Abstract
Hepatoblastomas are neoplasms that originate from putative pluripotential stem cells of the liver. A hepatic mass from an 8-year-old Abyssinian cat was composed of cords and sheets of neoplastic cells, with scattered rosettes and small ductal structures. Most neoplastic cells had a pale eosinophilic cytoplasm and a round to ovoid nucleus. The tumor also had short spindle cells with an oval nucleus. Immunohistochemically, neoplastic cells were weakly positive for embryonic hepatocellular markers, such as alpha-fetoprotein and cytokeratin (CK) 8/18, but negative for the hepatocellular marker Hepatocyte Paraffin 1. The cells were also positive for CD56/neural cell adhesion molecule and for the biliary epithelial markers CK 7, CK 8/18, CK CAM5.2, and vimentin, but negative for CK 20. Some neoplastic cells expressed neuroectodermal or neuroendocrine markers, such as protein gene product 9.5 and synaptophysin, but were negative for chromogranin A and not argyrophilic by the Grimelius technique. The cat died soon after the biopsy without clinical improvement.
Hepatoblastoma, derived from putative pluripotential stem cells of the liver, is the most common hepatic tumor in children.1,2,5,7,9 It is less common in juvenile or adult domestic animals,3,5,8,9 including mice, horses, sheep, and dogs, but has not, to the authors' knowledge, been reported in the cat. The purpose of this report is to present the histologic and immunohistochemical features of a feline hepatoblastoma.
Case History and Clinical Presentation
An 8-year-old spayed female Abyssinian cat was presented for vomiting and anorexia in September 2007. Serum biochemical abnormalities included moderate elevation of glutamic oxaloacetic transaminase (200 U/L), glutamate pyruvate transaminase (797 U/L), and alkaline phosphatase (884 U/L). Upon echography, bile ducts were dilated throughout the liver. Despite supportive therapy for hepatic dysfunction, there was no apparent improvement in the cat’s condition. The cat died shortly after laparotomy and resection of a hepatic nodule. Necropsy was not performed.
Pathologic Findings
The hepatic biopsy specimen, fixed in 10% phosphate-buffered formalin (pH 7.4) and submitted to Marupi Lifetech Company, contained a well-demarcated, off-white nodule, about 8 mm in diameter. The specimen was processed routinely for histologic examination; paraffin sections, 4 μm in thickness, were stained with hematoxylin and eosin (H&E). The Grimelius technique was used to detect neuroendocrine granules.
Immunohistochemistry was performed by the avidin-biotin-peroxidase complex technique with primary antibodies against human cytokeratin (CK) 7, CK 20, CK 8/18, CK CAM5.2 (CAM5.2), vimentin, alpha fetoprotein (AFP), Hepatocyte Paraffin 1 (HepPar-1), CD56/neural cell adhesion molecule (NCAM), protein gene product (PGP) 9.5, synaptophysin, and chromogranin A (Table 1). All antibodies were diluted in DAKO antibody diluent.
Primary Antibodies for Immunohistochemistry and Reactivity of Hepatoblastoma
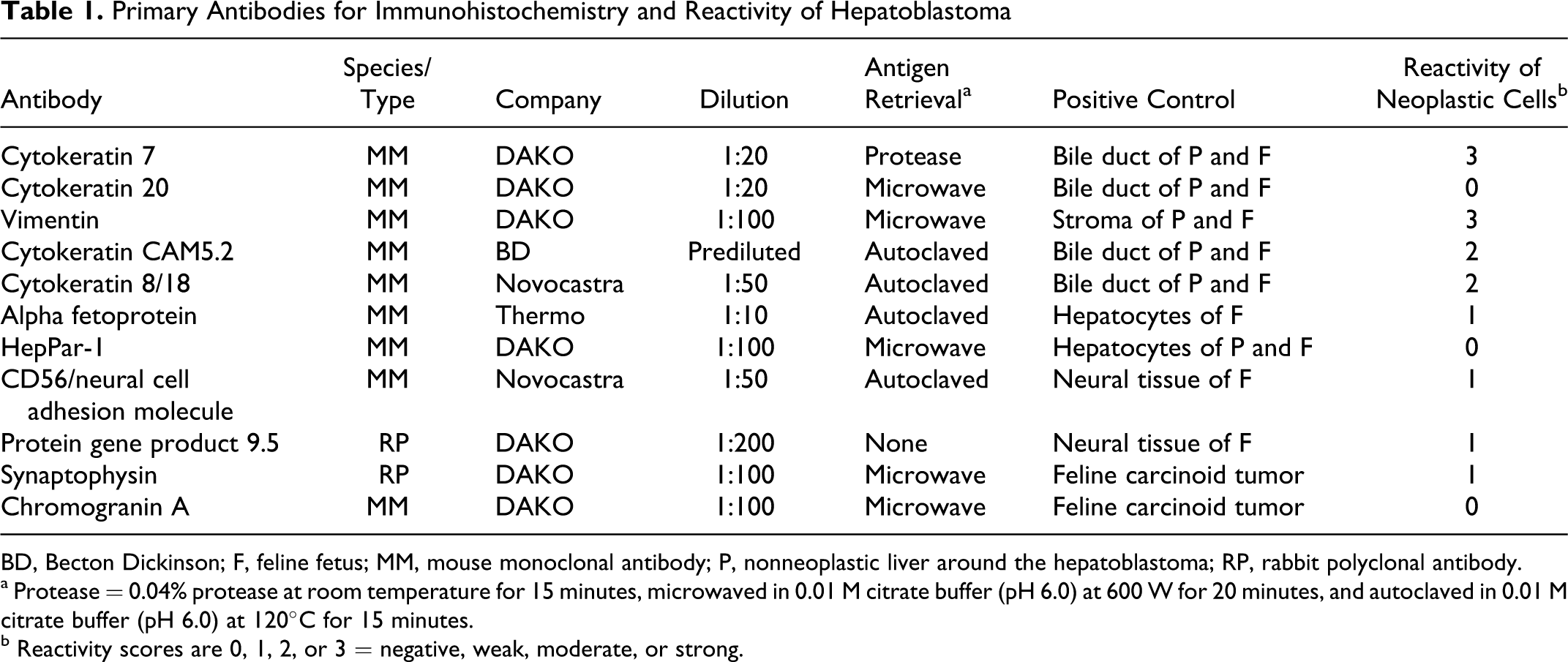
BD, Becton Dickinson; F, feline fetus; MM, mouse monoclonal antibody; P, nonneoplastic liver around the hepatoblastoma; RP, rabbit polyclonal antibody.
a Protease = 0.04% protease at room temperature for 15 minutes, microwaved in 0.01 M citrate buffer (pH 6.0) at 600 W for 20 minutes, and autoclaved in 0.01 M citrate buffer (pH 6.0) at 120°C for 15 minutes.
b Reactivity scores are 0, 1, 2, or 3 = negative, weak, moderate, or strong.
Adjacent nonneoplastic hepatic tissues from the surgical biopsy specimen and feline fetal liver were used as control tissues. Normal rabbit serum was used for the negative controls for PGP9.5 and synaptophysin. Mouse immunoglobulin of the same isotype as the primary antibody was used for negative controls for CK 20, vimentin, CAM5.2, CK 8/18, AFP, HepPar-1 Hepatocyte Paraffin 1, CD56 (NCAM), and chromogranin A. Secondary antibody was N-Histofine simple stain MAX PO (M or R) kit (Nichirei, Tokyo, Japan).
Histologically, the hepatic nodule was demarcated from adjacent hepatic tissue (Fig. 1) but unencapsulated and consisted of cords, sheets, and nests of neoplastic cells (Fig. 2) that resembled immature hepatocytes and were small and round to ovoid or spindle shaped. The cells had an indistinct cell membrane, sparse eosinophilic cytoplasm, and a round to oval nucleus with finely granular chromatin. Anisokaryosis was mild, without remarkable atypia or mitotic activity. Nests of neoplastic cells were separated by delicate fibrovascular stroma. Several vascular spaces were dilated with a sinusoidal appearance. The transition from neoplastic tissue to fibrous stroma was abrupt. Focally, the neoplastic cells formed rosettes and ductal structures (Fig. 3). The nuclear/cytoplasmic ratio decreased toward the periphery of the mass. In the peripheral rim of the neoplasm, mesenchymal spindle cells with a large ovoid nucleus were prominent and associated with fibrous to mucinous stroma (Fig. 1).
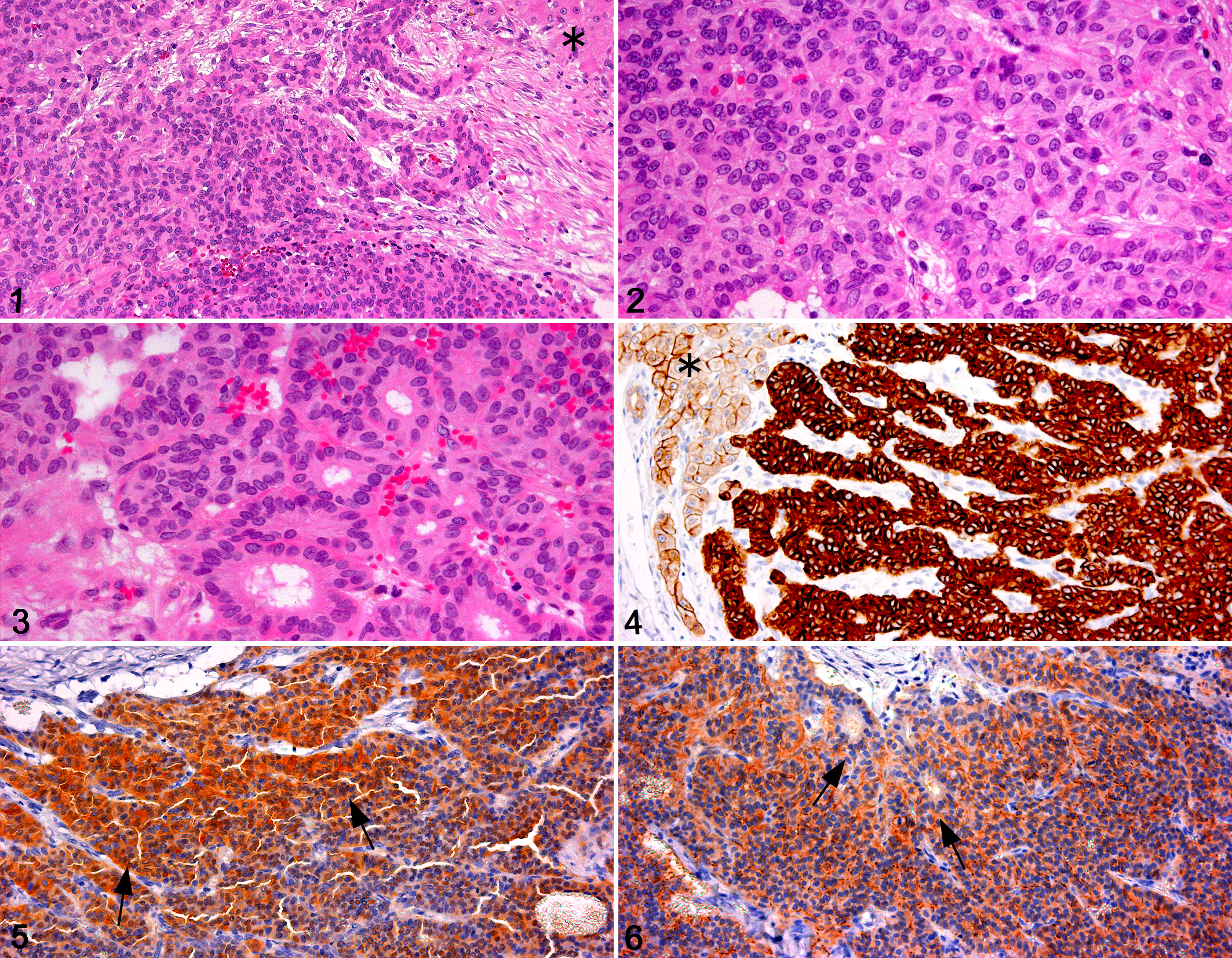
Immunohistochemistry methods and reactivity scores (0, 1, 2, or 3 = negative, weak, moderate, or strong) are summarized in Table 1. The neoplastic cells with uniform round nuclei were weakly positive for AFP and variably positive for CK 8/18 but did not express HepPar-1. The neoplastic cells, including those in ductal structures, were also positive for CK 7 (Fig. 4), CK 8/18, CAM5.2, and vimentin but negative for CK 20. Some neoplastic cells expressed neural markers such as PGP 9.5 (Fig. 5), CD56 (NCAM) (Fig. 6), and synaptophysin but were negative for chromogranin A and lacked argyrophilic granules with the Grimelius technique. In adjacent nonneoplastic liver and in fetal feline liver, hepatocytes were variably positive for AFP and positive for HepPar-1, and some fetal hepatocytes expressed vimentin. Nonneoplastic adult, but not fetal, feline hepatocytes were positive for CK7, CK 8/18, and CAM5.2. Bile duct epithelial cells of adult and fetal liver expressed CK 7, CK 8/18, CAM5.2, vimentin, and CD56 (NCAM). Oval cells were positive for AFP, CK 7, CK 20, CAM5.2, vimentin, and CD56 (NCAM). CK 20 was expressed in fetal hepatocytes, bile duct epithelial cells, and oval cells but only in bile duct epithelial cells in adult liver. No immunoreactivity for PGP 9.5 or synaptophysin was detected in nonneoplastic adult or fetal liver.
Discussion
Hepatoblastomas are variably composed of embryonic or fetal hepatocytes with or without components of fibrous tissue, osteoid, or skeletal muscle.1–3,5–9 Small, round cells are arranged in sheets, irregular cords, trabeculae, pseudorosettes, acini, or nests; the neoplastic cells resemble fetal or embryonal hepatocytes and blastemal cells.1,3,5–9 Hepatoblastoma is most frequently reported in the mouse, in which the tumors consist of short spindle-shaped cells, with formation of organoid structures and rosettes. 6 The tumor in this cat was also composed of cords, sheets, and rosettes of small round or ovoid cells that resembled immature hepatocytes. Immunohistochemically, the neoplastic cells expressed markers of fetal (embryonic) hepatocytes, biliary epithelial cells, and oval cells. Thus, this tumor was diagnosed as hepatoblastoma.
Human hepatoblastoma is diagnosed most often in children.1,5,7–9 In contrast, hepatoblastoma of animals is not always diagnosed in the young. It has been reported in juvenile and mature sheep, neonatal and young adult horses, a 13-year-old dog, and in many aged mice.3,5,6,8,9 The cat of this report was 8 years old.
The differential diagnosis for a hepatic mass in a cat includes hepatocellular carcinoma, cholangiocellular carcinoma, hepatocholangiocarcinoma, and carcinoid (neuroendocrine carcinoma). Hepatocellular carcinomas are composed of neoplastic cells that resemble mature hepatocytes with trabecular or plate-like architecture 3 but express markers of immature hepatocytes (AFP and HepPar-1). However, in the present case, much of the tumor lacked apparent trabecular patterns, and immunoreactivity for markers of immature hepatocytes was weakly positive in only some neoplastic cells, whereas almost all neoplastic cells expressed markers of biliary epithelium (CK 7, CK 8/18, and CAM5.2).
Cholangiocellular carcinomas have tubular and/or papillary patterns in ample fibrous stroma. 3 The neoplastic cells diffusely express biliary epithelial markers. Although neoplastic cells in the present case formed focal tubular structures, the pattern in most areas differed from that of cholangiocarcinoma, and the fibrous stroma appeared more like primitive mesenchyme with large nuclei and an edematous matrix.
Hepatocholangiocarcinomas have histologic characteristics of both hepatocellular carcinoma and cholangiocarcinoma, including relatively differentiated hepatocellular and biliary components. Although some neoplastic cells in the present case expressed markers of both hepatocytes and biliary epithelium, the tumor lacked differentiated hepatocytes or biliary epithelium.
Carcinoids consist of cords or ribbons of polygonal to rounded uniform cells with or without rosette formation and separated by fine fibrovascular stroma. 3 Immunohistochemically, neoplastic cells of the present case expressed several neuroendocrine markers and focally resembled those of carcinoid in cytologic features and histologic pattern. However, the negative argyrophil reaction for neuroendocrine granules with the Grimelius technique and the negative immunoreactivity for chromogranin A, along with the primitive mesenchymal component, are more consistent with hepatoblastoma than with carcinoid.
In both humans and animals, the neoplastic cells of hepatoblastoma variably express markers of hepatocytes, biliary epithelium, and neuroectodermal tissue.1,2,5,7,8 Although AFP is considered the most consistent immunohistochemical marker in hepatoblastoma1,5,8 and was positive in the present case (and in feline fetal liver), the neoplastic cells of mouse hepatoblastoma did not express AFP. 6 The immunohistochemical expression of AFP, CK 7, CK 8/18, CD56 (NCAM), CAM5.2, and vimentin in the neoplastic cells of this feline hepatoblastoma was similar to that in oval cells of the feline fetal liver. CK 7 and CD56 may be useful as immunohistochemical markers of oval cells, which are considered hepatic progenitor cells that can differentiate into hepatocytes and bile duct epithelial cells.4,10 CK 7 expression appeared in small cells in the canal of Hering in normal feline liver and in small- to intermediate-sized cells of the ductular reaction to feline hepatic disease. 4 The expression by the neoplastic cells of both CK7 and CD56 suggests origin from hepatic progenitor cells (oval cells).
Footnotes
The author(s) declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
The author(s) declared that they received no financial support for their research and/or authorship of this article.
