Abstract
Bovine parainfluenza virus-3 (BPIV-3) is a recognized respiratory pathogen of cattle, and it has also been identified in aborted fetuses. However, little is known of this agent as a reproductive pathogen and detailed descriptions of fetal pathology on natural cases are lacking in the scientific literature. This article describes and illustrates lesions in a fetus spontaneously aborted by a first-calving Holstein heifer, naturally infected with BPIV-3 genotype A, broadening the current knowledge on fetal pathology by this virus. Fetal autopsy revealed diffusely reddened, rubbery and unexpanded lungs. Histologically, there was necrotizing bronchiolitis/alveolitis with intraluminal fibrin exudate and syncytial cells in the bronchiolar/alveolar spaces, and non-suppurative peribronchiolitis and perivascular interstitial pneumonia. In the small intestine there was multifocal necrotizing cryptitis and occasional necrotic syncytial enterocytes. Intralesional and extralesional BPIV-3 antigen was detected by immunohistochemistry in the lung and small intestine, and BPIV-3a was identified in fetal tissues by RT-PCR and sequencing.
Bovine parainfluenza virus-3 (BPIV-3, Respirovirus, Paramyxoviridae) was first isolated from the nasal discharge of cattle with respiratory disease. It was named after its human counterpart, as it is antigenically and genetically related to human parainfluenza virus-3 (HPIV-3). 8 It has a single-stranded, negative-sense RNA genome, and a spherical to pleomorphic virion of 150-200 nm diameter, comprising the nucleocapsid and lipid envelope. Three distinct genotypes, namely BPIV-3a through c, have been identified. 9,13
BPIV-3 causes respiratory disease, and far less commonly, abortion. The virus spreads aerogenously, and even though infection is widespread in young dairy and beef cattle, spontaneous disease caused solely by BPIV-3 is rare. 3 However, the virus compromises the mucociliary apparatus and causes bronchitis, bronchiolitis and alveolitis, with necrosis of the respiratory epithelium, and changes in the local and systemic inflammatory response that contribute to the establishment of bacterial infections, typical of enzootic pneumonia and bovine respiratory disease complex. 3,7
Information on BPIV-3 as an abortifacient is scant, 19 and descriptions of fetal lesions in spontaneously aborted fetuses in the scientific literature are succinct and limited to pulmonary lesions in one case. 1 Here we provide a pathological description of pulmonary and enteric lesions in an aborted fetus naturally infected with BPIV-3a, broadening the current knowledge on fetal pathology caused by this agent.
The case occurred in April 2017 in a herd of ∼315 Holstein cattle at a Brucella abortus-free dairy farm in Uruguay. The aborting animal was a 25.5-month-old, first-calving heifer of unknown vaccination history that had been purchased pregnant, and introduced into the herd 48 days before. The heifer aborted a well-preserved female fetus, 83 cm in crown-to-rump length consistent with 240-270 days gestation. 14
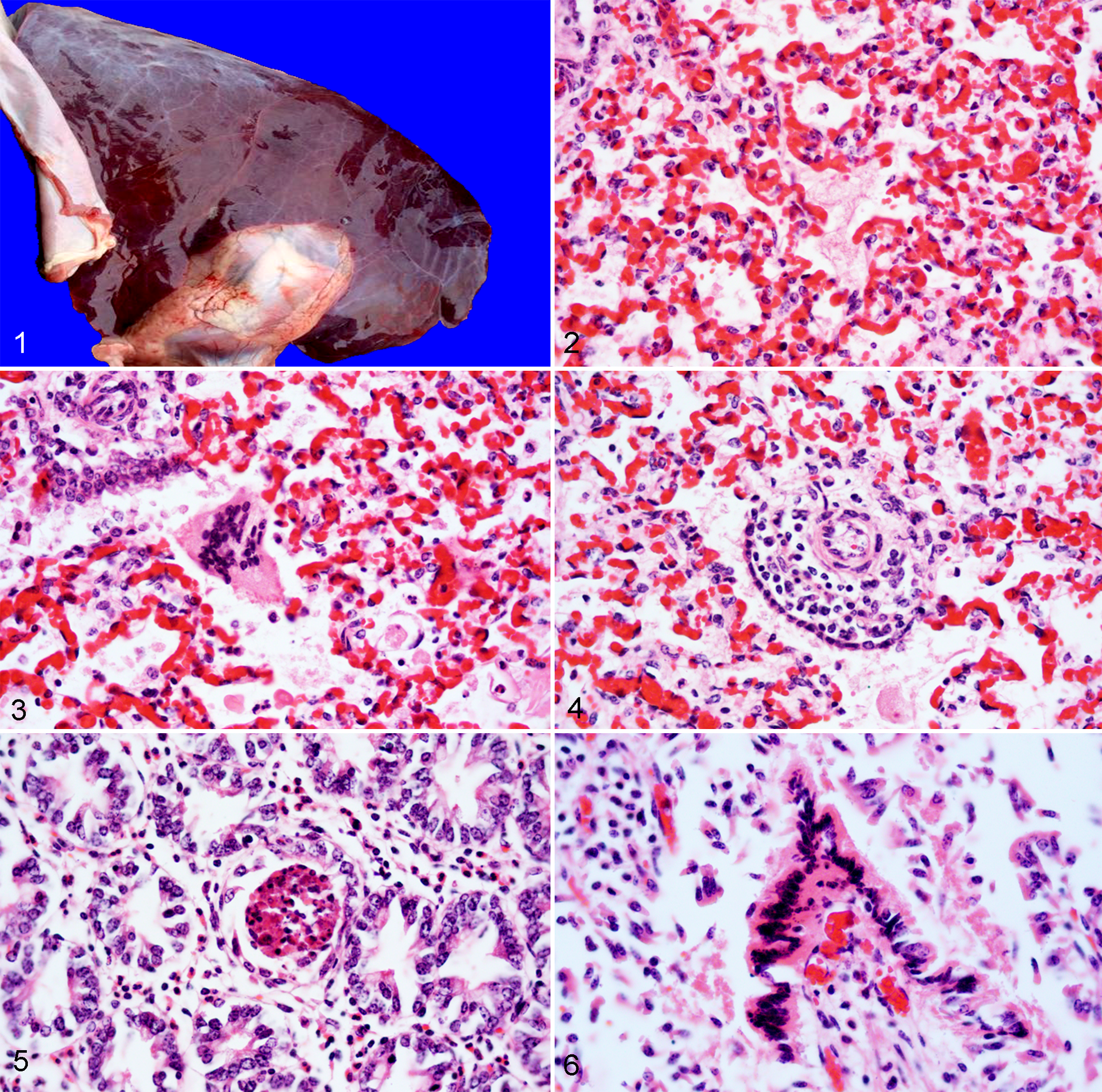
The fetal autopsy revealed diffusely reddened, rubbery and unexpanded lungs (Fig. 1). No other gross abnormalities were observed. The placenta was not available for examination.
Tissues including lung, kidney, spleen, heart, adrenal gland, liver, skeletal muscle, trachea, tongue, brain, thymus, lymph node, abomasum, forestomachs, small intestine, colon, and eyelid were immersion-fixed in 10% neutral buffered formalin. Sections from all lung lobes sank when immersed in formalin. Tissues were embedded in paraffin, sectioned at 4-5 µm, and stained with hematoxylin and eosin for histologic examination.
Sections of lung, trachea, small intestine, abomasum, liver, kidney, heart, thymus, lymph node, skeletal muscle and brain were processed by immunohistochemistry for the detection of BPIV-3 antigen. Slides were immersed in citrate buffer and heat-induced antigen retrieval was performed in a decloaking chamber at 121°C for 10 min, after quenching the endogenous peroxidase with 3% hydrogen peroxide for 10 min. Goat polyclonal antibody against BPIV-3 (VMRD, #210-70-PI3, Pullman, WA) was applied as a primary antibody at a 1:500 dilution for 15 min, and anti-goat horseradish peroxidase (HRP)-labeled polymer (Biocare, #GHP516, Pacheco, CA) was used as the detection system (15 min incubation), with 3-amino-9-ethylcarbazole as the chromogen substrate solution (ThermoScientific, #TA-125-SA, Fremont, CA). A formalin-fixed paraffin-embedded section of archived lung from a naturally-infected bovid that had tested positive for BPIV-3 by PCR and immunohistochemistry was used as a positive control. As negative controls, serial sections of all tissues were processed in parallel as described above but the primary antibody was replaced by normal goat serum (Gibco™ Normal Goat Serum, #10098792) at the same dilution.
Frozen lung (–20°C) was processed for BPIV-3 detection by reverse transcriptase (RT)-PCR. 9 Nucleic acid extraction was accomplished by TRIzol™ reagent (ThermoFisher, USA). RNA quality and integrity was assessed by NanoDrop™ (ThermoFisher, USA) and RT-PCR by amplifying a region within the beta actin gene. 17 BPIV-3 RT-PCR product was cloned into pJET1.2 Vector (ThermoFisher, USA) and 5 positive clones were sequenced. The plasmids containing the amplified region were purified and sequenced directly in both directions (Macrogen, Korea). Sequence analysis and phylogenetic reconstruction was performed with MEGA software. Supplementary Table 1 summarizes other ancillary test results.
Microscopically in the lungs, there was multifocal infiltration of neutrophils and macrophages in the alveolar spaces (alveolitis) with scattered intraluminal fibrin exudate and pyknotic or karyorrhectic cellular debris (necrosis), and a moderate number of syncytial cells with up to 40 nuclei within the alveolar and bronchiolar spaces (Figs. 2-3). The interstitium surrounding the epithelium of the terminal bronchioles and small-caliber arterioles and venules was multifocally expanded by moderate numbers of lymphocytes, macrophages and fewer plasma cells occasionally forming layers of up to 5 cells thick (peribronchiolitis with perivascular interstitial pneumonia) (Fig. 4). In the small intestine, there was scattered crypt dilation with epithelial necrotic debris filling the lumen (multifocal necrotizing cryptitis) (Fig. 5) with few neutrophils occasionally infiltrating the lamina propria. In addition, the superficial enterocytes occasionally formed syncytial cells that sloughed into the intestinal lumen, and often had hypereosinophilic cytoplasm and hyperchromatic/pyknotic nuclei (necrosis) (Fig. 6). No lesions were found in the other tissues. No intracytoplasmic inclusions were identified. Immunohistochemistry revealed moderate multifocal widespread granular intracytoplasmic BPIV-3 antigen in bronchiolar epithelial cells, pneumocytes, syncytial cells and alveolar macrophages, and in the alveolar and bronchiolar proteinic exudate (Fig. 7), as well as the necrotic crypts and syncytial enterocytes in the intestine. Thus, the immunoreactivity colocalized with the lesions. BPIV-3 antigen was also present in non-lesioned areas of the lung (pneumocytes), small intestine (crypt enterocytes and leukocytes/fibroblasts in the lamina propria) (Fig. 8), and superficial and glandular epithelium of the trachea. No significant intracytoplasmic immunoreactivity was observed in the other tissues.
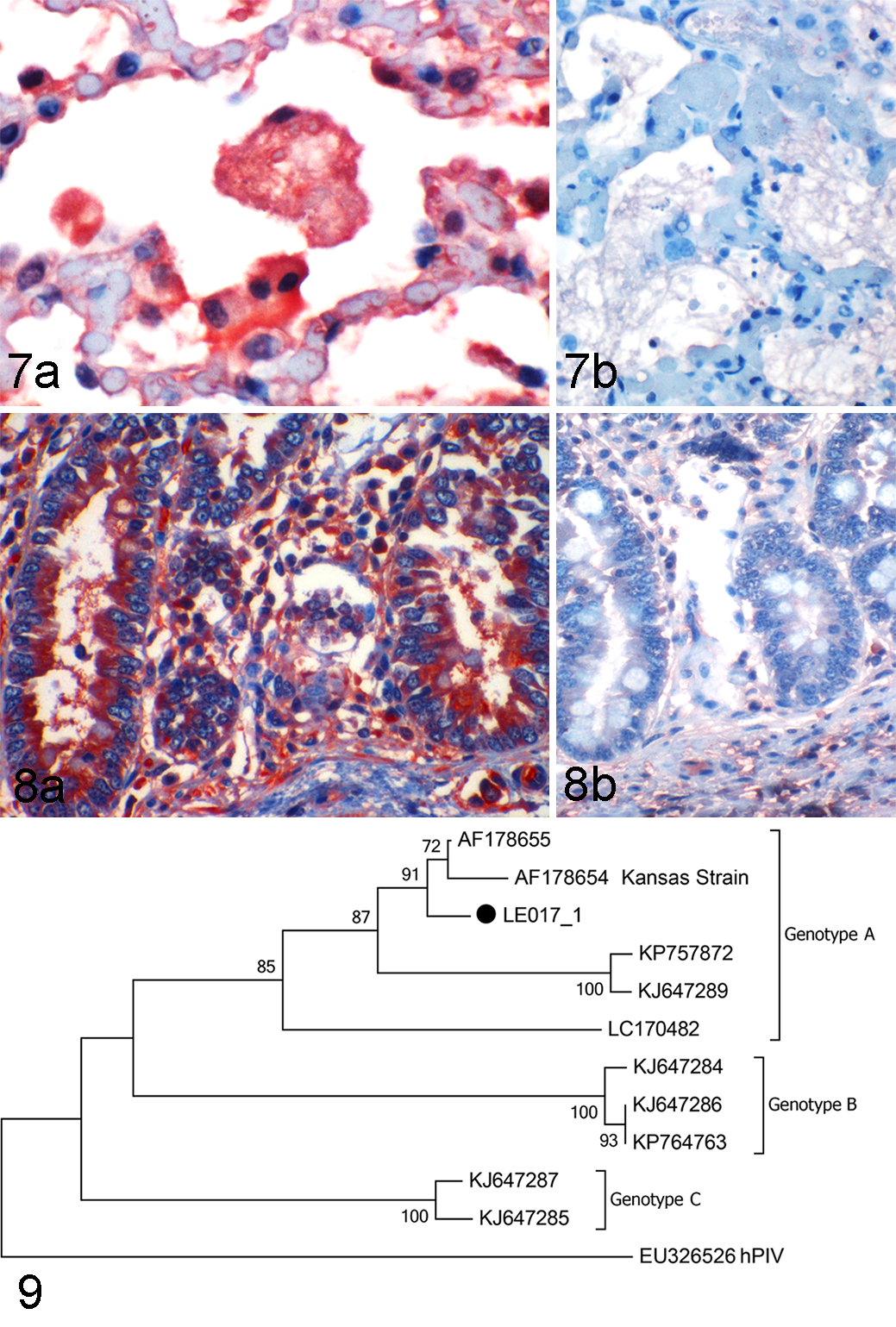
BPIV-3 genome was detected by RT-PCR. Sequences from the 5 clones were identical. Sequence analysis of the 274-bp matrix gene region confirmed the BPIV-3 identity. The identified strain, named LE017_1, was classified within genotype A by phylogenetic reconstruction (Fig. 9). The sequence was deposited in GenBank (#MG976794).
The diagnosis of BPIV-3a abortion in this case was based on pathological findings in the fetus, coupled with intralesional detection of the agent by immunohistochemistry, and identification of the viral genome in the lung. 3 Other infectious abortifacients were ruled out by specific testing (Supplementary Table 1).
The etiologic diagnosis of bovine abortion is complex. As it may be caused by many bacterial, viral, parasitic, toxic, metabolic, or genetic factors, identification of the specific cause can be challenging. Even at laboratories with experienced pathologists that routinely apply a broad panel of ancillary tests, most to detect infectious agents, the cause remains undetermined in a large proportion of cases. A subset of these have inflammatory lesions suggesting an infection, although in many cases, a specific pathogen is not identified. 1,2,4,10 Viral causes of bovine abortion are generally represented in low frequencies. An early study aiming at isolating viruses from aborted fetuses in Ohio identified the first BPIV-3 fetal isolate. 15 No results of histopathologic examination were provided.
In a 10-year study that examined 8,962 abortions and stillbirths submitted to a diagnostic laboratory in South Dakota, a viral etiology was determined in 948 (10.57%) cases. BPIV-3, detected in only one case, was considered a miscellaneous infection, and fetal lesions were not described. 10,11
In a case series from California that investigated 468 bovine abortion cases in 1985-1989, viral causes of abortion were diagnosed in 26 (5.6%) cases, including one BPIV-3 case. 1 In a second series that included 709 cases of abortion submitted to the same laboratory in 2007-2012, no BPIV-3 cases were recorded. 4 Altogether, these works indicate that BPIV-3 is infrequently associated with bovine abortion in the USA.
In case series from South America, including Uruguay, 7 Brazil, 5 and Argentina, 2 that collectively evaluated several hundred laboratory submissions, BPIV-3 was not reported as a cause of bovine abortion. However, BPIV-3a was isolated from an aborted bovine fetus in Brazil, 6 although no pathologic information was provided.
The pathogenesis of BPIV-3 as a respiratory pathogen in postnatal life is relatively well understood; 3,8 however, the mechanisms associated with abortion are largely unknown. We can infer that the virus is not transmitted aerogenously in fetal life. Experimentally, in utero inoculation of bovine fetuses with a BPIV-3 strain isolated from an aborted fetus produced both fetal lesions and immune response. 19 Neutralizing BPIV-3 antibody in pregnant heifers parenterally inoculated with BPIV-3 appears to prevent fetal infection. 19 However, viremia by BPIV-3 has been demonstrated, 20 and hematogenous transplacental transmission seems plausible in naïve dams. It has been hypothesized that in areas where most of the adult cattle have antibodies against BPIV-3, this agent is not likely to be a major abortifacient; seronegative pregnant females might instead be susceptible to abortion. 19
Necrotizing bronchiolitis, interstitial pneumonia and peribronchiolar lymphoid stimulation (“lymphoreticular hyperplasia/proliferation” and “peribronchiolar lymphoid nodules”) were demonstrated in surgically removed fetuses inoculated in utero with BPIV-3 at mid-gestation. 18,19 Under these experimental conditions, some fetuses died and were aborted, while others survived the infection and were born weak, unable to rise, underweight, or died in the perinatal period. 18,19 Descriptions of fetal lesions in spontaneous abortion cases are scarce and restricted to lymphocytic bronchointerstitial pneumonia in one case. 1 In the case presented here, the spectrum of pulmonary lesions included not only bronchointerstitial inflammation, but also necrosis, inflammatory and fibrinous exudate and epithelial syncytial cells in the bronchiolar/alveolar spaces. Although bronchial/alveolar syncytial cells are occasionally found in pneumonia induced by postnatal BPIV-3 infection, 3 to our knowledge this lesion has not been described in bovine fetuses. Interestingly, histological examination of the lung of a human fetus infected with HPIV-3 revealed multifocal pneumonia with necrosis, mononuclear and polymorphonuclear infiltrates, and “giant cells” 16 that might have represented epithelial syncytia. In addition, the fetus in our case had multifocal necrotizing cryptitis and syncytial enterocytes in the small intestine, which also represent previously undescribed lesions for this virus. In a diagnostic setting, identification of these lesions should encourage the pathologist to pursue specific BPIV-3 testing.
Current knowledge on BPIV-3 molecular epidemiology is somewhat limited. Sequencing analysis of BPIV-3 strains from the USA revealed that, in addition to the BPIV-3a that had been previously identified in the country, there were two additional genotypes circulating that, until then, had been described only in Australia (BPIV-3b) and Asia (BPIV-3c). 13 American genotypes B-C showed some divergence from the Australian and Asian strains, and cross-neutralization studies indicated that there were antigenic differences between these genotypes and the BPIV-3a included in commercial vaccines. 13 Information from South America is scant. In Argentina, BPIV-3a and BPIV-3c have been reported from cattle, and BPIV-3b was identified in water buffalo, 12 while BPIV-3a was identified in cattle in Brazil. 6 No information is available from Uruguay, since this case report represents the first scientific documentation of BPIV-3 in this country.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818798117 - Fetal Pathology in an Aborted Holstein Fetus Infected With Bovine Parainfluenza Virus-3 Genotype A
Supplemental Material, DS1_VET_10.1177_0300985818798117 for Fetal Pathology in an Aborted Holstein Fetus Infected With Bovine Parainfluenza Virus-3 Genotype A by Melissa Macías-Rioseco, Santiago Mirazo, Francisco A. Uzal, Martín Fraga, Caroline Silveira, Leticia Maya, Franklin Riet-Correa, Juan Arbiza, Rodney Colina, Mark L. Anderson, and Federico Giannitti in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank Yisell Perdomo, Cecilia Monesiglio, and Bruno López from INIA and Karen Sverlow from CAHFS for technical assistance, and graduate students from INIA for support. We also thank Drs Leticia Zarantonelli from the “Institut Pasteur de Montevideo,” Alejandra Suanes and Néstor D’Anatro (in memoriam) from the “División de Laboratorios Veterinarios” (DILAVE), Uruguay, for providing some of the laboratory test results shown as supplementary information.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by grant FSSA_X_2014_1_105696 from the Uruguayan “Agencia Nacional de Investigación e Innovación” (ANII).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
