Abstract
Membranoproliferative glomerulonephritis was observed in a 2-year-old male Japanese domestic cat with clinical renal failure. In the glomeruli, moderate mesangial hypercellularity with an increased mesangial matrix and thickening of the capillary walls were prominent. In addition, frequent duplication of the capillary walls, splitting, and spike formation were observed in the glomerular basement membrane. Granular cat IgG and complement component deposition were detected globally along the glomerular capillary walls and in the mesangium. Transmission electron microscopy revealed dense deposits in the subendothelial and subepithelial regions and the mesangium. Mesangial interposition was also observed. These glomerular lesions are also found in humans with membranoproliferative glomerulonephritis type III, which has not been reported in animals.
Naturally occurring immune complex–mediated glomerulonephritis (ICGN) is well documented in the cat. Membranous nephropathy is the most frequently observed glomerulonephritis in cats and is often associated with feline leukemia virus (FeLV) infection. 2 In addition, a form of glomerulonephritis with dense deposits in the subendothelial, subepithelial, and mesangial areas of the glomeruli has been reported in cats with malignant lymphoma, myeloproliferative disorders, and infection with FeLV. 2
Membranoproliferative glomerulonephritis (MPGN) is a chronic and progressive renal disease. The morphologic characteristics of MPGN are an expansion of mesangial areas by hypercellularity and an increased mesangial matrix and a thickening of the glomerular capillary walls with double contours. According to the World Health Organization classifications of human glomerular diseases, MPGN can be divided into three subtypes based on morphologic appearance. 1 Type I is characterized by subendothelial deposits, type II (dense deposit disease) is characterized by intramembranous dense deposits, and type III is characterized by both subendothelial and subepithelial deposits and by spike formation, similar to membranous glomerulonephritis. In this report, we describe a feline case of atypical MPGN that shares some morphologic characteristics with MPGN type III.
A 2-year-old male Japanese domestic cat had diarrhea, syntexis, and gastromegaly. The results of a blood test showed azotemia and hypoalbuminemia, indicative of a renal glomerular disorder (Table 1). Because ascites was suspected by radiography, ventrotomy was performed. The approximately 200 ml of ascitic fluid was serous. The kidneys and the mesenteric lymph nodes were swollen, but other organs appeared to be normal. Urine in the urinary bladder had high protein concentration (2, 110 mg/dl).
Blood test results for cat with membranoproliferative glomerulonephritis.
An open renal biopsy was performed, and the specimen was fixed in 10% formalin. The paraffin-embedded sections (3 µm) were stained with hematoxylin and eosin, periodic acid–Schiff (PAS), periodic acid–methenamine silver staining (PAM), Heidenhein's azan, and acid fuchsin orange G (AFOG). Additionally, immunohistochemical staining by the streptavidin–biotin peroxidase method (Histofine kit, Nichirei Corp., Tokyo, Japan; VECTASTAIN ABC kit, Vector Laboratories, Burlingame, CA) was carried out on the paraffin-embedded sections using primary antibodies against α-smooth muscle actin (α-SMA) (clone1A4, DAKO, Glostrup, Denmark), cat IgG (Kirkegaard & Perry Laboratories, Gaithersburg, MD), and cat complement component 3 (C3) (Biogenesis, Poole, UK). The immunoreaction was visualized by a diaminobendine–hydrogen peroxide solution, and the sections were counterstained with hematoxylin.
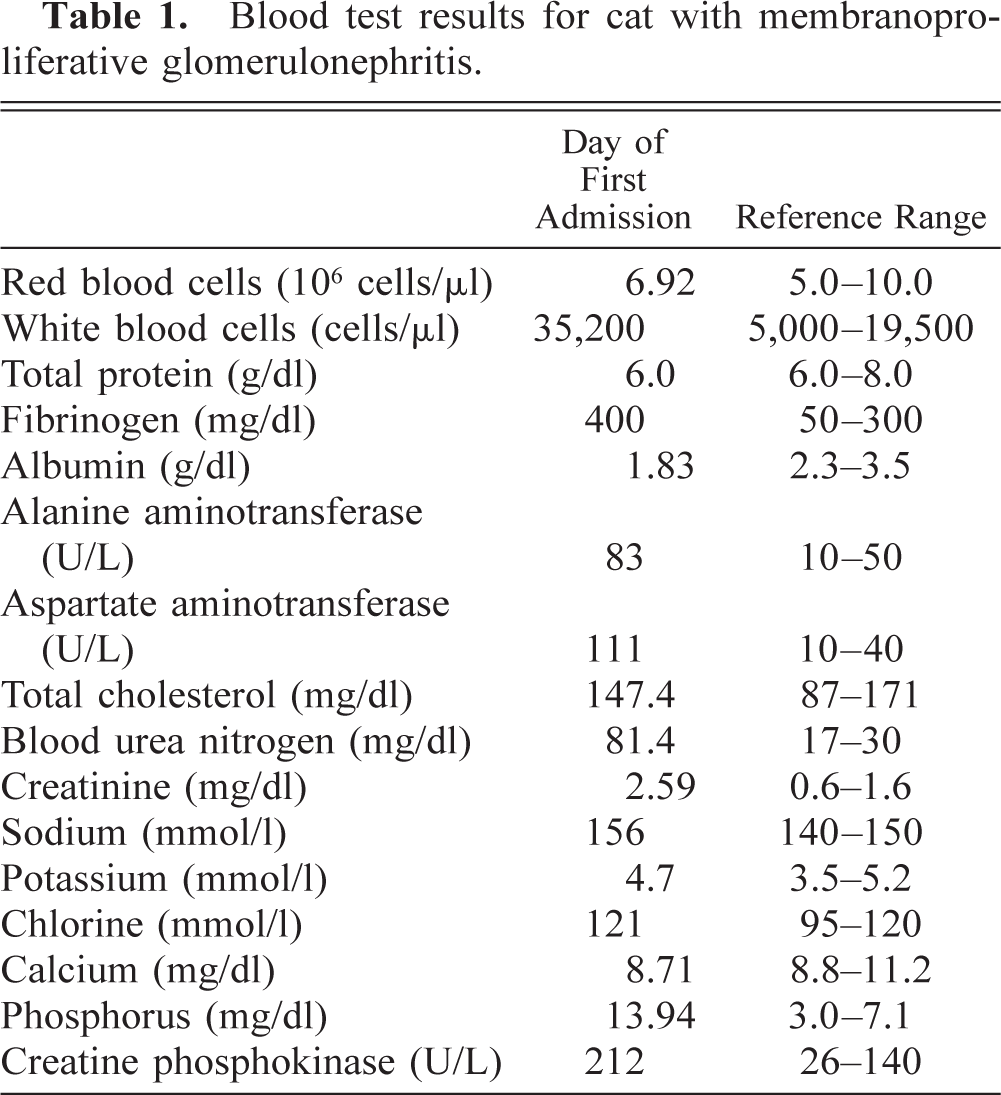
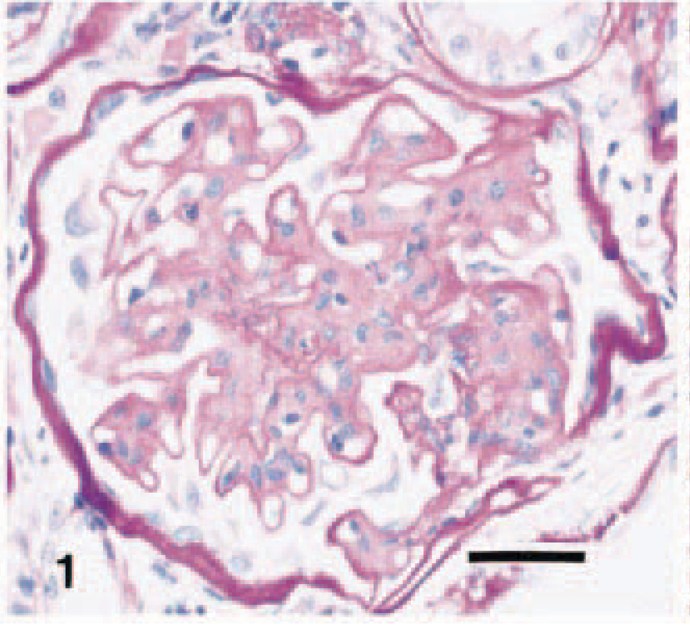
Histomorphologic examination revealed that all glomeruli in the specimen had moderate mesangial hypercellularity with an increased mesangial matrix (Fig. 1). In some glomeruli, a severe focal increase of mesangial cells and matrix encroached and compromised the patency of the capillary lumina. Thickening of the glomerular capillary walls was also prominent. Examination of the tissues stained with PAS and PAM revealed an occasional duplication of capillary walls, splitting, and the formation of spikes in the glomerular basement membrane (GBM) (Fig. 2). By azan and AFOG stains, granular protein deposits were seen along the capillary loops and within mesangial areas. A diffuse infiltration of mononuclear cells, predominantly lymphocytes and plasma cells, was observed in the interstitium. There were proteinaceous casts in some tubular lumina.
Renal glomerulus; cat. Mesangial expansion with moderate hypercellularity and increased mesangial matrix resulting in reduction of capillary spaces. PAS. Bar = 5 µm.
Renal glomerulus; cat. Spike formation (arrowhead) in the glomerular basement membrane. Glomerular capillary wall was thickened by mesangial interposition resulting in reduction of capillary spaces (arrow). PAM. Bar = 50 µm. Inset: Portion with characteristic changes indicated by large arrow. Bar = 0.5 µm.
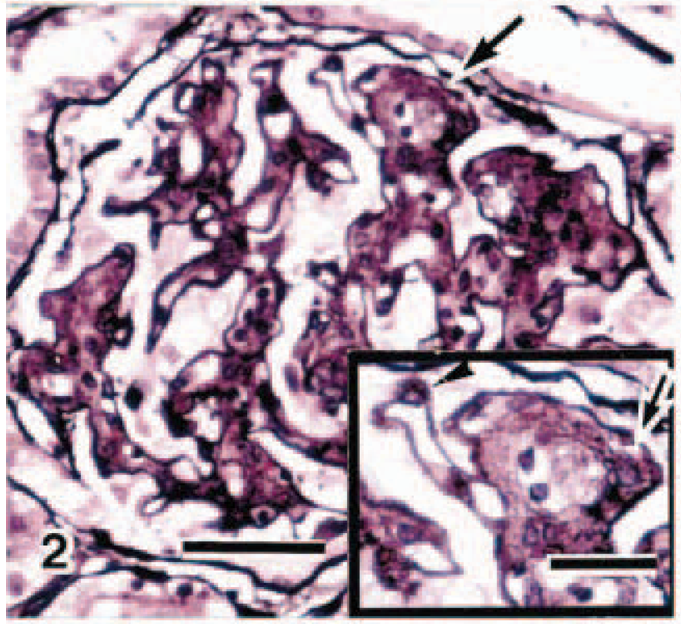
Immunohistochemically, granular deposits of cat IgG and C3 were detected globally along the capillary walls and in the mesangial areas (Fig. 3), and intense α-SMA expression was seen globally in the mesangial areas.
Renal glomerulus; cat. Granular deposits of cat complement component 3 were along the glomerular capillary walls (arrows) and in the mesangium. Streptavidin–biotin peroxidase method, Mayer's hematoxylin counterstain. Bar = 10 µm.
Electron microscopic evaluation of the formalin-fixed specimen revealed a large number of electron-dense deposits mainly in the subepithelial and mesangial areas and less in the subendothelial spaces (Figs. 4,5). A spikelike protrusion of GBM around the dense deposits was characteristic in the subepithelial region (Fig. 4). Interposition of mesangial cells between endothelial cells and GBM was also noted. The epithelial foot processes were widely effaced. Cytoplasmic vacuoles of various sizes and increased number of microvilli on the free cell surfaces were frequently seen in glomerular epithelial cells (Fig. 4). Portions of capillary lumina were narrowed, and platelet aggregation, monocytes, and neutrophils were sometimes observed in the capillary lumina.
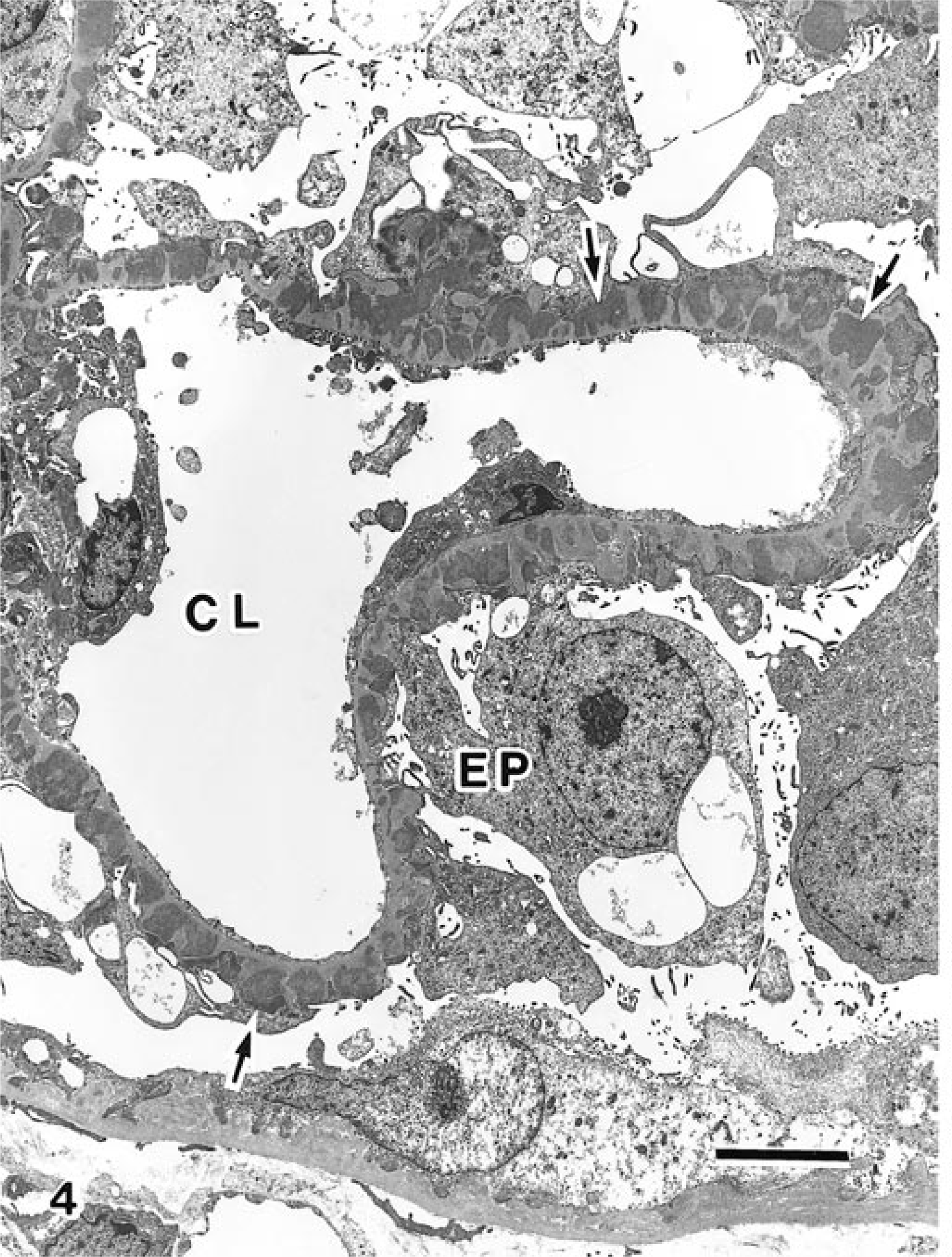
Transmission electron micrograph. Renal glomerulus; cat. Subepithelial electron-dense deposits (arrows) with spikelike protrusion of GBM were found in the glomerulus. Various size of cytoplasmic vacuoles were seen in the epithelial cells. CL = capillary lumen; EP = epithelial cell. Bar = 5 µm.
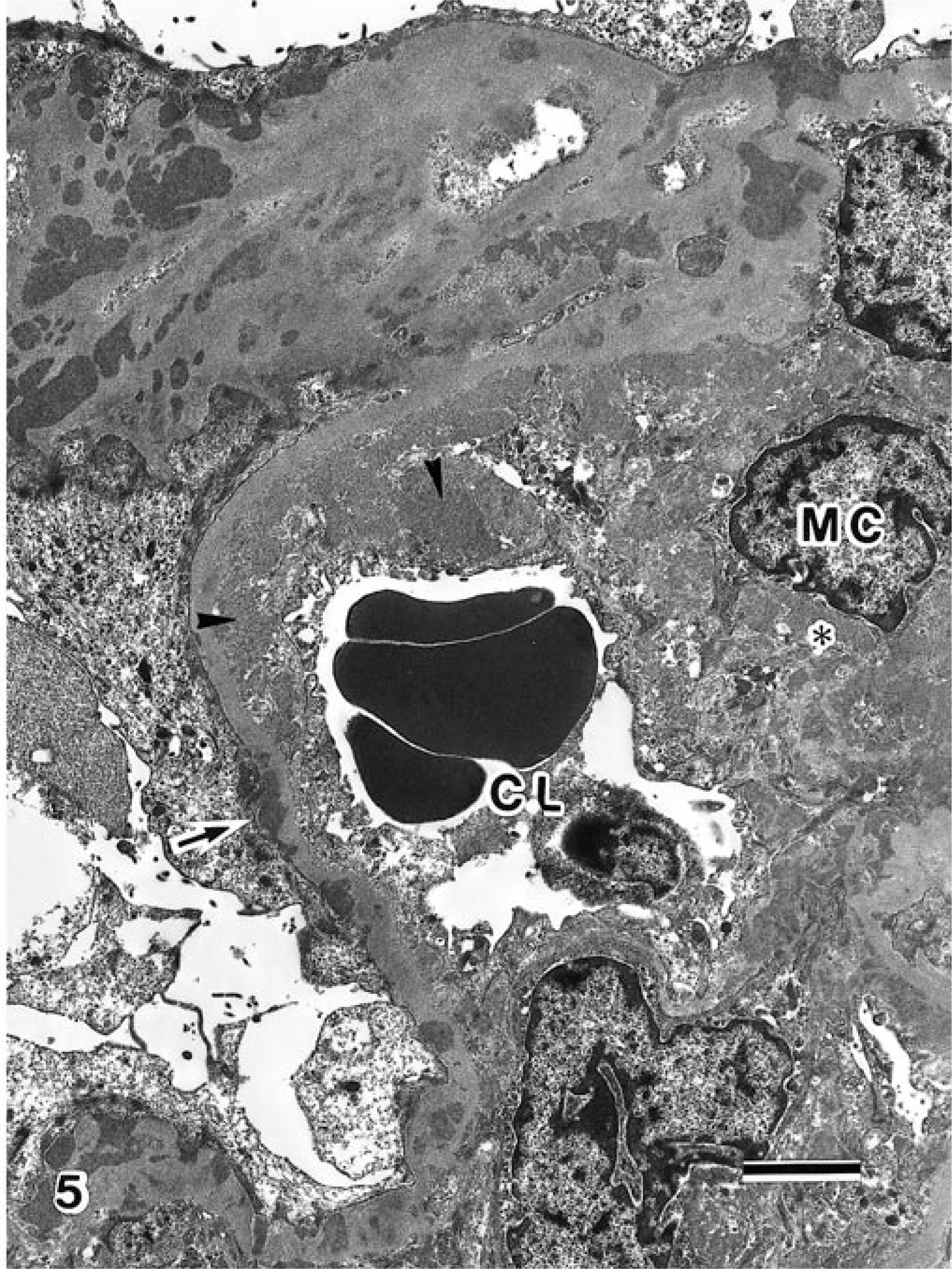
Transmission electron micrograph. Renal glomerulus; cat. Electron dense deposits were found in the subendothelial (arrowheads), mesangial (asterisk), and subepithelial (arrow) areas, leading to narrowing of the capillary lumens. CL = capillary lumen; MC = mesangial cell. Bar = 2.5 µm.
The clinical, histopathologic, immunohistochemical, and electron microscopic findings for the glomerular lesion in this cat share characteristics with those of MPGN type III in humans. 1 Spontaneous and experimental MPGN have been reported in cats, dogs, horses, pigs, and lambs. 4,5 Most of these cases have been type I, type II, or not otherwise classified.
By immunohistochemistry, C3 is almost invariably present, particularly as prominent granular mesangial deposits, in MPGN. 1 Hypocomplementemia and H-factor deficiency, which induce local complement activation, have been noted in swine with hereditary MPGN. 4 In the present study, we used immunohistochemistry to confirmed the granular deposition of C3 and IgG along the capillary walls and in the mesangial areas.
Antibodies against both feline infectious peritonitis virus (FIPV) and feline immunodeficiency virus were found in this cat. ICGN in cats is often associated with FeLV 2 and FIPV. 3 However, the relationship between these viral infections and glomerulonephritis in this cat was not clear.
