Abstract
A 7-y-old backyard Leghorn chicken (Gallus domesticus) was submitted to the California Animal Health and Food Safety Laboratory System (CAHFS)–Turlock branch for postmortem examination, with a history of unexpected death. At postmortem examination, a hemorrhagic soft tissue mass was observed in the cervical region. Microscopically, a densely cellular neoplasm of polygonal epithelial cells and small lymphocytes was observed. The microscopic features of the neoplasm in combination with positive immunohistochemistry for pancytokeratin and CD3 were used to classify the lesion as a thymoma. Thymoma was diagnosed in only 5 birds submitted to CAHFS from 1990 to 2019. Thymoma has been described only rarely in birds, and is an unusual diagnosis in backyard chickens.
Thymoma is a neoplasm that originates from epithelial cells of the thymus. Thymomas are slow growing and benign, and are classified as thymic carcinoma when malignancy is evident. 14 Thymomas occur uncommonly in adult mammals, with the exception of adult female goats who have an increased likelihood of developing thymomas. 14 Thymomas have been reported in various aged birds, ranging from an 8-wk-old chicken (Gallus domesticus) to a 49-y-old scarlet macaw (Ara macao).2,8,15 In chickens, the thymus is located parallel to the vagus nerve and jugular veins and extends from the third cervical vertebra to the cranial celom, in 7–8 separate lobes located on either side of the neck.5,12
Thymomas in avian species have been described only rarely, with cases documented in 2 domestic chickens, a myna (Acridotheres tristis), an owl (Athene cunicularia), a macaw, a Senegal parrot (Poicephalus senegalus), a budgerigar (Melopsittacus undulatus), a finch (Haemorhous sp.), a Java sparrow (Padda oryzivora), and an American robin (Turdus migratorius).2,4,6–10,13,16,17 We describe the clinical history, gross lesions, and microscopic lesions of a thymoma in a backyard Leghorn chicken.
A 7-y-old female Leghorn chicken was presented dead to the California Animal Health and Food Safety Laboratory System (CAHFS)–Turlock branch for postmortem examination. The hen resided with other backyard chickens with access to a coop and an outdoor run, had no previous medical history, and normal behavior was reported in the days prior to death.
The hen was in good body condition, weighed 2 kg, and had a pale comb and wattles. A 5-cm diameter mass was palpated in the subcutis of the neck, cranial and left of the celomic inlet (Fig. 1). Clotted blood was present in multiple cavities of the mass (Fig. 2). Three small (< 1 cm in length) thymic lobes were present cranial to the mass. Mild petechial hemorrhages were observed bilaterally in the liver capsule.
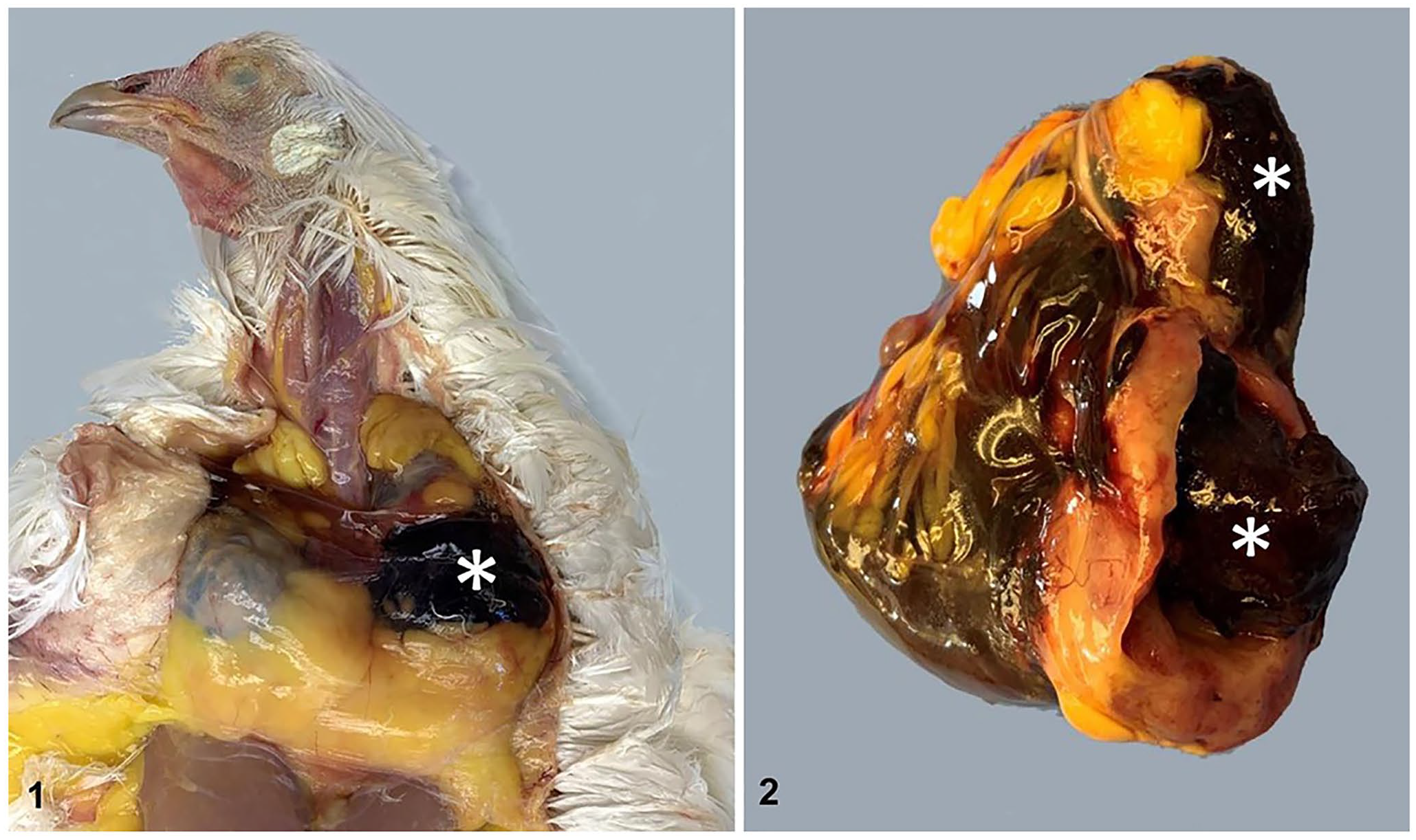
Thymoma in a 7-y-old backyard Leghorn chicken.
Tissue sections of trachea, liver, lung, kidney, spleen, thyroid, parathyroid, thymus, tumor, oviduct, ovary, brain, heart, proventriculus, intestine, and pancreas were processed routinely and stained with hematoxylin and eosin. Immunohistochemistry (IHC) was performed on tumor sections for pancytokeratin (monoclonal mouse antibody; Biocare Medical), cytokeratin 5/6 (monoclonal mouse antibody; Biocare Medical), cytokeratin 14 (monoclonal mouse antibody; Thermo Fisher), cytokeratin 18 (monoclonal mouse antibody; BioGenex), synaptophysin (chief cells, monoclonal mouse antibody; Agilent), chromogranin (chief cells, monoclonal rabbit antibody; LifeSpan BioSciences), and CD3 (T cells; monoclonal rat antibody; Leukocyte Antigen Biology Laboratory, Davis, CA).
Histologically, a partially encapsulated, densely cellular neoplasm composed of 2 cell types effaced the thymic tissue. The neoplastic cells were large polygonal epithelial cells with abundant finely granular eosinophilic cytoplasm, round nuclei with open chromatin, and a prominent nucleolus (Fig. 3). Occasionally, these cells formed concentrically arranged keratinizing bodies suggestive of Hassall corpuscles (Fig. 4). The second cell population consisted of abundant small lymphocytes (Fig. 5). The mitotic count was zero per 2.4 mm 2 (equivalent to 10 FN22/40× fields). Necrosis, hemorrhage, and pseudocysts were present, with accumulation of small amounts of eosinophilic fluid and fibrin (Fig. 6). The cytoplasm of thymic epithelial cells had strong cytoplasmic immunoreactivity to pancytokeratin (Fig. 7), and the lymphocytes had strong membranous and cytoplasmic immunoreactivity to CD3 (Fig. 8). There was no immunoreactivity to cytokeratin 5, 6, 14, and 18 (data not shown). Additionally, there was no immunoreactivity to chromogranin or synaptophysin in the epithelial cells. Mild subcapsular hemorrhages were observed in liver sections.
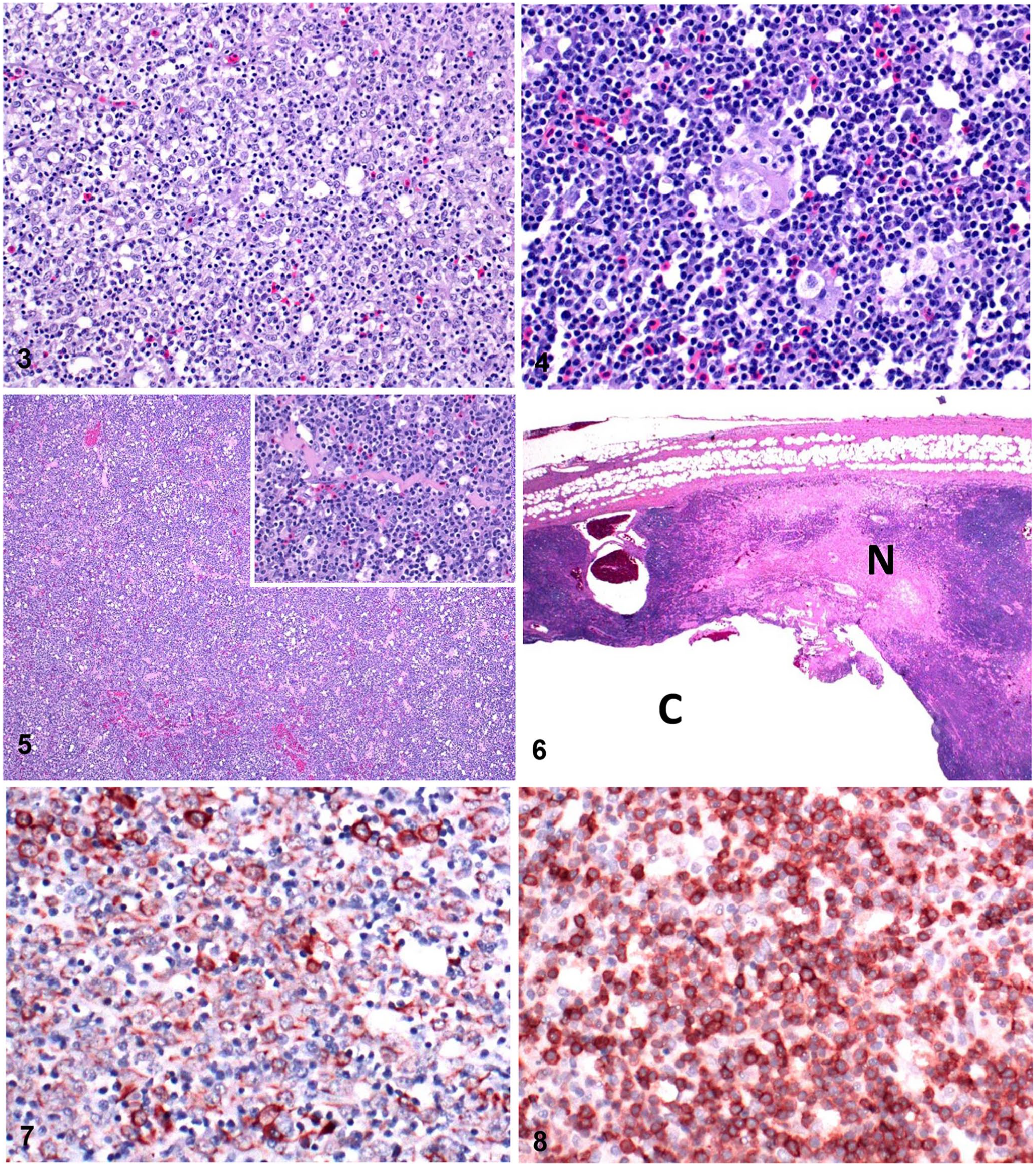
Microscopic examination of a thymoma in a 7-y-old Leghorn chicken.
Liver cultures performed to rule out sepsis yielded no bacterial growth after 48 h. An oropharyngeal swab was collected and tested negative for the presence of influenza A virus and virulent Newcastle disease virus (Avian orthoavulavirus 1) by reverse-transcription real-time PCR, according to the CAHFS routine surveillance protocol (National Animal Health Laboratory Network protocol, CAHFS, Davis laboratory). The final diagnosis of thymoma was based upon postmortem lesions, histologic examination, and immunoreactivity with pancytokeratin and CD3.
We searched the electronic database of CAHFS for postmortem cases of thymoma in birds from January 1, 1990 to December 31, 2019. Data analyzed for each case included date of submission, species, and age. Five cases were discovered; an adult lovebird (Agapornis sp.), a 30-y-old Moluccan cockatoo (syn. salmon-crested cockatoo; Cacatua moluccensis), an adult Muscovy duck (Cairina moschata), a 3-y-old backyard chicken, and the Leghorn chicken described in our report. In all cases, thymoma was diagnosed by the appearance of the gross lesions and microscopic examination. Neoplasia was diagnosed in 6,971 of 67,736 (10.3%) avian cases accessioned at CAHFS system-wide from 1990 to 2019.
In chickens, the thymic lobes reach a maximum size of ~10 mm at 3–4 mo of age and then begin physiologic involution; hence the presence of thymic tissue is abnormal in the 7-y-old hen in our case. 3 Metastasis to internal organs has been described in many cases of canine thymoma, and was observed in a macaw in which the tumor metastasized to the lung.1,8 Evidence of metastasis was not observed in the hen in our case.
Thymomas in veterinary medicine have been described as predominantly epithelial, lymphocytic, or mixed in appearance. 14 In our 7-y-old hen, the regions of positive staining for pancytokeratin and CD3 verified the mixed nature of the thymoma. 13 The World Health Organization (WHO) classifies human thymic epithelial tumors as type A, type B1-3, type AB, micronodular, thymic carcinoma, type X, and combined thymoma, based on histologic characteristics of the neoplastic epithelial cells and proportion of non-neoplastic lymphocytes.11,14 Based on the presence of epithelial cells and abundant small lymphocytes, we classified the tumor in our case as a type B1 thymoma.
IHC targeting pancytokeratin and CD3 was useful for diagnosis and differentiation from other causes of cervical enlargement in birds, such as thymic lymphoma, thymic carcinoma, and a fibriscess.6,13 Synaptophysin and chromogranin IHC were utilized to rule out parathyroid neoplasia. The cytokeratin 5, 6, 14, and 18 IHC that we utilized in this case were not developed or validated for use in avian species, which may explain the lack of immunoreactivity. Based on the 30-y retrospective search of the CAHFS database and published cases, thymomas have been diagnosed in domestic chickens, parrots, a budgerigar, a myna, an owl, a finch, a Java sparrow, a robin, and a duck; thymoma is a rare finding in birds.2,4,6–10,13,16,17
Footnotes
Acknowledgements
We thank the CAHFS and the UCDavis Veterinary Medicine Teaching Hospital histology departments for their support in performing the immunohistochemistry in our case.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
