Abstract
An experimental transmission study aimed at fulfilling Koch's postulates for a herpesvirus-associated stomatitis-rhinitis in Mediterranean tortoises is presented. Clinical, pathologic, serologic, and molecular studies were performed linking tortoise herpesvirus with the pathogenesis of stomatitis-rhinitis. Four adult Greek tortoises received either intranasally or intramuscularly two tortoise herpesvirus isolates by primary experimental infection and secondary challenge 11 months later. After the primary experimental infection and the secondary challenge, clinical signs of illness developed, which included conjunctivitis, diphtheritic oral plaques, and oral discharge. At 4 weeks after the secondary challenge, all tortoises were humanely euthanatized and evaluated. Although neutralizing antibodies developed after the primary experimental infection, they apparently did not prevent the later development of recurrent clinical signs. Polymerase chain reaction (PCR) and reverse transcription-PCR analyses allowed sensitive characterization of the systemic distribution of the herpesvirus DNA sequences and their presence in the cranial nerves and brains of the infected tortoises. Despite the failure to recover the herpesviruses used in the transmission study, the findings support the premise that tortoise herpes-virus is a primary pathogen of Greek tortoises.
A disease characterized by necrotizing stomatitis and rhinitis has been reported in several species of recently imported and captive tortoises worldwide. This disease is particularly problematic in Greek (Testudo graeca) and Hermann's (T. hermanni) tortoises. Clinical signs range from mild conjunctivitis and clear nasal discharge to severe stomatitis and mucopurulent rhinitis. Signs of lower–respiratory tract and central nervous system (CNS) disease also have been seen. 7,22 Although Mycoplasma agassizii has been documented as a cause of rhinitis (upper–respiratory tract disease) in gopher (Gopherus polyphemus) 2,13 and desert (G. agassizii) 2 tortoises, stomatitis is not a feature of this disease. Several reports have described eosinophilic intranuclear inclusions in Mediterranean and other tortoises with stomatitis and rhinitis. Herpesvirus-like particles have been seen using electron microscopy. 3–7,9,10,11,14,18,25 Herpesviruses have also been isolated from several of these cases. 11 Despite numerous descriptive reports, fulfillment of Koch's postulates demonstrating a causal relationship between herpesvirus and stomatitis-rhinitis in tortoises has not been reported.
To determine the role of tortoise herpesvirus in the pathogenesis of stomatitis-rhinitis disease in tortoises, and eventually fulfill Koch's postulates, we conducted a transmission study on Greek tortoises using two herpesviruses that had been isolated from Hermann's tortoises. Pathologic, serologic, and molecular diagnostic tests were used to confirm the pathogenicity of the viruses used in this study and thus clearly demonstrate their role in stomatitis-rhinitis of tortoises.
Materials and Methods
Animals
Five male Greek tortoises, that were used in a previous study designed to validate an enzyme-linked immunosorbent assay (ELISA) test for herpesvirus exposure detection in tortoises, 16 were used in this study.
Virus preparation and viral DNA purification
An American (HV 1976) and a European (HV 4295/7R/95) isolate of tortoise herpesvirus previously purified and characterized 17 were selected for use in this study. The two isolates were grown, harvested, and sucrose purified according to protocols described elsewhere. 16,17 Viral DNA was extracted from the purified pelleted herpesvirus isolates as follows. Each of the purified pelleted herpesviruses was resuspended in 0.5 ml of cold TE (pH 8.0), and 500 µl of 2× virus extraction buffer (1% Sarkosyl [IBI, New Haven, CT], Tris-HCl 10 mM, 30 mM β-mercaptoethanol, 100 mM NaCl, 10 mM ethylenediaminetetraacetic acid, 26% sucrose [Fisher, Pittsburgh, PA]) was added to the mixture and incubated at 39 C for 30 minutes. Two hundred micrograms of proteinase K and 100 µg of ribonuclease A (Invitrogen, Carlsbad, CA) were added to the viral suspension and incubated overnight at 39 C. After incubation the DNA was phenolchloroform extracted and ethanol precipitated. 20 The purified DNA pellet was then dissolved in TE buffer (pH 8.0) and stored at 4 C until use.
Inoculation of viruses and evaluation of tortoises
As previously described, 16 of the five tortoises used in this study, four were randomly assigned to one of two treatment groups: group 1 tortoises (Nos. 1 and 3) were inoculated with the isolate from Germany (HV 4295/7R/95), and group 2 tortoises (Nos. 2 and 4) were inoculated with the Washington State isolate (HV 1976). The fifth tortoise served as an uninfected control. The inoculum administered during the primary experimental infection consisted of 1.5 × 104 tissue culture infectious dose 50 (TCID50) that was administered either intramuscularly (IM; tortoise Nos. 3 and 4) or intranasally (IN; tortoise Nos. 1 and 2). The control tortoise (No. 5) was given istotonic phosphate-buffered saline (PBS) both IN and IM. The blood-sampling schedule that was followed immediately after the primary experimental infection, up to the secondary challenge, has been described elsewhere. 16 At 43 and 45 weeks (IM and IN inoculation, respectively) after the primary experimental infection, the tortoises were challenged (secondary challenge) with 1.5 × 105 TCID50 of live virus according to the same protocol described above. Blood samples were obtained from each tortoise 2 days before and 5, 12, and 19 days after the secondary challenge. The last blood sample was collected just before necropsy (4 weeks after the secondary challenge). Plasma samples were tested for the presence of total (ELISA) and neutralizing (serum neutralization [SN] test) anti-herpesvirus antibodies (Fig. 1A, B, Table 1). Starting 7 days after the primary experimental infection and immediately after every blood sampling, nasal saline flushes and pharyngeal swabs were collected for viral isolation attempts. After the secondary challenge, saline nasal flushes and pharyngeal swabs were collected at 1, 2, 5, 7, 9, 12, 15, 16, 19 days after inoculation. Occasionally, ocular exudates were also collected for viral isolation attempts. Attempts were made to isolate virus from the nasal flushes and pharyngeal and ocular swabs. In contrast to the isolation protocol adopted previously, 16 the supernatant of the samples collected after the challenge was not filtered to enhance the sensitivity of the cell line used for the isolation of the virus.
SN results for tortoises immunized with HV4295/7R/95 and HV1976 IN and IM.∗

∗ SN titers obtained for the plasma samples collected from the tortoises during the secondary challenge are given. The antigens used for the SN and for the challenge are listed at the top and at the bottom of the table, respectively. A total viral amount of 102 TCID50 was used in the SN. The control tortoise (No. 5) remained seronegative for the duration of the study and has not been included in the table.
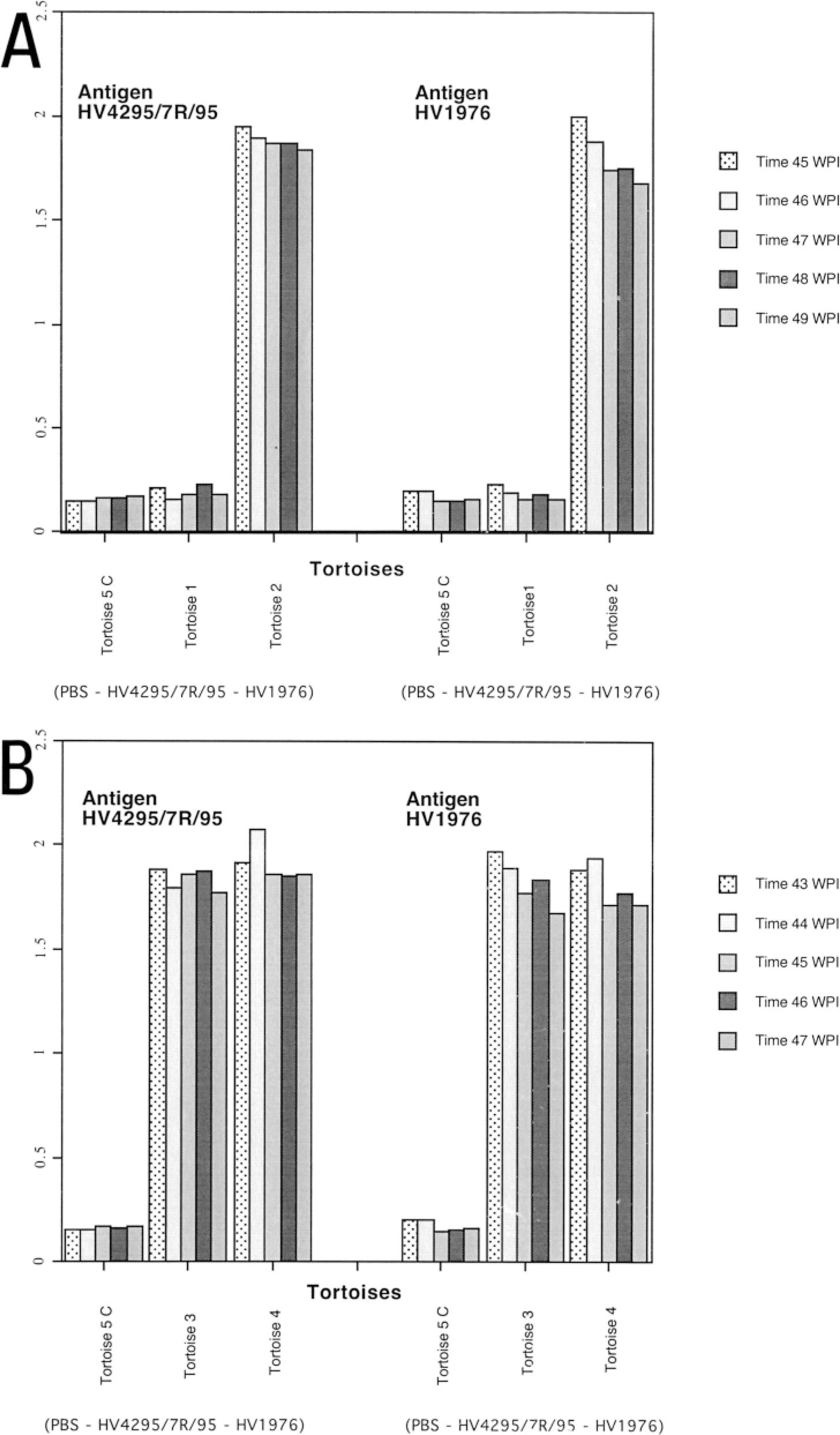
ELISA results for tortoises infected with HV1976 and HV4295/7R/95.
As previously described, 16 the tortoises in the study were checked daily for the first 2 weeks after the primary experimental infection and then every 3–4 days until the secondary challenge (43–45 weeks after infection [IM and IN, respectively]). After the secondary challenge the tortoises were examined twice a day for the duration of the study. A thorough physical examination was performed every time a blood sample was collected.
Serologic tests
All blood samples collected from the tortoises were tested for the presence of anti-herpesvirus antibody using an SN and a recently described ELISA. 16
To investigate the role of Mycoplasma spp., known to be pathogenic for tortoises, an aliquot of the plasma collected from each tortoise was tested by ELISA for mycoplasmal exposure, 21 at the beginning and at the end of the study.
Pathology
All the tortoises were humanely euthanatized at the end of the study with an intracoelomic injection of sodium pentobarbital (47 and 49 weeks [IM and IN, respectively] after the primary experimental infection 16 [4 weeks after the secondary challenge]). A thorough necropsy of the tortoises was conducted. During deep anesthesia and before euthanasia, blood was collected, and plasma was separated by centrifugation and stored at − 80 C. Cerebrum, medulla, cerebellum, cranial nerves, palate, tongue, oral mucosa, oral plaques, glottis, pharynx, trachea, lungs, esophagus, stomach, small and large intestines, kidneys, bladder, testes, spleen, liver, heart, brachial plexus, feces, urine, and lymphocytes were collected for histopathology, electron microscopy, viral isolation attempts, and DNA or RNA extraction for polymerase chain reaction (PCR) and reverse transcription–PCR (RT-PCR) detection of herpesvirus genomes and viral messenger RNA (mRNA) sequences, respectively. Tissues collected for histopathology were fixed in 10% neutral formalin, embedded in paraffin, sectioned at 5 µm, and stained with hematoxylin and eosin (HE). Five-micrometer tissue sections were also prepared for immunohistochemical staining. Tissue samples collected for electron microscopy were fixed in Trump's solution 12 (formaldehyde and glutaraldehyde mixture) and stored at 4 C. Tissue samples for viral isolation attempts were collected aseptically and placed in sterile plastic tubes (Fisher) and stored at −80 C. For RNA and DNA isolation, the samples were aseptically removed and placed into RNase/DNase–free plastic containers (Fisher), kept on ice until completion of the necropsy, and then stored at −80 C until use.
Immunohistochemistry
Direct immunohistochemical staining for demonstrating herpesvirus antigen in the infected tortoise tissues was performed as described previously. 8,17
All tissues that were collected from each tortoise were examined for herpesvirus antigen by immunohistochemistry. Pooled preimmunized rabbit sera served as negative control primary serum. A portion of paraffin-embedded glottis obtained from a Hermann's tortoise that died with clinical signs consistent with tortoise herpesvirus and that was histologically positive for the presence of herpesvirus intranuclear inclusions (HE) was used as the positive control. 17 Additionally, paraffin-embedded lung, trachea, and liver from a normal Boa constrictor constrictor served as a negative control.
Postmortem virus isolation
Virus isolation was attempted from all the other PCR-positive tissues (Table 2), except the CNS and cranial nerves because of the very limited amount of tissue available (they were all used for PCR and RT-PCR testing). The tissue extracts that were obtained (10% wt/vol homogenate in complete 16 Dulbecco modified Eagle medium/F12 medium [Invitrogen]) were inoculated onto TH-1 cell confluent monolayers. Cells were checked daily, and the isolation attempt was considered unsuccessful if no cytopathic effects were detectable after two blind passages repeated at 7-day intervals. 11
Summary of the semiquantitative PCR analysis.∗

∗ The values presented in this table are expressed in viral units (arbitrary measure not necessarily corresponding to an equivalent number of viral genomes).
† Abbreviations: Rx = right; Lx = left; CN = cranial nerve.
Preparation of DNA and RNA samples
DNA samples
Total DNA from infected TH-1 cell cultures or from the tissues of the experimental animals was isolated using a commercially available kit (DNeasy, tissue kit, Qiagen, Valencia, CA) according to the instructions of the manufacturer, and it was stored at −80 C.
RNA samples
Total RNAs were extracted (Trizol, Invitrogen) from virus-infected cell cultures and from the tissues of the experimental animals and treated with DNAase I (1 U for each microgram of RNA [Invitrogen]). The DNA-free total RNAs were then alcohol precipitated, 20 resuspended in RNase-free water, and stored at −80 C until use.
Subgenomic library
A subgenomic library of tortoise herpesvirus isolate HV 1976 was obtained by digesting 4 µg of purified herpesviral DNA with the restriction endonuclease HindIII (Invitrogen) and shotgun cloned into the HindIII restriction site of the plasmid pGEM 13 Zf+ (Promega, Madison, WI) according to standard protocols. 19 Sixteen of the 31 HindIII clones that were identified after purification and screening 19,29 were submitted for partial or complete sequencing using the Big Dye Terminator technique (Perkin–Elmer, Applied Biosystems, Foster City, CA) to the sequencing laboratory of the Center for Mammalian Genetics, University of Florida. A HindIII clone of 1.413 kb (No. 11), containing the partial sequence of the large and small subunits of the ribonucleotide reductase (RR) homologous gene (UL 39 and UL 40) of HSV1, was selected as a target for the development of the PCR and RT-PCR techniques. Full amplification and sequencing of the same gene fragment from the HV 4295/7R/95 herpes-viral genome followed.
Polymerase chain reaction
The sense (5′-TGCACTTTGATGCGTGGGAT-3′) and the antisense (5′-TTGATCGTATTCGAATGCCG-3′) primers for the PCR reaction used in this study were designed using the sequencing information obtained from the 1.413-kb HindIII clone (No. 11). The expected PCR product was a 386–base pair fragment (GenBank Accession AY338245) located within the coding region of the large subunit of the tortoise herpesvirus homolog of the HSV-1 RR gene (UL 39). The reaction mixture (final volume of 100 µl) contained 500 ng of DNA template; 200 µM of each deoxyguanosine triphosphate (dGTP), deoxyadenosine triphosphate (dATP), deoxycytidine triphosphate (dCTP), and deoxythymidine triphosphate (dTTP); 20 mM Tris-HCl (pH 8.4); 50 mM KC1; 3 mM MgCl2; 40 pmol of each primer; and 2.5 units of Taq polymerase (Invitrogen) in deionized sterile water. The amplification reaction was carried out in a PTC 100 thermocycler (MJ Research, Watertown, MA) and consisted of 35 cycles. After initial denaturation of the DNA template for 1 minute at 94 C, 35 amplification cycles consisting of a denaturation step (1 minute at 94 C), an annealing step (30 seconds at 54 C), and an extension step (30 seconds at 72 C) were performed. During the last amplification cycle the extension step was extended to 8 minutes to exhaust the DNA polymerase. The samples of the resulting PCR products were routinely sequenced to verify the specificity of the PCR amplification.
PCR sensitivity assay
The dilutions of known amounts of plasmid DNA carrying the target viral sequence were either amplified alone or spiked into 500 ng of total cellular DNA as a means of evaluating the sensitivity of the PCR under ideal or “field” conditions, respectively.
Semiquantitative PCR analysis
All tissue samples collected from the tortoises used in this study were tested for the presence of herpesviral DNA by PCR. Five hundred nanograms of total DNA was used for each PCR reaction. The negative samples were then retested using increased amounts of DNA. Failure to detect a positive PCR signal using 1 µg of total DNA as template was considered evidence of a “confirmed negative” sample. All the tortoise tissue samples that were PCR positive for the presence of herpesviral DNA were subjected to semiquantitative analysis to determine the relative amount of viral DNA present in the tissues. Starting with 500 ng of total DNA from each sample, 10-fold dilutions were prepared and used in the reactions. The total amount of viral DNA in the tissue samples was measured in viral units (VU; 1 VU = one DNA target plasmid copy) and was determined by 1) detection of visible DNA bands corresponding to the PCR products of 10-fold–diluted total DNA samples (TDT) resolved in agarose gel and 2) densitometric reading (UVP Image Store 7500, UVP Inc., Upland, CA) of the DNA bands corresponding to the PCR products of 10-fold–diluted total DNA samples resolved in agarose gel (DDT). The theoretic VUs present in 500 ng of total DNA from each tissue sample were then determined. Samples that tested PCR positive for herpesviral DNA only when 1 µg of total DNA was used as template were considered as containing ≤50 VU/500 ng DNA and were not subjected to further analysis.
Reverse transcription–polymerase chain reaction
The target RNA sequence for the RT-PCR reaction was the RNA transcript corresponding to the DNA sequence used as target for the PCR reaction. The 20-µl mixture for the RT reaction contained 5 µg of total RNA; 0.5 µl of 1.5 A260 random hexamer primers; 500 µM of each of dGTP, dATP, dCTP, and dTTP; 50 mM Tris-HCl (pH 8.3); 75 mM KC1; 3 mM MgC12; 10 mM dithiothreitol; and 1 µl (200 U) of Superscript II (Invitrogen) (not added to the reaction mixture of the negative controls), and RNase-free sterile water was prepared according the instruction of the manufacturer. The reaction was carried out at 42 C for 50 minutes. Five microliters of the first-strand complementary (cDNA) mixture obtained in the RT reaction was used as template DNA for the PCR reaction.
RT-PCR sensitivity assay
The sensitivity of the RT-PCR was evaluated using the synthetic RNA transcript of the target sequence, synthesized using T7 RNA polymerase (Invitrogen) according to the instruction of the manufacturer. Tenfold dilutions of the synthetic target RNA transcript (starting at 1010 copies), either pure or spiked into 5 µg of total cellular RNA, were prepared for the RT-PCR sensitivity assay.
RT-PCR strategy
The tissue samples from all tortoises that were PCR positive for herpesviral DNA (Table 2) were tested by RT-PCR for detection of mRNA viral transcripts. The RNA samples from a single tortoise containing less than 5 µg of total RNA were sequentially pooled with other samples until 5 µg of total RNA was obtained.
Housekeeping gene as an internal control for the RT-PCR reaction
The sense (5′-TGCTTCTAAACCGGACTG-3′) and antisense (5′-AAATGTACAATCAAAGTCC-3′) DNA primers based on the sequence of a cDNA clone of Gallus gallus cytoplasmic β-actin (GenBank Accession: AW355616, gi: 6857629) were designed to detect and use the homologous tortoise gene as the internal control housekeeping gene for the RT-PCR reaction. The expected product was a 238–base pair fragment. The amplification reaction was carried out in a PTC 100 thermocycler (MJ Research, Watertown, MA) and consisted of 35 cycles. The reaction mixture was prepared as described for the RR PCR amplification described previously (except for the specific primers). After initial denaturation of the DNA template for 1 minute at 94 C, 35 amplification cycles consisting of a denaturation step (30 seconds at 94 C), an annealing step (30 seconds at 46 C), and an extension step (30 seconds at 72 C) were performed. During the last amplification cycle the extension step was prolonged up to 8 minutes to exhaust the DNA polymerase.
Results
Clinical signs
As already reported in a previous study, 16 after the primary experimental infection, and before the secondary challenge, the only consistent clinical sign detectable in all the infected animals was either a transient unilateral or a bilateral recurrent, mild to severe conjunctivitis. Attempts at virus isolation from multiple samples of ocular exudates were unsuccessful. Overall, the episodes of conjunctivitis that developed in tortoises that received an IM inoculum appeared to be more severe than in those receiving the inoculum IN.
Tortoise No. 5, the control (Fig. 2A), never showed any sign of conjunctivitis. Its eyes were always clear and free of discharge.
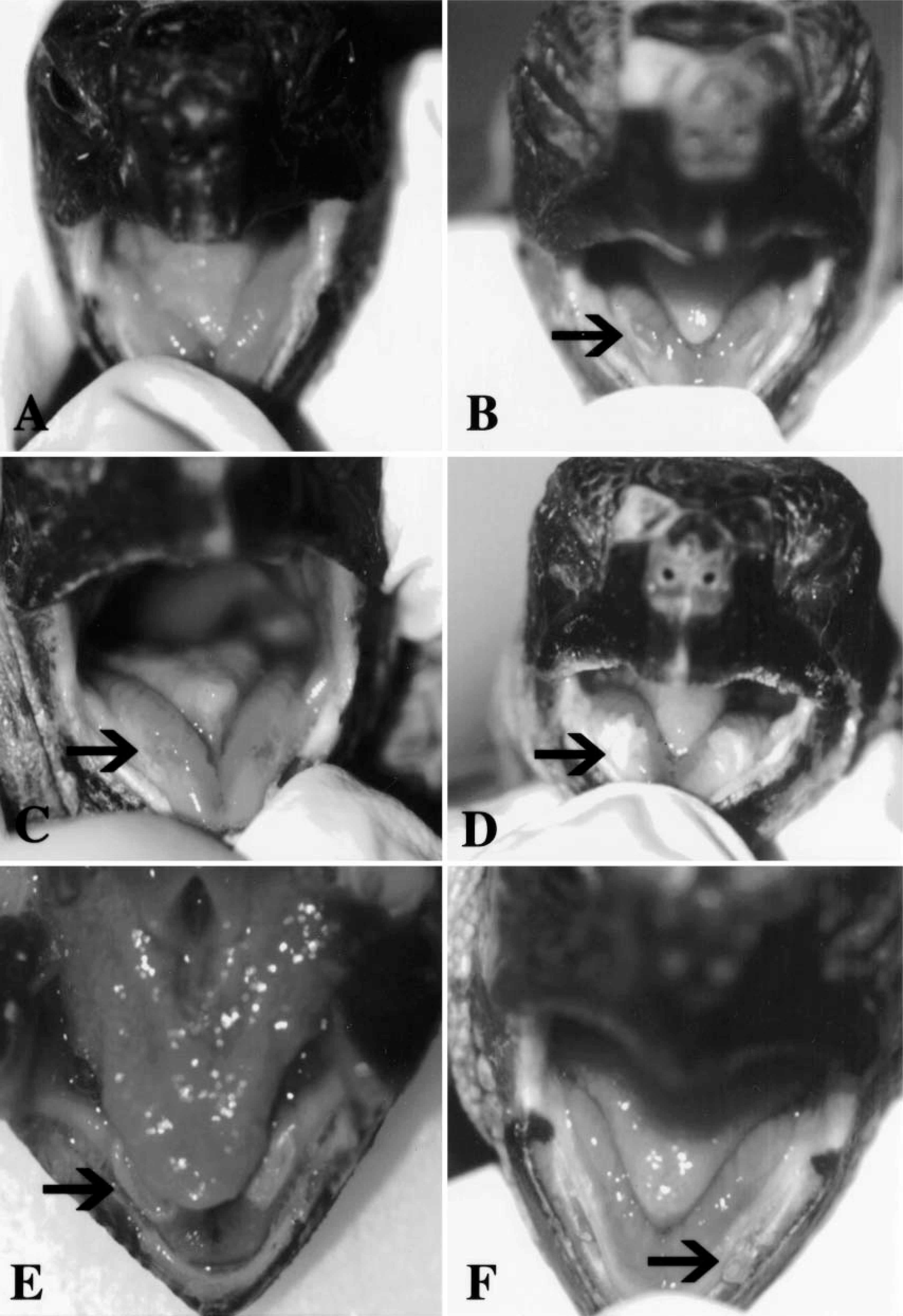
Progression of the oral lesions seen in the tortoises during the secondary challenge study.
After the secondary challenge, signs of conjunctivitis were observed in all four infected tortoises, and each tortoise developed a discoloration of the tip of the tongue, which appeared 1 or 2 days after the inoculation and lasted for no more than 72 hours (Fig. 2B). Tortoise No. 1 developed a yellowish plaque 1.5 × 3 mm in diameter in the cranial portion of the dorsum of the tongue at postsecondary challenge (PC) day 1. At PC day 12, tortoise No. 1 developed a bilateral symmetric hemorrhagic inflammation of the oral mucosa (Fig. 2C), which was soon followed (at PC day 15) by the development of two yellowish bilateral plaques on the mucosa lining the salivary gland area located laterally to the tongue (Fig. 2D). The plaques were reduced in size by PC day 20 (Fig. 2E) and completely resolved by PC day 24 (Fig. 2F). At PC day 8, lesions similar to those described above were observed in tortoise No. 4. After their appearance the plaques enlarged slightly and became ulcerated at PC day 11. Clear oral discharge was also observed. The oral plaques quickly regressed to one fourth of the initial size at PC day 17 and relapsed at PC day I22. Tortoise Nos. 2 and 3 never developed oral plaques similar to those observed for tortoise Nos. 1 and 4.
Serology
Serum neutralization test
Whereas tortoise Nos. 2–4 seroconverted against both the homologous and the heterologous isolates after the primary experimental infection, tortoise No. 1 remained negative for the presence of serum-neutralizing antibody for the duration of the study. Detailed results of the SN testing of the tortoises in the study are reported in Table 1.
Enzyme-linked immunosorbent assay
The ELISA results paralleled those of SN. Detailed results of the ELISA testing are reported in Fig. 1A, B. The mean A 405 ± SD of the negative control was 0.159 ± 0.02 and 0.145 ± 0.02 when tested against HV 4295/7R/95 and HV 1976, respectively. The mean A 405 readings ± SD for the positive control were 1.42 ± 0.05 and 1.27 ± 0.27 when tested against HV 4295/7R/95 and HV 1976, respectively. Specificity and sensitivity parameters of the ELISA have been reported elsewhere. 16
All the tortoises were seropositive for exposure to M. agassizii when tested by ELISA at the beginning and at the end of the transmission study.
Gross pathology
Oral lesions were seen only in one tortoise (No. 4) at necropsy. This tortoise had two 2-mm-diameter symmetric yellowish diphtheritic plaques in the oral cavity on the lower jaw, adjacent to the tongue. No other gross abnormalities were detected in the oral cavity of any of the other tortoises in the study. No ocular or periocular lesions were observed.
Histopathology and immunohistochemistry
The oral mucosa of tortoise No. 5 (Fig. 3A) was normal in appearance. In contrast, hyperplasia and hyperkeratosis were observed in the mucosa overlying the salivary glands of tortoise No. 4. Bacterial colonies were present on the epithelial surface and extended into the submucosa. Large numbers of degranulated heterophils were seen (Fig. 3B). Microthrombi that completely or partially occluded the lumina of affected vessels were a common finding along with the presence of severe vasculitis. Diffuse necrosis and edema were observed. A focal lymphoid infiltrate was present in the epithelium of the tongue of tortoise No. 4. Rare heterophils infiltrated the epithelium of the glottis of tortoise No. 3. No lesions were observed in the oral mucosa of tortoise Nos. 1 and 2. The hepatic cells from tortoise Nos. 5 (control), 1, 2 (Fig. 4A), and 4 were normal in appearance, but, in contrast, they exhibited nuclear degeneration changes in tortoise No. 3 (Fig. 4B). The largest nuclei were pale staining and appeared empty, whereas those smaller in size stained with a lightly basophilic stain (HE) (Fig. 4B). Several melanomacrophages were also present (more abundant than those observed in the other tortoises in the study). Some of these were in areas of focal necrosis.
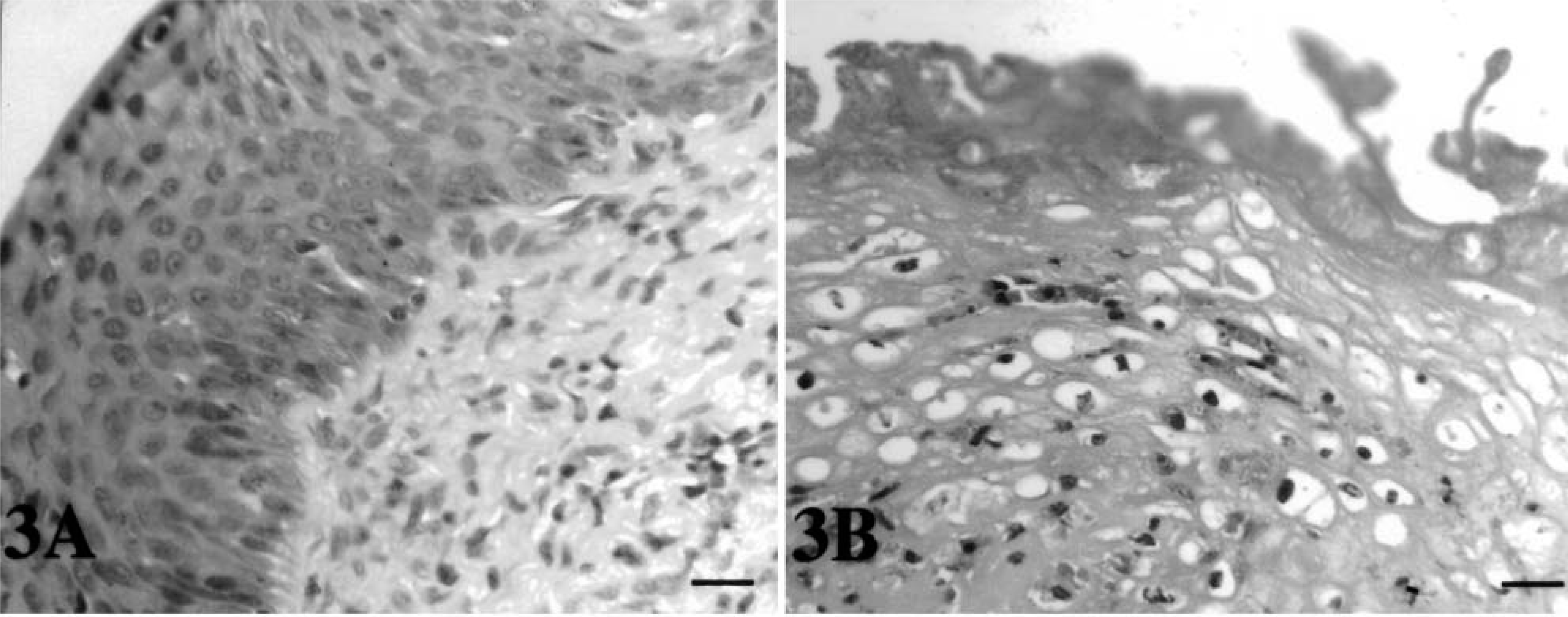
Histologic findings.
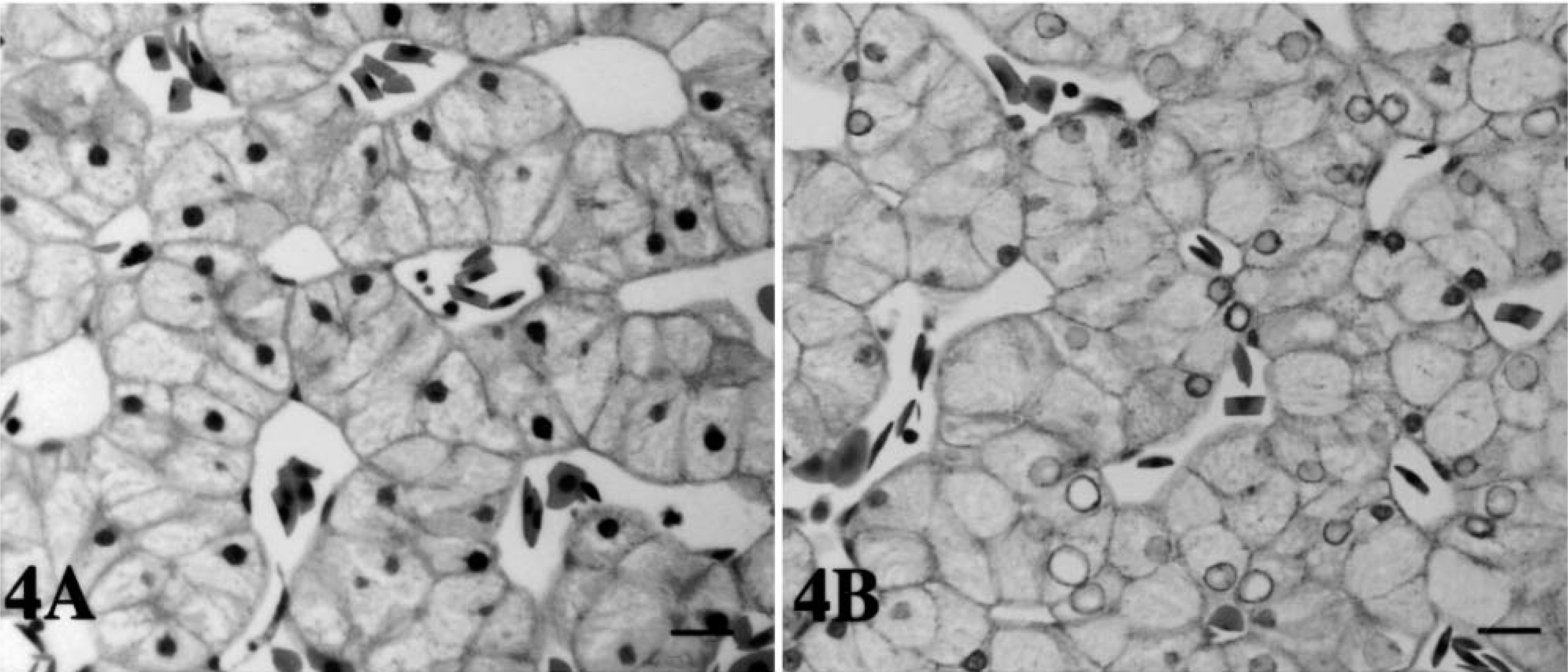
Histologic findings.
The red pulp of the spleen of tortoise No. 5 was normal in appearance, and the lymphoid cells were homogeneously distributed (Fig. 5A). In contrast, the red pulp of the spleen of tortoise Nos. 1–3 was depleted of lymphoid cells, which appeared to be mainly clustered in the areas surrounding the central arteries (Fig. 5B). Tortoise No. 4 exhibited features closer to those of tortoise No. 5. The spaces left free by the lymphoid cells were occupied by red blood cells in tortoise Nos. 1–3 (Fig. 5A, B). Ballooning degeneration of the epithelium of the bladder, stomach, and small and large intestines was a common finding in all five tortoises in the study. Mild subepithelial and sub-mucosal lymphoid infiltrates, with rare heterophils, were observed in tortoise Nos. 1–3. Intranuclear inclusions were not observed in any of the tissues examined.
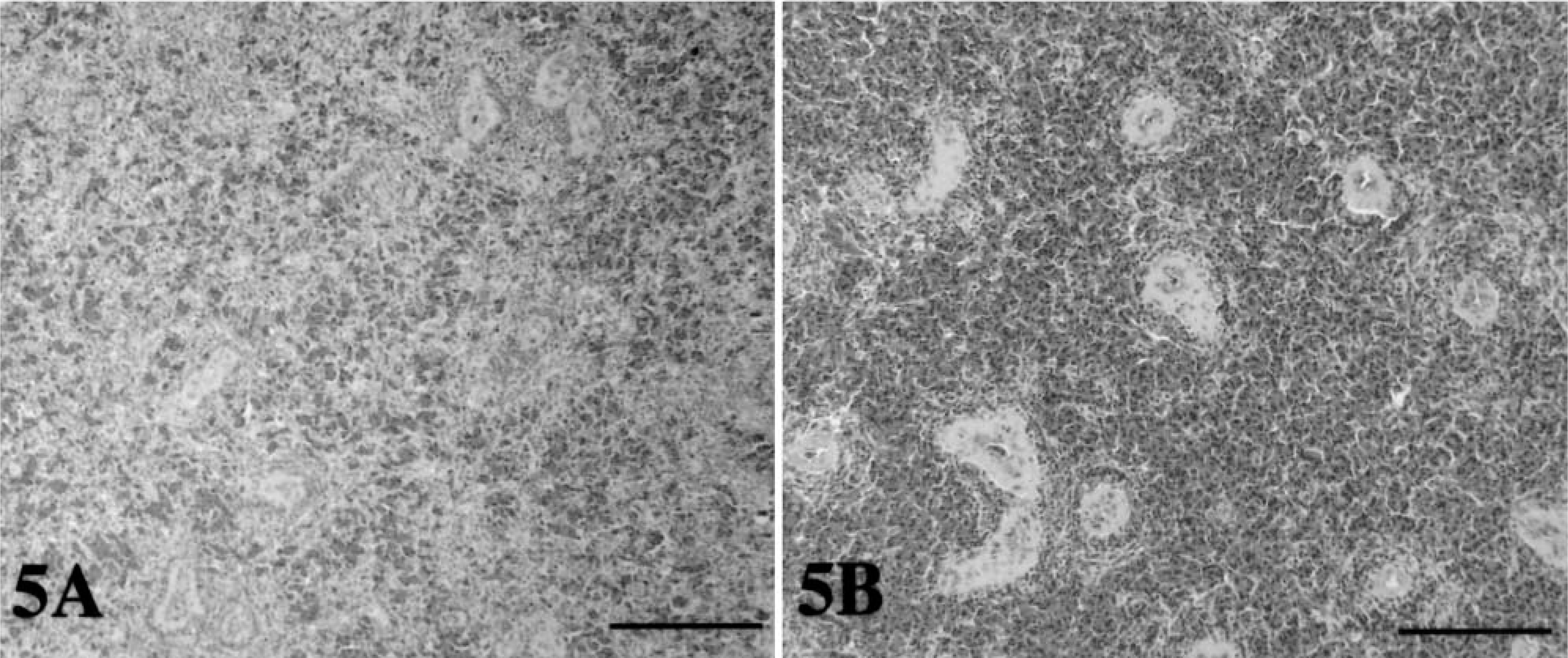
Histologic findings.
A total of five immunoperoxidase studies (one for each tortoise) were carried out. None of the tissue sections (n = 21 for each tortoise) stained positive for detectable herpesviral antigen.
Virus isolation
Despite several attempts neither of the two herpes-virus isolates used in this study was recovered from nasal washes, pharyngeal swabs, or multiple tissues collected from any of the infected tortoises in the study.
Virus cloning
Of the 31 HindIII clones obtained from the shotgun cloning of genomic DNA from the herpesvirus isolate HV 1976, clone No. 11 contained the partial coding sequence of the RR gene, which is a very highly conserved gene among herpesviruses. 28 It was shown that this 1.413-kb HindIII clone contained the 3′ portion (746 nucleotides) of the large subunit of the RR gene and the 5′ portion (645 nucleotides) of the small subunit of the same gene. A region of the large RR subunit, which was present in clone No. 11, was selected for the development of the PCR reaction. The homologous region of the genome from the HV 4295/7R/95 isolate was also sequenced, and it demonstrated 100% identity.
Molecular analyses
PCR titration under optimal and field conditions
The PCR assay using primers specific for a region of the RR gene could detect as few as 10 (0.5 fg) or 100 copies of the target plasmid when used as purified DNA template or spiked into total cellular DNA (field conditions), respectively.
PCR analysis of pharyngeal swabs and nasal washes for the presence of viral DNA
All nasal washes and the pharyngeal swabs collected during the secondary challenge phase of the study were PCR negative for herpesviral DNA.
Semiquantitative PCR analysis of tissue samples for the presence of viral DNA
A total of 162 tissue samples were collected from the five tortoises in the study. None of the tissues (n = 31) collected from the control tortoise (No. 5) were positive by PCR for herpesviral DNA. Of the 131 tissue samples collected from the four infected tortoises in the experimental study and tested by PCR for herpesviral DNA, 40 samples (30.5%) were positive. Twenty of the 40 positive samples (50%) were obtained from nervous tissue (CNS and brachial plexus), 13 (32.5%) from the respiratory and digestive tracts, 5 (12.5%) from the urogenital tract, and 2 (5%) from miscellaneous organs (spleen, heart). All the four experimentally infected tortoises were PCR positive for tortoise herpesvirus DNA sequences in two or more tissues. The detailed results are reported in Table 2.
Quantification of the relative amount of viral DNA in the positive tissue samples
For 18 of 25 samples (Table 2) the VU content determined by TDT were in agreement with that determined by DDT. For seven samples the two techniques exhibited a 10-fold difference in the result.
RT-PCR to detect viral transcripts in the various tissue samples
The RT-PCR reaction could detect as little as 2 pg (2 pg = 106 copies of the target RNA transcript) of pure RNA transcripts and as little as 20 pg when it was spiked into total cellular RNA (field conditions).
RT-PCR on tissue samples
All the samples were negative for detectable viral mRNA target at the time of sampling (4 weeks after secondary challenge).
Internal mRNA control
Of all the cDNA samples obtained from the RT step of the RT-PCR reactions (n = 31 samples in total, comprising single total RNA and pooled RNA samples), 26 (83%) were positive for the presence of the tortoise β-actin, whereas five samples remained negative despite additional pooling. The sequencing of the tortoise RT-PCR product indicated that it was slightly shorter (214 nucleotides, GenBank Accession AY344078) than the homologous sequence from chicken (238 nucleotides) but was 89% homologous with the corresponding chicken sequence (GCG program, University of Wisconsin, Madison, WI).
Discussion
Each of the challenged tortoises in the study was successfully infected with either the American or the European herpesvirus isolate. In contrast with data reported in the literature indicating mortality due to natural infection in naive collections of tortoises, 11 no animals died spontaneously after either the primary experimental infection or the secondary challenge. The higher doses of infective virus and the “constant” challenge that captive tortoises could encounter, especially when large numbers of tortoises are housed or shipped together, might account for this observation. Protection by SN antibodies, the possible reduced virulence of the “laboratory isolates” used in this study, the overall health of the experimental tortoises at the beginning of the study, and their immune “makeup” 26 might have also played an important role.
In this study we were able to reproduce the most common clinical signs reported in tortoises infected with herpesvirus. Recurrent unilateral or bilateral conjunctivitis was observed in each of the tortoises during the course of the study. Characteristic diphtheritic oral plaques were observed in tortoises infected either IN or IM with both the European and the American isolate, suggesting that both routes and isolates resulted in a productive infection. Herpesviral DNA was detected by PCR in similar organs in tortoises inoculated by either IM or IN. Therefore, the route of inoculation did not influence the distribution of viruses in tissues of infected tortoises.
At the time the tissue samples from the oral lesions were analyzed by PCR for herpesviral DNA (4 weeks after secondary challenge), no positive signal was detected. The extensive oral lesions might have been dependent on the indirect action of the virus with the release of cytotoxic compounds by the inflammatory cells rather than the direct effects of the virus itself. 27 The severe necrosis present at the site of the oral plaques might have been a consequence of local vascular compromise. Several intravascular thrombi that might have been directly or indirectly induced by the herpesviral activity 15 were identified.
In this study, although detectable amounts of circulating SN antibodies were identified, this was not sufficient to prevent the development of recurrent clinical signs. However, it might have been sufficient to prevent death of the tortoises and allow recovery. The cell-mediated immune system might also play an important role in tortoises for protection against stomatitis-rhinitis of tortoises, as has been reported for herpesvirus infections in other animals and in man. 26
As in reports of certain herpesviruses in other animals and in man, 24,26,27 we were able to demonstrate a prevalent neurotropism of the tortoise herpesviruses. Sequences of viral DNA were detected in the cranial nerves of three of the four infected tortoises in our study. The detection of herpesviral DNA in the CNS 4 weeks after the secondary challenge is consistent with viral latency. The absence of detectable viral mRNA by RT-PCR and the failure to concurrently detect the presence of herpesviral structural protein by immunoperoxidase further support this interpretation. The positive amplification of the mRNA internal control (chicken β-actin) suggested that sufficient RNA was recovered from the majority of the tortoise tissue samples to allow detection of transcripts by RT-PCR analysis. This and the sensitivity of the viral RR transcript primers suggested that failure to detect viral RNA in the tissue sections was due to either the absence of productive RNA transcripts or the presence of extremely low amounts in the tissue samples at the time points assayed.
We were unable to recover herpesvirus from the inoculated tortoises. The delay in collecting the nasal washes and pharyngeal swabs from the infected tortoises and the subsequent development of circulating neutralizing antibodies in three of the four tortoises may explain the failure to recover herpesvirus. Although one of the four experimental tortoises (tortoise No. 1) did not develop SN or ELISA antibodies, herpesvirus sequences were detected in two of its organs (Table 2) by PCR. Interestingly, this seronegative tortoise was the only one whose CNS was PCR negative for herpes DNA sequences. Negative PCR findings for all nasal and pharyngeal samples collected PC were consistent with the unsuccessful recovery of the virus from the same samples.
In this study we also assessed the exposure of the tortoises to another known pathogen of chelonians, M. agassizii. 1,2,13,21 Clinical mycoplasmosis of tortoises is generally characterized by palpebral edema, conjunctivitis, and rhinitis. However, oral lesions have not been described in tortoise mycoplasmosis. Tortoises at the beginning of the study were found by ELISA to be serpositive for exposure to M. agassizii. However, rhinitis was not seen before experimental infection or after experimental infection. The oral lesions seen in the experimentally infected tortoises in this study were different from the lesions seen in mycoplasma transmission studies. 1,2 Although examples of mycoplasma-herpesvirus productive interaction in the same host are not reported in the literature, it is known that M. hyopneumoniae can potentiate the severity of the pneumonia induced by porcine reproductive and respiratory syndrome virus, an arterivirus of swine. 23 Despite some preliminary data demonstrating the possibility of the coexposure of Mediterranean tortoises to herpes-virus and mycoplasma (Karina Mathes, personal communication), no evidence of a productive interaction between the two microorganisms is currently available. The possible interaction between both pathogens on the host needs further study.
In summary, the pathogenesis of a new tortoise herpesvirus infection was experimentally investigated. The virus demonstrated neurotropic behavior, with a less specific infection of other systems. SN antibodies did not appear to protect the tortoises from reinfection or from the recurrence of clinical signs. Tortoise herpesvirus appears to remain silent or at an undetectable level of activity in the tissues where it localizes. The unsuccessful reisolation of the two herpesviruses permits only partial fulfillment of Koch's postulates for the association of tortoise herpesvirus and stomatitis-rhinitis of tortoises. Nevertheless, we have presented strong evidence suggesting the role of this tortoise herpesvirus as one of the primary causative agents of stomatitis-rhinitis in Greek tortoises.
Footnotes
Acknowledgements
This research has been partially supported by two grants (DACA 05-99-P-0661 and 1434-HQ-97-RU-01544) from the US Department of Defense, Corps of Engineers. We would like to thank Dr. Rachel Marschang for providing the herpesvirus isolates HV 4295/7R/95 and HV 1976, Mrs. Diane Duke for the development of the monoclonal antibody HL1546, and Dr. Paola Bonfanti for her field work.
