Abstract
Two otherwise healthy adult cats were presented with progressive cerebellar signs of different severity. Owners requested euthanasia. Necropsy disclosed whole cerebellum and pontine atrophy, with a severity paralleling the neurologic dysfunction. We used cell type-specific immunolabelings to characterize the lesions. The severity of the cerebellar cortex atrophy followed a general gradient from the midvermis toward the hemispheres and a local gradient from the depth of the folia toward their tip. Along these gradients, Purkinje cells were the first to disappear, followed by basket, Golgi, and stellate cells, and eventually by granule cells. Bergmann glia cells and unipolar brush cells were preserved. Pontine nuclei and the olivary complex were also severely depopulated. Neurons in the cerebellar nuclei, vestibular nuclei, and other cerebellar system-associated structures were preserved, as well as substantia nigra. Olivopontocerebellar atrophy (OPCA) in a domestic animal species was rarely reported. Some features allow tentative linking to either familial or sporadic OPCA of humans. However, the ordered disappearance of all cortical neuronal types has never been described before. Either this entity is cat specific or it might pinpoint the need for increased knowledge about differential gene expression depending on genetic background, i.e., among different species. It also would open prospects about gene product interactions within neurons.
Congenital ataxia in kittens has been recognized from 1885. 5,24 All these animals presented with an early and stable cerebellar ataxia. In 1966, congenital ataxia in cats was related to fetal or neonatal infection by a virus, 19 later identified as the feline panleukopenia virus. 8,17 A single, infectious entity in the cat contrasts with the dog, where numerous neurodegenerative diseases involving the cerebellum were reported. 35
More recently, a heritable, early-onset cortical cerebellar degeneration (CCD) has been described in Japan. 1,16 Two separate descriptions of single adult-onset progressive CCD cases have also been published, with one case being associated with retinal degeneration. 2,33 A single case of olivopontocerebellar atrophy (OPCA) in an adult cat was recorded in 1946 32 as well as a familial occurrence in 1934. 3
This article reports two OPCA cases occurring in possibly related feral cats. Although little clinical history was available, progressive symptoms began during adulthood. Pathologic features in these two cases differed from all previously described entities in either man or animal.
Materials and Methods
Case history
The two cats were feral animals monitored and fed by cat lovers.
Cat No. 1 was a fully adult castrated male, which became reluctant to move and exhibited subtle disequilibrium. The cat performed poorer than expected in visually guided tasks like jumping on furniture. The head and neck movements were sometimes jerky. Neurologic tests were, however, judged normal. Blood tests for feline retrovirus (FeLV), feline lentivirus (FIV), Toxoplasma gondii (TG), and feline corona virus (FIP) were performed. The cat had a high antibody titer (1/2,800) for FIP. The cat was euthanatized.
Cat No. 2 was a young adult female with a severe gait disturbance. She was caught 1 month previously for ovar-iohysterectomy, at which time she displayed some slight gait abnormality. On presentation, the cat was alert, and responsive. She displayed a wide-based stance and exhibited variable oscillations from side to side and back to fore, exaggerated jerky limb flexion, and excessive bobbing head and neck movements characterized her gait. Visual tracking of moving targets was delayed, and attempts to catch them were seldom successful. Hopping and placing reaction times were normal, although hypermetric. Cranial nerve and segmental reflexes were normal. Dyssynergia in balance and movement suggests diffuse cerebellar involvement. The cat was seronegative for FeLV, FIV, FIP, and TG. The cat was euthanatized.
Histopathology
Brain and cranial cervical spinal cord were immediately removed from both animals after death and fixed in 4% paraformaldehyde, in 0.1-M phosphate buffer, pH 7.4 for 3 days at 4 C, followed by a 24-hour rinse in 0.1-M phosphate buffer, and embedded in paraffin. Specimens were cut semiserially at 5 µm;, every 20th section was stained with the Klüver–Barrera technique, 20 and some of them were stained with a periodic acid–Schiff (PAS) technique.
Immunohistochemistry
The peroxidase–anti-peroxidase (PAP) technique was used. The deparaffinized sections were treated for 30 minutes with 1% hydrogen peroxide to block endogenous peroxidase activity, boiled in citrate buffer, pH 6 for 15 minutes in a microwave oven to enhance immunoreactivity, and rinsed two times with phosphate-buffered saline (PBS) 0.01 M for 5 minutes. Neutralizing of unspecific protein linkages was carried out with 20% normal sheep serum. The sections were incubated in primary antibody at 4 C for 36 hours. They were then rinsed two times with PBS 0.01 M for 5 minutes, incubated for 15 minutes in anti-rabbit or anti-mouse IgG (Nordic Immunological Lab, Maidenhead-Berks, UK), rinsed two times with PBS 0.01 M for 5 minutes, incubated in PAP complex (Dako, Glostrup, Denmark), and rinsed again with PBS. Peroxidase activity was visualized by reaction with diaminobenzidine. Between each step the sections were rinsed two times with PBS 0.01 M for 5 minutes. Lectin Ricinus communis agglutinin IA (RCA I) was visualized using the ABC technique (Vector, Burlingame, CA).
Control sections were selected from paraffin-embedded encephalon samples collected in the same conditions from cats euthanatized for untreatable conditions unrelated to the nervous system as well as for unrelated encephalitis.
Nine primary antisera were used: polyclonal rabbit serum against calbindin (courtesy of Dr E. Lawson, 1/5,000), calretinin (SWannt, Bellinzona, Switzerland 1/1,000), microtubule-associated protein (MAP2) (courtesy of Dr JP Brion, 1/1,500), tau (courtesy of Dr JP Brion, 1/1,000), glial fibrillary acidic protein (GFAP) (Dako, 1/10,000), S100 (Dako, 1/5,000), monoclonal mouse antibodies against phosphorylated neurofilaments (clone RT97, Roche, Basel, Switzerland 1/200), synaptophysin (Sigma, Saint Louis, MI, 1/200), and ubiquitin (courtesy of Dr JP Brion, 1/5,000). RCA I (Vector, 1/200) was also used as a marker for microglial cells.
Results
Macroscopic findings
As compared with a control cat, the rostrocaudal dimension of the cerebellum was 70% in cat No. 1 and 25% in cat No. 2 (Fig. 1). Hemispheres and vermis were similarly affected. Folia were moderately (cat No. 1, Fig. 1b) or severely (cat No. 2, Fig. 1c) atrophic. The pons was underdeveloped in cat No. 1 and extremely flat in cat No. 2. The remainder of the brain and the spinal cord were normal in both cats.
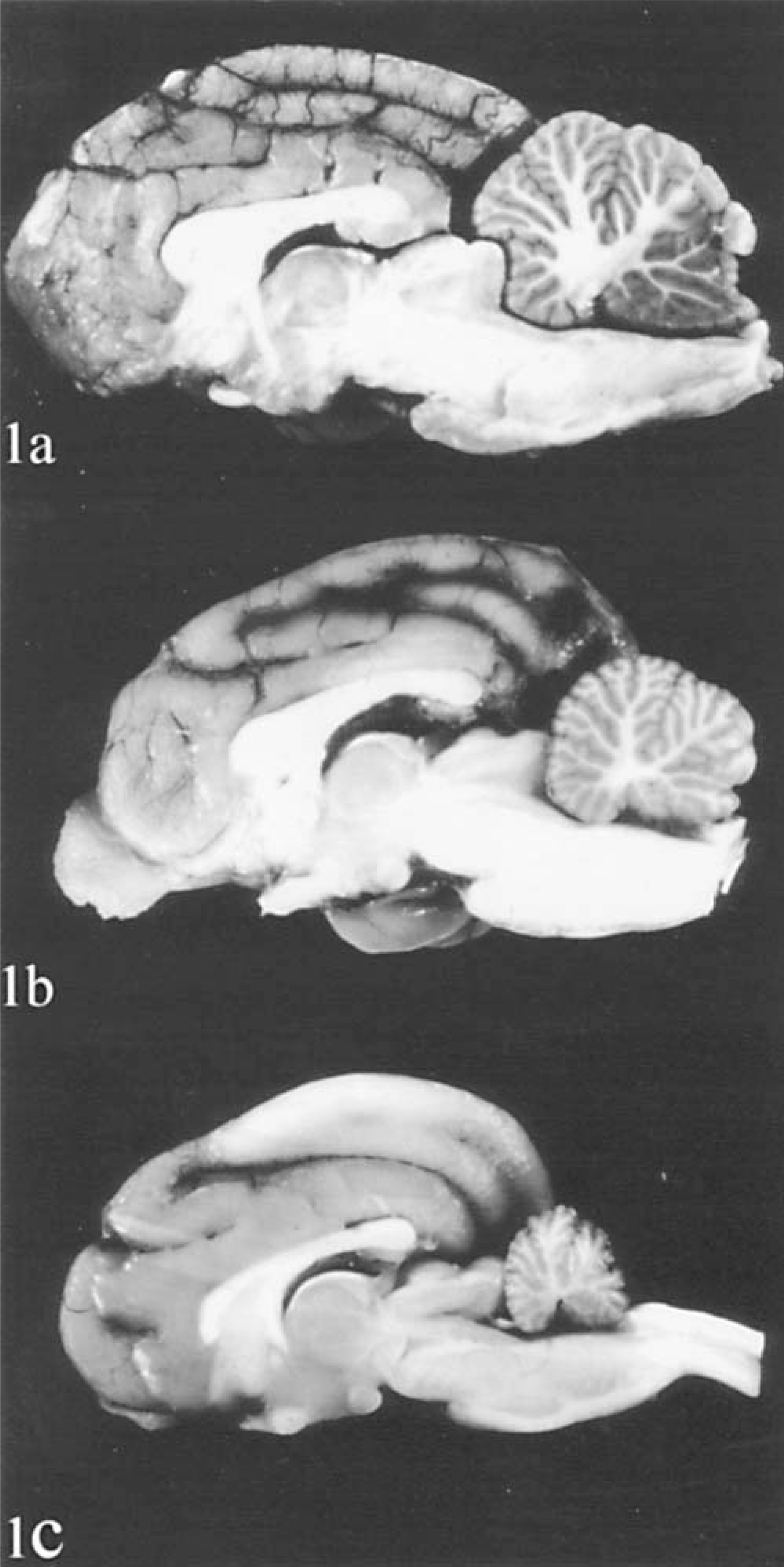
Brain in sagittal plane.
Immunolabelings specificity
The reaction of the immunesera used in this study was similar to that in previous reports on mice and rats. 4,6,11,22,25,30,39 Anti-calbindin immunopositivity identified Purkinje cells and neurons of the olive. Anti-calretinin–stained unipolar brush cells, olivary neurons, and pontine nuclei neurons. Anti-MAP2 stained all neuron pericarya and most of the main dendrites. Anti-tau identified oligodendrocytes and granule cell axons. Clone RT97 identified phosphorylated neuro-filaments in general and basket cell axons in particular. Anti-synaptophysin identified synapses. GFAP and S100 were expressed in glial cells, and lectin RCA I reacted with microglial cells.
Microscopic analysis
The cerebellar cortex
The whole neuron population was examined with Klüver–Barrera staining and MAP2 immunolabeling. The cerebellar cortex appeared markedly atrophic in both animals with a similar pattern of progressive thinning, which followed two gradients: 1) a general gradient of increasing severity from the midvermis toward the hemispheres, and 2) a gradient of increasing severity from the depth of the folia toward their tips. In cat No. 1, the deepest third of the vermal folia approached normal: granule cells appeared normally packed inside a thick granular layer and Purkinje cells were present (Fig. 2). More distally in the folia, and more laterally in the hemisphere, the Purkinje cells disappeared rapidly followed by progressive loss of granule cells resulting in an overall thinning of the granular layer (Fig. 3). However, even in the most dorsal and lateral parts of the cortex, a granular layer remained present (Fig. 3). In cat No. 2, the degenerative sequence was similar along the folia but more rapidly progressive: at mid-level of the medial part of the vermal folia and even in the depth of the lateral part of the vermal folia the granule cells were definitely reduced in number, the granular layer markedly thinned, and the Purkinje cells infrequent (Fig. 4). Cortical degeneration increased progressively, culminating with complete disappearance of the Purkinje cells, granule cells, Golgi cells, and eventually basket and stellate cells, with almost absence of cortex at the tip of the vermal folia and in the hemispheric surface (Fig. 5). The granule cell axons, parallel fibers in the molecular layers, and their synapses (in the granular and molecular layers) were stained, respectively, by anti-tau and anti-synaptophysin: their decrease paralleled strictly the disappearance of the granule cells.
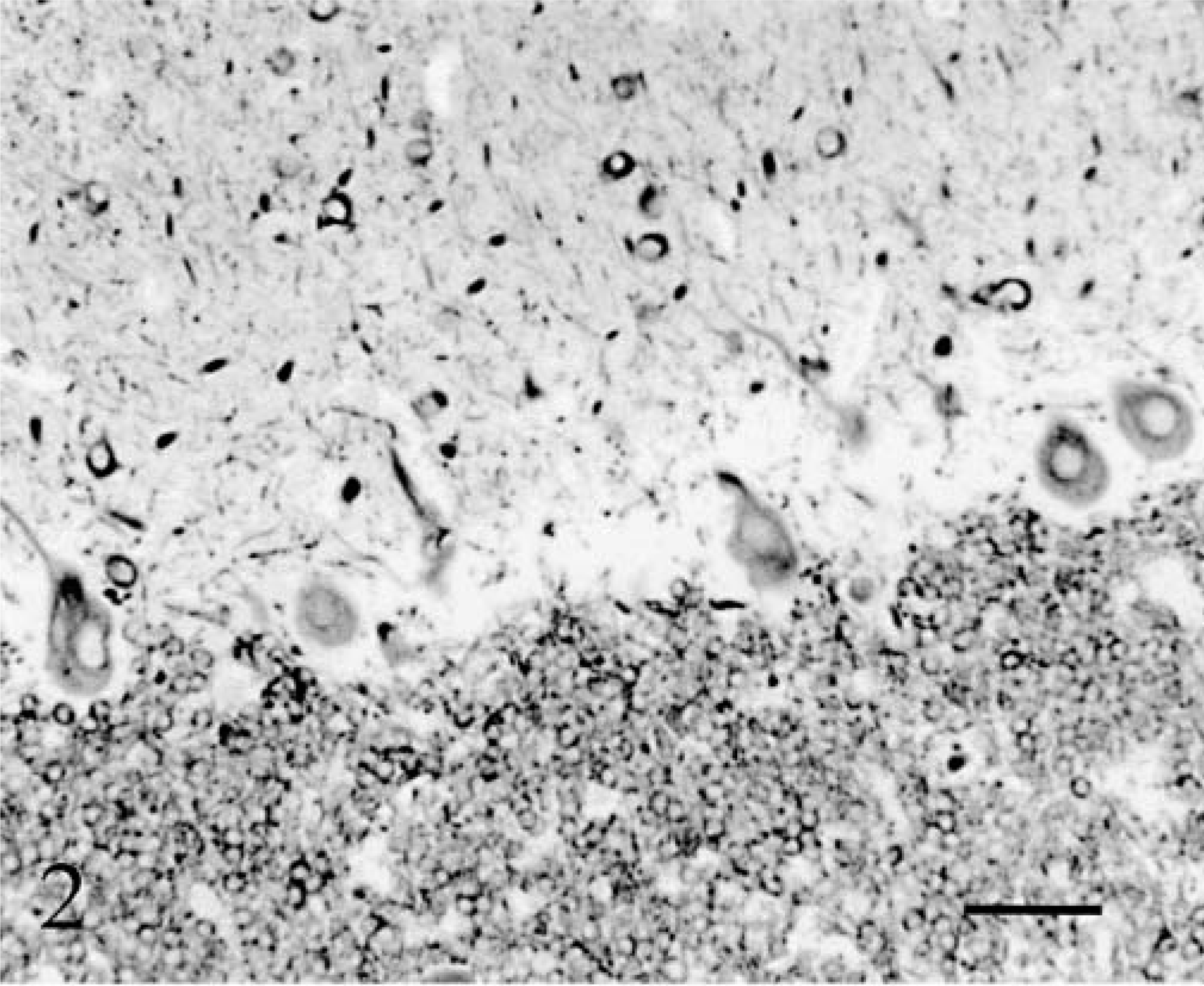
Cerebellum; cat No. 1. Cortex in the depth of the midvermis. Anti-MAP2. Bar = 55 µm.
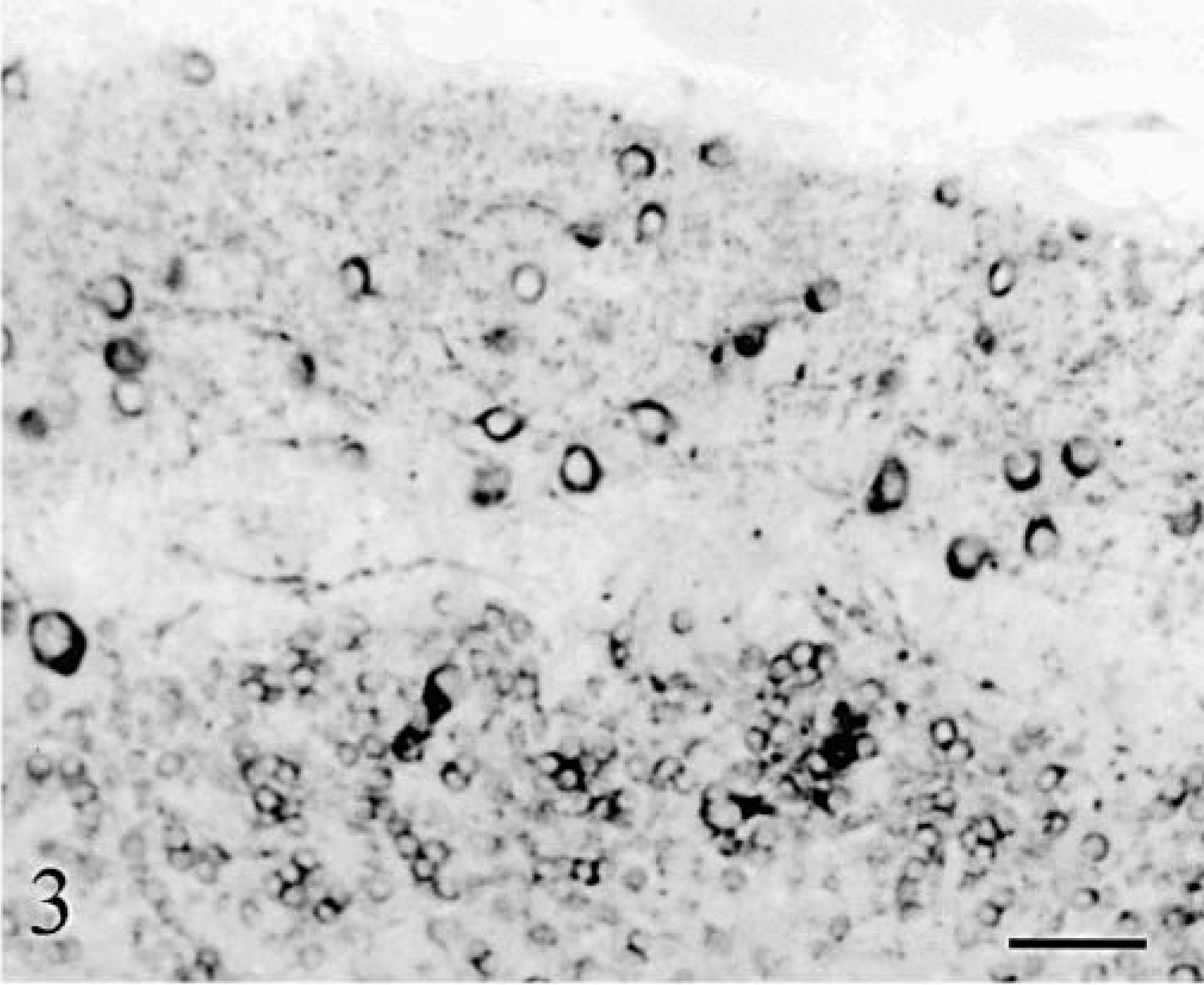
Cerebellum; cat No. 1. Cortex at the surface of the lateral vermis. Note the absence of Purkinje cells and the low number of granule cells. Anti-MAP2. Bar = 55 µm.
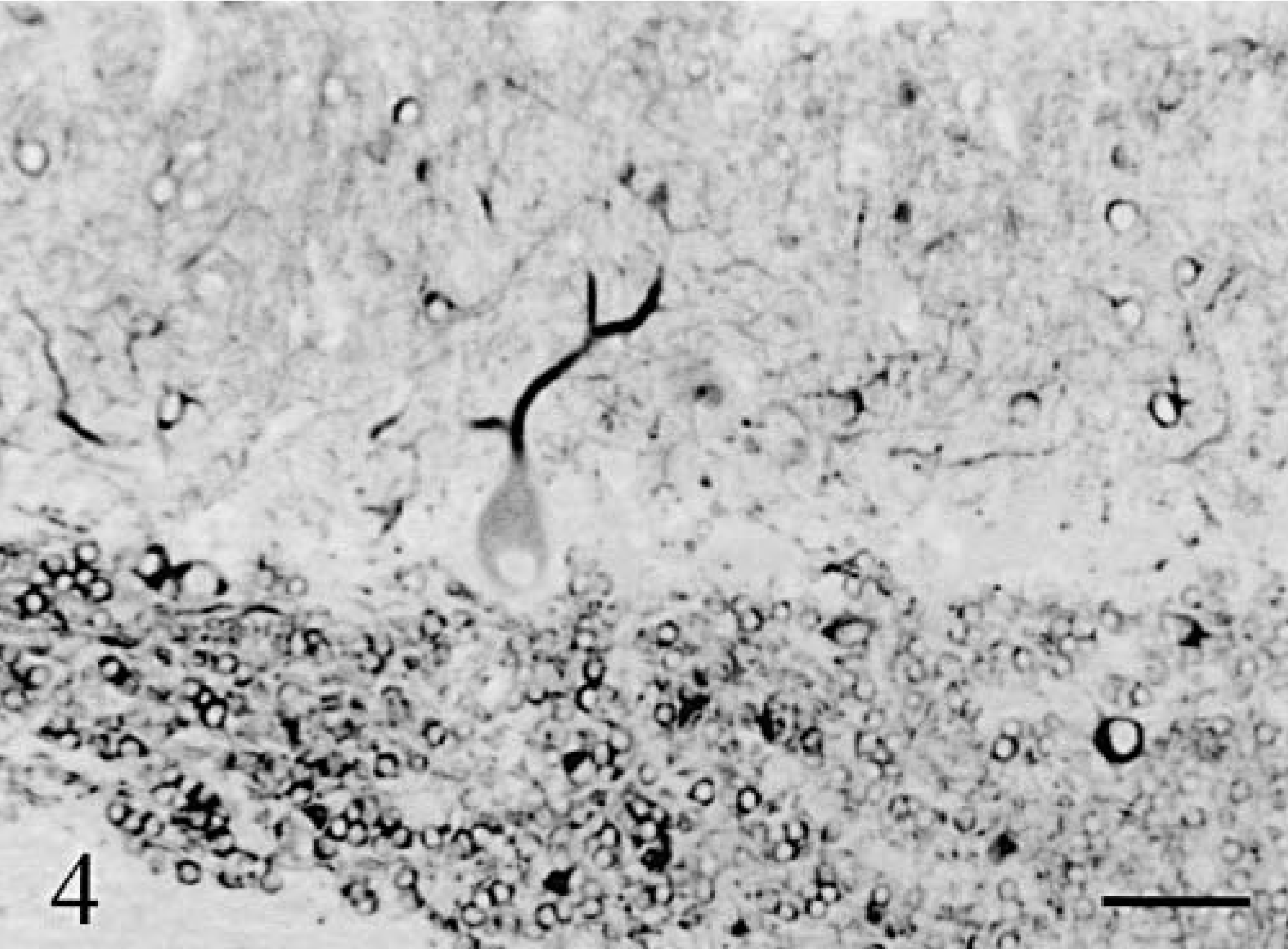
Cerebellum; cat No. 2. Cortex in the depth of a lateral vermis folium. Anti-MAP2. Bar = 55 µm.
Cerebellum; cat No. 2. Cortex at the tip of an hemispheric folium. Complete degeneration of all cortical layers. Anti-MAP2. Bar = 55 µm.
In both cats, the nodulus and flocculus were degenerated to the same extent as the other parts of the cerebellum.
Purkinje cells
Purkinje cells (Figs. 6–8) were observed only in regions with a relatively well-preserved cortex, i.e., in a large moiety of the deep vermis in cat No. 1 (Figs. 6, 8) and along a small length of the deepest part of vermal folia in cat No. 2 (Fig. 7). None of them were positive for phosphorylated neurofilaments (RT97 immuneserum). They had a normal distribution inside the cortex and appeared normal except for a high number of cells harboring two main dendrites (Fig. 8). Axonal spheroids, arciform axons reentering the internal granular layer, or abnormal nuclear inclusions were never observed. At more distal positions in the folia, Purkinje cells disappeared abruptly, at comparable levels in two adjacent folia (Figs. 6, 7). In the mediolateral direction also, the transition between the presence and absence of Purkinje cells took place at similar levels in adjacent folia. The positions of the most lateral surviving Purkinje cells in the hemispheric lobules (cat No. 1) or the lateral vermis (cat No. 2) formed a defined band oriented parallel to the longitudinal axis of the cerebellum. In cat No. 1 but not in cat No. 2, small PAS-positive lipofuscin inclusions were frequently seen in the cytoplasm of the Purkinje cells and in many neurons of the brainstem and hippocampus. Such inclusions are frequently encountered in normal cats more than 4 years old.
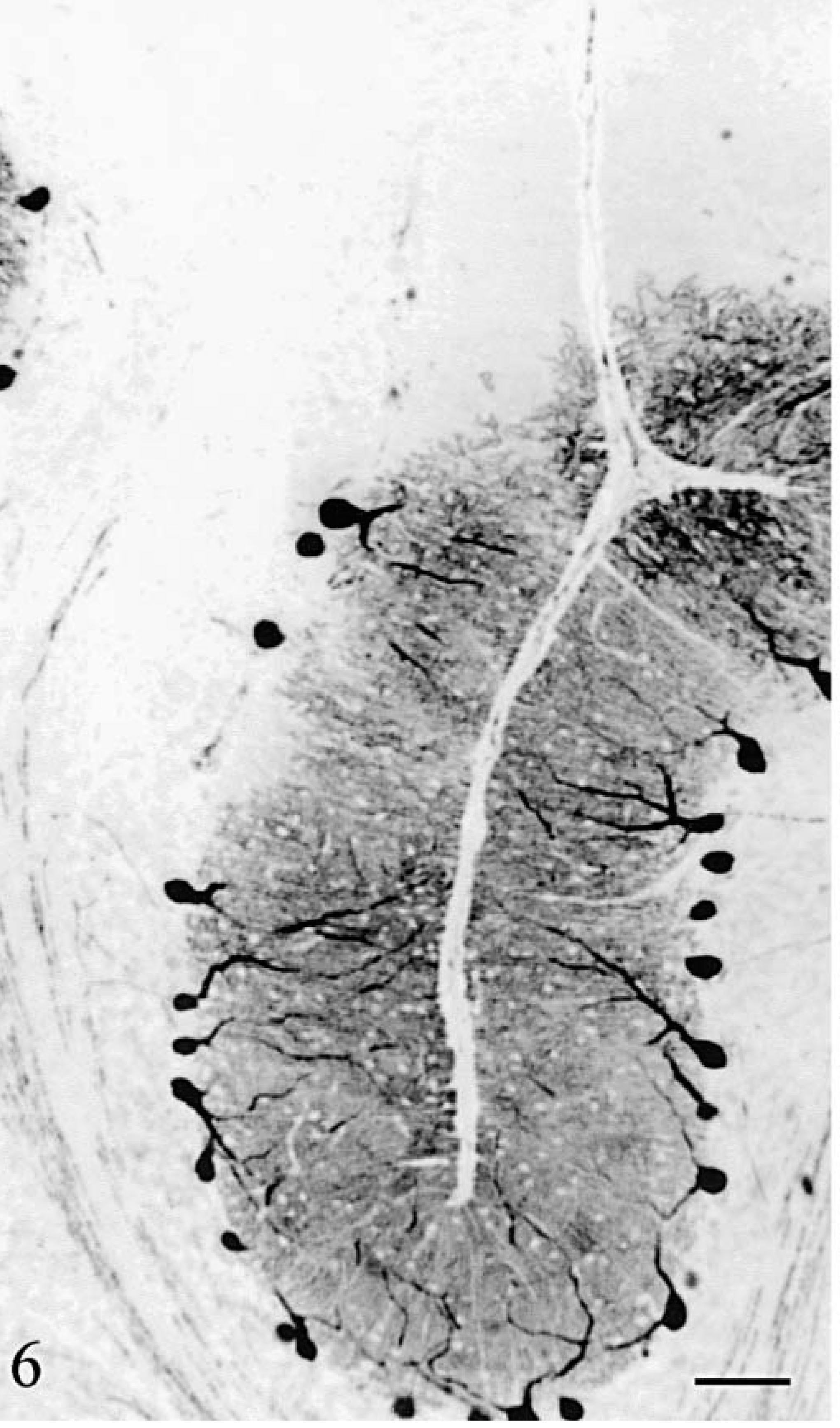
Cerebellum; cat No. 1. Sagittal section of the vermis. Purkinje cells are present only in the lower two third of the folia. Anti–calbindin. Bar = 150 µm.
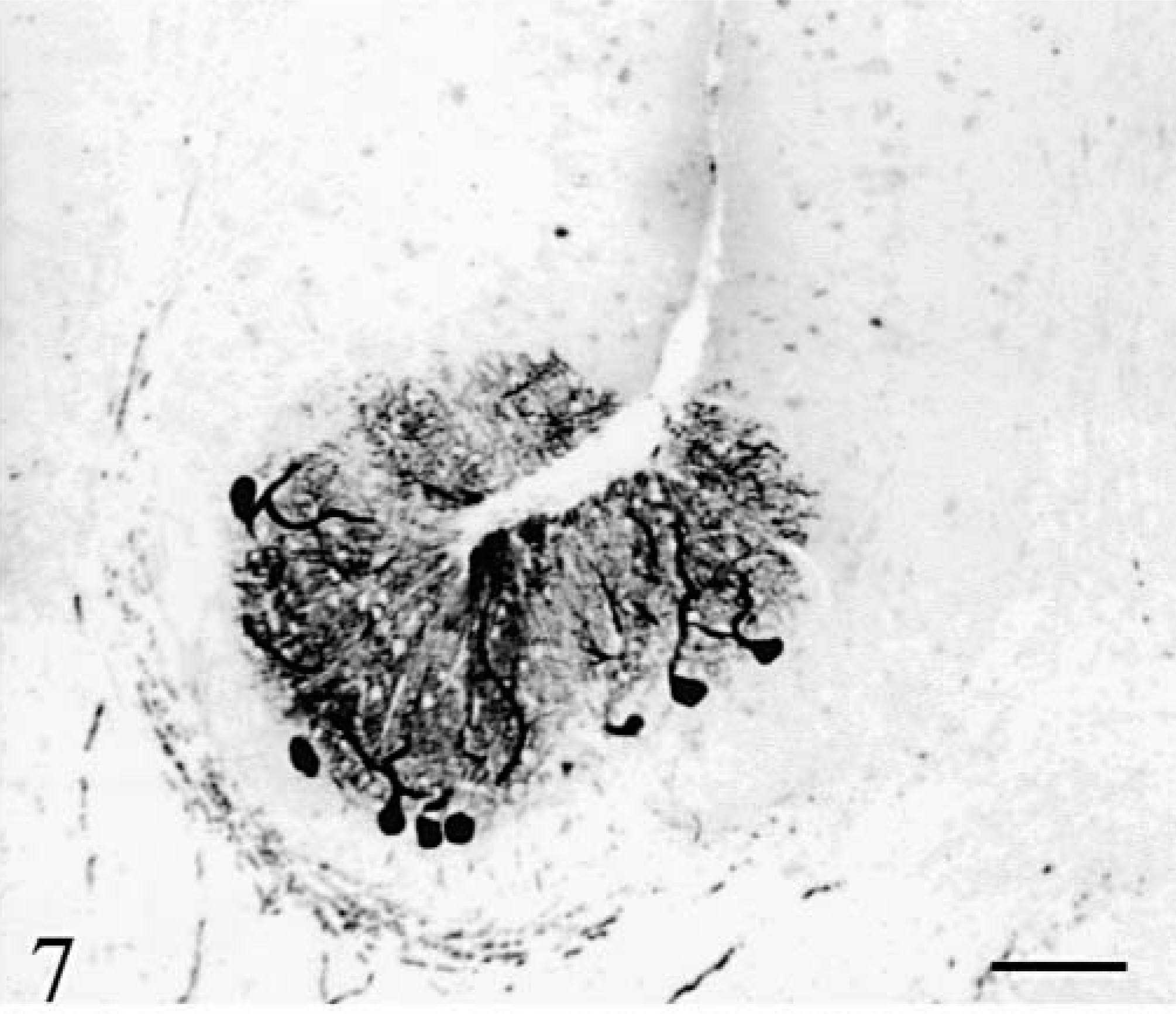
Cerebellum; cat No. 2. Sagittal section of the vermis. Purkinje cells are present only in the depth of the folia. Anti-calbindin. Bar = 200 µm.
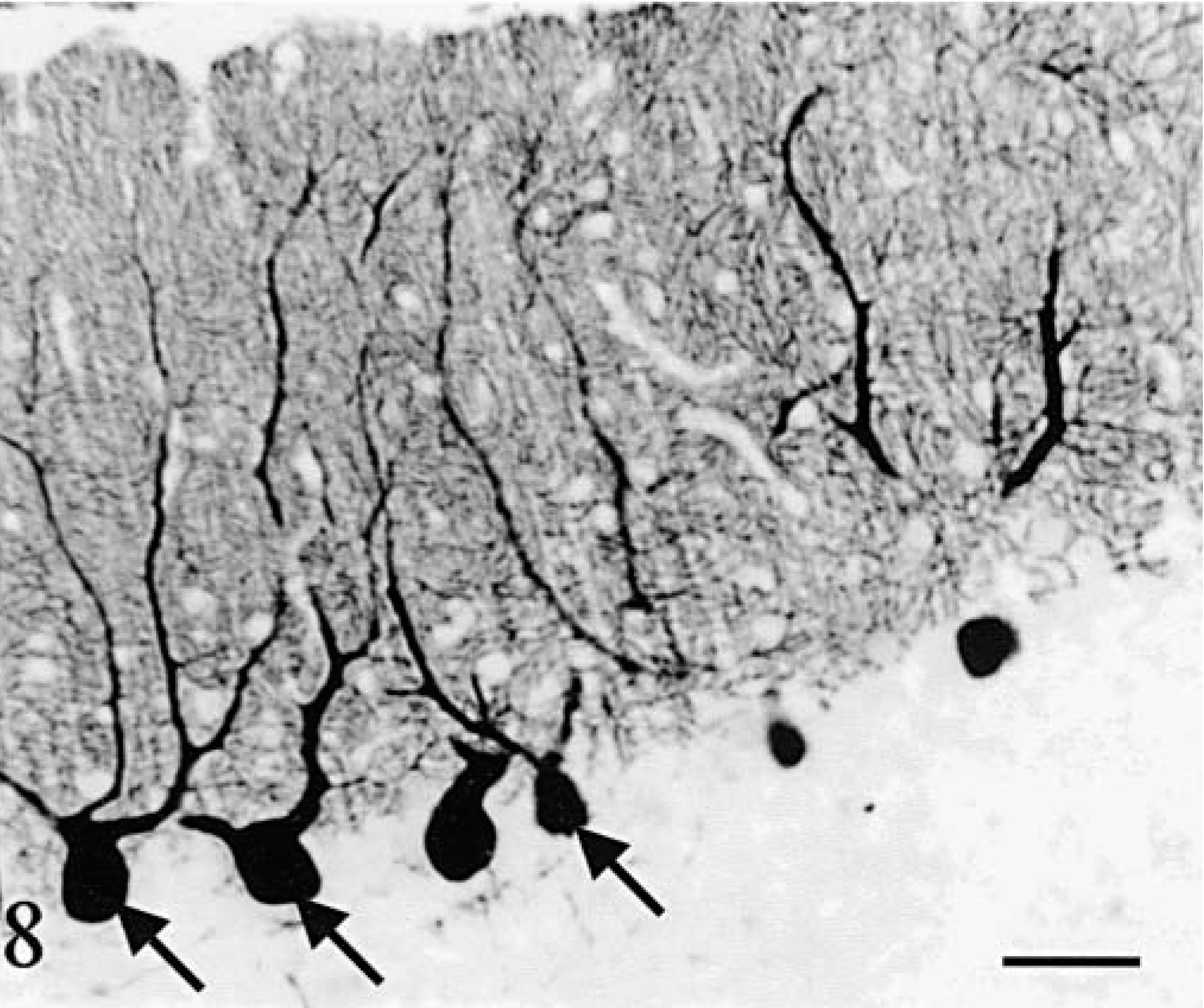
Cerebellum; cat No. 1. Purkinje cells in a sagittal section of the vermis. Arrows point out abnormal multiple main dendrites. Anti-calbindin. Bar = 75 µm.
Basket cell
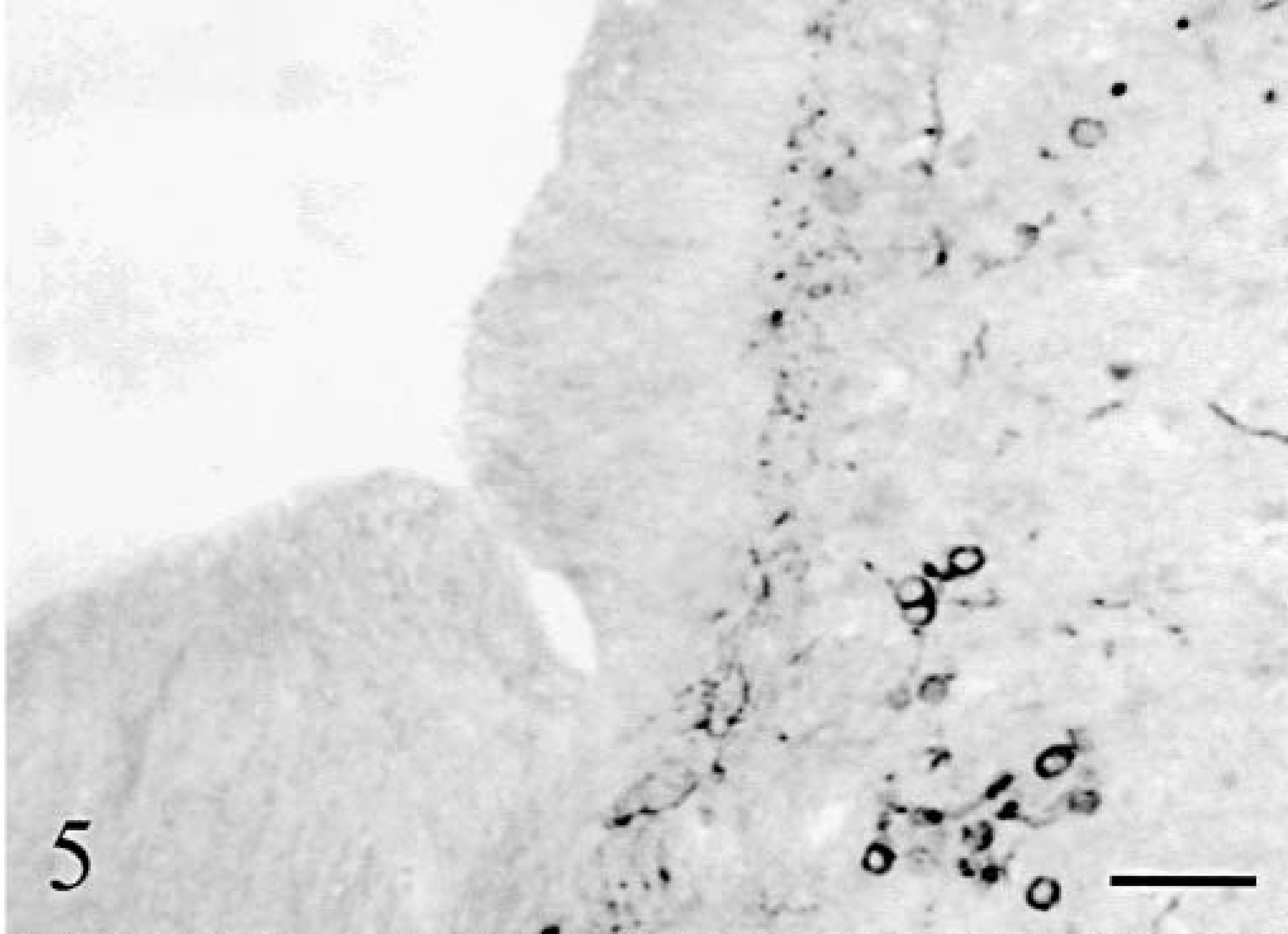
Basket cell axons were identified using antiphosphorylated neurofilaments (RT97) and their cell bodies were also stained by anti-MAP2. As did all other cortical cells, basket cells disappeared progressively in the bottom-surface (Fig. 9) and medial–lateral directions. The baskets surrounding the surviving Purkinje cells were normal. When the latter became scattered, a mixture of normal and shrunken empty baskets was seen, progressively replaced by empty baskets only. At the tip of the vermis folia in cat No. 2 and in its hemispheres, empty baskets were increasingly rare and disorganized (Fig. 10), and eventually entirely absent. At the same time, the number of thick RT97-positive parallel processes running at the superficial border of the molecular layer diminished and the number of MAP2-positive basket cells decreased also until absent (Fig. 5).
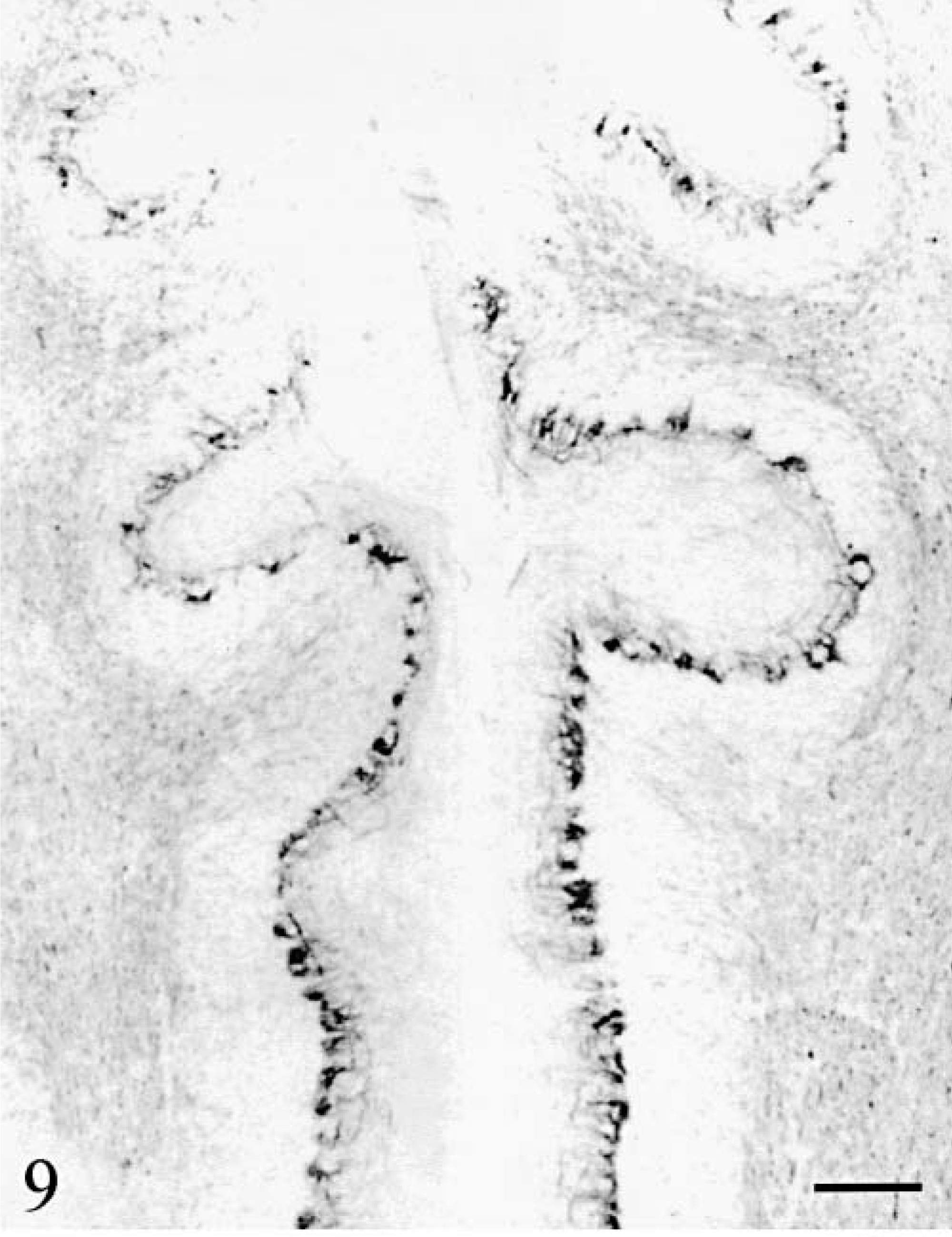
Cerebellum; cat No. 1. Sagittal section of the vermis. Note the progressive disappearance of the baskets. Anti-phosphorylated neurofilaments. Bar = 280 µm.
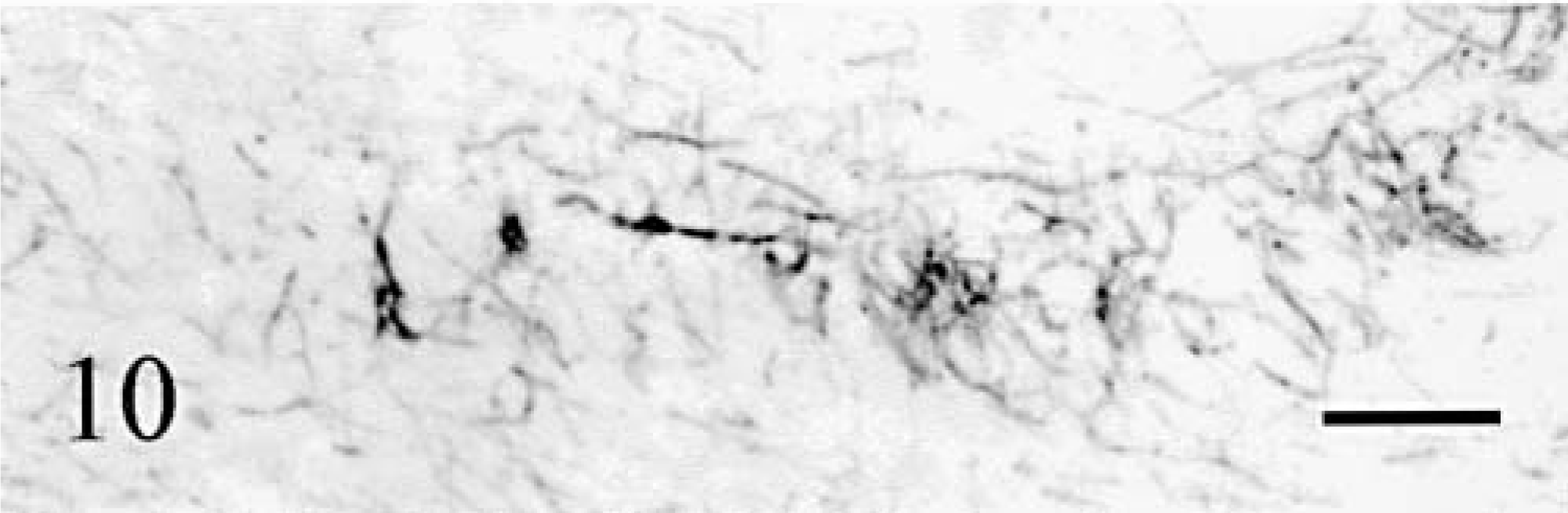
Cerebellum; cat No. 2. Remaining basket cell axons in sagittal sections of the midvermis. Detail at the tip of the folia. Anti-phosphorylated neurofilaments. Bar = 60 µm.
Unipolar brush cells
Unipolar brush cells marked with anti-calretinin, were the most resistant cells in the cerebellar cortex. They were present and morphologically normal in the caudal vermis (Fig. 11) and flocculus even when few other cortical neurons were still recognizable.
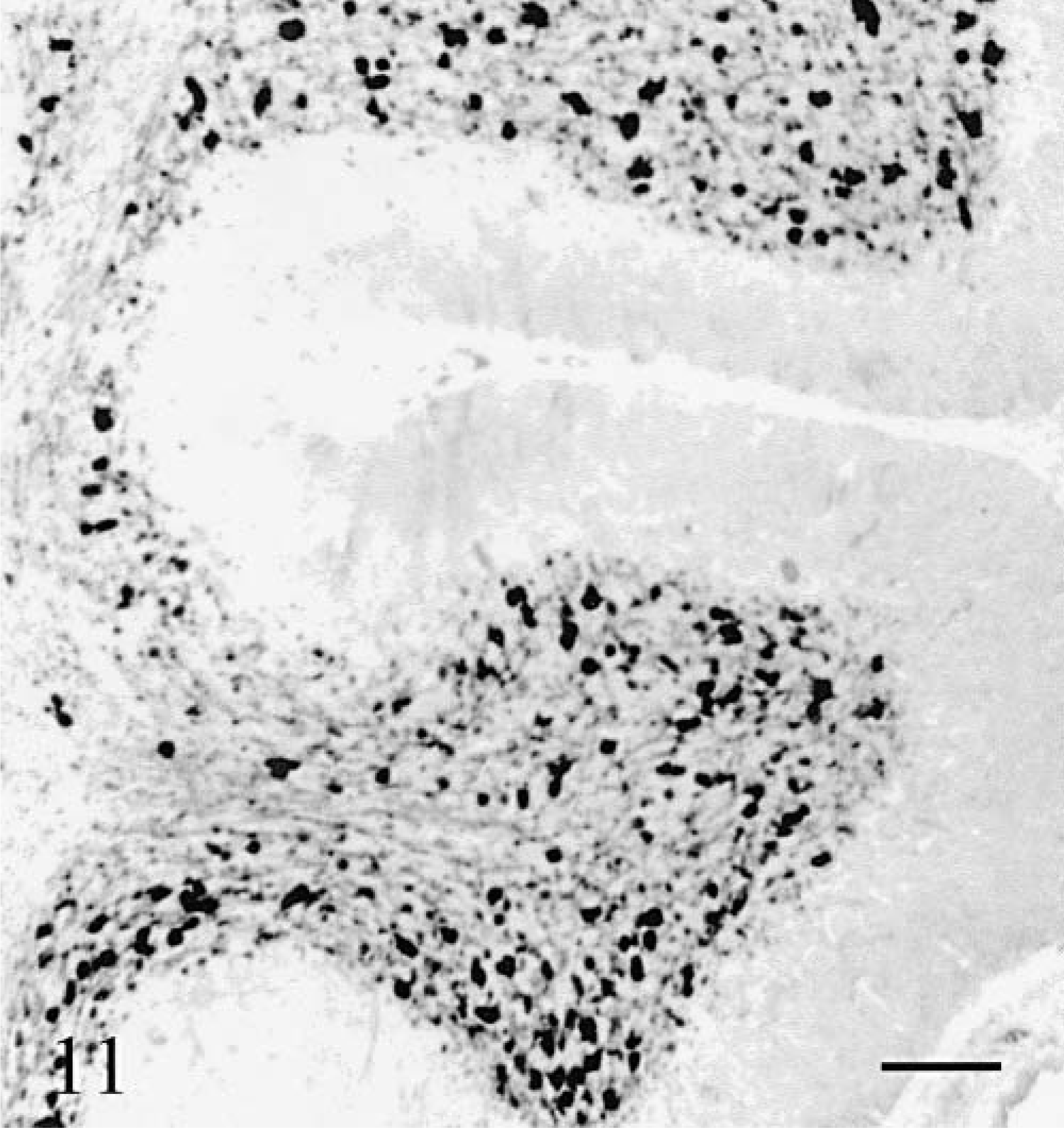
Cerebellum; cat No. 2. Unipolar brush cells in the caudal vermis. Both cell aspect and density are normal. Anti-calretinin. Bar = 100 µm.
Cerebellar white matter
The white matter degeneration of the folial axes was proportional to CCD. No active demyelination was visible. Loss of myelinated fibers was most conspicuous around the blood vessels especially in cat No. 2.
Glial cells
Glial cells appeared moderately increased in number. However, their processes were abnormally numerous. They were the only cell types still present at the tips of the folia in cat No. 2 (Fig. 12). Anti-GFAP–staining showed significant increase in glial fibers randomly oriented (Fig. 13). This change was proportional to the degeneration of the cortex. In addition, anti-GFAP stained a dense matrix of thin fibers around the blood vessels in the white matter. Anti-S100 stained the pericarya of the Bergmann glial cells in all cortical regions, but appropriate labeling of the processes was seen only where Purkinje cells were present (Fig. 14). S100 immunoreactivity in glial processes disappeared in areas devoid of Purkinje cells (compare the distribution of calbindin [Fig. 6] and S-100 [Fig. 14] immunoreactivities). Again, the topographic modifications of GFAP and S100 immunopositivity in cat No. 2 was identical to the one in cat No. 1 but with a more rapid worsening.
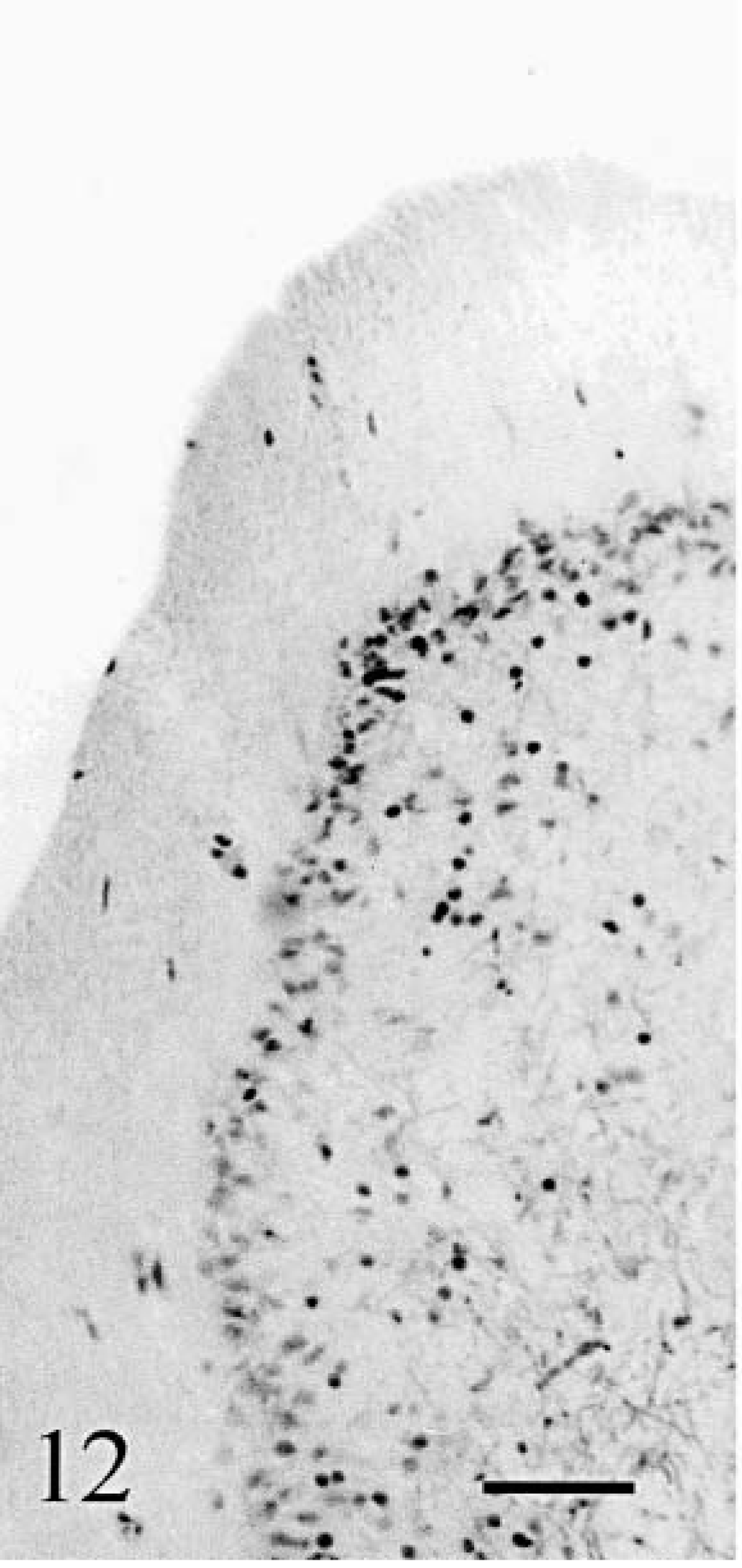
Cerebellum; cat No. 2. Many cortical glial cells are present but no neuron. Klüver–Barrera. Bar = 80 µm.
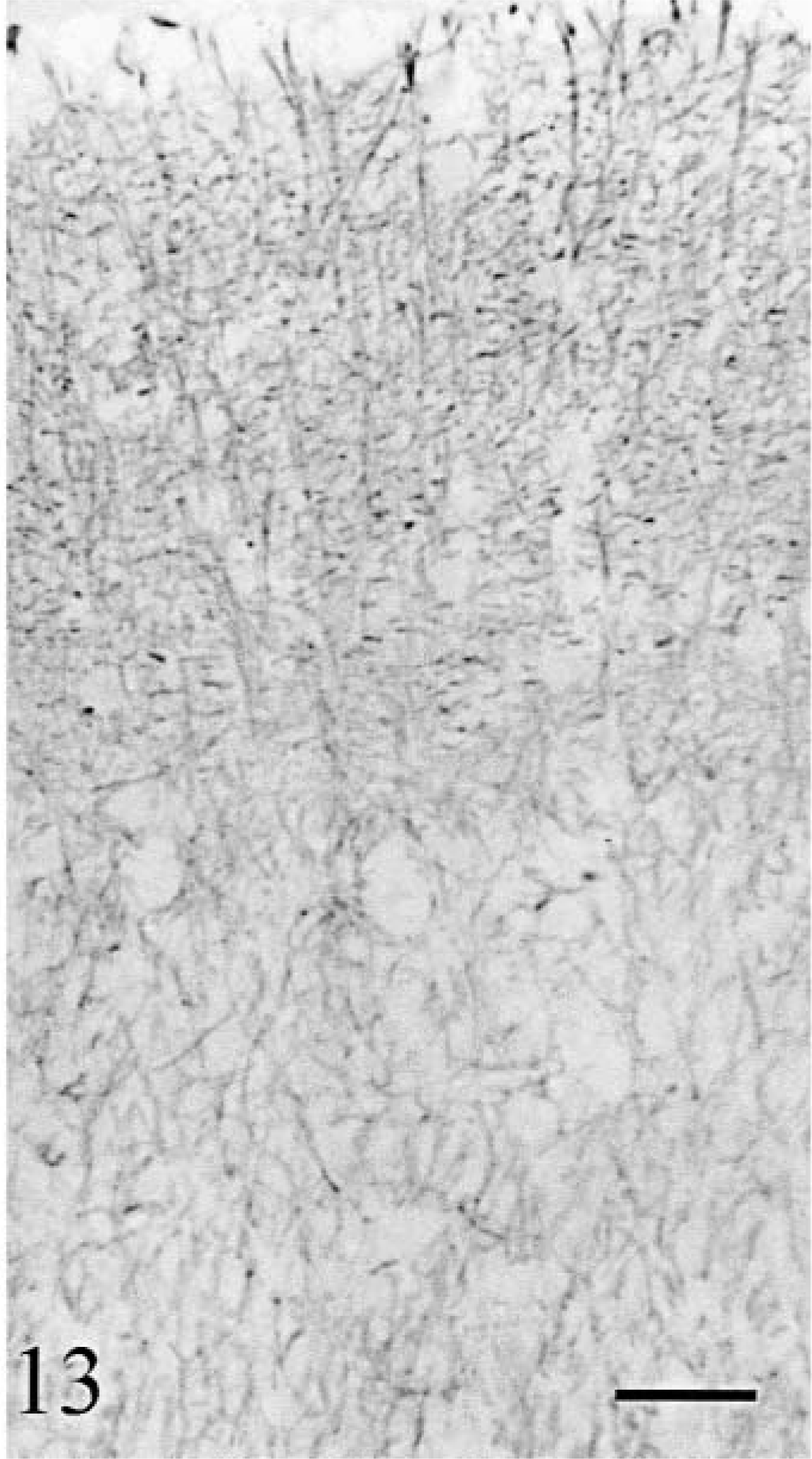
Cerebellum; cat No. 1. Cortical glial cells. Dense abnormal glial network. Anti-GFAP. Bar = 50 µm.

Cerebellum; cat No. 1. Both the cell body and the processes of the glial cells are labeled in the depth of the folia but only the cell body more superficially. Anti-S100. Bar = 155 µm.
Cerebellar and lateral vestibular nuclei
Cerebellar and lateral vestibular nuclei were composed of morphologically normal MAP2-positive neurons. In cat No. 1, as in the normal controls, these neurons were embedded in a dense network of calbindin-positive fibers (Fig. 15a) and their surface was covered by calbindin-positive synaptic “boutons” (Fig. 15a) emanating mainly from Purkinje cell axons. These fibers and synaptic endings were extremely decreased in number in cat No. 2 (Fig. 15b). The rostral cerebellar peduncle was normal in both cats.
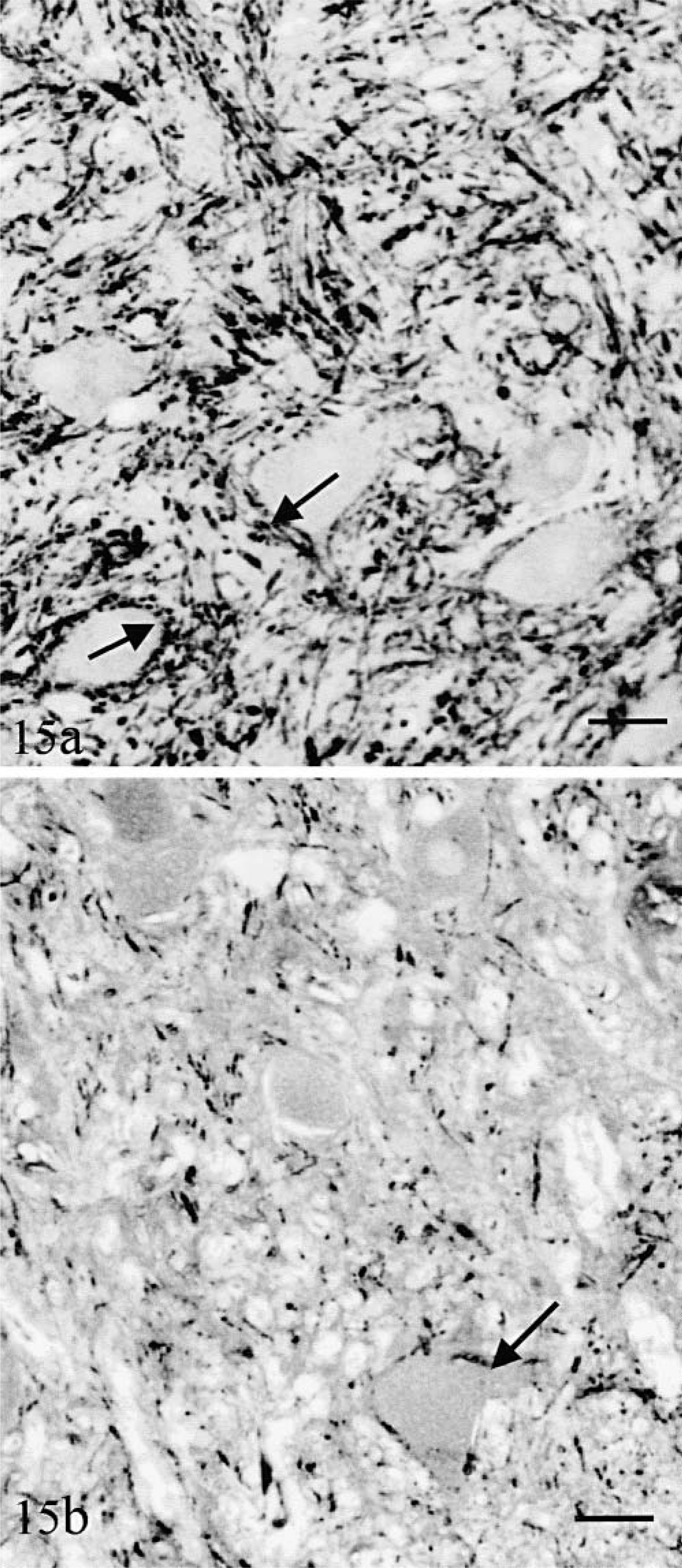
Lateral vestibular nucleus.
Olivary nuclei
Neurons were calretinin-, MAP2-and calbindin-positive. When compared with normal controls (Fig. 16a), cat No. 1 had many calbindin-positive neurons in its dorsal accessory nucleus but neuronal loss (Fig. 16b) and gliosis was severe in the olivary and medial accessory olivary nuclei. A significant decrease in calbindin-positive olivarocerebellar axons was also observed. In cat No. 2, all the olivary nuclei were nearly devoid of neurons (Fig. 16c), replaced by dense gliosis. No arcuate fibers were visible leaving the hilus of the nuclei. In the caudal cerebellar peduncle the decrease in calbindin-positive processes emanating from the olivary nuclei was important, especially in cat No. 2, but atrophy was only moderate because of the persistence of normal spinocerebellar afferents.
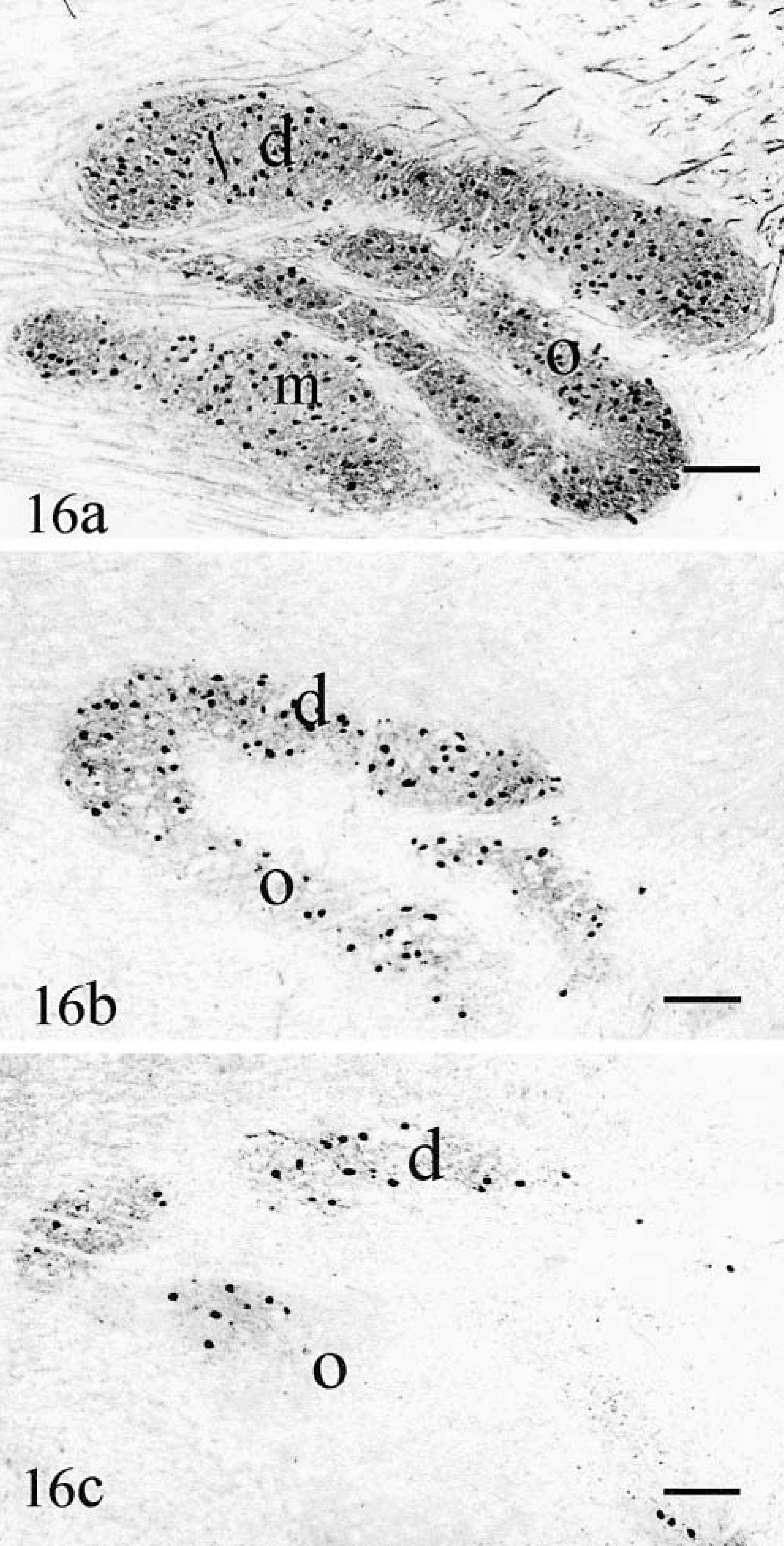
Olivary nuclei.
Pontine nuclei
Neurons were MAP2- and calretin-in-positive. Compared with normal (Fig. 17a), the loss of calretinin-positive neurons in both cat No. 1 (Fig. 17b) and cat No. 2 (Fig. 17c) was significant. In both cats, few neurons were present in subnuclei within the longitudinal fibers of the pons. The ventral subnucleus was markedly diminished in cat No. 1 and almost absent in cat No. 2. Cat No. 2 had no remaining transverse fibers along the ventral surface of its pons. The middle cerebellar peduncle of cat No. 2 was severely atrophic, hypomyelinated, and exhibited the similar degrees of perivascular gliosis as the cerebellar white matter.
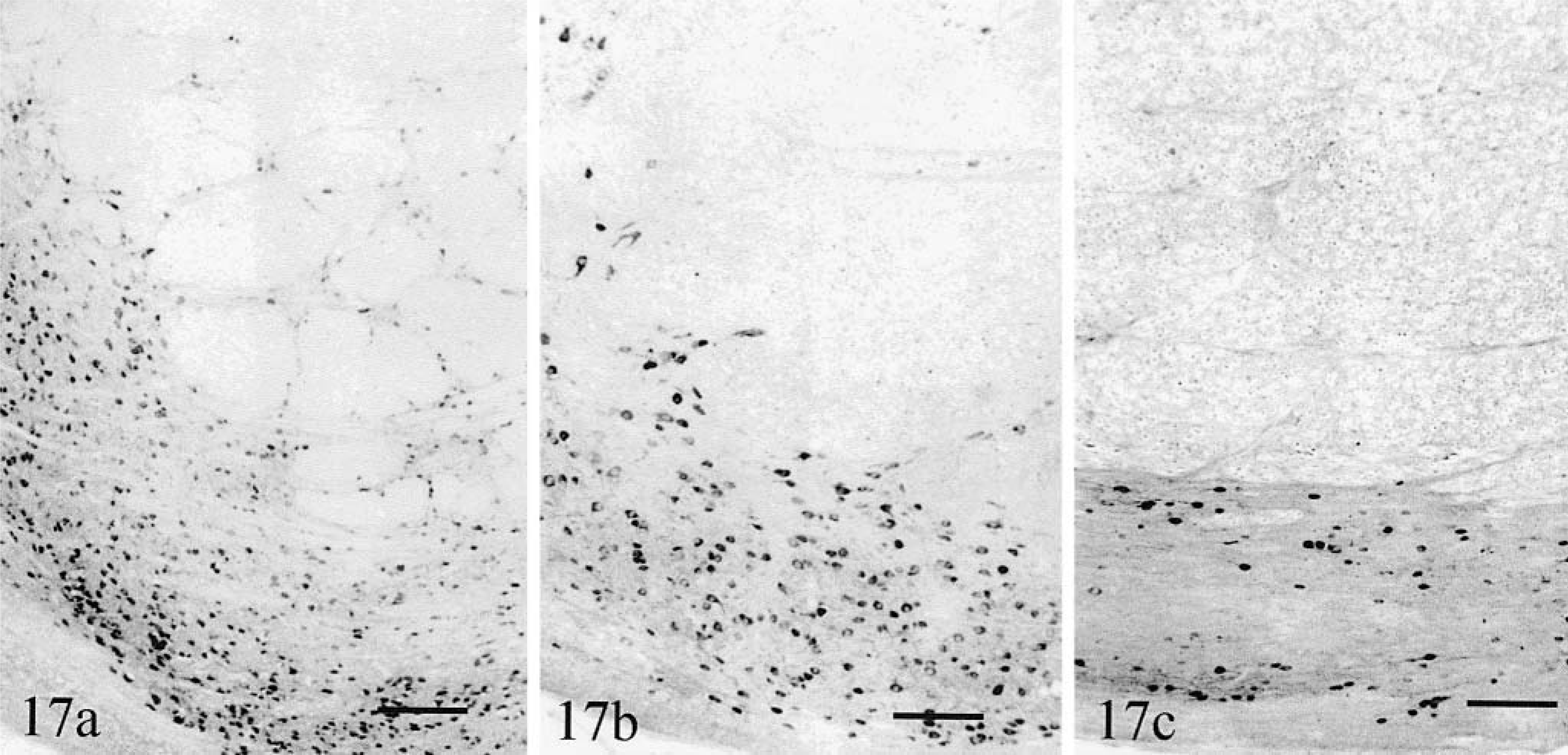
Pontine nuclei. Transverse sections.
Other brainstem and spinal cord structures were normal.
Discussion
In this report we described the cerebellar and brainstem lesions of two cats with very different clinical signs but similar lesions. Although differing in severity, both cats exhibited prominent degeneration of Purkinje cells and other corticocerebellar cells, with the exception of unipolar brush cells. The topographic distribution of lesions was similar in both cats. Olivary and pontine nuclei were degenerate, whereas other mesencephalic, medullary, or spinal systems were intact. Active inflammation was not apparent in either cat. These facts strongly suggest a similar disease process (OPCA) possibly at early (cat No. 1) or late (cat No. 2) phase of evolution.
Cat No. 1, in a subclinical status, was at least 4 to 5 years old as attested by the lipofuscin inclusions in many of its brainstem neurons, and the lesions were still evolving as indicated by the degenerating Purkinje cells at the border of the abnormal areas. Cat No. 2 was observed with mild signs 1 month before presentation; moreover the final disabling was most probably incompatible with a long outdoors survival. Consequently, available history and histopathology substantiated an adult onset, evolving entity. The mild clinical changes in cat No. 1 were consistent with data showing that a loss of 50–75% of the Purkinje cells was necessary to generate clinical signs in mice. 14 These feral cats were living at 4.5 km from each other and might be genetically related. Indeed, the largest distance in roaming area of dominant male cats during mating seasons can be two to three times this distance. 23 The hypothesis that they were suffering a same genetic disease is thus a real possibility, although coincidental sporadic cases also are possible.
The lesions in these two cats contrast with previously reported cases of juvenile- 1,16,36 or adult-onset 2,33 feline cerebellar degeneration. These entities were purely cortical, and despite severe Purkinje cell loss, the granule cells were spared; moreover, the olivary nuclei were only moderately degenerate and the pontine nuclei were normal. This is in sharp contrast to the parvovirus-associated congenital ataxia in cats where the uniform granular layer atrophy is accompanied by Purkinje cell heterotopia and where pontine nuclei are preserved. 34 Accompanying olivary and pontine destruction link the present cases to feline cases reported in 1946 and 1934. 3,32
Many breed-related cerebellar degenerations (abiotrophies) have been described in the dog species. 35 Most are purely cortical with some olivary nuclei involvement. In some reports, the cerebellar hemispheres were more affected than the vermis, 38,41 but few other points of comparison with the cats of this report are available. More complex primary cerebellar degenerations have been reported in dogs, including spinocerebellar degeneration, 15 and a multisystem degeneration involving the substantia nigra, caudate nuclei 12 which in some breeds exhibits a concurrent cerebral cortex degeneration. 9
Purkinje cell degeneration along a strictly organized spatial pattern has been reported in strong shaker mutant rats; 37 however, in these rats, the topography of Purkinje cell death was different, and the Purkinje cell decay did not lead to complete cerebellar cortex loss.
Sporadic or dominantly inherited late-onset OPCAs are observed in humans. 13,21,26,27,31,40 Most of the latter are caused by expanded triplet repeat in one of the ataxin genes. 28,29 Differences in onset age and severity for the two cats of the present report are reminiscent of the anticipation phenomenon, i.e., earlier onset and faster evolution among generations observed in these cases. However, in human entities, no significant attrition of the granular layer was present despite dramatic loss of Purkinje cells, contrasting with the cases reported here. In addition, affected Purkinje cells harbor axon torpedoes and nuclear inclusions, made of ubi-quitinated pathologic ataxin. Both characteristics were absent in the present cats. However, this does not preclude comparison because axonal dilatations were absent in transgenic mice bearing an 82-polyglutamic repeat (ataxin-1[82Q]), 7 and because nuclear aggregates depended on the genetic background of the mice. 10 Abnormal expression of phosphorylated neurofilaments and synaptophysin reported in the main dendrites of the abnormal Purkinje cells in humans 18 with sporadic OPCA was not observed in our cats.
Very few descriptions of an OPCA were made in a domestic animal species. They are by far more often encountered in humans, but few features in our cats allow tentative linking to these. The ordered disappearance of all cortical neuronal types has never been described before. Either this entity is cat specific, or it might pinpoint the need for increased knowledge about differential gene expression depending on genetic background, i.e., among different species. It also would open prospects about gene product interactions within neurons.
Footnotes
Acknowledgements
We are grateful to Dr J.P. Brion from the Faculty of Medicine of the Free University of Brussels who gave us several of the immunesera used in this report. This investigation was supported by grants of the “Fonds de la Recherche Scientifique Medicale Belge” (3.4411.88 to AR and 1.5025.02 to LP).
