Abstract
A hereditary cerebellar degenerative disorder has emerged in Scottish Terriers. The aims of this study were to describe and quantify polyglucosan body accumulation and quantify Purkinje neurons in the cerebellum of affected and control dogs. The brains of 6 affected Scottish Terriers ranging in age from 8 to 15 years and 8 age-matched control dogs were examined histopathologically. Counts of Purkinje neurons and polyglucosan bodies were performed in control and affected dogs on cerebellar sections stained with periodic acid–Schiff. Affected dogs showed a significant loss of Purkinje neurons compared with control dogs (vermis: P < .0001; hemisphere: P = .0104). The degeneration was significantly more pronounced dorsally than ventrally (P < .0001). There were significantly more polyglucosan bodies in the ventral half of the vermis when compared with the dorsal half (P < .0001) in affected dogs. In addition, there were more polyglucosan bodies in the ventral half of the vermis in affected dogs than in control dogs (P = .0005). Polyglucosan bodies in all affected dogs stained positively with toluidine blue and alcian blue. Immunohistochemically, polyglucosan bodies in affected dogs were positive for neurofilament 200 kD and ubiquitin and negative for glial fibrillary acidic protein, synaptophysin, neurospecific enolase, vimentin, and S100; the bodies were negative for all antigens in control dogs. Ultrastructurally, polyglucosan bodies in 1 affected dog were non–membrane-bound, amorphous structures with a dense core. This study demonstrates significant Purkinje cell loss and increased polyglucosan bodies in the cerebellum of affected Scottish Terriers.
Keywords
Hereditary cerebellar degeneration is a common neurodegenerative disorder that affects a broad range of species. 9 This disorder, often also known as cerebellar abiotrophy, is a major concern in several dog breeds. 9 Generally, affected dogs show cerebellar signs characterized by nystagmus, a base-wide stance, hypermetric gait, ataxia, and intention tremors. 10 The lesion typically is localized to the cerebellar cortex. Primary loss of Purkinje neurons with depletion of molecular and granular layers has been identified as a hereditary trait in American Staffordshire Terriers, 29 Kerry Blue Terriers, 11 Gordon Setters, 33 Old English Sheepdogs, 34 Beagles, 23 and Rhodesian Ridgebacks. 5 Extensive loss of granular neurons with sparing of Purkinje neurons has also been described in Border Collies. 31 Lesion distribution, clinical signs, age of onset, and severity vary among breeds, likely reflecting a range of underlying causes. In Scottish Terriers, the disorder is inherited by an autosomal recessive mode, and affected dogs exhibit purely cerebellar signs with a relatively mild phenotype. 40 Histopathologically, Purkinje neurons are lost, the molecular and granular layers are atrophied, and polyglucosan bodies are present in the molecular layer. 40 However, there has not been a systematic quantification and mapping of Purkinje neurons and polyglucosan bodies. The aims of this study were to quantify Purkinje neurons and polyglucosan bodies as well as to describe the immunohistochemical and ultrastructural features of the polyglucosan bodies in affected and control dogs.
Materials and Methods
Case Histories and Samples
Brains were obtained from Scottish Terriers with clinical signs and a history consistent with cerebellar degeneration and from age-matched control dogs. Scottish Terriers were recruited through the Scottish Terrier Club of America; owners consented to examination of their dogs’ brains following euthanasia. These dogs were the same individuals described in our previous work. 40 Control dogs were recruited through the pathology service at the Veterinary Teaching Hospital, North Carolina State University; to be included they had to be aged 10 years or older with no history of neurological abnormalities.
Brains were obtained from 6 affected Scottish Terriers ranging from 8 to 15 years of age (case Nos. 1-6, mean 11.5 years, SD 2.26, 4 males, and 2 females). Four of the dogs were euthanatized for various reasons including kidney failure, liver failure, Cushing disease, Lyme disease, lung neoplasia, and bladder neoplasia. The remaining 2 dogs were euthanatized as a result of their cerebellar signs. At the time of euthanasia, all dogs displayed either moderate or severe neurological signs. 40 Brains were also obtained from 4 male and 4 female age-matched control dogs (a 10-year-old Scottish Terrier, a 11-year-old Labrador Retriever, a 14-year-old Dachshund, a 12-year-old Labrador Retriever, a 10-year-old Dachshund, a 12-year-old Labrador Retriever, a 12-year-old mixed breed, and a 13-year-old Dalmatian, case Nos. 7-14, respectively). Case Nos. 7 and 14 were analyzed by immunohistochemistry and electron microscopy but not morphometry.
All brains were removed and placed in 10% neutral buffered formalin. The brains of 3 affected and 7 control dogs were removed within 12 hours after death. The brains of 3 affected and 1 control dog were removed within 12 to 24 hours after death. For 1 affected dog, additional tissues were evaluated, including cervical spinal cord, sciatic nerve, muscle, heart, lung, liver, spleen, pancreas, thyroid gland, adrenal gland, and popliteal and mandibular lymph nodes. Following fixation, the cerebellum was sectioned in the midsagittal plane. One-half of the cerebellum was cut into sequential sagittal sections, approximately 3 mm in thickness. The other half was sectioned transversely, again in 3-mm-thick sequential sections. Sections were embedded in paraffin using routine techniques. Transverse sections of the brain stem, cerebrum, and thalamus were also embedded as were the other tissues available in 1 dog. Five-micrometer histological sections were stained with hematoxylin and eosin, periodic acid–Schiff (PAS), toluidine blue, and Alcian blue.
Morphometry
The midsagittal section was selected for morphometric analysis of the vermis. To map the distribution of Purkinje neurons, Purkinje neurons were counted along systematically drawn lines in the midsagittal cerebellar section from control and affected dogs. A microscope (Nikon AZ100, Nikon Instruments Inc, Melville, New York) was connected to a digital camera (Nikon DS-Ri1, Nikon Instruments Inc) to capture and display the image on a computer screen. The cerebellar tissue section was divided into regions by a series of regularly spaced parallel and perpendicular counting lines (Fig. 1). A middle dorsoventral (MDV) line was drawn first, starting just rostral to the primary fissure and extending ventrally to the nodulus. This line divided the cerebellum into rostral and caudal halves. A second line was drawn perpendicular to and bisecting the first line, called the middle rostrocaudal (MRC) line. This line divided the cerebellum into dorsal and ventral halves. Two additional dorsoventral lines were drawn parallel to the first line to divide the MRC line equally into 4 segments, producing a rostral dorsoventral (RDV) line and a caudal dorsoventral (CDV) line, which together with the MDV line defined 4 dorsoventral cerebellar regions. Finally, 2 lines were drawn parallel to the MRC line to divide the MDV line equally into 4 segments, producing the dorsal rostrocaudal (DRC) line and the ventral rostrocaudal (VRC) line, which defined 4 rostrocaudal regions. Nikon-Elements AR 3.0 software (Nikon Instruments Inc) was used to place a 90 000-μm2 grid over the image viewed at 20× magnification. This counting grid was placed at specific locations along the counting lines. The grid was centered over 1 end of each counting line and was moved sequentially down the line to count all neurons along that line. All Purkinje neurons in the counting grid field with a visible nucleus that were located at the junction between the molecular and granular layer were counted. The counts were performed by 1 observer (G.U.). When viewing PAS-stained sections, polyglucosan bodies with a diameter larger than 3 μm found in the molecular layer were counted in the same way. The polyglucosan bodies’ diameter was measured using the Nikon-Elements AR 3.0 measurement tool. Smaller fragments of PAS-positive material identified at the junction between the molecular and granular layers could not be conclusively distinguished from nonpolyglucosan, PAS-positive, cellular components and were excluded from the counting process. Purkinje neuron and polyglucosan body counts per 90 000 μm2 were performed for each of the 6 counting lines in affected and control dogs. Because each line was divided into 4 sections, the mean number of Purkinje neurons and polyglucosan bodies per 90 000 μm2 was calculated for each of those sections and reported in tabular form to demonstrate regional gradients in counts. The mean counts along each line (RDV, MDV, CDV, DRC, MRC, and VRC) and the mean total counts in affected and control dogs were compared using the Wilcoxon rank sum test. The differences in the regions of the vermis (dorsal vs ventral; the mean of all counts above vs below the MRC line and rostral vs caudal; the mean of all counts rostral vs caudal to the MDV line) were compared within and between control and affected dogs in the same way. Simple linear regression analysis was performed to investigate the relationship between the number of Purkinje neurons (independent variable) and the number of polyglucosan bodies (dependent variable) per 90 000 μm2 in affected dogs.
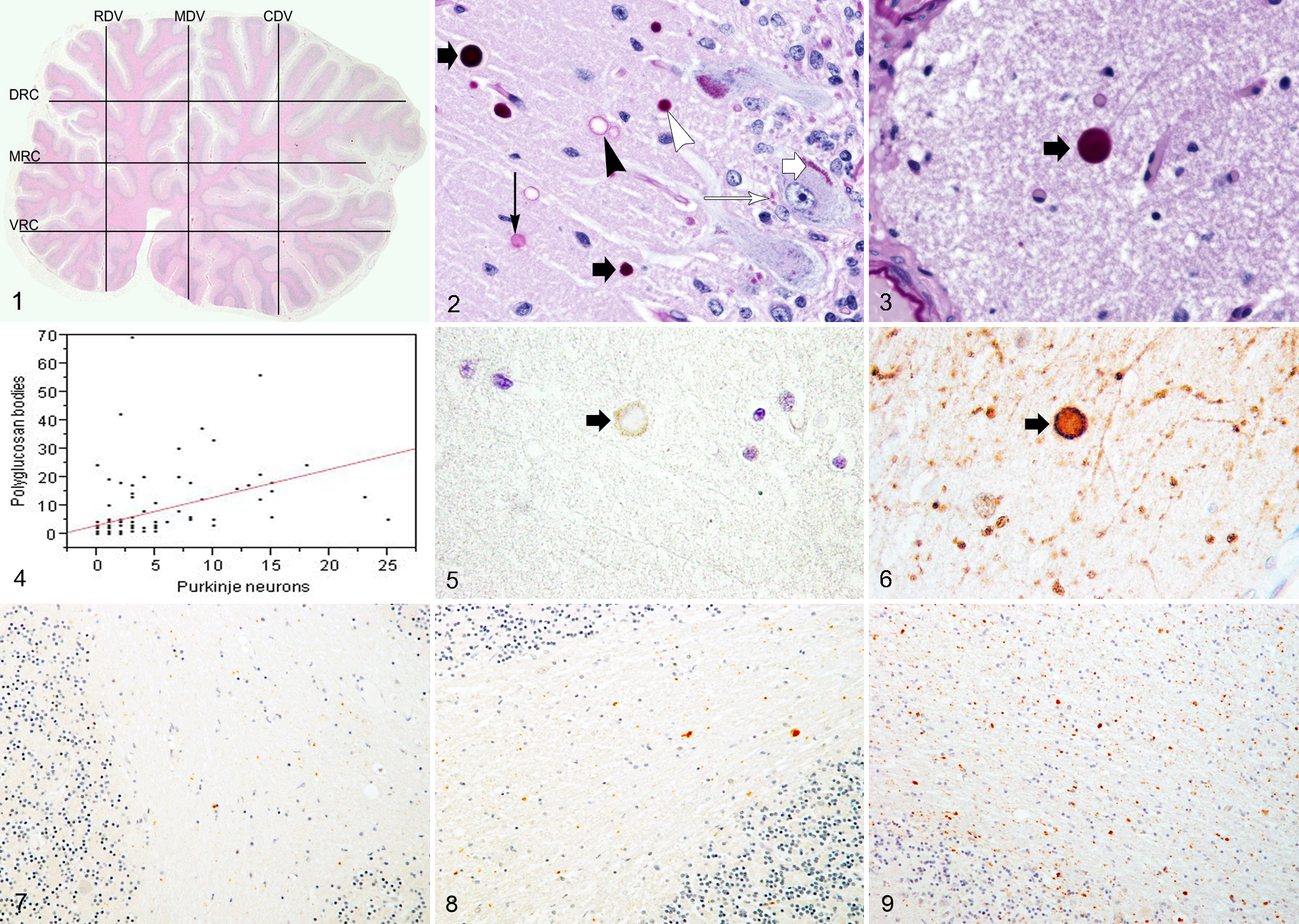
The midsagittal cerebellar section of an 11-year-old Scottish Terrier with hereditary cerebellar degeneration (case No. 3) showing the counting lines superimposed. RDV, rostral dorsoventral line; MDV, middle dorsoventral line; CDV, caudal dorsoventral line; DRC, dorsal rostrocaudal line; MRC, middle rostrocaudal line; VRC, ventral rostrocaudal line.
To quantify Purkinje neurons and polyglucosan bodies in the cerebellar hemispheres, a transverse section of one-half of the cerebellum was evaluated. The hemispheres have a much greater proportion of white matter than the vermis, with correspondingly fewer Purkinje neurons, thus limiting the ability to map dorsoventral and rostrocaudal gradients. Purkinje neurons and polyglucosan bodies were counted in 2 nonoverlapping, 90 000-μm2 grid fields located in the deepest area of each sulcus of the dorsal folia of the cerebellar hemisphere. Four consecutive sulci were counted in each dog starting with the sulcus of the folia adjacent to the vermis and the results expressed as a mean of all 8 fields counted. These folia were selected for counting because they were present in every case. The mean cerebellar hemisphere counts of Purkinje neurons and polyglucosan bodies were compared in affected and control dogs using Wilcoxon rank sum test. In addition, the mean counts in the vermis and in the hemispheres were compared within and between affected and control dogs. P values <.05 were taken as significant. The statistical analysis was performed on JMP version 8.0 (SAS Institute Inc, Cary, North Carolina).
Immunohistochemistry
To further characterize the polyglucosan bodies, immunohistochemical staining for glial fibrillary acidic protein (GFAP), synaptophysin, neuron-specific enolase (NSE), vimentin, S100, neurofilament 200 kD (NF-200), and ubiquitin was performed. Briefly, 5-μm histological sections were dewaxed in xylene and rehydrated through a gradient series of ethanol baths. Endogenous peroxide activity was quenched by incubation in 3% hydrogen peroxide for 10 minutes. Sections were then rinsed with a wash solution (an automation buffer) and digested with ready-to-use Proteinase K for 5 minutes for epitope retrieval. After a second buffer rinse, sections were incubated with the primary antibody (Supplemental Table 1, found at http://vet.sagepub.com/supplemental), washed, and incubated with the secondary antibody-biotinylated goat anti-rabbit or mouse immunoglobulin (LSAB2 System-HRP) as appropriate for 10 minutes. After washing with buffer, sections were incubated in peroxidase-labeled streptavidin (LSAB2 System-HRP) for 10 minutes, developed with diaminobenzoic acid substrate–chromogen solution for 5 minutes, and counterstained with hematoxylin. Reagents were obtained from Dako North America, Inc (Carpentaria, CA), except where otherwise stated in Supplemental Table 1.
Electron Microscopy
Transmission electron microscopy (TEM) was performed using formalin-fixed cerebellum embedded in paraffin blocks and using fresh cerebellum when available. Sections of paraffin-embedded cerebellum containing polyglucosan bodies were cut from the cerebellar cortex of the vermis, deparaffinized, rehydrated, placed in 1% osmium tetroxide (EMS 19110, Hatfield, PA) in 0.1 M phosphate buffer for 1 hour, rinsed twice with distilled water, and dehydrated in a concentration gradient series of ethanol baths, followed by 100% acetone double rinse. When fresh brain or formalin-fixed tissue was available, pieces of cerebellar cortex from the vermis and hemisphere were cut into approximately 1-mm2 cubes and placed in McDowell’s and Trump’s 4F:1G fixative. Tissue samples were rinsed twice with 0.1 M sodium phosphate buffer (pH 7.2) prior to incubation in 1% osmium tetroxide for 1 hour. The samples were then placed in a mixture of Spurr resin (EMS Spurr resin kit 14300, Hatfield, PA) and acetone (1:1) for 30 minutes, followed by 2 changes of 100% resin for 2 hours each. Subsequently, the samples were placed in resin that was polymerized at 70°C for 8 hours. Semi-thin sections (0.25 μm) were cut, stained with 1% toluidine blue-O in 1% sodium borate, and used to identify areas of interest. Ultrathin sections (70-90 nm) of these areas, stained with methanolic uranyl acetate (EMS 22400), followed by lead citrate, were examined by TEM (FEI/Philips EM208S TEM, Hillsboro, OR). Reagents were obtained from Fisher Scientific (Pittsburgh, PA) unless otherwise indicated.
Results
Histopathological Findings
There was degeneration of all 3 layers of the cerebellar cortex of affected dogs, especially at the tips of the folia. Histochemical staining of the brains of affected dogs with PAS revealed polyglucosan bodies ranging from 3 to 30 μm in diameter located in the molecular layer of the cerebellum that were most numerous at the base of sulci. Sometimes there were linear chains of 2 to 4 bodies in a row. There were also smaller PAS-positive fragments (<3 μm) at the junction of the molecular and granular layers (Fig. 2). Four different PAS staining patterns were identified; the first had a strong PAS-positive homogenous core; the second had light, uniform PAS-positive staining with lighter peripheral staining; the third had faint central staining with a peripheral darker ring of PAS-positive staining; and the last had negative staining at the center but had a peripheral ring of PAS-positive staining (Fig. 2). All polyglucosan bodies stained positively with toluidine blue and alcian blue. Polyglucosan bodies with 4 different staining patterns were present in the molecular layer of the controls (Fig. 3). Occasional polyglucosan bodies were also found in the hippocampus (CA1 and CA3) and sporadically throughout the brain in both affected and control dogs. No polyglucosan bodies were found in other visceral tissues in the 1 case examined.
The molecular layer of the cerebellar cortex of an affected 11-year-old Scottish Terrier (case No. 2). The round dense core structure of a polyglucosan body is shown by transmission electron microscopy. 7100×.
Morphometry
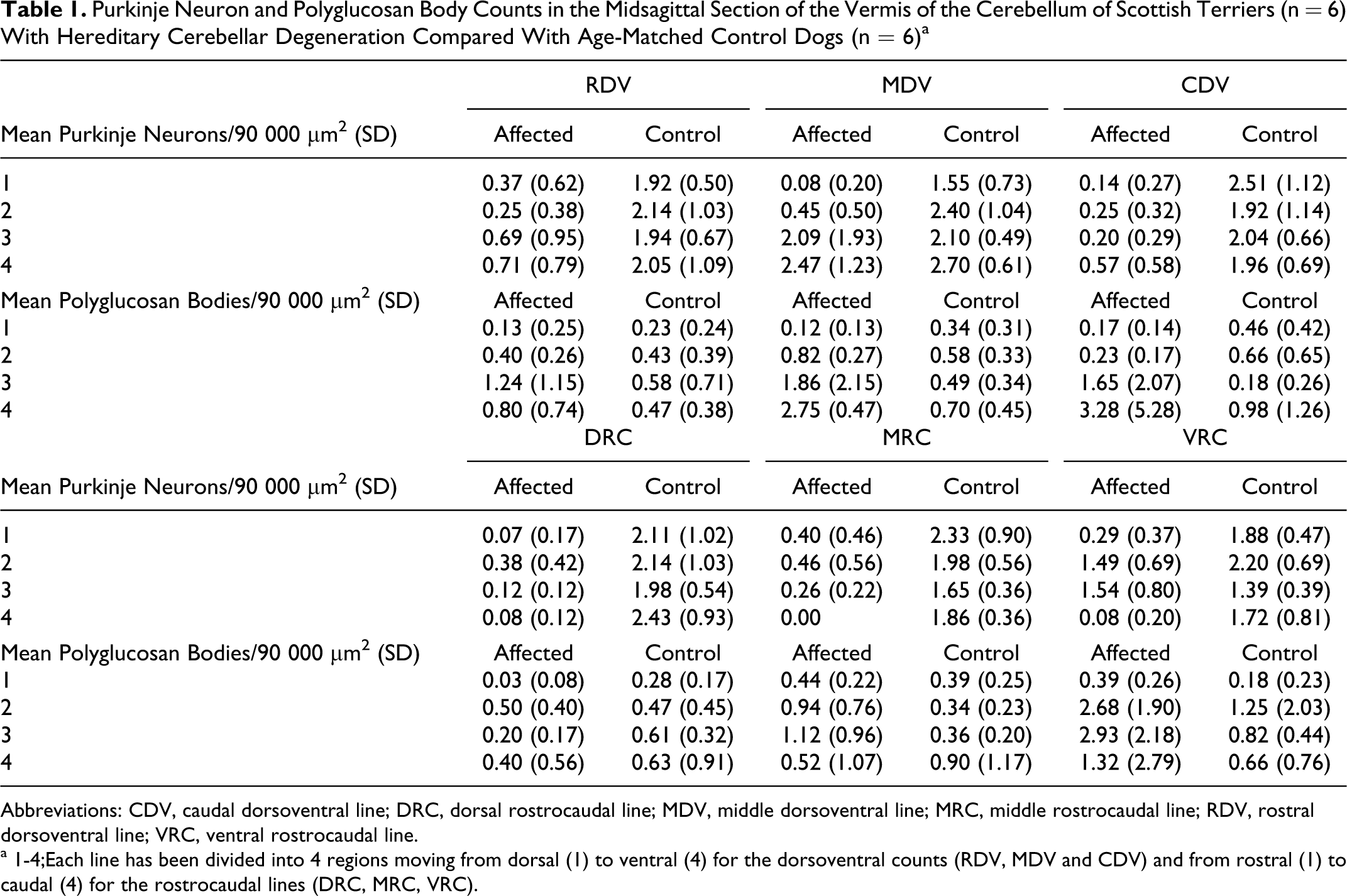
Details of the Purkinje neuron counts in the midsagittal section of the vermis are given in Table 1 and clearly show an increasing gradient in neuron and polyglucosan body density from dorsal to ventral in affected dogs. The mean number of Purkinje neurons per 90 000 μm2 in the vermis was 2.04 (range, 0.50-4.17; SD 0.78) in control dogs and 0.56 (range, 0.00-5.00; SD 0.88) in affected dogs. Compared with the controls, there were significantly fewer Purkinje neurons per 90 000 μm2 along each line, in each region (dorsal, ventral, rostral, and caudal), and in the whole vermis of affected dogs (P < .0050). In affected dogs, there were significantly more Purkinje neurons per 90 000 μm2 in the ventral half (mean 0.98; range, 0.00-4.80; SD 1.03) than the dorsal half (mean 0.20; range, 0.00-1.30; SD 0.31) of the vermis (P < .0001), but there was no difference between the rostral (mean 0.52; range, 0.00-2.50; SD 0.61) and caudal (mean 0.35; range, 0.00-1.71; SD 0.47) halves of the vermis (P = .2173). Control dogs showed no difference in the number of neurons per 90 000 μm2 between rostral and caudal (P = .8755) or dorsal and ventral (P = .5637) halves of the vermis. The mean number of Purkinje neurons per 90 000 μm2 in the cerebellar hemisphere was 0.65 (range, 0.00-1.75; SD 0.77) in affected dogs and 2.58 (range, 1.38-3.13; SD 0.64) in control dogs. Affected dogs had significantly fewer Purkinje neurons per 90 000 μm2 than control dogs (P = .0104). The mean number of Purkinje neurons per 90 000 μm2 in the hemispheres was not significantly different from the mean number in the vermis in both control (P = .0604) and affected (P = .6780) dogs.
Purkinje Neuron and Polyglucosan Body Counts in the Midsagittal Section of the Vermis of the Cerebellum of Scottish Terriers (n = 6) With Hereditary Cerebellar Degeneration Compared With Age-Matched Control Dogs (n = 6) a
Abbreviations: CDV, caudal dorsoventral line; DRC, dorsal rostrocaudal line; MDV, middle dorsoventral line; MRC, middle rostrocaudal line; RDV, rostral dorsoventral line; VRC, ventral rostrocaudal line.
a 1-4;Each line has been divided into 4 regions moving from dorsal (1) to ventral (4) for the dorsoventral counts (RDV, MDV and CDV) and from rostral (1) to caudal (4) for the rostrocaudal lines (DRC, MRC, VRC).
Counts of polyglucosan bodies in the vermis are reported in Table 1. The mean number of polyglucosan bodies per 90 000 μm2 was 1.04 (range, 0.00-13.80; SD 1.73) in affected dogs and 0.54 (range, 0.00-5.33; SD 0.68) in control dogs. However, there was no significant difference between affected and control dogs when the mean counts for the whole vermis were compared (P = .1417). In the ventral half of the vermis, affected dogs had significantly more polyglucosan bodies per 90 000 μm2 than control dogs (P = .0005), whereas in the dorsal half of the vermis, affected dogs had significantly fewer polyglucosan bodies per 90 000 μm2 than control dogs (P = .0206). There was no difference in the mean number of polyglucosan bodies in rostral (P = .0564) and caudal (P = .8789) halves of the vermis between control and affected dogs. Control dogs showed no difference in the number of polyglucosan bodies per 90 000 μm2 between rostral and caudal (P = .0777) or dorsal and ventral (P = .5910) halves of the vermis. In affected dogs, there was no difference in the number of polyglucosan bodies between the rostral (mean 0.72; range, 0.00-3.50; SD 0.81) and caudal (mean 1.25; range, 0.00-13.80; SD 2.35) halves of the vermis (P = .8192). However, there were significantly more polyglucosan bodies per 90 000 μm2 in the ventral half (mean 1.91; range, 0.00-9.78; SD 2.16) than the dorsal half (mean 0.30; range, 0.00-1.50; SD 0.32) of the vermis (P < .0001). Thus, compared with controls, there was a relative gain in polyglucosan bodies in the ventral aspect of the vermis of affected dogs and a relative loss in the dorsal aspect. Moreover, there was evidence of a significant linear relationship between the number of Purkinje neurons and polyglucosan bodies in affected dogs (P < .0001, R 2 = 0. 2053) (Fig. 4). The mean number of polyglucosan bodies per 90 000 μm2 in the cerebellar hemispheres was 0.48 (range, 0.25-1.13; SD 0.37) in affected dogs and 0.60 (range, 0.25-1.63; SD 0.52) in control dogs: there was no significant difference between the 2 groups (P = .5714). The mean number of polyglucosan bodies per 90 000 μm2 in the hemispheres was not significantly different from the mean number in the vermis in both control (P = .4832) and affected (P = .7267) dogs.
Immunohistochemistry and Electron Microscopy Findings
Immunohistochemically, polyglucosan bodies in affected dogs had positive immunoreactivity for NF-200 and ubiquitin (Figs. 5 and 6) and were negative for GFAP, NSE, S-100, vimentin, and synaptophysin. In control dogs, polyglucosan bodies were negative for all immunohistochemistry reactions performed. Positive ubiquitin immunoreactivity was detected as granules and globules of various sizes in the white matter in both control and affected dogs, but subjectively there was more positive staining of the white matter in affected dogs (Figs. 7, 8, and 9).
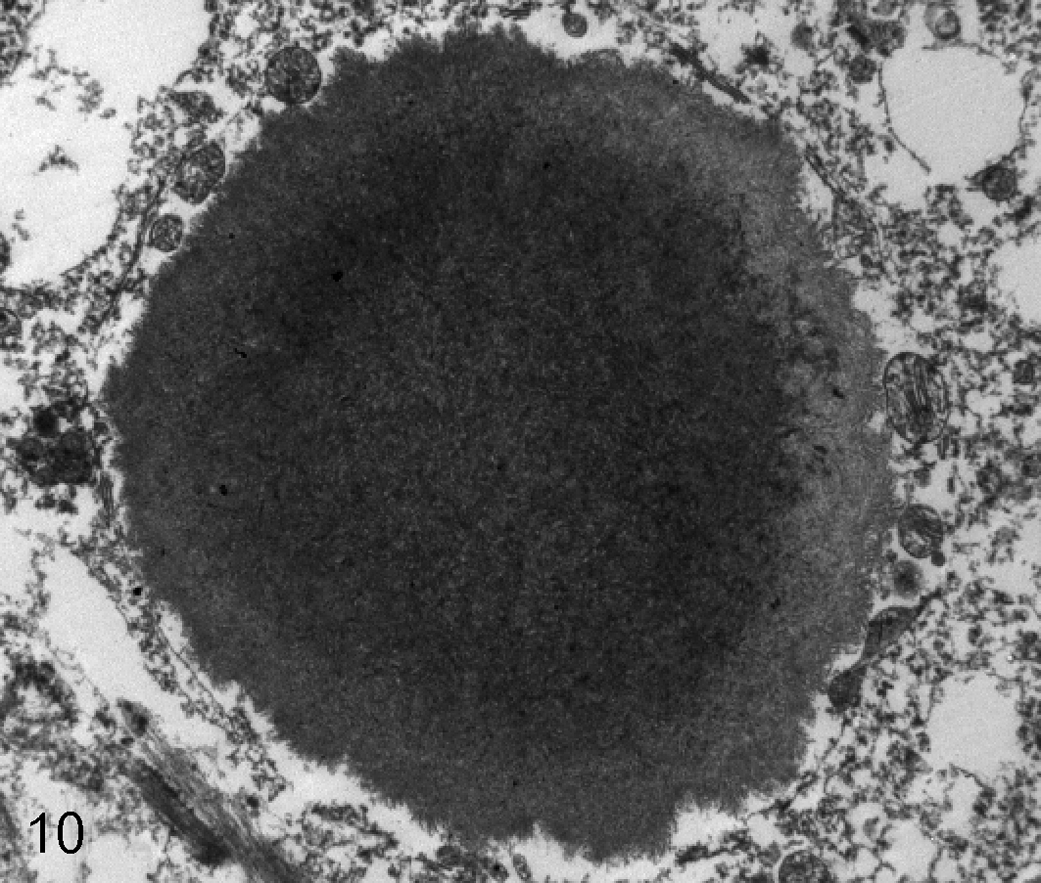
Cerebellar tissues from 2 affected dogs (case Nos. 2 and 6) (1 from a paraffin block and 1 from formalin-fixed tissue) and 2 control dogs (case Nos. 7 and 14) (1 from a paraffin block and 1 from fresh tissue) were examined using electron microscopy. Polyglucosan bodies were only identified from a paraffin block of 1 affected dog. Ultrastructurally, the bodies were non–membrane bound with a dense core structure containing amorphous, moderately electron dense, granular material (Fig. 10). Failure to identify bodies in the other dogs was likely related to fixation artifact and/or the relative paucity of the bodies.
Discussion
Hereditary cerebellar cortical degeneration has become relatively common in Scottish Terriers with an estimated minimum prevalence of 1 in 1335 among AKC-registered Scottish Terriers. 40 The main histopathological finding on evaluation of the cerebellum of diseased dogs is loss of Purkinje neurons with thinning of the granular and molecular layers. 40 Quantification of Purkinje neurons in our study confirmed that there is significant loss of these neurons in affected dogs compared with control dogs. More specifically, Purkinje neuron degeneration in the vermis of the cerebellum was more profound dorsally, especially at the convexity of the folia, than ventrally, but there was no rostrocaudal gradient. Counts of Purkinje neurons in the cerebellum of 6 control dogs did not reveal any geographic differences in cell distribution, confirming that the changes seen in affected dogs are likely due to their underlying disease.
The pattern of Purkinje neuron loss may reflect functional or metabolic differences in dorsally versus ventrally located neuronal populations, although details of such differences are not apparent at this time. The flocculonodular lobe, located ventrally, is part of the phylogenetically oldest part of the cerebellum that controls vestibular function. 38 The sparing of this region is consistent with the lack of vestibular signs in affected Scottish Terriers. 40 A similar distribution of cerebellar pathology has been described in Kerry Blue Terriers that suffer from multiple system atrophy 10 but not in other dog breeds. A similar pattern of relative sparing of Purkinje neurons in the nodulus and uvula is reported in humans with multiple system atrophy, 42 idiopathic late cortical cerebellar atrophy, 30 and spinocerebellar ataxia type 6. 37 However, there is no report of polyglucosan bodies in these diseases. In humans and rodent models, some disorders have been described in which there is differential loss of Purkinje neurons in the hemispheres versus the vermis, although the reasons for this differential loss are unknown. 27,42 In Scottish Terriers, there was no evidence of differential loss of Purkinje neurons between the vermis and hemispheres, and the significance of the dorsoventral gradient of Purkinje neuronal loss is unclear. Attempts to correlate the degree of neuronal loss with the severity of neurological signs were not made because there was no opportunity to accurately quantify their neurological signs at time of euthanasia.
Polyglucosan bodies are accumulations of insoluble complex glycoprotein. They can be associated with normal aging (termed corpora amylacea) or with neurodegenerative diseases such as Lafora disease (termed Lafora bodies) in which the polyglucosan body is a prominent feature of the disease, although their pathological role is still debated. 1,39 The number of corpora amylacea has been reported to increase in association with several other neurodegenerative disorders, in which the polyglucosan body does not appear to be the primary cause of neurodegeneration. 22,44 To determine whether the polyglucosan bodies previously described in affected Scottish terriers 40 were a consequence of their neurodegenerative disease or simply of aging, we quantified the distribution of these bodies and described their staining characteristics and ultrastructural features in both control and affected dogs. Although the density of polyglucosan bodies in affected and age-matched control dogs was not significantly different, in the affected dogs, contrary to expectations, there were significantly more bodies in the ventral half of the cerebellum with surviving Purkinje neurons than in regions in which the Purkinje neuron population was depleted. Although the data points were clustered between 0 and 5, there were enough data to establish that the relationship between the number of Purkinje neurons and polyglucosan bodies was statistically significant. A much larger cohort of affected dogs euthanatized at different stages of their disease would have to be evaluated to determine whether the distribution of polyglucosan bodies is temporally related to the evolution of neural degeneration and to investigate the pattern of Purkinje neuron loss at different disease stages. However, given the relatively mild clinical course, it is unlikely that this opportunity will present itself unless affected dogs are euthanatized at a younger age because of other health problems.
Evaluation of PAS-stained sections of the brain revealed a heterogeneous population of circular bodies that stained positive for PAS within the molecular layer of the cerebellum in both affected and control dogs. Other groups have described canine polyglucosan bodies with a variety of different PAS staining patterns comparable to the patterns identified in our dogs. 8,16 -18,26,35 However, reports of polyglucosan bodies in dogs tend to be inconsistent, with variable descriptions of the distribution in the brain, intracellular location, and immunohistochemical staining characteristics. For example, one group reported that polyglucosan bodies in aged dogs are located in neuronal perikarya and their processes, 45 but another reported that polyglucosan bodies are located in astrocytic processes. 2 Polyglucosan bodies have been reported in many different parts of the brain, 8,16 –18,21,26,35 but the molecular layer of the cerebellum is the most common location of finding these bodies in both control dogs and dogs with Lafora-like disease. 2,7,16,18 One group reported that polyglucosan bodies stained negative for NSE and GFAP in dogs with Lafora-like disease and positive in age-matched control dogs. 26 In our study, the immunohistochemical staining characteristics of polyglucosan bodies in affected dogs were consistent with Lafora-like bodies described previously in dogs, 26 but the polyglucosan bodies found in our control dogs were not positive for any of the antigens evaluated. Although most groups have reported polyglucosan bodies associated with aging as staining positive with ubiquitin in both humans and dogs, 3,6,19,26 one has reported negative staining in aged dogs. 2 A similarly confusing picture has been described in different human reports of corpora amylacea associated with aging, 3 and it is unclear whether bodies with different staining characteristics represent a spectrum of the same body or distinct entities.
The ultrastructure of the polyglucosan bodies of an affected dog in our study differed from the typical fibrillar-like structure of Lafora-like bodies described in dogs and humans. 18 , 20 , 26 , 35 , 45 This may simply reflect artifacts of fixation, because the brain tissue used was obtained well after euthanasia, was fixed in formalin, and was embedded in paraffin prior to being trimmed for TEM. Additional TEM studies in freshly fixed tissue are needed to further characterize the ultrastructural features of this disease.
Ubiquitin plays an important role in intracellular protein degradation through the ubiquitin–proteasome system. Detecting an aggregation of ubiquitin-positive material can imply increased protein degradation and/or insufficient removal of a target protein. 36 Although there is an increase of ubiquitin-positive granules in the brains of aged dogs, 2,14 the apparently higher density of staining in the affected dogs in this study may indicate that there is a higher rate of protein degradation within the white matter than is normal for dogs of this age.
In humans, the presence of polyglucosan bodies has been associated with Lafora disease, an autosomal, recessive, severe neurodegenerative disease characterized by progressive myoclonic seizures with extensive accumulation of polyglucosan material and bodies throughout the brain, especially in the mesencephalon, thalamus, and cerebellum, 3,17,26,45 and in other organs. 3,17 Two underlying mutations in genes EPM2A and B have been identified, 4,28 and their protein products (laforin and malin) have been proposed to play roles in autophagy, 1 maintenance of soluble glycogen, 12 formation of branched glycogen molecules, 24 and the ubiquitin–proteasome degradative system. 15 Similar disorders have also been described in dogs, 16,18,20,32 and a mutation in EPM2B causes myoclonic epilepsy in Wire Haired Dachshunds. 25,41 In contrast to these disorders, the distribution of polyglucosan bodies in Scottish Terriers with cerebellar degeneration is restricted to the cerebellum. Polyglucosan bodies have also been associated with mutations in glycogen branching enzyme, causing weakness, cognitive impairment, and gait abnormalities in humans. 43 A single case report of an early-onset spinocerebellar ataxia associated with polyglucosan bodies was postulated to result from glycogen branching enzyme deficiency. 13 The detailed description of polyglucosan bodies provided in this report suggests that they are an important component of cerebellar degeneration in Scottish Terriers, but their restricted distribution and relatively low density are unique to this disease, distinguishing it from previous reports of Lafora-like disease in dogs.
Footnotes
The work was completed at North Carolina State University, Raleigh, NC.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the American Kennel Club Canine Health Foundation and supported by the Health Trust Fund of the Scottish Terrier Club of America.
