Abstract
Macrorhabdus ornithogaster (M. ornithogaster) is an anamorphic ascomycetous yeast found only in the stomach of birds. Infection is often benign but has also been associated with disease in some species of birds under some circumstances. In vitro efforts to grow M. ornithogaster have been largely unsuccessful. In this report, multiple liquid and solid media of varying pH, sugar concentration, and fetal bovine serum (FBS) concentrations, incubated at various temperatures in room air or microaerophilic conditions, were examined for their ability to support the growth of M. ornithogaster, obtained from a budgerigar (Melopsittacus undulatus). Optimum growth conditions were found to be Basal Medium Eagle's, pH 3 to 4, containing 20% FBS, and 5% glucose or sucrose under microaerophilic conditions at 42°C. Using these conditions, M. ornithogaster was repeatedly passaged without loss of viability. Polyclonal isolates of M. ornithogaster consistently assimilated glucose, sucrose, and trehalose. M. ornithogaster did not grow with prolonged exposure to atmospheric oxygen, but growth in microaerophilic conditions was moderately enhanced by preincubation with atmospheric oxygen for 24 hours. An isolate of M. ornithogaster was found to be infective to day-old chickens, reduce their rate of weight gain, and induce a mild to moderate heterophilic inflammation of the isthmus. M. ornithogaster was reisolated from the chicks 7 days after infection, fulfilling Koch's postulates. A 761-bp sequence of 18S rDNA from this isolate was compared to the originally reported M. ornithogaster sequence and was found to be 97% identical.
Keywords
Introduction
Macrorhabdus ornithogaster is an anamorphic ascomycetous yeast that is the only known member of its genus. 15 M. ornithogaster colonizes the narrow junction (isthmus) of the glandular stomach (proventriculus) and grinding stomach (ventriculus) of birds and has not been identified elsewhere in the body or in the environment. 10 In mucosal scrapings and in the feces, the organism is a stiff, straight rod that is 20 to 80 μm long and 2 to 3 μm wide with rounded ends. It is gram positive, but only the cytoplasm stains with the Gram stain. 1 Infection has been described in a range of species of birds including chickens, turkeys, ostriches, several species of parrots, and both captive-bred and wild finches. 1–3 , 5 , 7–10 , 12 , 14 , 16 , 17
The prevalence of M. ornithogaster infection is highest in captive-bred budgerigars (Melopsitticus undulatus), parrotlets (Forpus spp.), and canaries (Serinus canaria) and can be found in these birds throughout the world. 1–3 , 10 , 16 Most M. ornithogaster infections cause little detectable disease. However, M. ornithogaster has been associated with a chronic wasting disease in budgerigars, canaries, and finches and an acute hemorrhagic gastritis in budgerigars and parrotlets. 1–3 , 5 , 10 , 16 A stunting syndrome was noted in chickens naturally infected with M. ornithogaster, but these birds were also affected by a number of other pathogens. 7 , 8 Experimentally infected chickens did not show clinical signs of disease but had a reduced feed-conversion ratio as compared to controls. Microscopically, the lamina propria of the isthmus of the infected chickens contained greater numbers of lymphocytes and plasma cells than did the controls. 11
Efforts to grow M. ornithogaster on traditional bacterial and fungal media have largely been unsuccessful. Gerlach reported isolating this organism once on MRS medium, a medium used to isolate Lactobacillus spp., and other investigators have reported that it can be grown in liquid media containing minimum essential media, 20% fetal bovine serum (FBS), and 5% sucrose. 5 , 13 In the liquid medium, M. ornithogaster is said to grow slowly, taking 2 weeks to double its concentration. Because it cannot be readily and consistently cultured, M. ornithogaster has been characterized only genetically, and all drug susceptibility trials have been done in vivo. 10
This report demonstrates that M. ornithogaster can be readily grown and repeatedly passaged in a micro-aerophilic environment. Its optimum growth characteristics, its ability to assimilate various carbon compounds, and its resistance to short-term oxygen exposure are described. Furthermore, an M. ornithogaster isolate is shown to be able to colonize the isthmus of the chicken and can be reisolated.
Materials and methods
Initial isolation attempts. Budgerigars were obtained from a local budgerigar breeder whose flock had previously been shown to have approximately 60% prevalence of M. ornithogaster infection. Five birds were anesthetized with an intramuscular injection of 5 mg of ketamine a and 1 mg of xylazine. b When birds were no longer responsive to a toe pinch, they were killed by a 0.1 ml intracerebral dose of euthanasia solution. c The combined proventriculus and ventriculus were dissected free and opened longitudinally. The isthmus was then gently rinsed with Dulbecco's phosphate-buffered saline d (PBS) to remove ingesta, the mucosa was scraped with a scalpel blade, and scrapings were suspended in 500 μl of PBS. Wet mounts were examined microscopically, and the sample from the bird with the highest concentration of M. ornithogaster was used.
To prepare the inoculum, the suspension was strained through 4 layers of surgical gauze to eliminate large debris. Microscopic examination showed that along with M. ornithogaster, some bacteria and host cells were still present in the filtrate. The concentration of M. ornithogaster was determined by counting the organisms on a hemocytometer, and approximately 100,000 cells were inoculated in 2 ml of broth or streaked onto agar in 24-well tissue culture plates. Liquid media tested included MRS broth, e Y M broth, f Niger seed broth supplemented with biphenol, 18 and basal medium Eagle g (BME) supplemented with 20% FBS and 1% or 5% glucose or 1% or 5% sucrose. Solid media were made by dissolving agar (1 g/100 ml) in 20% millet seed extract or BME containing 20% FBS and 1% or 5% of either glucose or sucrose. Millet seed extract was made by grinding 25 g of millet seed in 125 ml of water, heating it to 110°C for 10 min, and straining it through 4 layers of cheese cloth. Biphenol, 100 μl of a 1% solution of biphenol in 95% ethanol, was added to the millet seed extract. Each medium was tested at pH 4.0, 6.0, and 7.0 and 25°C, 38°C, and 40°C both aerobically and under microaerophilic conditions. h All media contained 100 U/ml penicillin and 100 μg/ml streptomycin. Each inoculated sample was monitored daily for evidence of elongation, branching, or increase in the number of organisms (liquid media) or the development of microscopic or grossly visible colonies (solid media) for 20 days.
Growth in semisolid agar, optimization of growth requirements, and assimilation assays using cultivated M. ornithogaster. Twelve wells of a 24-well plate were filled with BME containing 20% FBS and either 5% glucose or 5% sucrose and 0.3% agar. Approximately 1,000 cultivated M. ornithogaster organisms were mixed with the semisolid agar. The plate was incubated at 40°C under microaerophilic conditions and observed daily for growth.
To determine if concentrations of M. ornithogaster in liquid media could be monitored without directly counting the organism, multiple dilutions of M. ornithogaster ranging from 1 × 105 to 2 × 108 in 100 μl of BME containing 20% FBS and 5% sucrose were placed in triplicate in a 96-well, flat-bottom enzyme-linked immunosorbent assay plate. The optical density (OD) of each well was determined using a microplate reader i at a wavelength of 570 nm. OD values obtained were subtracted from a blank well containing media with no organisms and, the concentration versus optical density was graphed.
Optimization of liquid media conditions were carried out in 24-well plates containing 2 ml BME, pH 3.0. FBS was initially held constant at 20%, and glucose and sucrose concentrations tested at 1% and 5%. Then, sucrose concentrations were held at 5%, and concentrations of 1%, 5%, 10%, 15%, and 20% FBS were compared. To determine the effects of pH on growth rates, BME containing 20% FBS and 5% sucrose was adjusted to pH 2, 3, 4, 5, and 6. The effect of temperature on growth rate was determined with BME (pH 3.0) containing 20% FBS and 5% sucrose. Media were incubated at room temperature, 25°C, 38°C, and 42°C. Replicates (15 to 21) of each growth environment were used in each experiment. Growth of M. ornithogaster was monitored by determining the OD of 100 μl of 3 replicates from each experimental condition daily.
Assimilation assays were run using a commercial assimilation assay strip. j Initial trials were done to determine the initial optimum concentration of organisms and the duration of the incubation (data not shown). When the optimum experimental conditions were determined, assimilation assays were then repeated 3 times. BME containing 20% FBS was substituted for the minimal media provided with the kit because M. ornithogaster cannot be grown without serum Assimilation chambers were inoculated with 0.3 ml containing 1.0 × 105 organisms and incubated at 42°C under microaerophilic conditions for 14 days. After 14 days, the contents of each chamber were mixed with 500 ml of PBS, and the concentration of the organisms was determined by counting them with a hemocytometer. Assimilation reactions were considered positive if there was a greater than 100-fold increase in the number of organisms.
Repeated passaging and sensitivity to exposure to oxygen concentrations in room air. Flasks containing BME, 20% FBS, and 5% sucrose were inoculated with M. ornithogaster to an approximate concentration of 2 × 105 cells/ml. Flasks were allowed to grow in microaerophilic conditions at 42°C until the organisms covered the bottom of the flask (an approximate concentration of 2 × 106), at which time the cells were diluted 1:20 in fresh media and grown until they covered the bottom of the flask (approximately 4 days). The process was repeated 18 times in 72 days. A second isolate was passaged 22 times in 90 days.
M. ornithogaster cannot sustain growth under atmospheric oxygen concentrations. Diagnostic specimens are commonly shipped to diagnostic laboratories overnight, with the possibility of prolonged oxygen exposure occurring before cultures can be initiated. To test the ability of M. ornithogaster to withstand oxygen exposure, 21 aliquots of M. ornithogaster (a concentration of 5 × 105/ml) in BME containing 20% FBS and 5% sucrose were exposed to atmospheric oxygen at 5°C, 25°C, and 42°C for 24 hr before incubating them at 42°C in a microaerophilic environment. Optical density readings of 100 μl of the culture media were determined at 5 days after inoculation in triplicate. Controls included organisms preincubated at the same temperature but in a microaerophilic environment and organisms inoculated without exposure to air and incubated for an identical time.
Infection trials in white leghorn chicks. Day-old male white leghorn chickens were obtained from a local commercial breeder. k Chicks were provided with a heat lamp and supplied with chicken starter and water ad libitum. Chicks were divided randomly between control and treatment groups and allowed to adjust to their new environment for 8 hr. The control group was then administered 0.5 ml of BME containing 20% FBS and 5% sucrose by gavage. The treatment group was administered the same volume of media containing 1 × 105 organisms. Seven days after inoculation, birds were weighed and killed by an injection of 10 mg of ketamine and 2 mg of xylazine, and when they were no longer responsive to a toe pinch, a 0.3 ml intracerebral dose of euthanasia solution. The mean weight of each group was compared with the 2-tailed Student's t-test. The combined proventriculus and ventriculus of each bird was dissected free and bisected longitudinally. One half of the isthmus was scraped, the scrapings were suspended in 0.5 ml of PBS, and an aliquot was examined microscopically for the presence of organisms in a wet mount. Organisms from 1 bird were reisolated as described above. The second halves of the proventriculus and ventriculus of 4 control birds and 4 infected birds were fixed in 10% formalin, paraffin embedded, sectioned at 4 μm, and stained with hemotoxylin and eosin. All animal use including the method of euthanasia was approved by the Texas A&M University Laboratory Animal Care Committee.
PCR identification of the isolated organism. The fourth passage of M. ornithogaster isolated from the budgerigar and the second passage of M. ornithogaster isolated from an infected chick were pelleted and their DNA extracted by mechanical disruption and a Puregene DNA isolation kit. l Approximately 25% of the 18S rDNA (739 bp) from these organisms was amplified and sequenced as previously described. 15
Results
Isolation and growth requirements of M. ornithogaster. In this experiment, multiple culture media incubated at varying temperatures and oxygen concentrations were tested for their ability to support the growth of M. ornithogaster. M. ornithogaster grew in BME supplemented with 20% FBS and either glucose (1% or 5%) or sucrose (1% or 5%) at 37°C and 40°C and at pH 4.0 in the microaerophilic environment but not in any of the other solid or liquid media tested. Within 3 days, the organisms could be seen to elongate and branch. As they continued to grow, they formed dense clusters (Fig. 1). In the first 2 passages, fully differentiated epithelial cells from the budgerigar proventriculus were seen but were not present in subsequent passages. A linear relationship was found between the OD reading at a wavelength of 570 nm and the log of the concentration of M. ornithogaster in the range of 105 and 108 organisms per milliliter (data not shown), allowing growth to be monitored by spectroscopy in subsequent experiments.
Semisolid media (0.3% agarose in BME) containing 20% FBS and 5% sucrose or glucose in a microaerophilic environment also supported M. ornithogaster growth. Microscopically, growth was clearly evident by 4 days, and distinct, grossly visible colonies were detected by the 10th day of incubation. Colonies were white, opaque, raised, and had smooth to variably irregular margins. More and larger colonies were seen in the semisold agar containing sucrose as compared to the semisolid agar containing glucose. By 14 days, organisms had spread diffusely into the semisolid media.
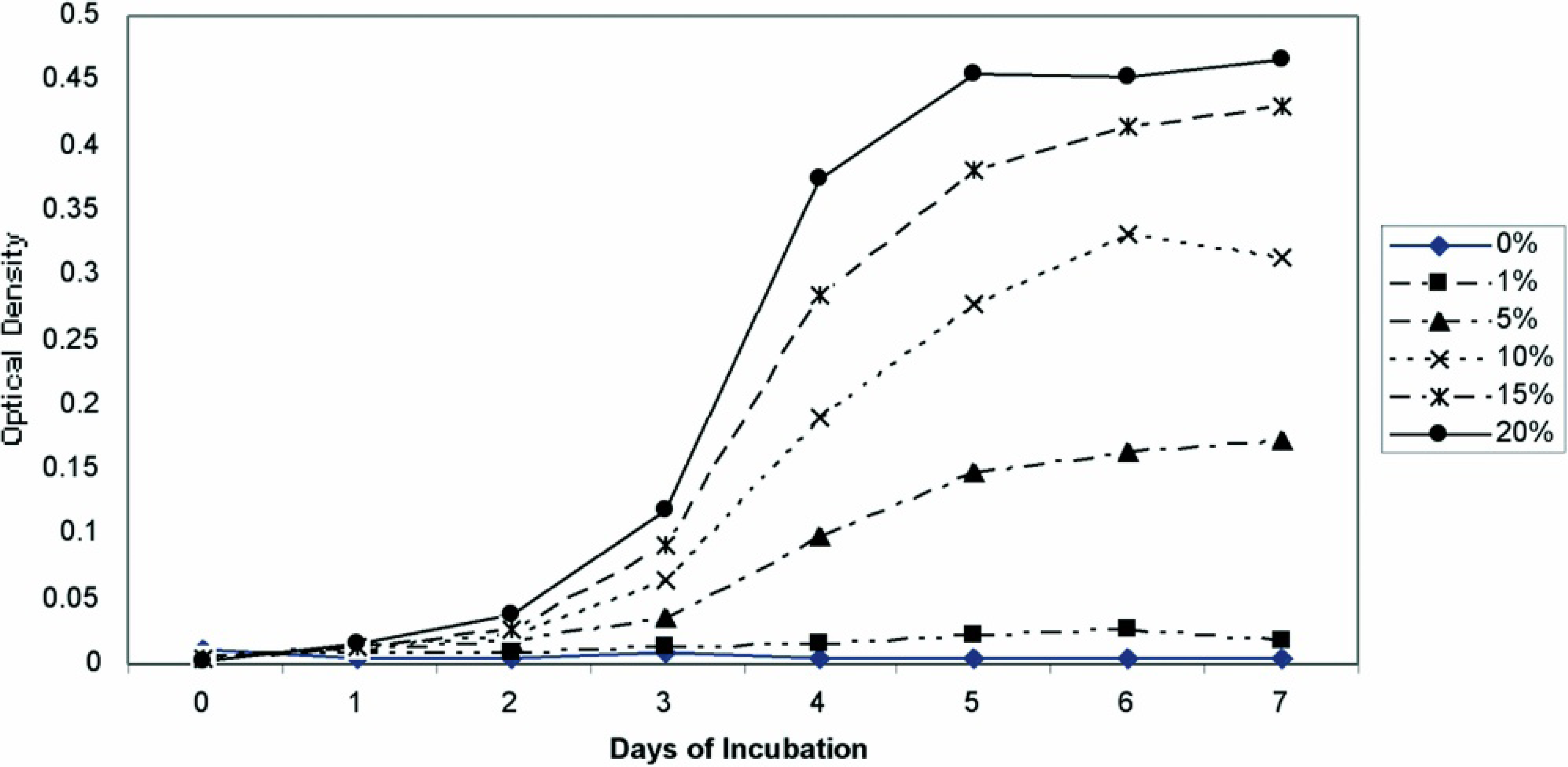
Optimization of growth conditions. M. ornithogaster grew optimally (an approximately 1,000-fold increase in 5–7 days) at pH 3 and 4, and growth could be detected with spectroscopy by 48 hours. Minimal growth was observed at pH 1, 5, and 6. Growth in the pH 2 medium was not observed until day 4, at which time it grew at a similar rate to organisms in the pH 3 and pH 4 media (Fig. 2). The pH of the medium that was initially pH 2 was rechecked on day 4 and was found to be pH 3. An FBS concentration of 20% was found to provide optimal growth, but growth in FBS concentrations of 5%, 10%, and 15% also occurred (Fig. 3). The optimum temperature for growth was 42°C, although sustained growth also occurred at 38°C. Minimal or no growth occurred at 25°C (Fig. 4). M. ornithogaster grew at a similar rate in 1% and 5% concentrations of glucose and sucrose but had a more sustained growth in 5% glucose and sucrose and reached maximal concentrations in 5% sucrose. A much smaller (10-fold) increase in organisms was observed in the medium that contained unsupplemented FBS (Fig. 5).
Assimilation assays. As expected, an approximately 10-fold increase was observed in the control sample (BME with FBS and no other substrate). A substrate was considered to be positive for assimilation if the concentration of M. ornithogaster in the well containing the carbon source exceeded 107 organisms per milliliter (at least a 100-fold increase in concentration) after 2 weeks. In all 4 experiments, M. ornithogaster assimilated glucose, sucrose, and trehalose. In 1 experiment, raffinose was intermediately positive (3 times the concentration of the control; Table 1).

Kohler illumination of a cluster of Macrorhabdus ornithogaster grown in optimized medium under microaerophilic conditions at 42°C. Bar = 60 μm.
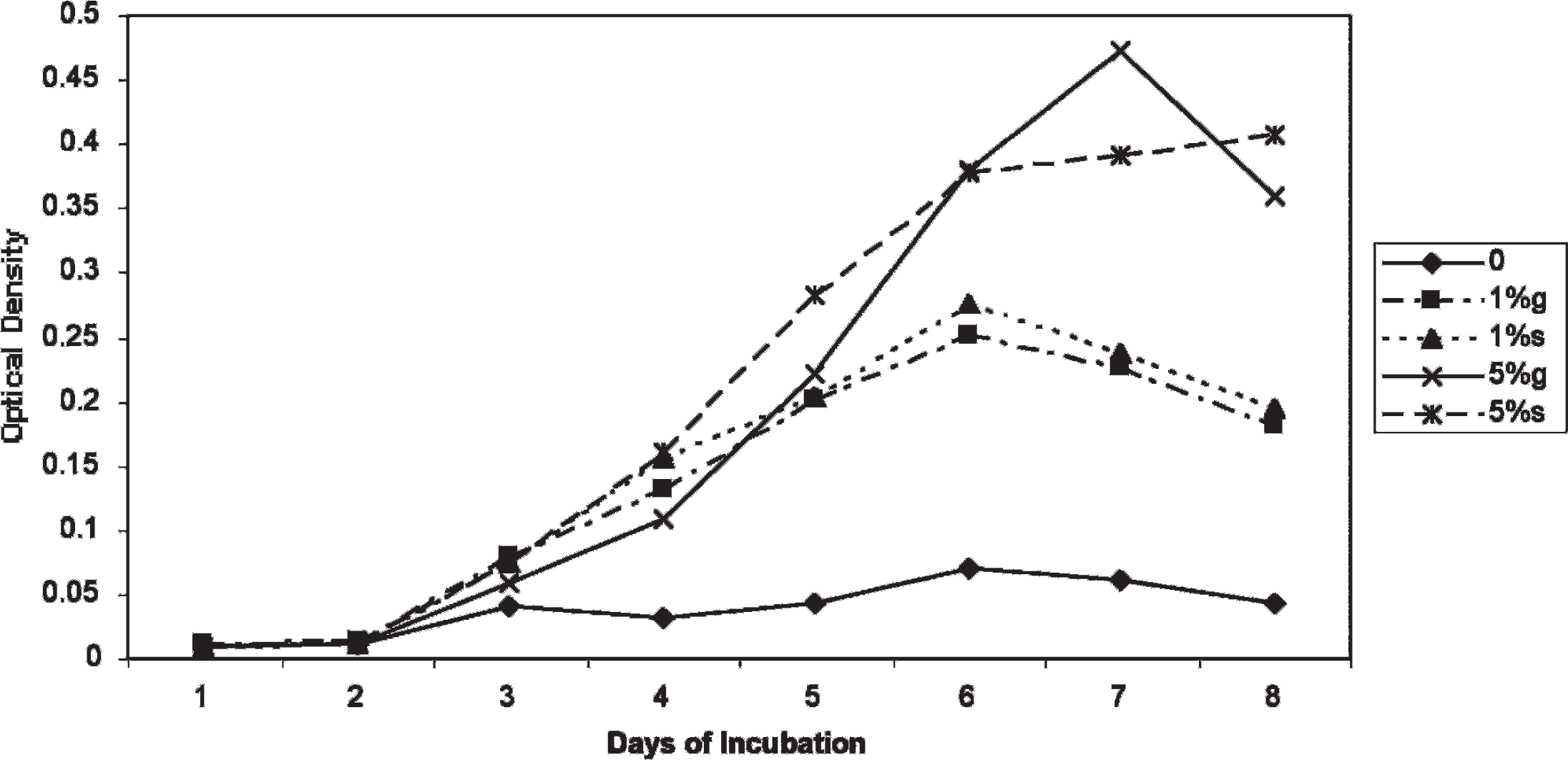
Impact of 24 hours of oxygen exposure. In this experiment, the ability of M. ornithogaster to withstand oxygen exposure was tested. M. ornithogaster in optimized culture medium was
The effect of pH on the grow rate of Macrorhabdus ornithogaster. The effect of fetal bovine serum concentration on the growth rate of Macrorhabdus ornithogaster.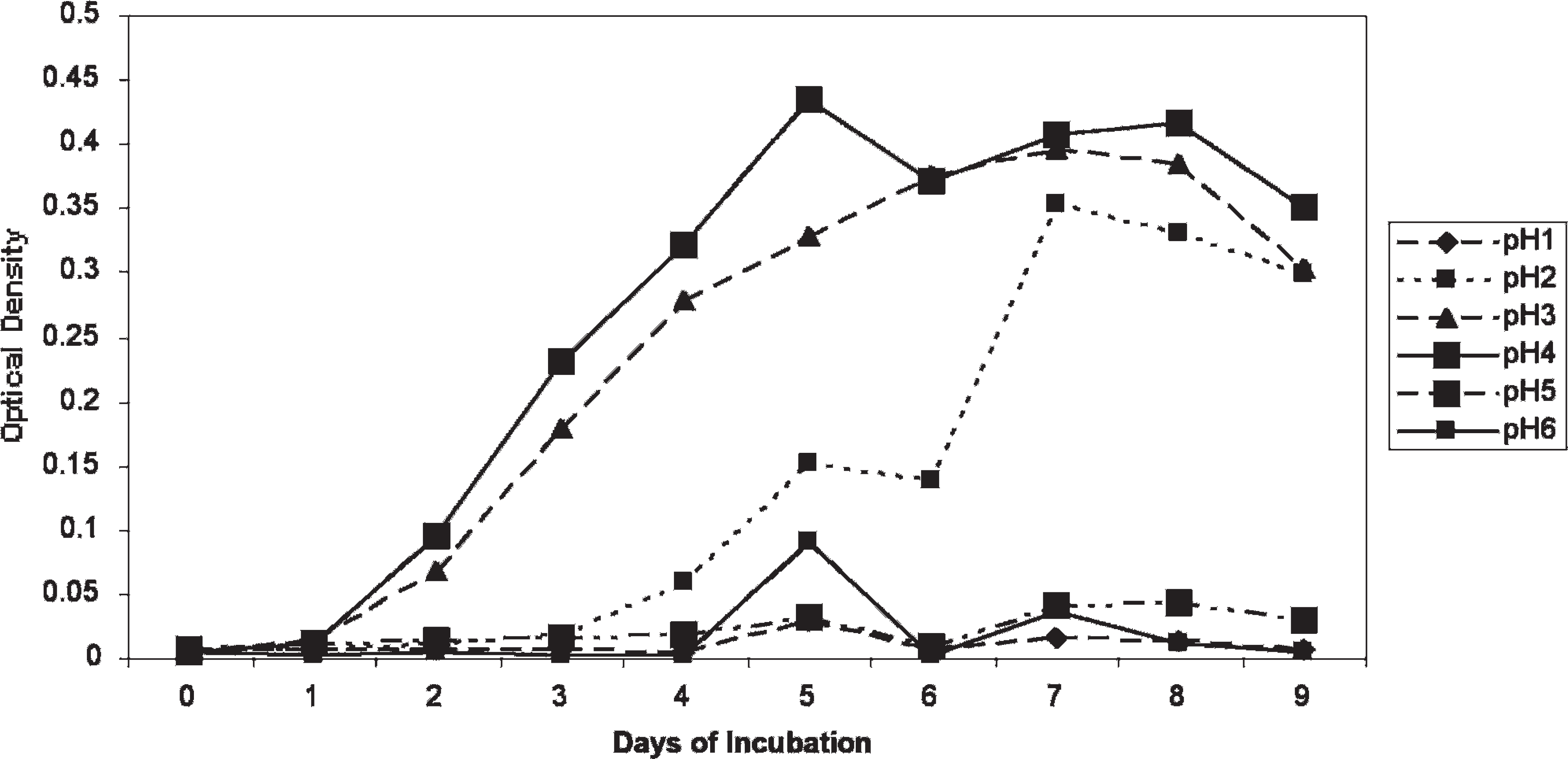
Chicken infection trial. M. ornithogaster was present in large concentrations in isthmus scrapings from the 11 infected chicks and was entirely absent from the 10 unaffected chicks. Infected chicks were observed to have diarrhea (soft, unformed stools), but the control chicks did not. The mean body weight of the infected chicks was 66.6 g (95% confidence interval, 62.3–70.9 g) and of the uninfected chicks was 72.9 g (95% confidence interval, 67.4–76.4 g; P = 0.061). Histologically, masses of M. ornithogaster were found in the transitional koilin, penetrating down to mucosal glands of the isthmuses of the infected chicks. The koilin was disrupted and fragmented as compared to the control birds, and there was a mild to moderate heterophil infiltrate in the lamina propria of the infected birds that was not present in the sham-infected control chicks (Figs. 7, Figs.8). Microabscesses were also present within the koilin, and in 1 bird, there was a localized lymphoplasmacytic infiltration into the lamina propria. M. ornithogaster from scrapings from the isthmus of an infected chick grew readily in the optimized medium.
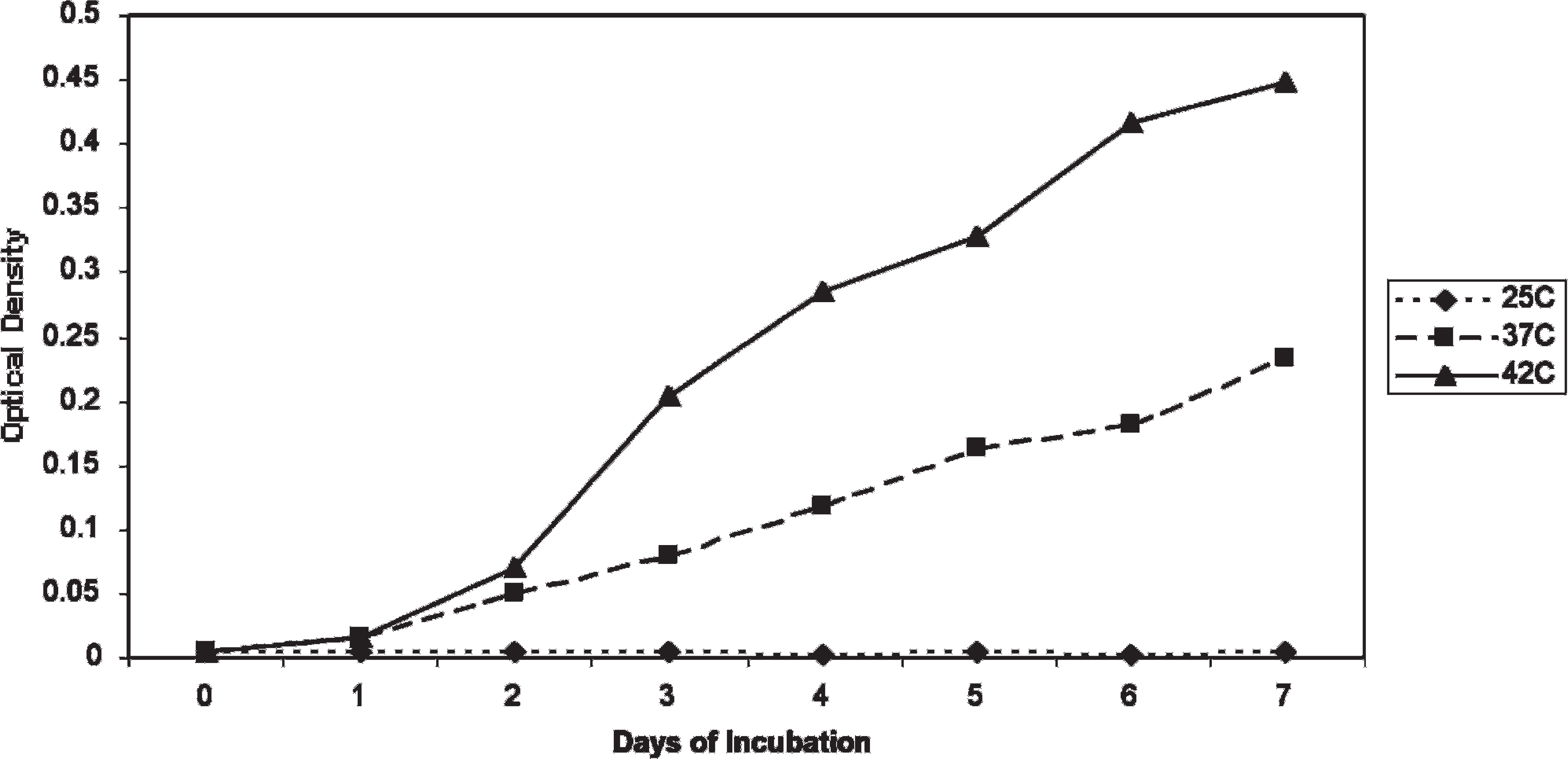
The effect of incubation temperature on the growth rate of Macrorhabdus ornithogaster.
Growth curves of Macrorhabdus ornithogaster in 1% and 5% glucose and sucrose and a medium not containing sugar.
Ability of Macrorhabdus ornithogaster to assimilate 19 carbon sources.

Each experiment was repeated 3 times.
One sample had a concentration that was less than the other positive carbon sources but was 5 times greater than the negative carbon sources.
Sequence comparisons. A 761-bp sequence comprising approximately 25% of the 18S rDNA was amplified from the M. ornithogaster isolate used to inoculate the day-old chicks and the organisms isolated from stomach scrapings of an infected chicks 7 days later. The sequences were compared and found to be identical. Alignment of this sequence to the originally reported M. ornithogaster sequence demonstrated 97% homology with 5 gaps and 17 point mutations. 15 The sequence was submitted to GenBank (accession number DQ231141).
Discussion
M. ornithogaster commonly infects many species of birds and under some circumstances is a significant pathogen. Study of this organism has been hampered because it could not be grown in vitro. The purpose of this study was to discover and optimize the conditions that would allow it to grow. The findings show that M. ornithogaster requires FBS, a narrow pH range of 3 to 4, and a microaerophilic environment in order to grow. Consistent with its growth in birds that have a high body temperature, it was also found to grow optimally at 42°C. Using optimized conditions, the organism was able to be subcultured repeatedly. This discovery means that it will now be possible to rapidly screen M. ornithogaster against potential therapeutic agents without having to use live animals. Using these culture conditions, it may be possible to grow M. ornithogaster directly from feces or from a proventricular lavage.
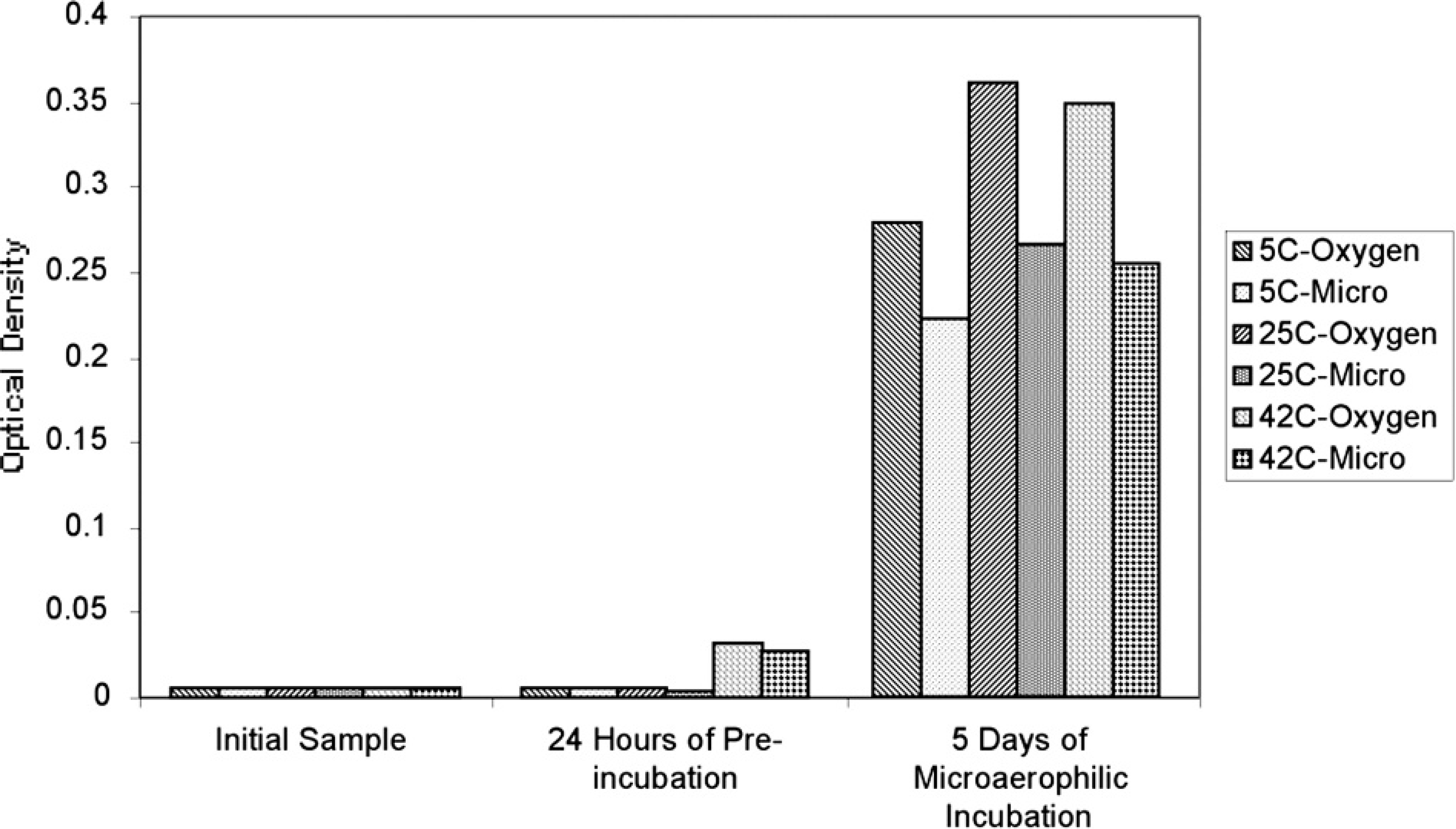
Effect of 24 hours of preincubation in microaerophilic conditions or atmospheric oxygen at 3 temperatures on the growth of Macrorhabdus ornithogaster.
Unexpectedly, both of the original isolates stopped growing after being subcultured 18 and 24 times, respectively. The specific cause of the isolates' loss of viability is not known. However, since they were growing robustly until the passage at which they stopped growing, it is suspected that air leakage into the microaerophilic chamber or an improper pH of the growth media and not inadequate nutrient supplies caused the loss of the isolates.
Previously, it was reported that M. ornithogaster could be isolated on MRS agar. 5 In this current study, M. ornithogaster did not grow on MRS agar, and given its sensitivity to oxygen and apparent need for some nutrient in FBS, it is unlikely that the organism isolated on MRS agar was in fact M. ornithogaster.
Exposure to oxygen for 24 hours at 5°C, 25°C, and 42°C improved the growth rate of M. ornithogaster over organisms incubated at the same temperature in microaerophilic environments. These data suggest that the organism can survive in the environment for at least limited periods of time and that organisms may still be recoverable from carcasses submitted for necropsy that have been dead for 24 hours or less.
These data also suggest that initial short exposures to atmospheric oxygen concentrations may enhance the growth of M. ornithogaster during attempts to isolate it. The short-term growth of M. ornithogaster in atmospheric oxygen explains the observations of other investigators who found that M. ornithogaster concentrations doubled when incubated at 38°C in medium containing FBS and sucrose with atmospheric oxygen. 13 It is theorized that what these investigators observed was a short period of growth that occurred in the first 1 to 2 days, followed by a cessation of growth and a separation of the newly formed branches.
During the pH experiment, M. ornithogaster was found to modify its environment, creating an environment that permitted its growth. When inoculated into a medium of pH 2, it increased the medium pH to 3, at which point it was able to grow. Given that M. ornithogaster lives in very dense colonies in the isthmus of the bird's stomach, this may mean that it may be able to locally increase the pH of its environment when conditions are not optimal. A previous investigation suggested that M. ornithogaster could not tolerate acid environments. In that report, the stomach pH in birds without infection ranged from 0.7 to 2.3 as compared to the stomach pH of infected birds, which ranged from 7.0 to 7.3. 16 It is not clear from that paper whether the investigators actually measured the pH of the microenvironment where the M ornithogaster grew. The data do not suggest that the pH change observed in this previous investigation could have been caused by M ornithogaster or that it would be beneficial for this organism to cause the pH to rise to these levels, as M ornithogaster can grow only in the range of pH of 3 to pH of 4. However, the data suggest that if the local pH at the isthmus could be raised above pH 4.0, growth might be inhibited. An attempt to inhibit M ornithogaster growth by raising the gastric pH with cimetidine has been done without success in 2 treated budgerigars. 4 However, the gastric pH of these birds was not measured, and it remains uncertain if the cimetidine was effective in increasing the gastric pH above 4.
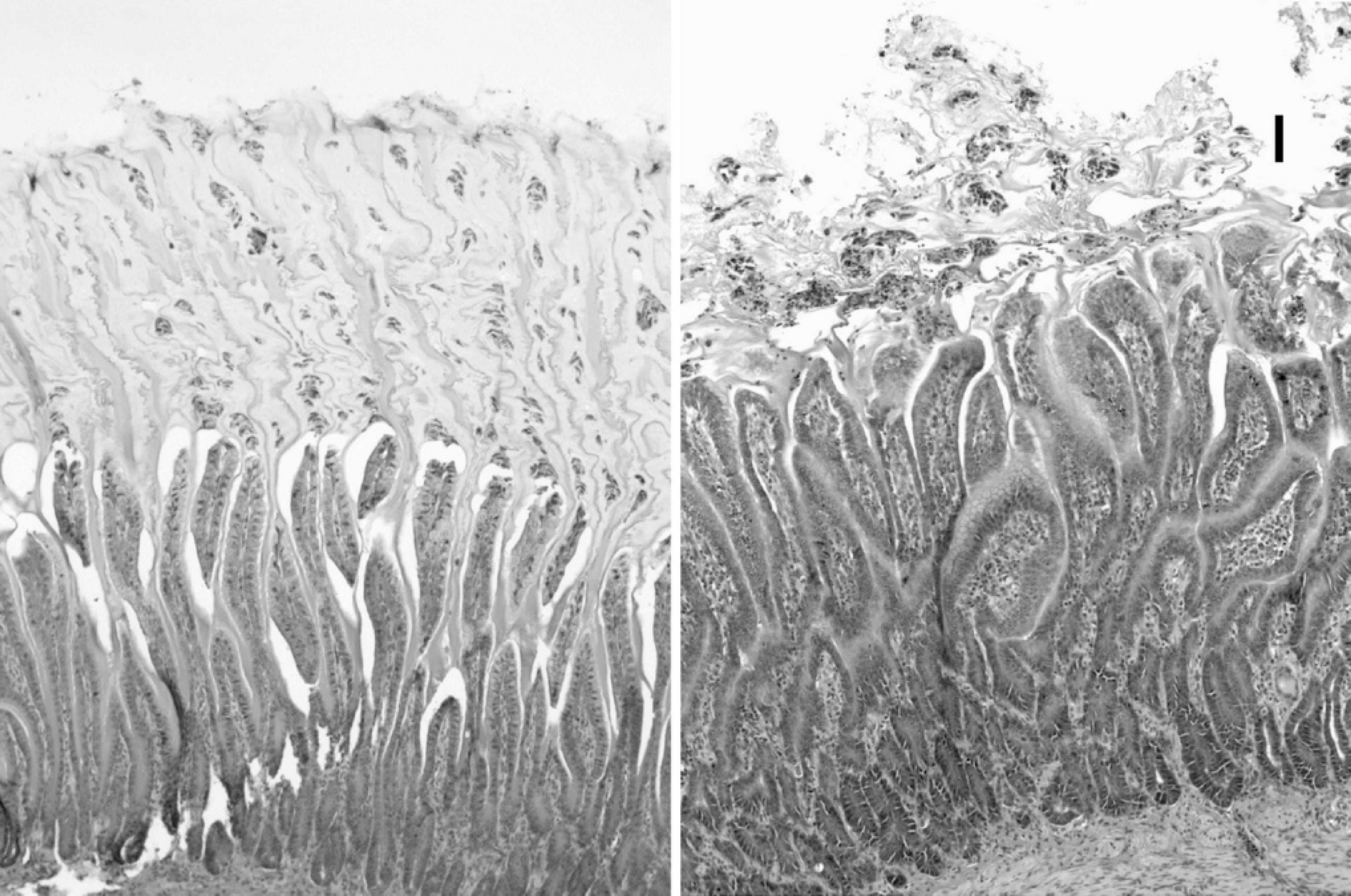
Photomicrographs of hemotoxylin and eosin-stained sections through the gastric isthmus of a 7-day-old control (
M. ornithogaster consistently assimilated glucose, sucrose, and trehalose. In 1 of 3 attempts, it grew better in raffinose than the controls but not as well as those in glucose, sucrose, and trehalose. This discrepancy may be the result of the fact that the organisms used in the assimilation assays were not grown from a single organism. Because M. ornithogaster could not be grown on solid media, it was not possible to use organisms from a single colony to inoculate the assimilation wells. It is likely, then, that a small percentage of the organisms inoculated may have had the ability to assimilate raffinose, but most organisms did not. As a result, when by chance, raffinoseutilizing organisms were in sufficient concentration in the initial inoculum, they grew to concentrations that were detectably different from the control samples.
Previously, it was shown that M. ornithogaster obtained directly from budgerigars could colonize the isthmus of chickens inoculated at 1 day of age. 11 These birds showed a mild to moderate lymphoplasmacytic infiltration of the isthmus when necropsied 28 days after infection. Although M. ornithogaster was suspected to cause these lesions, it could not be ruled out that other organisms present in the isthmus scrapings might have also elicited this response. In the current experiment, Koch's postulates were fulfilled by inoculating day-old chickens with a pure culture of M. ornithogaster, causing colonization of the isthmus and reisolating the same organism from the lesions. In contrast to the previous study in which a lymphoplasmacytic response was observed, a heterophilic response was observed in the 3 infected chickens in this study, and a combined heterophilic and lymphoplasmacytic response was observed in the fourth chicken. The heterophilic response most likely represents an acute response to infection as these birds were killed at 1 week, whereas the lymphoplasmacytic response seen in the previous experiment could represent a more chronic response seen in birds infected for 4 weeks.
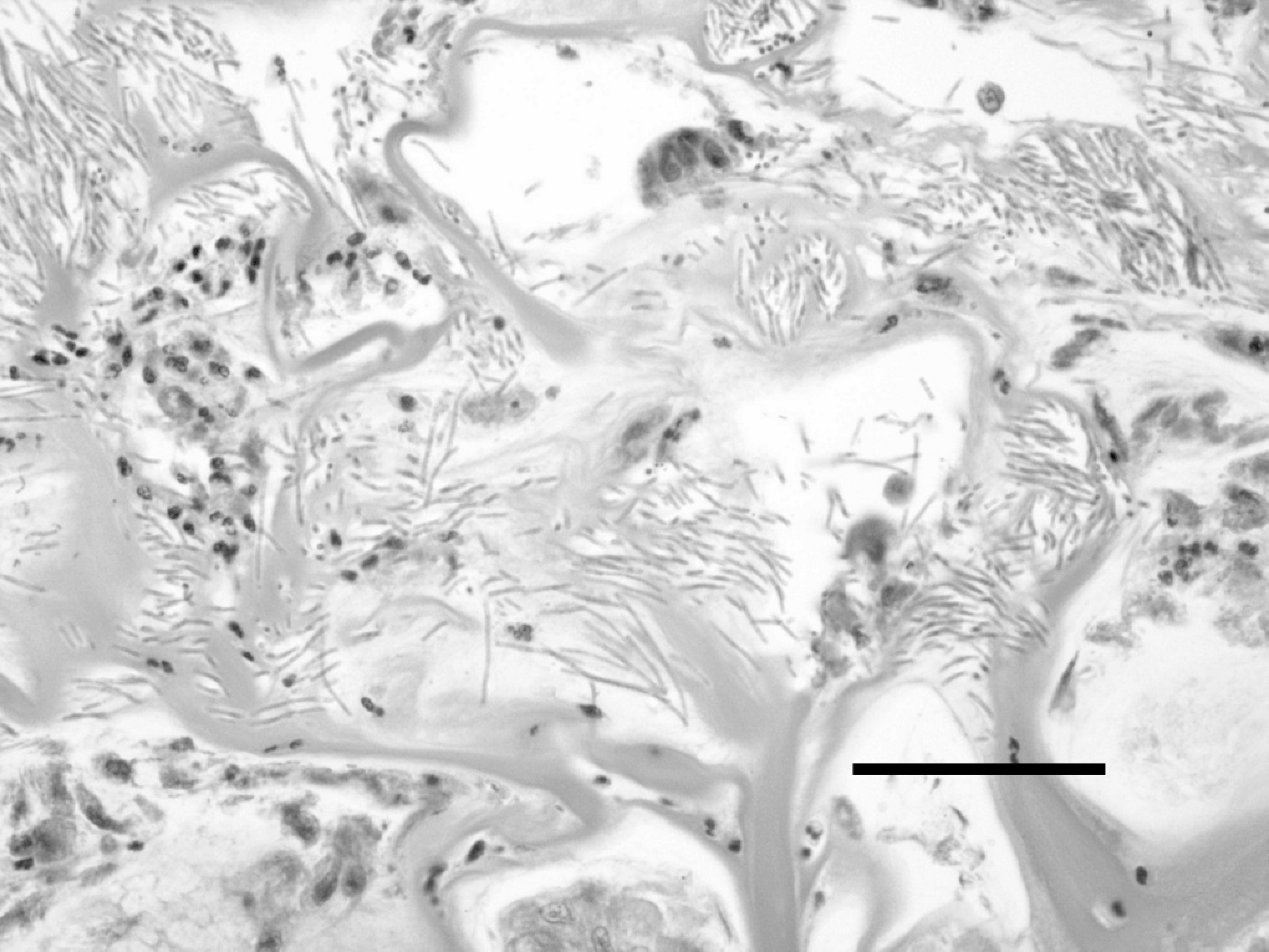
Photomicrograph of a hemotoxylin and eosin-stained section of the koilin layer of a 7-day-old chicken infected with Macrorhabdus ornithogaster. Numerous M. ornithogaster are present in the koilin. Bar = 100 μm.
Previous experiments have also suggested that M. ornithogaster had an effect on infected chickens by reducing the rate of their weight gain. 11 The duration of the infection trial was much shorter in this experiment (7 days compared to 28 days); however, a trend toward a reduced weight gain was also seen in infected birds as compared to controls. Diarrhea was seen in the infected chicks and not in the controls, suggesting that infection with M. ornithogaster was responsible for the diarrhea.
The sequence of the M. ornithogaster isolate used in this experiment is only the second reported sequence for this organism. The fact that it differed by 3% from the original sequence suggests that different genotypes of M. ornithogaster may exist and that additional sequence data may prove useful for epidemiologic studies and determining if there are different pathotypes of M. ornithogaster.
Acknowledgements
This work was funded by the National Institute of Animal Health, Japan, the Schubot Exotic Bird Health Center, and donations from the Central Indiana Bird Club and the National Parrot Rescue and Preservation Foundation. The authors thank Cletus Kurtzman, Kathleen Logan, Bradley R. Weeks, and Bruce Simpson for their advice and expertise.
Footnotes
a.
Phoenix Scientific, Inc., St. Joseph, MO.
b.
Bayer Corp., Shawnee Mission, KS.
c.
Fatal Plus; Vortech Pharmaceuticals, Dearborn, MI.
d.
Sigma Chemical Co., Saint Louis, MO.
e.
EM Science, Gibbstown, NJ.
f.
Becton Dickinson, Sparks, MD.
g.
MediaTech, Inc., Herdon, VA.
h.
Campygen Atmosphere Generation System; Oxoid Limited, Basingstoke, Hampshire, UK.
i.
Bio-Rad, Hercules, CA.
j.
Api 20 C AUX: bioMérieux, Hazelwood, MO.
k.
Hy-Line International, Bryan, TX.
l.
Gentra, Minneapolis, MN.
