and in the controls. At 3 days after BDL, the collagen fibers surrounding the proliferating bile ducts had increased significantly compared with fibers observed 1 day after BDL and in the controls in the portal areas. Moreover, at 2 to 4 weeks after BDL, the collagen fibers had infiltrated the hepatic parenchyma, accompanying a marked increase in bile ducts. In the controls, immunohistochemically, α-SMA–positive cells were observed in blood vessels in the portal areas. One day after BDL, a few α-SMA–positive cells were seen in the portal areas (Fig. 3A). At 3 days after BDL, numerous α-SMA–positive cells were observed around the proliferating bile ducts in the portal areas (Fig. 3B). Moreover, α-SMA–positive cells had infiltrated the hepatic parenchyma at 2 to 4 weeks after BDL (Fig. 3C).
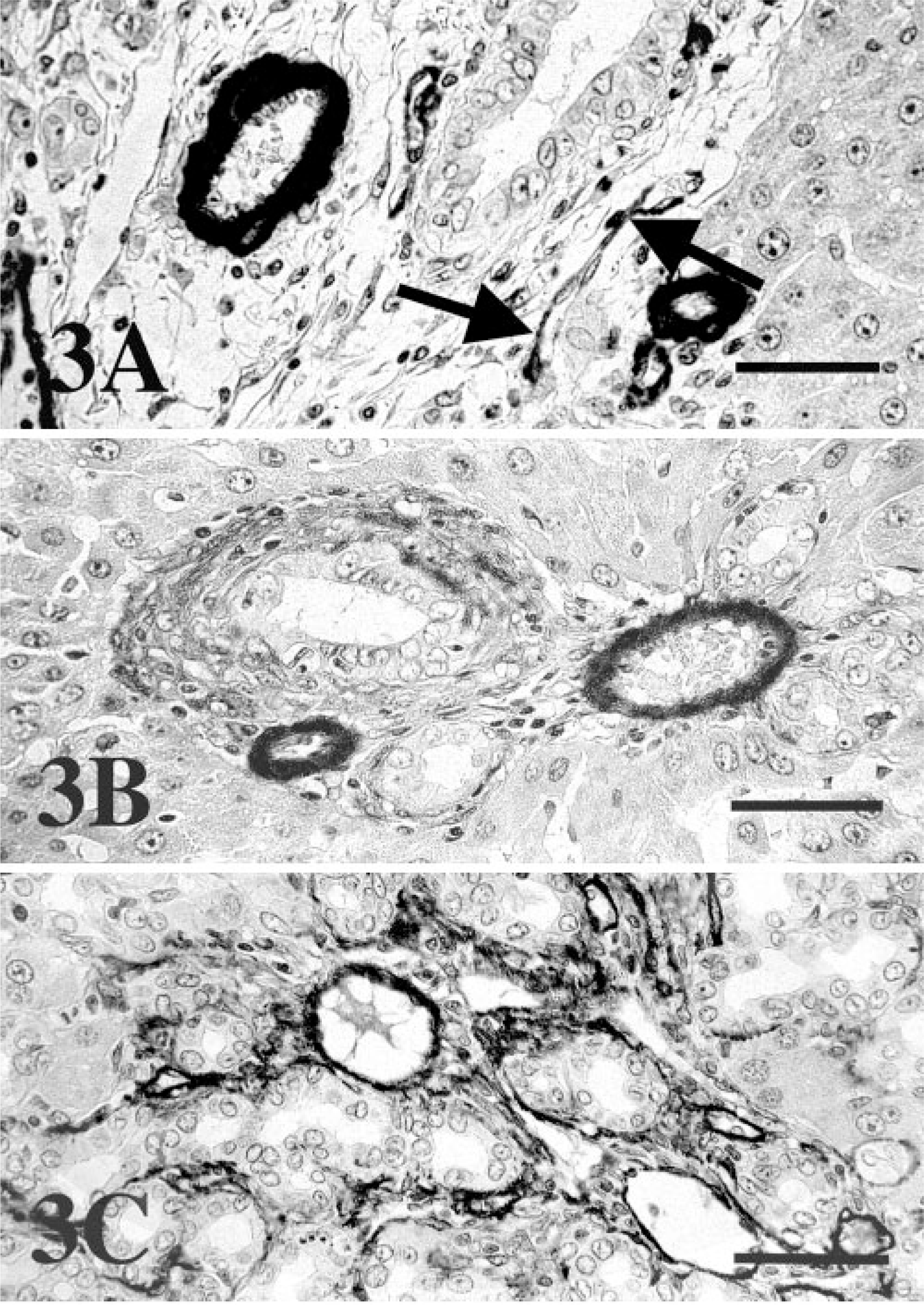
Liver; rat.
On an average, approximately 90% of the proliferating bile ducts were positive for PCNA 1 day after BDL. Their cell proliferation activity was significantly (P < 0.05) increased compared with controls. The immunoreactivity was slightly reduced 3 days after BDL and was significantly (P < 0.05) reduced 2 to 4 weeks after BDL. The same tendency was observed with immunohistochemistry using BrdU antibody (Table 1). An average of approximately 40% of the proliferating bile ducts were BrdU positive 1 day after BDL (Fig. 4A). However, at 3 days after BDL, the cell proliferation activity had decreased significantly (P < 0.05) compared with that observed 1 day after BDL (Fig. 4B). The BrdU-positive cells had decreased even further 2 to 4 weeks after BDL (Fig. 4C). The difference between control and BDL rats in the cell proliferation activity of hepatocytes was not significant.
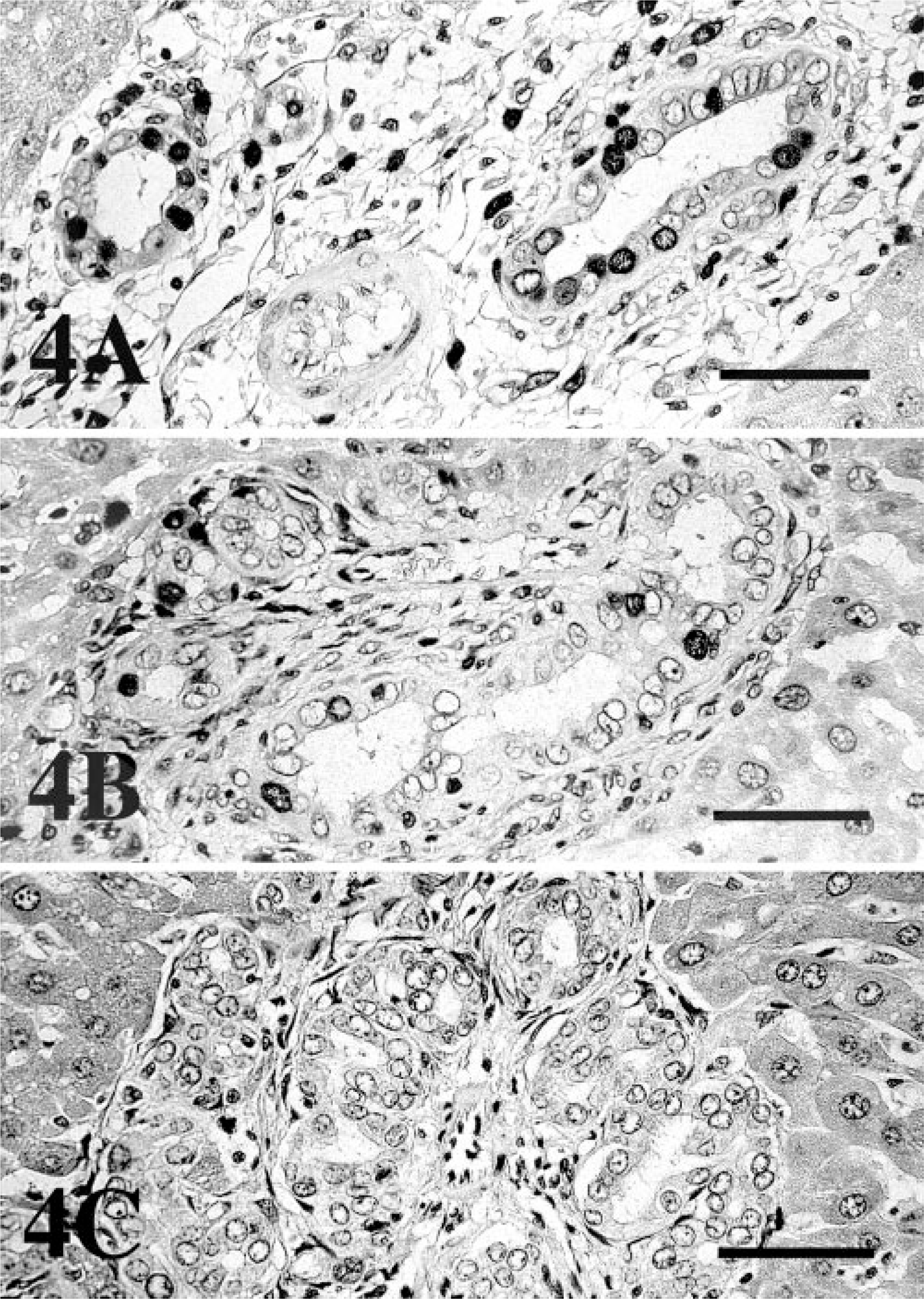
Liver; rat.
Cell proliferation activity of proliferating bile ducts in five experimental groups.
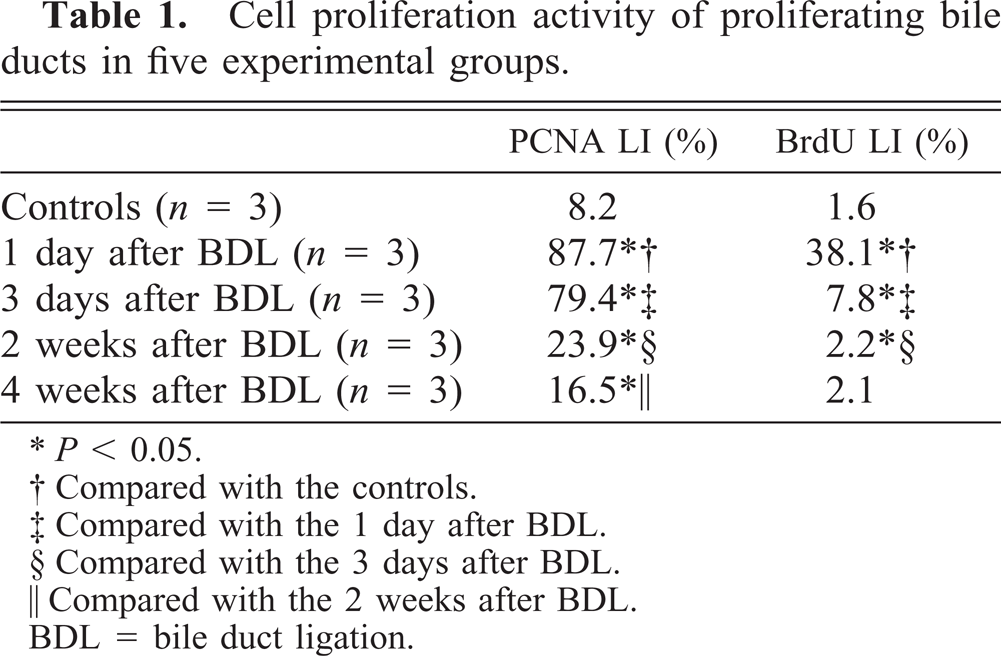
∗P < 0.05.
† Compared with the controls.
‡ Compared with the 1 day after BDL.
§ Compared with the 3 days after BDL.
‖ Compared with the 2 weeks after BDL.
BDL = bile duct ligation.
In this study, the immunohistochemical and ultrastructural features of proliferating bile ducts were similar to those of normal bile ducts, suggesting a typical ductular reaction resulting from the elongation and increased tortuosity of existing bile ducts within the periportal space. 13 Immunohistochemically, biliary epithelial cells are positive for cytokeratins 7, 8, 18, and 19, whereas hepatocytes are positive for cytokeratins 8 and 18. 12, 13 In the BDL rat, proliferating ducts retain the immunohistochemical, ultrastructural, and functional characteristics of their normal counterparts and appear morphologically as elongations of preexisting ducts. 3 In humans with extrahepatic biliary obstruction, proliferating parenchymal and bile duct cells express their respective normal cytokine profiles. 9 These reports show that typical ductular reactions, which induce an increase in the number of intrahepatic bile ducts (with well-defined lumens), result from the elongation of preexisting bile ducts located within the portal areas.
Our study demonstrates that the number of α-SMA–positive cells increases after BDL. These cells have been recognized as myofibroblast-like cells in several previous reports. 1, 7 The hepatic stellate cells (Ito cells, hepatic lipocytes, or fat-storing cells) have an important role in the pathogenesis of hepatic fibrosis, cirrhosis, and liver remodeling. 4, 10 After liver injury, hepatic stellate cells undergo activation and transdifferentiation to myofibroblast-like cells. 4, 10 α-SMA is a marker for myofibroblast-like cells. 1, 10 When activated by various cytokines, myofibroblast-like cells produce extracellular matrix. 1, 4, 7
PCNA is an accessory protein to DNA polymerase delta and is expressed from G1 phase into S phase in the cell cycle. 6 BrdU is a thymidine analog normally incorporated into new DNA. 2 Antibodies directed against these proteins were used as markers for cell proliferation activity. The cell proliferation activity of proliferating bile ducts tended to be high 1 and 3 days after BDL and decreased progressively at 2 to 4 weeks after BDL. On the other hand, α-SMA expression increased continuously after BDL. These findings indicate that there is a negative correlation between the cell proliferation activity of proliferating bile ducts and the numbers of myofibroblast-like cells, which increase with time after BDL (Table 1). Hepatic stellate cells secrete latent metalloproteinase-2 (MMP-2), and the accumulation of collagen may specifically contribute to the remodeling of extra-cellular matrix in fibrotic livers by inducing MMP-2 activation. 14 Activated human Ito cells produce an inhibitor of metalloproteinase-1 (TIMP-1), which decreases interstitial collagenase activity and contributes to extracellular matrix accumulation. 1, 8 Furthermore, the myofibroblast-like cells produce not only extracellular matrix, MMP, and TIMP but also a variety of cytokines and growth factors that act in paracrine and autocrine loops on parenchymal cells, untransformed hepatic stellate cells, and myofibroblast-like cells. 4 Our results indicate that the negative correlation simply may be attributed to the stimulus for bile duct proliferation occurring first; bile duct proliferation then stimulates the myofibroblast-like cell such that its response lags. These findings may indicate that myofibroblast-like cell products cause a decrease in the cell proliferation activity of cholangiocytes. It also suggests that this negative correlation may be attributed to feedback mechanisms of the myofibroblast-like cell on the biliary epithelium. Our study demonstrates that there is difference in that BrdU LI is less than PCNA LI in each experimental group. It may be the reason that PCNA is expressed from G1 phase into S phase, whereas BrdU is expressed only in S phase in the cell cycle. 2, 6
In conclusion, our data indicate that there is a negative correlation between the cell proliferation activity of proliferating bile ducts and numbers of myofibroblast-like cells, which increase with time after BDL.
